Determination of DPPH Radical Oxidation Caused by Methanolic Extracts of Some Microalgal Species by Linear Regression Analysis of Spectrophotometric Measurements
Abstract
:1. Introduction
2. Materials and methods
2.1. Microalgae and cultivation conditions
2.2 Preparation of the methanolic extract
2.3 Spectrophotometric measurements
2.4 α-Tocopherol
2.5 Preparation of reference and measuring cuvette
2.6 Experimental protocol
2.7 Data analysis
2.8 Determination of the effective concentration (EC50)
3. Results and discussion
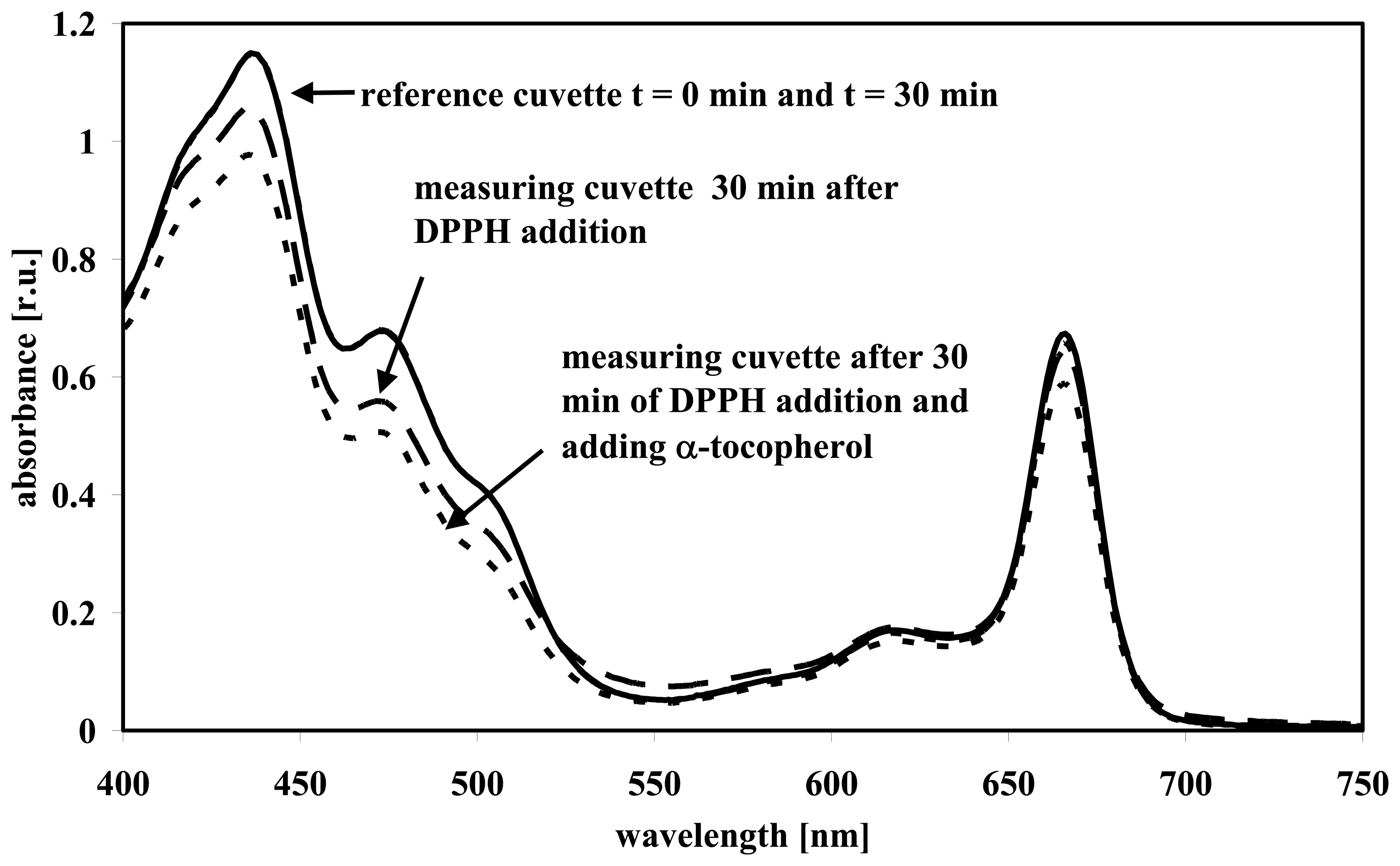
3.1 Wavelength scans
3.2 Absorbance measurements
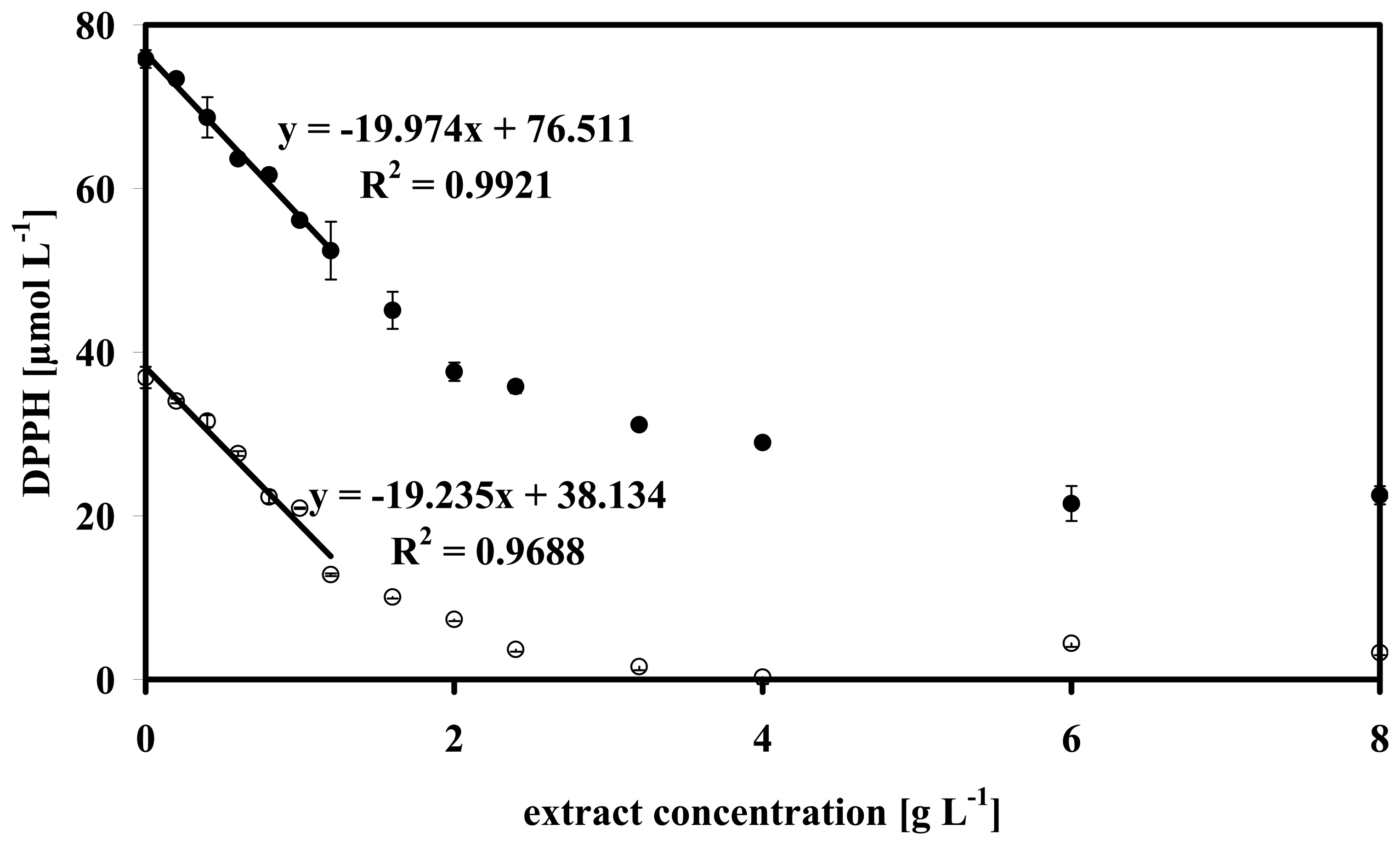
3.3 Theoretical background
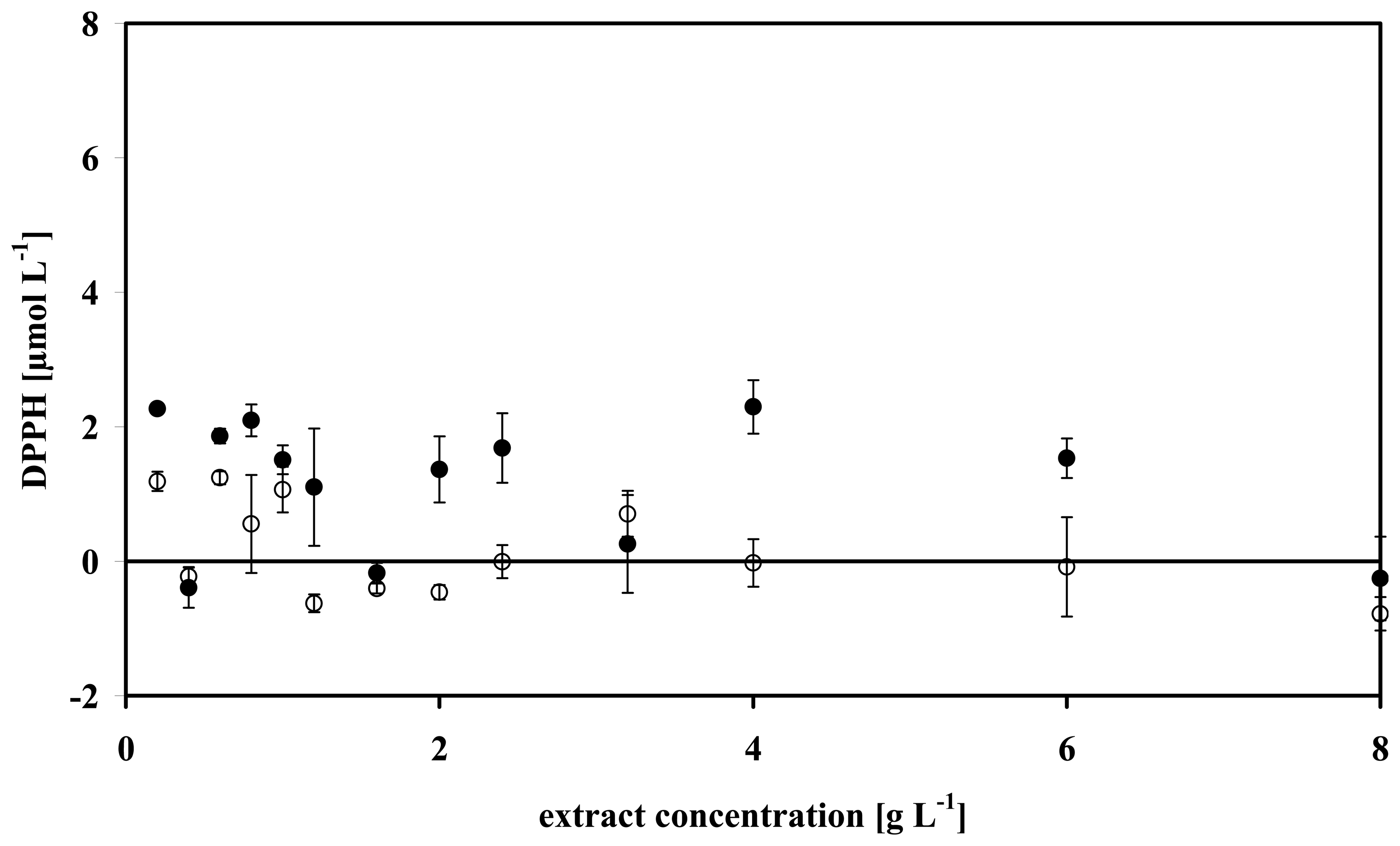
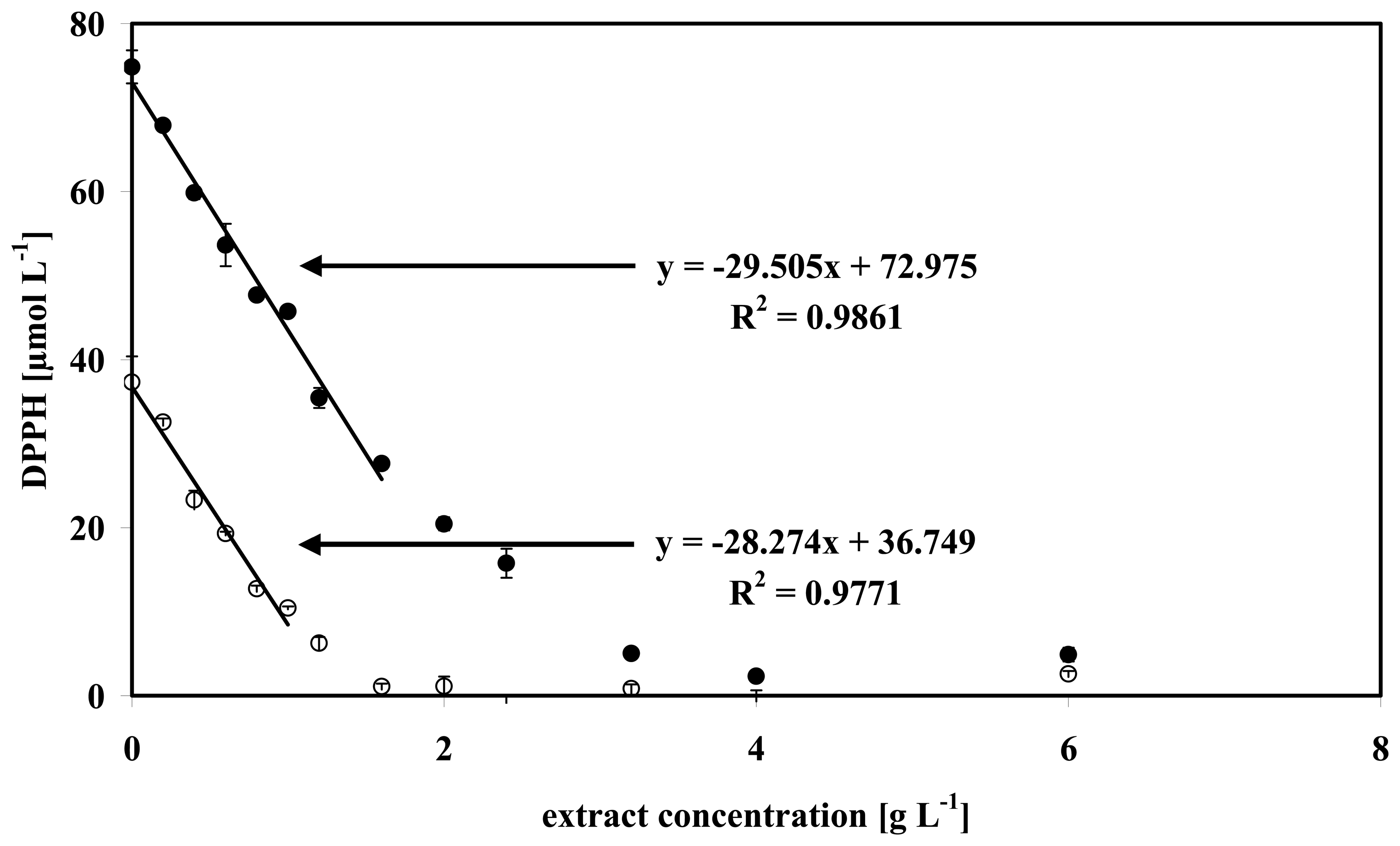
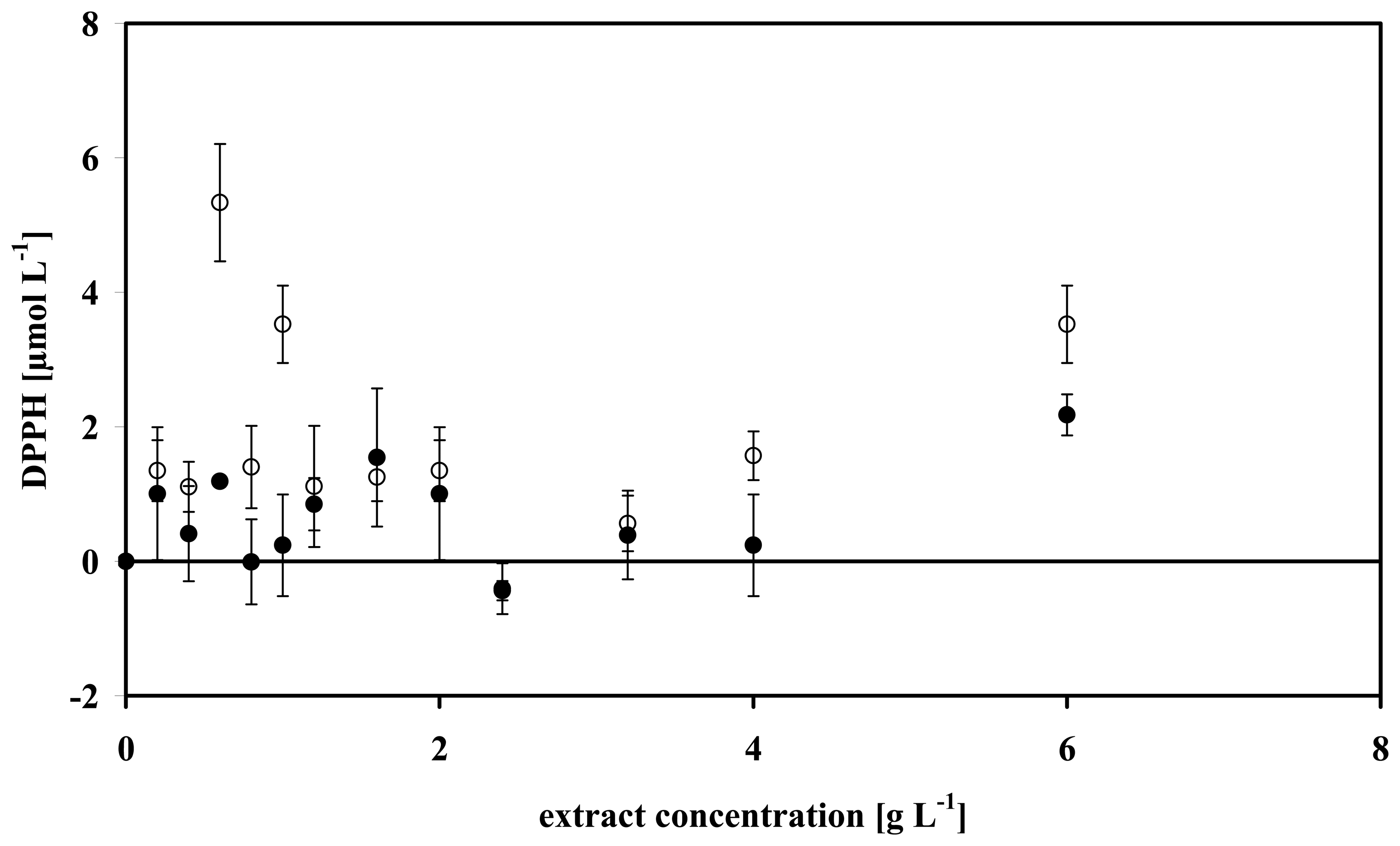
3.4 Data analysis of some microalgae
Acknowledgments
References and Notes
- Allakhverdiev, S.I.; Nishiyama, Y.; Suzuki, I.; Tasaka, Y.; Murata, N. Genetic engineering of the unsaturation of fatty acids in membrane lipids alters the tolerance of Synechocystis to salt stress. Proc. Natl. Acad. Sci. 1999, 96, 5862–5867. [Google Scholar]
- Anderson, J.A.; Padhye, S.R. Protein aggregation, radical scavenging capacity, and stability of hydrogen peroxide defense systems in heat-stressed vinca and sweet pea leaves. J. Am. Soc. Hortic Sci. 2004, 129, 54–59. [Google Scholar]
