Study of Interactions between Metallothionein and Cisplatin by using Differential Pulse Voltammetry Brdicka´s reaction and Quartz Crystal Microbalance
Abstract
:1. Introduction
2. Results and Discussion
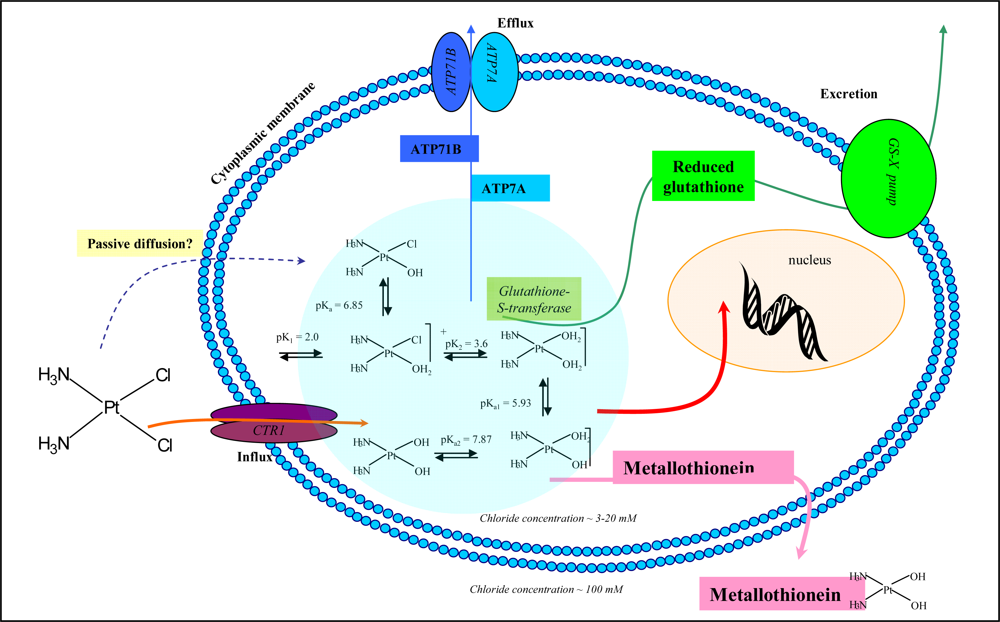
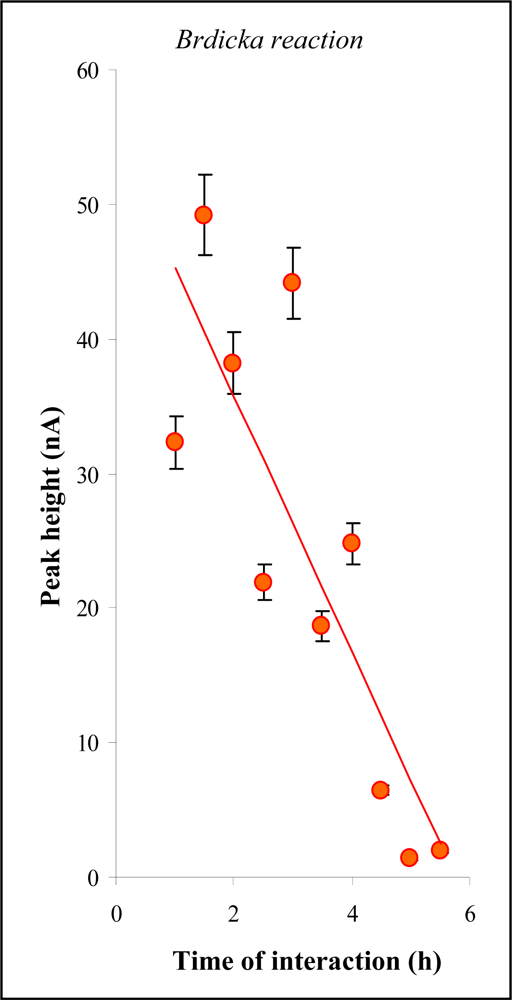
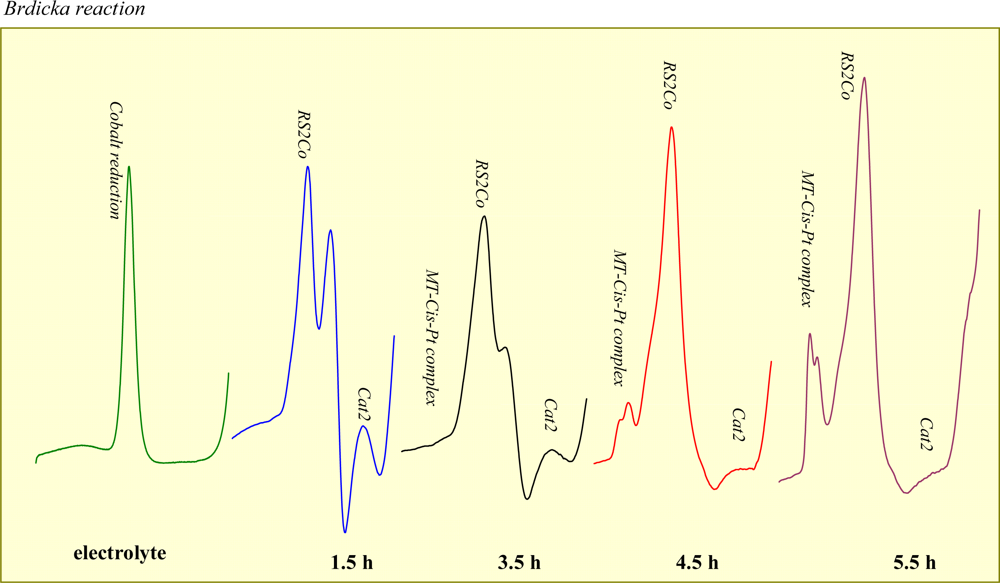
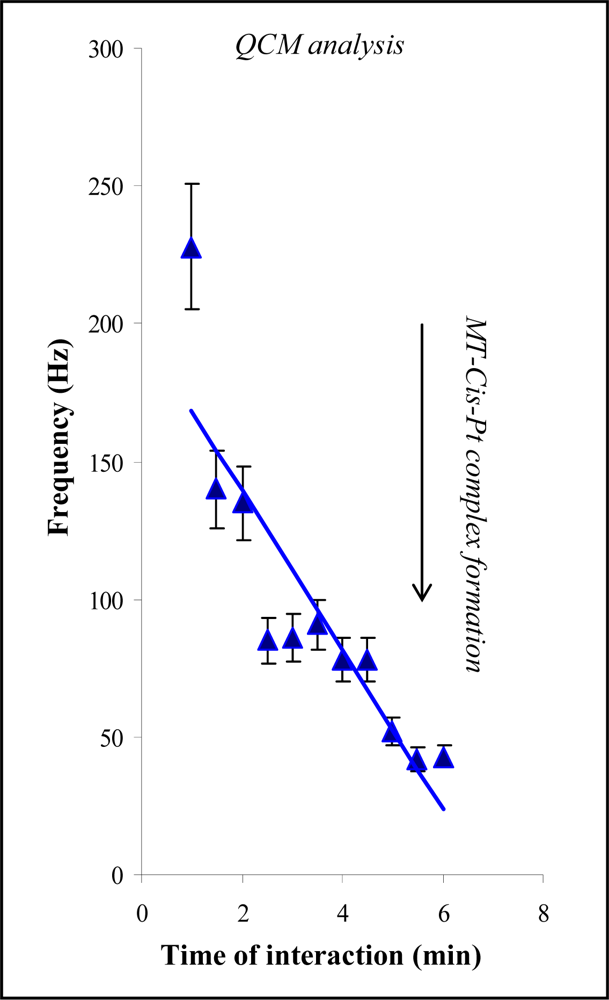
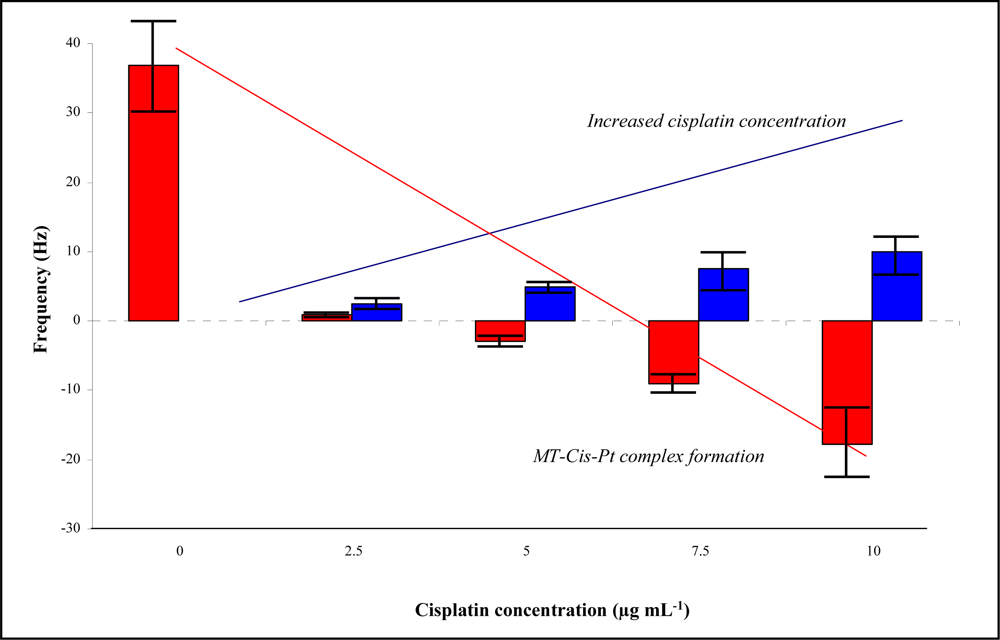
2.1. Metallothionein-cisplatin interaction
2.2. Electrochemical study of metallothionein-cisplatin complexes in rat blood
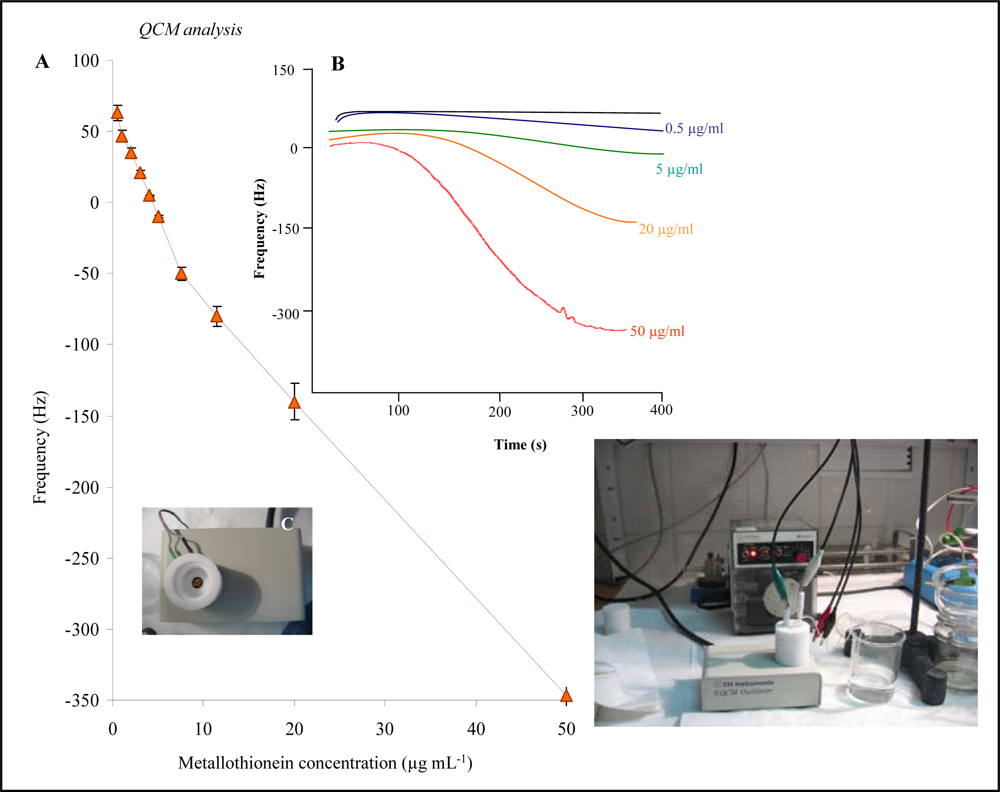
2.3. Detection of MT by quartz crystal microbalance
3. Materials and Methods
3.1. Chemicals, pH measurements and materials
3.2. Preparation of biological samples for metallothionein determination
3.3. Rats
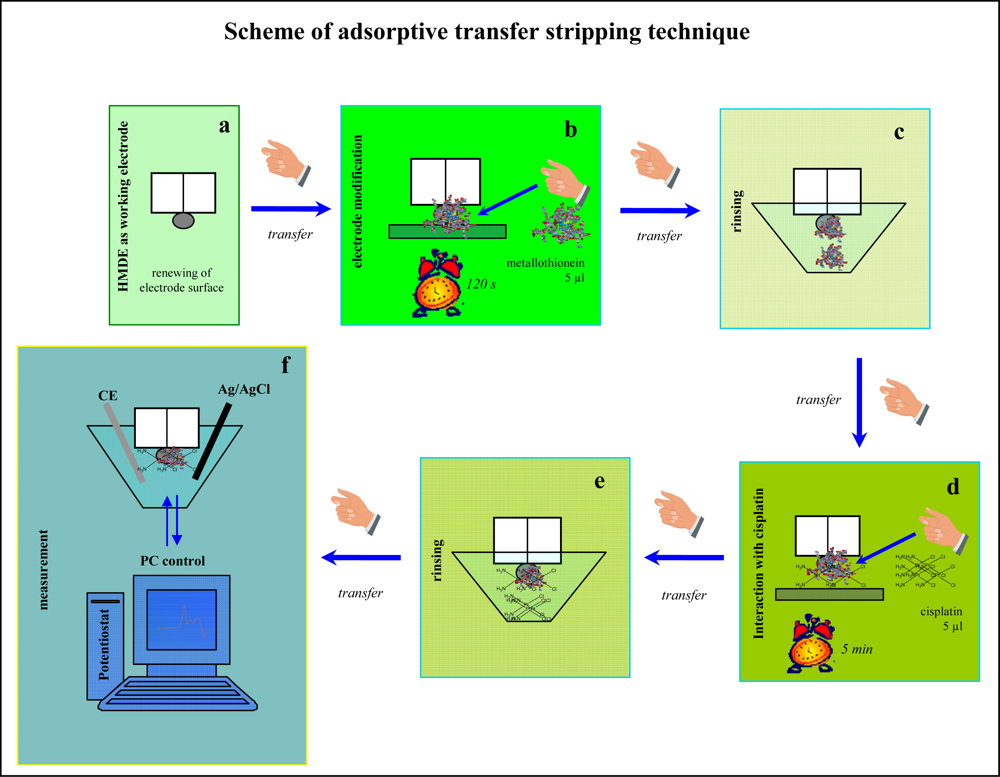
3.4. Electrochemical measurements – Brdickás reaction
3.5. Quartz crystal microbalance
3.6 Descriptive statistics
4. Conclusions
Acknowledgments
References
- Kagi, J.H.R.; Schaffer, A. Biochemistry of Metallothionein. Biochemistry 1988, 27, 8509–8515. [Google Scholar]
- Miles, A.T.; Hawksworth, G.M.; Beattie, J.H.; Rodilla, V. Induction, regulation, degradation, and biological significance of mammalian metallothioneins. Crit. Rev. Biochem. Mol. Biol 2000, 35, 35–70. [Google Scholar]
- Kukacka, J.; Tesfaye, H.; Malis, J.; Adam, V.; Fabrik, I.; Kotaska, K.; Stary, J.; Kizek, R.; Prusa, R. Carboblatin monitoring by flameless atomic absorption spectrophotometry and metallothionein levels in pediatric patients with solid tumors. Clin. Chem 2008, 54, A11–A11. [Google Scholar]
- Zhang, B.L.; Sun, W.Y.; Tang, W.X. Determination of the association constant of platinum(II) to metallothionein. J. Inorg. Biochem 1997, 65, 295–298. [Google Scholar]
- Huska, D.; Krizkova, S.; Beklova, M.; Havel, L.; Zehnalek, J.; Diopan, V.; Adam, V.; Zeman, L.; Babula, P.; Kizek, R. Influence of cadmium(II) ions and brewery sludge on metallothionein level in earthworms (Eisenia fetida) - Biotransforming of toxic wastes. Sensors 2008, 8, 1039–1047. [Google Scholar]
