A Determination of Metallothionein in Larvae of Freshwater Midges (Chironomus riparius) Using Brdicka Reaction
Abstract
:1. Introduction
2. Material and Methods
2.1 Chemicals and instruments
2.2 Larvae of freshwater midges
2.3 Heavy metals analysis
2.4 Preparation of biological samples for electrochemical analysis
2.5 Stationary electrochemical analyser – Adsorptive transfer stripping differential pulse voltammetry Brdicka reaction – MT content
2.6 Stationary electrochemical analyser coupled with autosampler – Differential pulse voltammetry Brdicka reaction – Total content of thiols
2.7 Statistical analyses
3. Results and Discussion
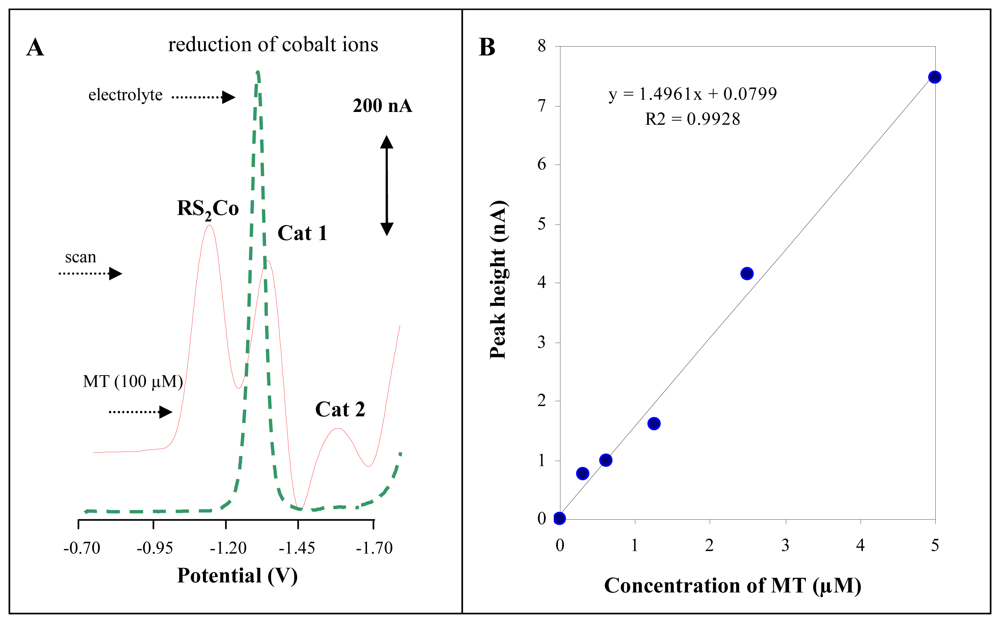
3.1 Stationary electrochemical analyser coupled with autosampler
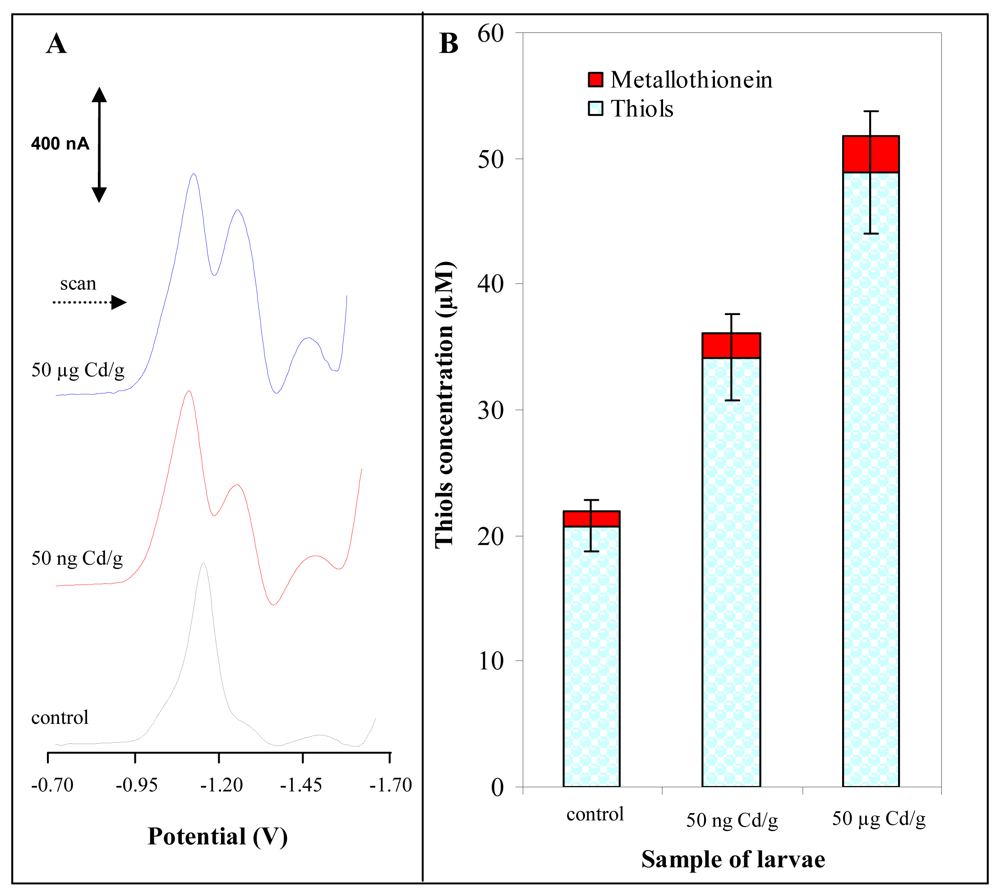
3.2 Determination of MT content in larvae of freshwater midges exposed to cadmium(II) ions
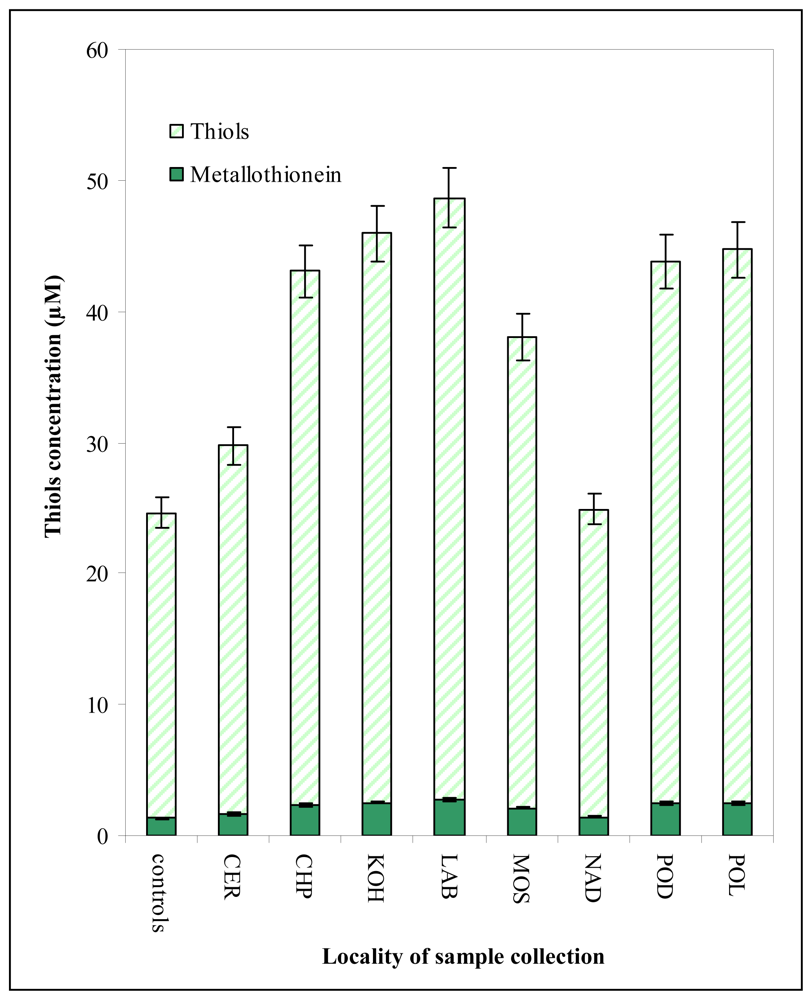
3.3 Determination of the content of heavy metals and thiols in chironomid larvae from the field study
4. Conclusion
Acknowledgments
References
- Houtman, C.J.; Booij, P.; van der Valk, K.M.; van Bodegom, P.M.; van den Ende, F.; Gerritsen, A.A.M.; Lamoree, M.H.; Legler, J.; Brouwer, A. Biomonitoring of estrogenic exposure and identification of responsible compounds in bream from Dutch surface waters. Environ. Toxicol. Chem. 2007, 26, 898–907. [Google Scholar]
- Dardenne, F.; Smolders, R.; De Coen, W.; Blust, R. Prokaryotic gene profiling assays to detect sediment toxicity: Evaluating the ecotoxicological relevance of a cell-based assay. Environ. Sci. Technol. 2007, 41, 1790–1796. [Google Scholar]
- Tripathi, A.K.; Gautam, M. Biochemical parameters of plants as indicators of air pollution. J.Environ.Biol. 2007, 28, 127–132. [Google Scholar]
- Ervin, G.N.; Herman, B.D.; Bried, J.T.; Holly, D.C. Evaluating non-native species and wetland indicator status as components of wetlands floristic assessment. Wetlands 2006, 26, 1114–1129. [Google Scholar]
- Misik, M.; Micieta, K.; Solenska, M.; Misikova, K.; Pisarcikova, H.; Knasmuller, S. In situ biomonitoring of the genotoxic effects of mixed industrial emissions using the Tradescantia micronucleus and pollen abortion tests with wild life plants: Demonstration of the efficacy of emission controls in an eastern European city. Environ. Pollut. 2007, 145, 459–466. [Google Scholar]