- Antolovic, M.; Prenzler, P.D.; Patsalides, E.; McDonald, S.; Robards, K. Methods for testing antioxidant activity. Analyst. 2002, 127, 183–198. [Google Scholar]
- Apel, K.; Hirt, H. Reactive oxygen species: metabolism, oxidative stress and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar]
- Arora, A.; Sairam, R.K.; Srivastave, G.C. Oxidative stress and antioxidative systems in plants. Curr. Sci. 2002, 82, 1227–1238. [Google Scholar]
- Bandoniene, D.; Murkovic, M.; Pfannhauser, W.; Venskutonis, P.R.; Gruzdiene, D. Detection and activity evaluation of radical scavenging compounds by using DPPH free radical and on-line HPLC-DPPH methods. Eur. Food Res. Technol. 2002, 214, 143–147. [Google Scholar]
- Bendich, A.; Olson, J.A. Biological actions of carotenoids. FASEB J. 1989, 3, 1927–1932. [Google Scholar]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann. Bot. 2003, 91, 179–194. [Google Scholar]
- Bondet, V.; Brand-Williams, W.; Berset, C. Kinetics and mechanisms of antioxidant activity using the DPPH free radical method. Lebensm. Wiss. Technol. 1997, 30, 609–615. [Google Scholar]
- Britton, G. Structure and properties of carotenoids in relation of function. FASEB J. 1995, 9, 1551–1558. [Google Scholar]