- Prusa, R.; Petrlova, J.; Kukacka, J.; Adam, V.; Sures, B.; Beklova, M.; Kizek, R. Study of interaction of glutathiones and metallothionein with cytostatics. Clin. Chem 2006, 52, A175–A175. [Google Scholar]
- Prusa, R.; Blastik, O.; Kukacka, J.; Zehnalek, J.; Adam, V.; Petrlova, J.; Kizek, R. The influence of platinum-based drugs on the amount of metallothionein. Toxicol. Lett 2005, 158, S66–S67. [Google Scholar]
- Prusa, R.; Blastik, O.; Kukacka, J.; Kizek, R.; Stuchlikova, H. Plasma metallothionein levels in lead poisoned child. Toxicol. Lett 2005, 158, S156–S157. [Google Scholar]
- Diopan, V.; Baloun, J.; Adam, V.; Macek, T.; Havel, L.; Kizek, R. Determination of expression of metallothionein at transgenic tobacco plants. Lis. Cukrov. Repar 2007, 123, 325–327. [Google Scholar]
- Supalkova, V.; Beklova, M.; Baloun, J.; Singer, C.; Sures, B.; Adam, V.; Huska, D.; Pikula, J.; Rauscherova, L.; Havel, L.; Zehnalek, J.; Kizek, R. Affecting of aquatic vascular plant Lemna minor by cisplatin revealed by voltammetry. Bioelectrochemistry 2008, 72, 59–65. [Google Scholar]
- Krizkova, S.; Fabrik, I.; Adam, V.; Kukacka, J.; Prusa, R.; Chavis, G.J.; Trnkova, L.; Strnadel, J.; Horak, V.; Kizek, R. Utilizing of adsorptive transfer stripping technique Brdicka reaction for determination of metallothioneins level in melanoma cells, blood serum and tissues. Sensors 2008, 8, 3106–3122. [Google Scholar]
- Eckschlager, T.; Adam, V.; Hrabeta, J.; Figova, K.; Kizek, R. Metallothioneins and cancer. Curr. Protein Pept. Sci 2009, in press.. [Google Scholar]
- Lobinski, R.; Chassaigne, H.; Szpunar, J. Analysis for metallothioneins using coupled techniques. Talanta 1998, 46, 271–289. [Google Scholar]
- Sestakova, I.; Navratil, T. Voltammetric methods in metallothionein research. Bioinorg. Chem. Appl 2005, 3, 43–53. [Google Scholar]
- Szpunar, J. Bio-inorganic speciation analysis by hyphenated techniques. Analyst 2000, 125, 963–988. [Google Scholar]