- Tomasevic, M.; Rajsic, S.; Dordevic, D.; Tasic, M.; Krstic, J.; Novakovic, V. Heavy metals accumulation in tree leaves from urban areas. Environ. Chem. Lett. 2004, 2, 151–154. [Google Scholar]
- Sasmaz, A.; Yaman, M. Distribution of chromium, nickel, and cobalt in different parts of plant species and soil in mining area of Keban, Turkey. Commun. Soil Sci. Plant Anal. 2006, 37, 1845–1857. [Google Scholar]
- Rusu, A.M.; Jones, G.C.; Chimonides, P.D.J.; Purvis, O.W. Biomonitoring using the lichen Hypogymnia physodes and bark samples near Zlatna, Romania immediately following closure of a copper ore-processing plant. Environ. Pollut. 2006, 143, 81–88. [Google Scholar]
- Huska, D.; Zitka, O.; Adam, V.; Beklova, M.; Krizkova, S.; Zeman, L.; Horna, A.; Havel, L.; Zehnalek, J.; Kizek, R. A sensor for investigating the interaction between biologically important heavy metals and glutathione. Czech J. Anim. Sci. 2007, 52, 37–43. [Google Scholar]
- Petrlova, J.; Potesil, D.; Mikelova, R.; Blastik, O.; Adam, V.; Trnkova, L.; Jelen, F.; Prusa, R.; Kukacka, J.; Kizek, R. Attomole voltammetric determination of metallothionein. Electrochim. Acta 2006, 51, 5112–5119. [Google Scholar]
- Kukacka, J.; Vajtr, D.; Huska, D.; Prusa, R.; Houstava, L.; Samal, F.; Diopan, V.; Kotaska, K.; Kizek, R. Blood metallothionein, neuron specific enolase, and protein S100B in patients with traumatic brain injury. Neuroendocrinol. Lett. 2006, 27, 116–120. [Google Scholar]
- Kizek, R.; Trnkova, L.; Palecek, E. Determination of metallothionein at the femtomole level by constant current stripping chronopotentiometry. Anal. Chem. 2001, 73, 4801–4807. [Google Scholar]
- Adam, V.; Krizkova, S.; Zitka, O.; Trnkova, L.; Petrlova, J.; Beklova, M.; Kizek, R. Determination of apo-metallothionein using adsorptive transfer stripping technique in connection with differential pulse voltammetry. Electroanalysis 2007, 19, 339–347. [Google Scholar]