- Di Mascio, P.; Murphy, M.E.; Sies, H. Antioxidant defense systems: the role of carotenoids, tocopherols, and thiols. Am. J. Clin. Nutr. 1991, 53, 194–200. [Google Scholar]
- Edge, R.; McGarvey, D.J.; Truscott, T.G. The carotenoids as anti-oxidants – a review. J. Photoch. Photobio. B. 1997, 41, 189–200. [Google Scholar]
- Ehling-Schulz, M.; Scherer, S. UV protection in cyanobacteria. Eur. J. Phycol. 1999, 34, 329–338. [Google Scholar]
- Eonseon, J.; Polle, J.E.W.; Lee, H.K.; Hyun, S.M.; Chang, M. Xanthophylls in microalgae: From Biosynthesis to biotechnological mass production and application. J. Microbiol. Biotechnol. 2003, 13, 165–174. [Google Scholar]
- Fang, Y.Z.; Yang, S.; Wu, G. Free radicals, antioxidants, and nutrition. Nutrition 2002, 18, 872–879. [Google Scholar]
- Gazi, M.R.; Kanda, K.; Yasuda, M.; Kato, F. Optimisation of cultural conditions and some properties of radical scavenging substances from sporobolomyces salmonicolor. Pak. J. Biol. Sci. 2004, 7, 1365–1370. [Google Scholar]
- Guillard, R.R.L.; Ryther, J.H. Studies of marine planktonic diatoms I. Cyclotella nana Hustedt and Detonula confervacea Cleve. Can. J. Microbiol 1962, 8, 229–239. [Google Scholar]
- He, Y.Y.; Häder, D.P. Reactive oxygen species and UV-B: effect on Cyanobacteria. Photochem. Photobiol. Sci. 2002, 1, 729–736. [Google Scholar]
