Flow-through Bulk Optode for Spectrophotometric Determination of Thiocyanate and Its Application to Water and Saliva Analysis
Abstract
:1. Introduction
2. Results and Discussion
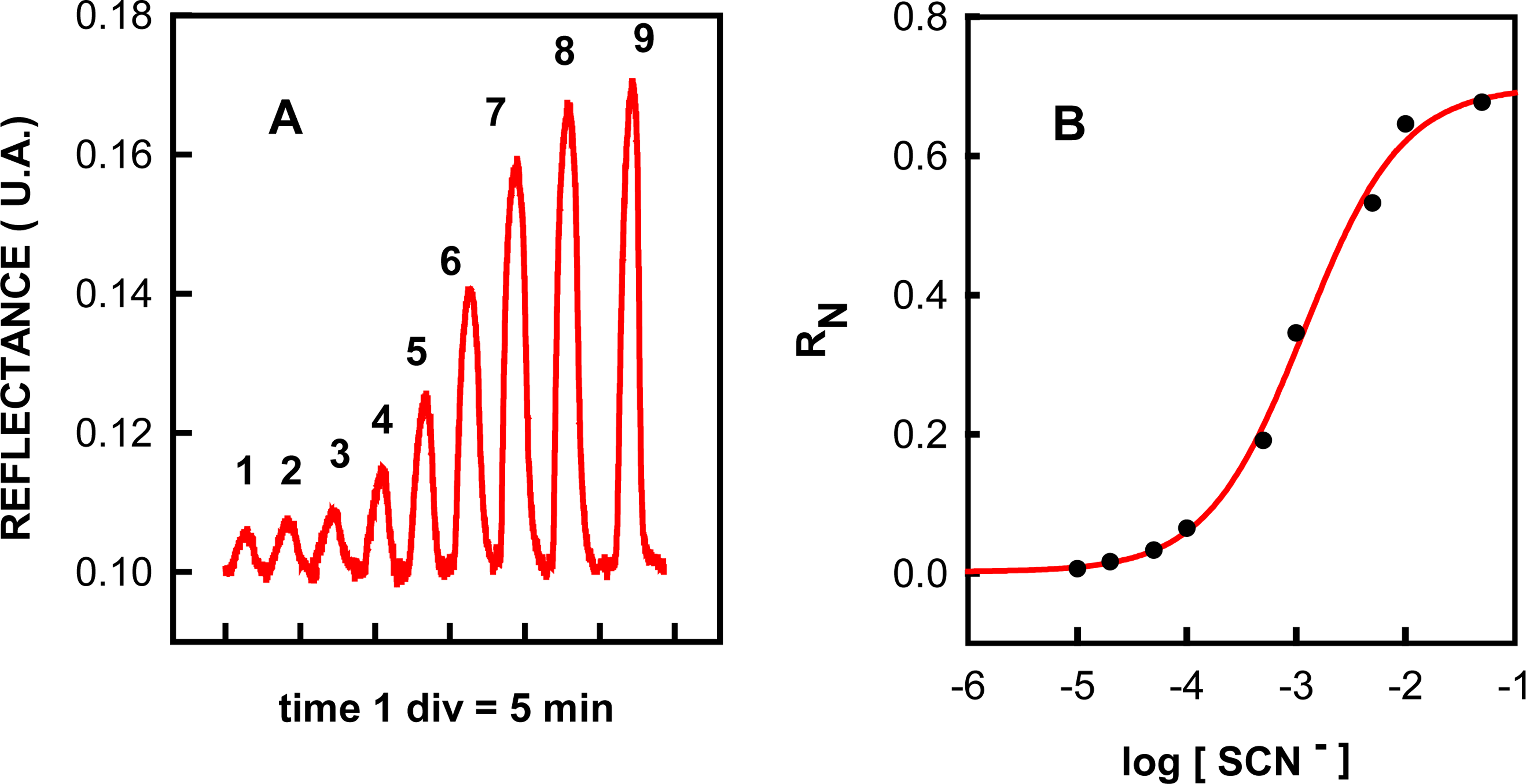
2.1 Principle of operation
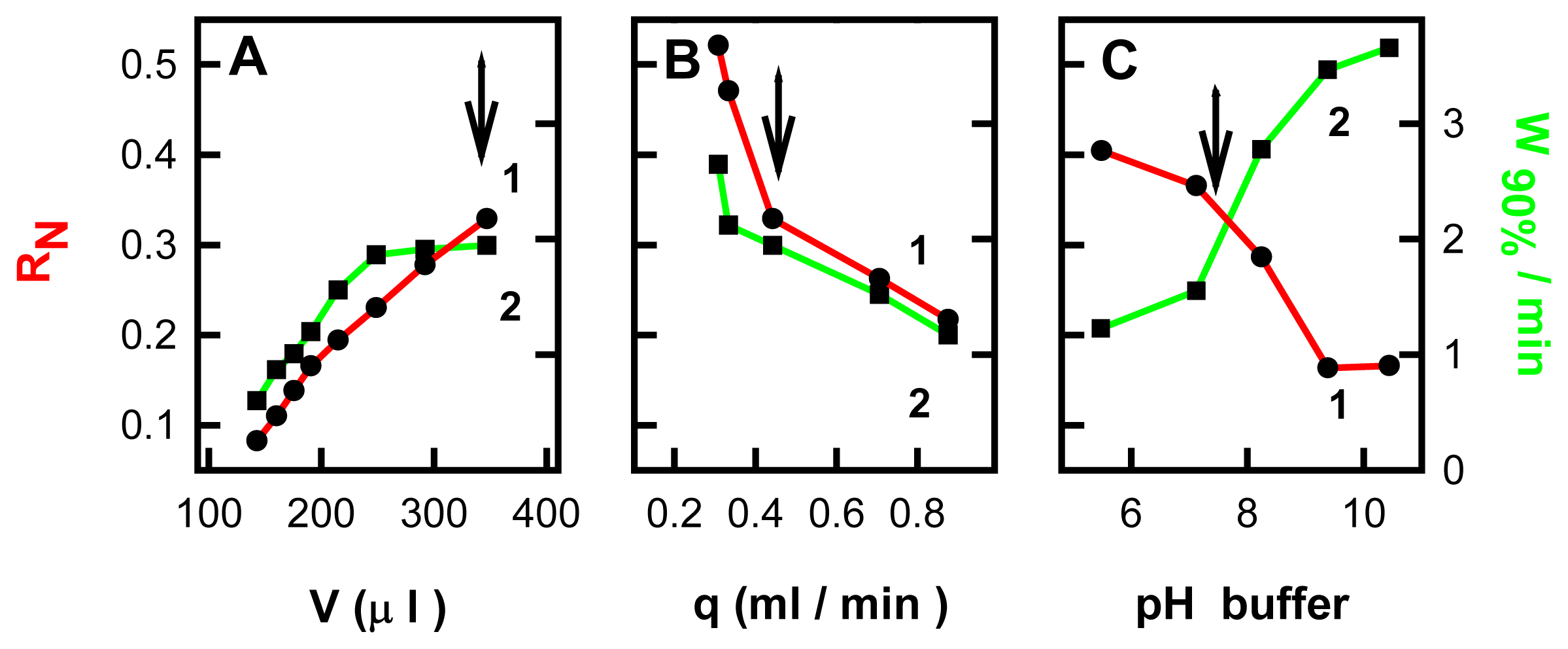
2.2 Study of variables
2.3 Features of the flow injection method