- Studnickova, M.; Turanek, J.; Zabrsova, H.; Krejci, M.; Kysel, M. Rat liver metallothioneins are metal dithiolene clusters. J. Electroanal. Chem. 1997, 421, 25–32. [Google Scholar]
- Tsujikawa, K.; Imai, T.; Kakutani, M.; Kayamori, Y.; Mimura, T.; Otaki, N.; Kimura, M.; Fukuyama, R.; Shimizu, N. Localization of Metallothionein in Nuclei of Growing Primary Cultured Adult-Rat Hepatocytes. FEBS Lett. 1991, 283, 239–242. [Google Scholar]
- Prusa, R.; Svoboda, M.; Blastik, O.; Adam, V.; Zitka, O.; Beklova, M.; Eckschlager, T.; Kizek, R. Increase in content of metallothionein as marker of resistence to cisplatin treatment. Clin. Chem. 2006, 52, A174–A175. [Google Scholar]
- Petrlova, J.; Potesil, D.; Zehnalek, J.; Sures, B.; Adam, V.; Trnkova, L.; Kizek, R. Cisplatin electrochemical biosensor. Electrochim. Acta 2006, 51, 5169–5173. [Google Scholar]
- Strouhal, M.; Kizek, R.; Vecek, J.; Trnkova, L.; Nemec, M. Electrochemical study of heavy metals and metallothionein in yeast Yarrowia lipolytica. Bioelectrochemistry 2003, 60, 29–36. [Google Scholar]
- Adam, V.; Baloun, J.; Fabrik, I.; Trnkova, L.; Kizek, R. An electrochemical detection of metallothioneins in nanolitres at zeptomole level. Sensors 2008, 8, 2293–2305. [Google Scholar]
- Adam, V.; Beklova, M.; Pikula, J.; Hubalek, J.; Trnkova, L.; Kizek, R. Shapes of differential pulse voltammograms and level of metallothionein at different animal species. Sensors 2007, 7, 2419–2429. [Google Scholar]
- Adam, V.; Blastik, O.; Krizkova, S.; Lubal, P.; Kukacka, J.; Prusa, R.; Kizek, R. Application of the Brdicka reaction in determination of metallothionein in patients with tumours. Chem. Listy 2008, 102, 51–58. [Google Scholar]
- Adam, V.; Fabrik, I.; Nakielna, J.; Hrdinova, V.; Blahova, P.; Krizkova, S.; Kukacka, J.; Prusa, R.; Kizek, R. New perspectives in electrochemical determination of metallothioneins. Tumor Biol. 2007, 28, 79–79. [Google Scholar]
- Adam, V.; Krizkova, S.; Fabrik, I.; Zitka, O.; Horakova, Z.; Binkova, H.; Hrabeta, J.; Eckschlager, T.; Kukacka, J.; Prusa, R.; Sykorova, E.; Kizek, R. Metallothioneins as a new potential tumour marker. Tumor Biol. 2007, 28, 43–43. [Google Scholar]