- Iwashima, M.; Mori, J.; Ting, X.; Matsunaga, T.; Hayashi, K.; Shinoda, D.; Saito, H.; Sankawa, U.; Hayashi, T. Antioxidant and antiviral activities of plastoquinones from the brown alga Sargassum micracanthum, and a new chromene derivative converted from the plastoquinones. Biol. Pharm. Bull. 2005, 28, 374–377. [Google Scholar]
- Jacob, R.A.; Burri, B.J. Oxidative damage and defense. Am. J. Clin. Nutr. 1996, 63, 985–990. [Google Scholar]
- Krinsky, N.I. The biological properties of carotenoids. Pure & Appl. Chem. 1994, 66, 1003–1010. [Google Scholar]
- Kobayashi, M.; Kakizono, T.; Nishio, N.; Nagai, S.; Kurimura, Y.; Tsuji, Y. Antioxidant role of astaxanthin in the green alga Haematococcus pluvialis. Appl. Microbiol. Biotechnol. 1997, 48, 351–356. [Google Scholar]
- Koleva, I.I.; van Beek, T.A.; Linssen, J.P.H.; de Groot, A.; Evstatieva, L.N. Screening of plant extracts for antioxidant activity: a comparative study on three testing methods. Phytochem. Analysis 2001, 13, 8–17. [Google Scholar]
- Kosakowska, A.; Lewandowska, J.; Ston, J.; Burkiewicz, K. Qualitative and quantitative composition of Phaedactylum tricornutum (Bacillariophyceae) stressed by iron. J. Biometals 2004, 17, 45–52. [Google Scholar]
- Kulisic, T.; Radonic, A.; Katalinic, V.; Milos, M. Use of different methods for testing antioxidative activity of oregano essential oil. Food Chem. 2004, 85, 633–640. [Google Scholar]