- Szpunar, J.; Lobinski, R.; Prange, A. Hyphenated techniques for elemental speciation in biological systems. Appl. Spectrosc 2003, 57, 102A–112A. [Google Scholar]
- Chassaigne, H.; Lobinski, R. Characterization of metallothionein isoforms by reversed-phase high-performance liquid chromatography with on-line post-column acidification and electrospray mass spectrometric detection. J. Chromatogr. A 1998, 829, 127–136. [Google Scholar]
- Chassaigne, H.; Lobinski, R. Characterization of horse kidney metallothionein isoforms by electrospray MS and reversed-phase HPLC-electrospray MS. Analyst 1998, 123, 2125–2130. [Google Scholar]
- Chassaigne, H.; Lobinski, R. Polymorphism and identification of metallothionein isoforms by reversed-phase HPLC with on-line ion spray mass spectrometric detection. Anal. Chem 1998, 70, 2536–2543. [Google Scholar]
- Kizek, R.; Trnkova, L.; Palecek, E. Determination of metallothionein at the femtomole level by constant current stripping chronopotentiometry. Anal. Chem 2001, 73, 4801–4807. [Google Scholar]
- Fabrik, I.; Krizkova, S.; Huska, D.; Adam, V.; Hubalek, J.; Trnkova, L.; Eckschlager, T.; Kukacka, J.; Prusa, R.; Kizek, R. Employment of electrochemical techniques for metallothionein determination in tumor cell lines and patients with a tumor disease. Electroanalysis 2008, 20, 1521–1532. [Google Scholar]
- Adam, V.; Blastik, O.; Krizkova, S.; Lubal, P.; Kukacka, J.; Prusa, R.; Kizek, R. Application of the Brdicka reaction in determination of metallothionein in patients with tumours. Chem. Listy 2008, 102, 51–58. [Google Scholar]
- Lim, D.; Koh, J.; Yip, G.; Tan, P.; Bay, B. Metallothionein as a potential molecular target in breast cancer. Breast 2007, 16, S16–S16. [Google Scholar]
- El Sharkarvy, S.L.; Farrag, A.R.H. Mean nuclear area and metallothionein expression in ductal breast tumors: Correlation with estrogen receptor status. Appl. Immunohistochem 2008, 16, 108–112. [Google Scholar]