- Adam, V.; Petrlova, J.; Potesil, D.; Zehnalek, J.; Sures, B.; Trnkova, L.; Jelen, F.; Kizek, R. Study of metallothionein modified electrode surface behavior in the presence of heavy metal ions-biosensor. Electroanalysis 2005, 17, 1649–1657. [Google Scholar]
- Diopan, V.; Baloun, J.; Adam, V.; Macek, T.; Havel, L.; Kizek, R. Determination of expression of metallothionein at transgenic tobacco plants. Listy Cukrov. Reparske 2007, 122, 325–327. [Google Scholar]
- Huska, D.; Krizkova, S.; Beklova, M.; Havel, L.; Zehnalek, J.; Diopan, V.; Adam, V.; Zeman, L.; Babula, P.; Kizek, R. Influence of cadmium(II) ions and brewery sludge on metallothionein level in earthworms (Eisenia fetida) – Bio-transforming of toxic wastes. Sensors 2008, 8, 1039–1047. [Google Scholar]
- Kizek, R.; Vacek, J.; Trnkova, L.; Klejdus, B.; Havel, L. Application of catalytic reactions on a mercury electrode for electrochemical detection of metallothioneins. Chem. Listy 2004, 98, 166–173. [Google Scholar]
- Krizkova, S.; Zitka, O.; Adam, V.; Beklova, M.; Horna, A.; Svobodova, Z.; Sures, B.; Trnkova, L.; Zeman, L.; Kizek, R. Possibilities of electrochemical techniques in metallothionein and lead detection in fish tissues. Czech J. Anim. Sci. 2007, 52, 143–148. [Google Scholar]
- Kukacka, J.; Petrlova, J.; Prusa, R.; Adam, V.; Sures, B.; Beklova, M.; Havel, L.; Kizek, R. Changes of content of glutathione and metallothionein at plant cells and invertebrate treated by platinum group metals. Faseb J. 2006, 20, A75–A75. [Google Scholar]
- Petrlova, J.; Krizkova, S.; Zitka, O.; Hubalek, J.; Prusa, R.; Adam, V.; Wang, J.; Beklova, M.; Sures, B.; Kizek, R. Utilizing a chronopotentiometric sensor technique for metallothionein determination in fish tissues and their host parasites. Sens. Actuator B-Chem. 2007, 127, 112–119. [Google Scholar]
- Prusa, R.; Blastik, O.; Potesil, D.; Trnkova, L.; Zehnalek, J.; Adam, V.; Petrlova, J.; Jelen, F.; Kizek, R. Analytic method for determination of metallothioneins as tumor markers. Clin. Chem. 2005, 51, A56–A56. [Google Scholar]
- Prusa, R.; Kizek, R.; Vacek, J.; Trnkova, L.; Zehnalek, J. Study of relationship between metallothionein and heavy metals by CPSA method. Clin. Chem. 2004, 50, A28–A29. [Google Scholar]