- Margalith, P.Z. Production of ketocarotenoids by microalgae. Appl. Micorbiolol. Biotechnol. 1999, 51, 431–438. [Google Scholar]
- Marxen, K.; Vanselow, K.H.; Lippemeier, S.; Hintze, R.; Ruser, A.; Hansen, U.P. A photobioreactor system for computer controlled cultivation of microalgae. J. Appl. Phycol. 2005, 17, 535–549. [Google Scholar]
- Masuda, T.; Yonemori, S.; Oyama, Y.; Takeda, Y.; Tanaka, T.; Andoh, T.; Shinohara, A.; Nakata, M. Evaluation of the antioxidant of environmental plants: activity of the leaf extracts from seashore plants. J. Agric. Food Chem. 1999, 47, 1749–1754. [Google Scholar]
- Miller, N.J.; Sampson, J.; Candeias, L.P.; Bramley, P.M.; Rice-Evans, C.A. Antioxidant activities of carotenes and xanthophylls. FEBS Letters 1996, 384, 240–242. [Google Scholar]
- Molyneux, P. The use of the stable free radical diphenylpicryl-hydrazyl (DPPH) for estimating antioxidant activity. Songklanakarin J. Sci. Technol. 2004, 26, 211–219. [Google Scholar]
- Naguib, Y.M.A. Antioxidant activities of astaxanthin and related carotenoids. J. Agri. Food Chem. 2000, 48, 1150–1154. [Google Scholar]
- Paiva, S.A.R.; Russell, R.M. β-carotene and other carotenoids as antioxidants. J. Am. Coll. Nutr. 1999, 18, 426–433. [Google Scholar]