- Liu, Z.M.; Chen, G.G.; Shum, C.K.Y.; Vlantis, A.C.; Cherlan, M.G.; Koropatnick, J.; van Hasselta, C.A. Induction of functional MT1 and MT2 isoforms by calcium in anaplastic thyroid carcinoma cells. FEBS Lett 2007, 581, 2465–2472. [Google Scholar]
- Ferrario, C.; Lavagni, P.; Gariboldi, M.; Miranda, C.; Losa, M.; Cleris, L.; Formelli, F.; Pilotti, S.; Pierotti, M.A.; Greco, A. Metallothionein 1G acts as an oncosupressor in papillary thyroid carcinoma. Lab. Invest 2008, 88, 474–481. [Google Scholar]
- Albrecht, A.L.; Singh, R.K.; Somji, S.; Sens, M.A.; Sens, D.A.; Garrett, S.H. Basal and metal-induced expression of metallothionein isoform 1 and 2 genes in the RWPE-1 human prostate epithelial cell line. J. Appl. Toxicol 2008, 28, 283–293. [Google Scholar]
- Yamasaki, M.; Nomura, T.; Sato, F.; Mimata, H. Metallothionein is up-regulated under hypoxia and promotes the survival of human prostate cancer cells. Oncol. Rep 2007, 18, 1145–1153. [Google Scholar]
- Athanassiadou, P.; Bantis, A.; Gonidi, M.; Athanassiades, P.; Agelonidou, E.; Grapsa, D.; Nikolopoulou, P.; Patsouris, E. The expression of metallothioneins on imprint smears of prostate carcinoma: Correlation with clinicopathologic parameters and tumor proliferative capacity. Tumori 2007, 93, 189–194. [Google Scholar]
- Lee, S.S.; Yang, S.F.; Ho, Y.C.; Tsai, C.H.; Chang, Y.C. The upregulation of metallothionein-1 expression in areca quid chewing-associated oral squamous cell carcinomas. Oral Oncol 2008, 44, 180–186. [Google Scholar]
- Liu, Y.B.; Li, L.N.; Gao, Y.X.; Chen, C.Y.; Li, B.; He, W.; Huang, Y.Y.; Chai, Z.F. Distribution of metalloproteins in hepatocellular carcinoma and surrounding tissues. Hepato-Gastroenterol 2007, 54, 2291–2296. [Google Scholar]
- Tao, X.; Zheng, J.M.; Xu, A.M.; Chen, X.F.; Zhang, S.H. Downregulated expression of metallothionein and its clinicopathological significance in hepatocellular carcinoma. Hepatol. Res 2007, 37, 820–827. [Google Scholar]
- Wang, D.; Lippard, S.J. Cellular processing of platinum anticancer drugs. Nat. Rev. Drug Discov 2005, 4, 307–320. [Google Scholar]