- Trnkova, L.; Kizek, R.; Vacek, J. Catalytic signal of rabbit liver metallothionein on a mercury electrode: a combination of derivative chronopotentiometry with adsorptive transfer stripping. Bioelectrochemistry 2002, 56, 57–61. [Google Scholar]
- Vajtr, D.; Kukacka, J.; Adam, V.; Kotaska, K.; Houstava, L.; Toupalik, P.; Kizek, R.; Prusa, R. Serum levels of metallothionein in expansive contusions, interleukine-6 and TNF-α during reparative phase of the blood brain barrier damage. Tumor Biol. 2007, 28, 44–44. [Google Scholar]
- Szpunar, J. Bio-inorganic speciation analysis by hyphenated techniques. Analyst 2000, 125, 963–988. [Google Scholar]
- Lobinski, R.; Chassaigne, H.; Szpunar, J. Analysis for metallothioneins using coupled techniques. Talanta 1998, 46, 271–289. [Google Scholar]
- Dabrio, M.; Rodriguez, A.R.; Bordin, G.; Bebianno, M.J.; De Ley, M.; Sestakova, I.; Vasak, M.; Nordberg, M. Recent developments in quantification methods for metallothionein. J. Inorg. Biochem. 2002, 88, 123–134. [Google Scholar]
- Vodickova, H.; Pacakova, V.; Sestakova, I.; Mader, P. Analytical methods for determination of metallothioneins. Chem. Listy 2001, 95, 477–483. [Google Scholar]
- Sestakova, I.; Vodickova, H.; Mader, P. Voltammetric methods for speciation of plant metallothioneins. Electroanalysis 1998, 10, 764–770. [Google Scholar]
- Werner, J.; Palace, V.; Baron, C.; Shiu, R.; Yarmill, A. A real-time PCR method for the quantification of the two isoforms of metallothionein in lake trout (Salvelinus namaycush). Arch. Environ. Contam. Toxicol. 2008, 54, 84–91. [Google Scholar]
- Ndayibagira, A.; Sunahara, G.I.; Robidoux, P.Y. Rapid isocratic HPLC quantification of metallothionein-like proteins as biomarkers for cadmium exposure in the earthworm Eisenia andrei. Soil Biol. Biochem. 2007, 39, 194–201. [Google Scholar]
- Brdicka, R. Polarographic Studies with the Dropping Mercury Kathode. -Part XXXII. - Activation of Hydrogen in Sulphydryl Group of Some Thio-Acids in Cobalt Salts Solutions. Coll. Czech. Chem. Commun. 1933, 5, 148–164. [Google Scholar]