- Pavlov, A.; Kovatcheva, P.; Georgiev, V.; Koleva, I.; Ilieva, M. Biosynthesis and radical scavenging activity of betalains during the cultivation of red beet (Beta vulgaris) hairy root cultures. Z. Naturforsch. 2002, 57c, 640–644. [Google Scholar]
- Pinchuk, I.; Lichtenberg, D. The mechanism of action of antioxidants against lipoprotein peroxidation, evaluation based on kinetic experiments. Prog. Lipid Res. 2002, 41, 279–314. [Google Scholar]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4303. [Google Scholar]
- Pulz, O.; Gross, W. Valuable products from biotechnology of microalgae. Appl. Microbiol. Biotechnol. 2004, 65, 635–648. [Google Scholar]
- Rippka, R.; Deruelles, J.; Waterbury, J.; Herdman, M.; Stanier, R. Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J. Gen. Microbiol. 1979, 111, 1–61. [Google Scholar]
- Rijstenbil, J.W. Assessment of oxidative stress in the planktonic diatom Thalassiosira pseudonana in response to UVA and UVB radiation. J. Plankton Res. 2002, 24, 1277–1288. [Google Scholar]
- Romay, C.; Armesto, J.; Remirez, D.; González, R.; Ledon, N.; García, I. Antioxidant and anti-inflammatory properties of C-phycocyanin from blue-green algae. Inflamm. Res. 1998, 47, 36–41. [Google Scholar]
- Sies, H.; Stahl, W. Vitamins E and C, β-carotene, and other carotenoids as antioxidants. Am. J. Clin. Nutr. 1995, 62 suppl, 1315–1321. [Google Scholar]