- Chaney, S.G.; Campbell, S.L.; Temple, B.; Bassett, E.; Wu, Y.B.; Faldu, M. Protein interactions with platinum-DNA adducts: from structure to function. J. Inorg. Biochem 2004, 98, 1551–1559. [Google Scholar]
- Krizkova, S.; Adam, V.; Petrlova, J.; Zitka, O.; Stejskal, K.; Zehnalek, J.; Sures, B.; Trnkova, L.; Beklova, M.; Kizek, R. A suggestion of electrochemical biosensor for study of platinum(II)-DNA interactions. Electroanalysis 2007, 19, 331–338. [Google Scholar]
- Petrlova, J.; Potesil, D.; Zehnalek, J.; Sures, B.; Adam, V.; Trnkova, L.; Kizek, R. Cisplatin electrochemical biosensor. Electrochim. Acta 2006, 51, 5169–5173. [Google Scholar]
- Krizkova, S.; Zitka, O.; Adam, V.; Beklova, M.; Horna, A.; Svobodova, Z.; Sures, B.; Trnkova, L.; Zeman, L.; Kizek, R. Possibilities of electrochemical techniques in metallothionein and lead detection in fish tissues. Czech J. Anim. Sci 2007, 52, 143–148. [Google Scholar]
- Kelland, L. The resurgence of platinum-based cancer chemotherapy. Nat. Rev. Cancer 2007, 7, 573–584. [Google Scholar]
- Timerbaev, A.R.; Hartinger, C.G.; Aleksenko, S.S.; Keppler, B.K. Interactions of antitumor metallodrugs with serum proteins: Advances in characterization using modern analytical methodology. Chem. Rev 2006, 106, 2224–2248. [Google Scholar]
- Baik, M.H.; Friesner, R.A.; Lippard, S.J. Theoretical study of cisplatin binding to purine bases: Why does cisplatin prefer guanine over adenine? J. Am. Chem. Soc 2003, 125, 14082–14092. [Google Scholar]
- Burda, J.V.; Zeizinger, M.; Leszczynski, J. Hydration process as an activation of trans- and cisplatin complexes in anticancer treatment. DFT and ab initio computational study of thermodynamic and kinetic parameters. J. Comput. Chem 2005, 26, 907–914. [Google Scholar]
- Zimmermann, T.; Zeizinger, M.; Burda, J.V. Cisplatin interaction with cysteine and methionine, a theoretical DFT study. J. Inorg. Biochem 2005, 99, 2184–2196. [Google Scholar]