- Brdicka, R. Polarographic Studies with the Dropping Mercury Kathode. -Part XXXI. - A New Test for Proteins in The presence of Cobalt Salts in Ammoniacal Solutions of Ammonium Chloride. Coll. Czech. Chem. Commun. 1933, 5, 112–128. [Google Scholar]
- Brdicka, R. Polarographic studies with the dropping mercury kathode.- Part XXXIII.- The microdetermination of cysteine and cystine in the hydrolysates of proteins, and the course of the protein decomposition. Coll. Czech. Chem. Commun. 1933, 5, 238–252. [Google Scholar]
- Dragun, Z.; Erk, M.; Raspor, B.; Ivankovic, D.; Pavicic, J. Metal and metallothionein level in the heat-treated cytosol of gills of transplanted mussels Mytilus galloprovincialis Lmk. Environ. Int. 2004, 30, 1019–1025. [Google Scholar]
- Raspor, B.; Dragun, Z.; Erk, M.; Ivankovic, D.; Pavicic, J. Is the digestive gland of Mytilus galloprovincialis a tissue of choice for estimating cadmium exposure by means of metallothioneins? Sci. Total Environ. 2004, 333, 99–108. [Google Scholar]
- Marijic, V.F.; Raspor, B. Metal exposure assessment in native fish, Mullus barbatus L., from the Eastern Adriatic sea. Toxicol. Lett. 2007, 168, 292–301. [Google Scholar]
- Marijic, V.F.; Raspor, B. Metal exposure assessment in native fish, Mullus barbatus, from the Eastern Adriatic Sea. Toxicol. Lett. 2006, 164, S156–S156. [Google Scholar]
- Marijic, V.F.; Raspor, B. Age- and tissue-dependent metallothionein and cytosolic metal distribution in a native Mediterranean fish, Mullus barbatus, from the Eastern Adriatic Sea. Comp. Biochem. Physiol. C-Toxicol. Pharmacol. 2006, 143, 382–387. [Google Scholar]
- Dragun, Z.; Raspor, B.; Erk, M.; Ivankovic, D.; Pavicic, J. The influence of the biometric parameters on metallothionein and metal level in the heat-treated cytosol of the whole soft tissue of transplanted mussels. Environ. Monit. Assess. 2006, 114, 49–64. [Google Scholar]
- Raspor, B.; Paic, M.; Erk, M. Analysis of metallothioneins by the modified Brdicka procedure. Talanta 2001, 55, 109–115. [Google Scholar]
- Kizek, R.; Vacek, J.; Trnkova, L.; Jelen, F. Cyclic voltammetric study of the redox system of glutathione using the disulfide bond reductant tris(2-carboxyethyl)phosphine. Bioelectrochemistry 2004, 63, 19–24. [Google Scholar]