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial application of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar]
- Vaya, J.; Aviram, M. Nutritional antioxidants: mechanism of action, analyses of activities and medical applications. Curr. Med. Chem.–Imm., Endoc. & Metab. Agents 2001, 1, 99–117. [Google Scholar]
- Wada, H.; Murata, N. Temperature-induced changes in the fatty acid composition of the cyanobacterium, Synechocystis PCC6803. Plant Physiol. 1990, 92, 1062–1069. [Google Scholar]
- Yen, G.C.; Duh, P.D. Scavenging effect of methanolic extracts of peanut hulls on free-radical and active-oxygen species. J. Agric. Food Chem. 1994, 42, 629–632. [Google Scholar]
- Yongmanitchai, W.; Ward, O.P. Growth of and omega-3 fatty acid production by Phaeodactylum tricornutum under different culture conditions. J. Appl. Environm. Microb. 1991, 57, 419–425. [Google Scholar]
- Yordabov, N.D.; Christova, A.G. Quantitative spectrophotometric and EPR-determination of 1,1-diphenyl-2-picryl-hydrazyl (DPPH). Fresen. J. Anal. Chem. 1997, 358, 610–613. [Google Scholar]
| High DPPH concentration (76 μM | Low DPPH concentration (38 μM) | |||||
|---|---|---|---|---|---|---|
| Microalgae | Slope [μmol DPPH g−1extract] | R | EC50 [g extract] | Slope [μmol D] | R2 | EC50 [g extract] |
| Anabaena sp. | 34.45 | 0.994 | 1.103 | 31.74 | 0.998 | 0.599 |