- Adam, V.; Baloun, J.; Fabrik, I.; Trnkova, L.; Kizek, R. An electrochemical detection of metallothioneins at the zeptomole level in nanolitre volumes. Sensors 2008, 8, 2293–2305. [Google Scholar]
- Petrlova, J.; Krizkova, S.; Zitka, O.; Hubalek, J.; Prusa, R.; Adam, V.; Wang, J.; Beklova, M.; Sures, B.; Kizek, R. Utilizing a chronopotentiometric sensor technique for metallothionein determination in fish tissues and their host parasites. Sens. Actuat. B-Chem 2007, 127, 112–119. [Google Scholar]
- Adam, V.; Beklova, M.; Pikula, J.; Hubalek, J.; Trnkova, L.; Kizek, R. Shapes of differential pulse voltammograms and level of metallothionein at different animal species. Sensors 2007, 7, 2419–2429. [Google Scholar]
- Adam, V.; Hanustiak, P.; Krizkova, S.; Beklova, M.; Zehnalek, J.; Trnkova, L.; Horna, A.; Sures, B.; Kizek, R. Palladium biosensor. Electroanalysis 2007, 19, 1909–1914. [Google Scholar]
- Huska, D.; Zitka, O.; Adam, V.; Beklova, M.; Krizkova, S.; Zeman, L.; Horna, A.; Havel, L.; Zehnalek, J.; Kizek, R. A sensor for investigating the interaction between biologically important heavy metals and glutathione. Czech J. Anim. Sci 2007, 52, 37–43. [Google Scholar]
- Adam, V.; Krizkova, S.; Zitka, O.; Trnkova, L.; Petrlova, J.; Beklova, M.; Kizek, R. Determination of apo-metallothionein using adsorptive transfer stripping technique in connection with differential pulse voltammetry. Electroanalysis 2007, 19, 339–347. [Google Scholar]
- Petrlova, J.; Potesil, D.; Mikelova, R.; Blastik, O.; Adam, V.; Trnkova, L.; Jelen, F.; Prusa, R.; Kukacka, J.; Kizek, R. Attomole voltammetric determination of metallothionein. Electrochim. Acta 2006, 51, 5112–5119. [Google Scholar]
- Adam, V.; Petrlova, J.; Potesil, D.; Zehnalek, J.; Sures, B.; Trnkova, L.; Jelen, F.; Kizek, R. Study of metallothionein modified electrode surface behavior in the presence of heavy metal ions-biosensor. Electroanalysis 2005, 17, 1649–1657. [Google Scholar]
- Grieshaber, D.; MacKenzie, R.; Voros, J.; Reimhult, E. Electrochemical biosensors - Sensor principles and architectures. Sensors 2008, 8, 1400–1458. [Google Scholar]