- Erk, M.; Ivanković, D.; Raspor, B.; Pavičić, J. Evaluation of different purification procedures for the electrochemical quantification of mussel metallothioneins. Talanta 2002, 57, 1211–1218. [Google Scholar]
- Lang, C.; Langdobler, B. Melimex, an Experimental Heavy-Metal Pollution Study - Oligochaetes and Chironomid Larvae in Heavy-Metal Loaded and Control Limno-Corrals. Schweizerische Zeitschrift Fur Hydrologie-Swiss Journal of Hydrology 1979, 41, 271–276. [Google Scholar]
- Bervoets, L.; Solis, D.; Romero, A.M.; Van Damme, P.A.; Ollevier, F. Trace metal levels in chironomid larvae and sediments from a Bolivian river: Impact of mining activities. Ecotox. Environ. Safe. 1998, 41, 275–283. [Google Scholar]
- Pourang, N. Heavy metal concentrations in surficial sediments and benthic macroinvertebrates from Anzali wetland, Iran. Hydrobiologia 1996, 331, 53–61. [Google Scholar]
- Gillis, P.L.; Reynoldson, T.B.; Dixon, D.G. Metallothionein-like protein and tissue metal concentrations in invertebrates (Oligochaetes and Chironomids) collected from reference and metal contaminated field sediments. J. Gt. Lakes Res. 2006, 32, 565–577. [Google Scholar]
- Bhattacharyay, G.; Sadhu, A.K.; Mazumdar, A.; Chaudhuri, P.K. Antennal deformities of chironomid larvae and their use in biomonitoring of heavy metal pollutants in the River Damodar of West Bengal, India. Environ. Monit. Assess. 2005, 108, 67–84. [Google Scholar]
- Mousavi, S.K.; Primicerio, R.; Amundsen, P.A. Diversity and structure of Chironomidae (Diptera) communities along a gradient of heavy metal contamination in a subarctic watercourse. Sci. Total Environ. 2003, 307, 93–110. [Google Scholar]
- Long, G.L.; Winefordner, J.D. Limit of Detection. Anal. Chem. 1983, 55, A712–A724. [Google Scholar]
- Martinez, E.A.; Moore, B.C.; Schaumloffel, J.; Dasgupta, N. The potential association between mental deformities and trace elements in Chironomidae (diptera) taken from a heavy metal contaminated river. Arch. Environ. Contam. Toxicol. 2002, 42, 286–291. [Google Scholar]