| Isochrysis galbana | 29.51 | 0.986 | 1.288 | 28.27 | 0.977 | 0.672 |
| Synechocystis sp. PCC6803 | 19.97 | 0.992 | 1.902 | 19.24 | 0.969 | 0.988 |
| Phaeodactylum triconutum | 20.58 | 0.968 | 1.847 | 18.71 | 0.996 | 1.016 |
| Porphyridium purpureum | 7.26 | 0.978 | 5.234 | 5.97 | 0.992 | 3.182 |
| α Tocopherol | 3300.3 | 0.999 | 0.0115 | |||
© 2007 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Marxen, K.; Vanselow, K.H.; Lippemeier, S.; Hintze, R.; Ruser, A.; Hansen, U.-P. Determination of DPPH Radical Oxidation Caused by Methanolic Extracts of Some Microalgal Species by Linear Regression Analysis of Spectrophotometric Measurements. Sensors 2007, 7, 2080-2095. https://doi.org/10.3390/s7102080
Marxen K, Vanselow KH, Lippemeier S, Hintze R, Ruser A, Hansen U-P. Determination of DPPH Radical Oxidation Caused by Methanolic Extracts of Some Microalgal Species by Linear Regression Analysis of Spectrophotometric Measurements. Sensors. 2007; 7(10):2080-2095. https://doi.org/10.3390/s7102080
Chicago/Turabian StyleMarxen, Kai, Klaus Heinrich Vanselow, Sebastian Lippemeier, Ralf Hintze, Andreas Ruser, and Ulf-Peter Hansen. 2007. "Determination of DPPH Radical Oxidation Caused by Methanolic Extracts of Some Microalgal Species by Linear Regression Analysis of Spectrophotometric Measurements" Sensors 7, no. 10: 2080-2095. https://doi.org/10.3390/s7102080