- Lucarelli, F.; Tombelli, S.; Minunni, M.; Marrazza, G.; Mascini, M. Electrochemical and piezoelectric DNA biosensors for hybridisation detection. Anal. Chim. Acta 2008, 609, 139–159. [Google Scholar]
- Sassolas, A.; Leca-Bouvier, B.D.; Blum, L.J. DNA biosensors and microarrays. Chem. Rev 2008, 108, 109–139. [Google Scholar]
- Cooper, M.A.; Singleton, V.T. A survey of the 2001 to 2005 quartz crystal microbalance biosensor literature: applications of acoustic physics to the analysis of biomolecular interactions. J. Mol. Recognit 2007, 20, 154–184. [Google Scholar]
- Olafson, R.W.; Sim, R.G. Electrochemical Approach to Quantitation and Characterization of Metallothioneins. Anal. Biochem 1979, 100, 343–351. [Google Scholar]
- Raspor, B. Elucidation of the mechanism of the Brdicka reaction. J. Electroanal. Chem 2001, 503, 159–162. [Google Scholar]
- Raspor, B.; Paic, M.; Erk, M. Analysis of metallothioneins by the modified Brdicka procedure. Talanta 2001, 55, 109–115. [Google Scholar]
- El Hourch, M.; Dudoit, A.; Amiard, J.C. Optimization of new voltammetric method for the determination of metallothionein. Electrochim. Acta 2003, 48, 4083–4088. [Google Scholar]
- El Hourch, M.; Dudoit, A.; Amiard, J.C. An optimization procedure for determination of metallothionein by square wave cathodic stripping voltammetry: application to marine worms. Anal. Bioanal. Chem 2004, 378, 776–781. [Google Scholar]
- Knipp, M.; Karotki, A.V.; Chesnov, S.; Natile, G.; Sadler, P.J.; Brabec, V.; Vasak, M. Reaction of Zn(7)Metallothionein with cis- and trans-[Pt(N-donor)(2)Cl-2] anticancer complexes: trans-Pt-II complexes retain their N-donor ligands. J. Med. Chem 2007, 50, 4075–4086. [Google Scholar]
- Romero-Isart, N.; Vasak, M. Advances in the structure and chemistry of metallothioneins. J. Inorg. Biochem 2002, 88, 388–396. [Google Scholar]
- Zhang, B.L.; Huang, H.; Tang, W.X. Interaction of Cis-Diamminedichloroplatinum and Trans-Diamminedichloroplatinum with Metallothionein in-Vivo. J. Inorg. Biochem 1995, 58, 1–8. [Google Scholar]