- Krizkova, S.; Fabrik, I.; Adam, V.; Kukacka, J.; Prusa, R.; Trnkova, L.; Strnadel, J.; Horak, V.; Kizek, R. Effects of reduced glutathione, surface active agents and ionic strength on detection of metallothioneinS by using of brdicka reaction. Electroanalysis 2008. submitted. [Google Scholar]
- Krizkova, S.; Fabrik, I.; Adam, V.; Kukacka, J.; Prusa, R.; Chavis, G.J.; Trnkova, L.; Strnadel, J.; Horak, V.; Kizek, R. Utilizing of adsorptive transfer stripping technique Brdicka reaction for determination of metallothioneins level in melanoma cells, blood serum and tissues. Sensors 2008, 8, 3106–3122. [Google Scholar]
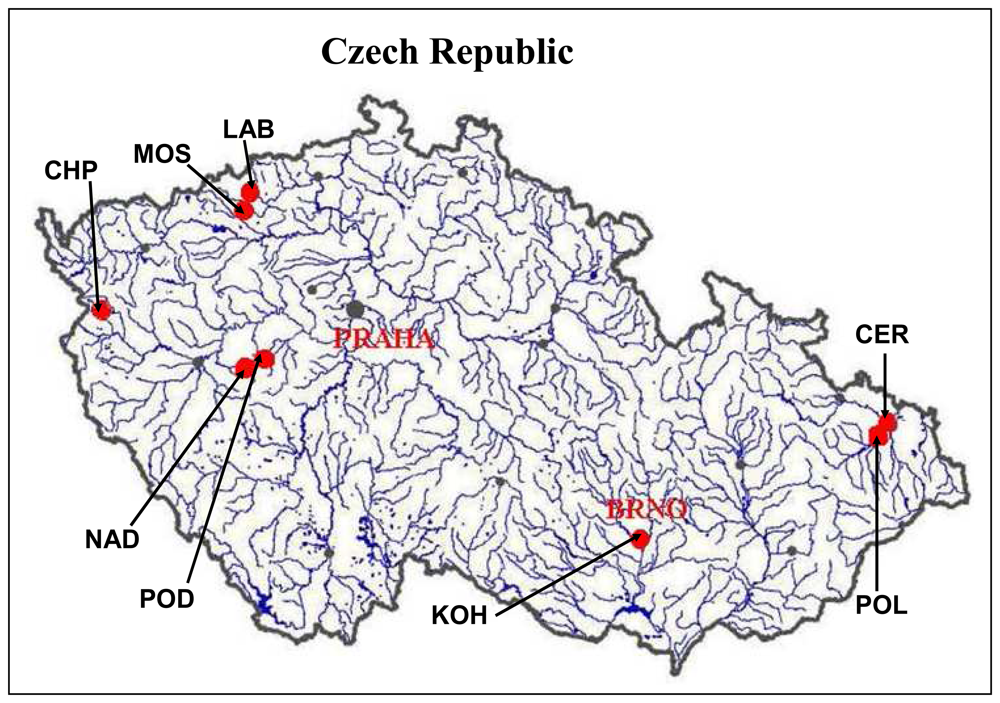

| Locality | Heavy metals (mg/kg)* | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Cr | Co | Ni | Cu | Zn | As | Mo | Cd | Sn | Pb | Sum | |
| MOS | 23.7 | 28.9 | 44.0 | 32.0 | 153 | 33.8 | 1.5 | 0.7 | 0.8 | 23.9 | 342.3 |
| LAB | 74.4 | 170* | 249* | 346* | 715* | 81.7* | 17.9 | 2.8* | 5.8* | 74.0 | 1736.6 |
| CER | 243* | 21.0 | 38.7 | 114 | 417 | 9.6 | 8.1 | 0.9 | 1.5 | 152 | 1005.8 |
| POL | 17.3 | 4.9 | 14.8 | 23.9 | 132 | 3.9 | 0.6 | 0.3 | 0.5 | 15.4 | 213.6 |
| NAD | 57.0 | 24.3 | 33.6 | 38.3 | 336 | 10.2 | 118 | 1.3 | 3.8 | 39.2 | 661.7 |
| POD | 51.1 | 20.3 | 41.6 | 46.9 | 264 | 19.2 | 133* | 2.5 | 3.4 | 41.6 | 623.6 |
| CHP | 66.6 | 10.0 | 39.9 | 43.1 | 567 | 7.6 | 3.3 | 0.6 | 1.0 | 22.7 | 761.8 |
| KOH | 121 | 23.3 | 41.7 | 93.1 | 667 | 5.9 | 4.0 | 0.6 | 2.2 | 86.9* | 1045.7 |
© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fabrik, I.; Ruferova, Z.; Hilscherova, K.; Adam, V.; Trnkova, L.; Kizek, R. A Determination of Metallothionein in Larvae of Freshwater Midges (Chironomus riparius) Using Brdicka Reaction. Sensors 2008, 8, 4081-4094. https://doi.org/10.3390/s8074081
Fabrik I, Ruferova Z, Hilscherova K, Adam V, Trnkova L, Kizek R. A Determination of Metallothionein in Larvae of Freshwater Midges (Chironomus riparius) Using Brdicka Reaction. Sensors. 2008; 8(7):4081-4094. https://doi.org/10.3390/s8074081
Chicago/Turabian StyleFabrik, Ivo, Zuzana Ruferova, Klara Hilscherova, Vojtech Adam, Libuse Trnkova, and Rene Kizek. 2008. "A Determination of Metallothionein in Larvae of Freshwater Midges (Chironomus riparius) Using Brdicka Reaction" Sensors 8, no. 7: 4081-4094. https://doi.org/10.3390/s8074081