- Kizek, R.; Vacek, J.; Trnkova, L.; Jelen, F. Cyclic voltammetric study of the redox system of glutathione using the disulfide bond reductant tris(2-carboxyethyl)phosphine. Bioelectrochemistry 2004, 63, 19–24. [Google Scholar]
- Murphy, S.; Boyle, F.M.; Davey, R.A.; Gu, X.Q.; Mather, L.E. Enantioselectivity of thalidomide serum and tissue concentrations in a rat glioma model and effects of combination treatment with cisplatin and BCNU. J. Pharm. Pharmacol 2007, 59, 105–114. [Google Scholar]
- Krizkova, S.; Fabrik, I.; Adam, V.; Kukacka, J.; Prusa, R.; Trnkova, L.; Strnadel, J.; Horak, V.; Kizek, R. Effects of reduced glutathione, surface active agents and ionic strength on detection of metallothioneins by using of Brdicka reaction. Electroanalysis 2009, 21, 640–644. [Google Scholar]
© 2009 by the authors; licensee MDPI, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huska, D.; Fabrik, I.; Baloun, J.; Adam, V.; Masarik, M.; Hubalek, J.; Vasku, A.; Trnkova, L.; Horna, A.; Zeman, L.; et al. Study of Interactions between Metallothionein and Cisplatin by using Differential Pulse Voltammetry Brdicka´s reaction and Quartz Crystal Microbalance. Sensors 2009, 9, 1355-1369. https://doi.org/10.3390/s90301355
Huska D, Fabrik I, Baloun J, Adam V, Masarik M, Hubalek J, Vasku A, Trnkova L, Horna A, Zeman L, et al. Study of Interactions between Metallothionein and Cisplatin by using Differential Pulse Voltammetry Brdicka´s reaction and Quartz Crystal Microbalance. Sensors. 2009; 9(3):1355-1369. https://doi.org/10.3390/s90301355
Chicago/Turabian StyleHuska, Dalibor, Ivo Fabrik, Jiri Baloun, Vojtech Adam, Michal Masarik, Jaromir Hubalek, Anna Vasku, Libuse Trnkova, Ales Horna, Ladislav Zeman, and et al. 2009. "Study of Interactions between Metallothionein and Cisplatin by using Differential Pulse Voltammetry Brdicka´s reaction and Quartz Crystal Microbalance" Sensors 9, no. 3: 1355-1369. https://doi.org/10.3390/s90301355