2.4 Selectivity
2.5 Applications
3. Experimental Section
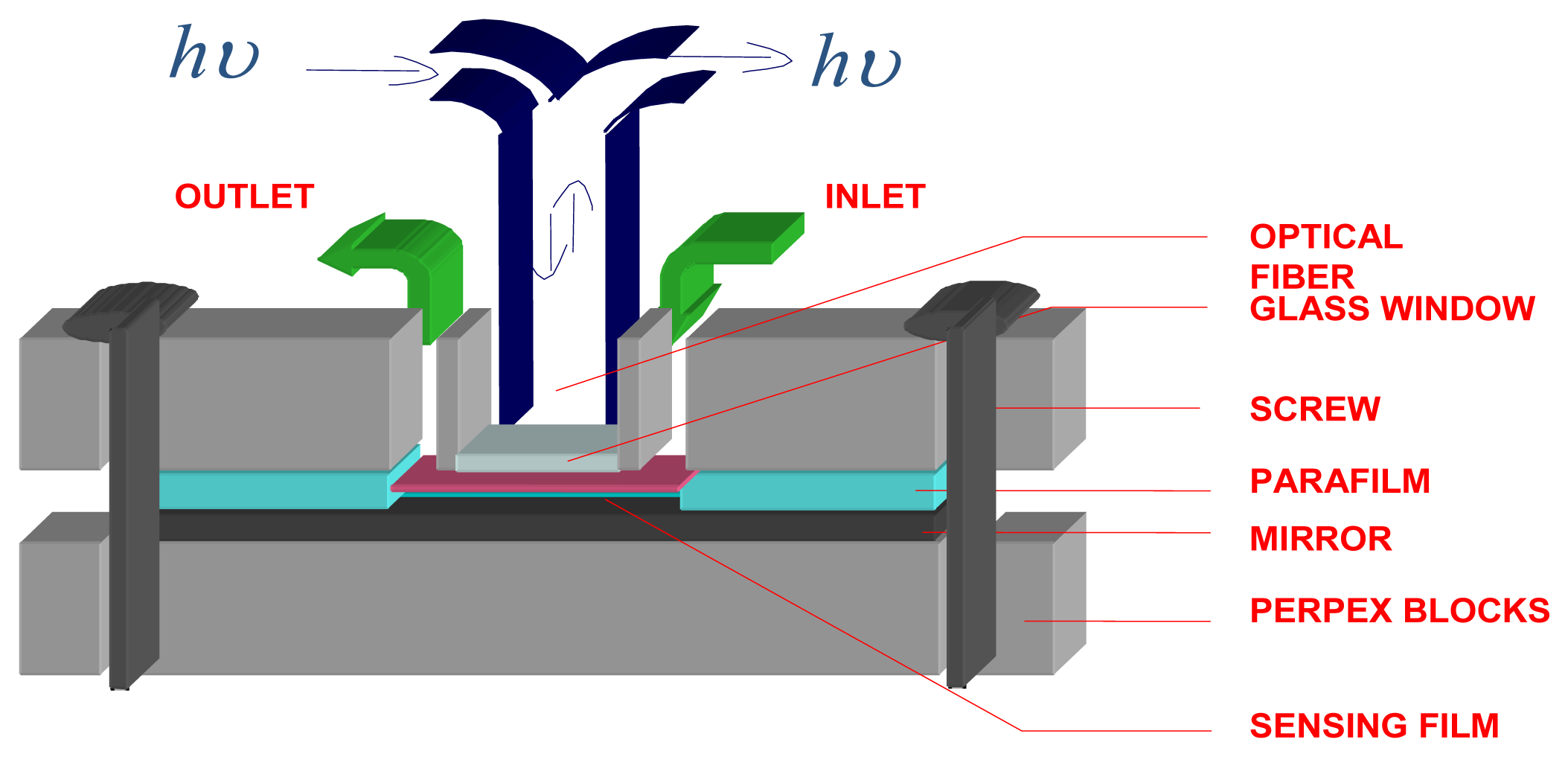
3.1 Apparatus
3.2 Materials and Reagents
3.3 Optode membrane preparation
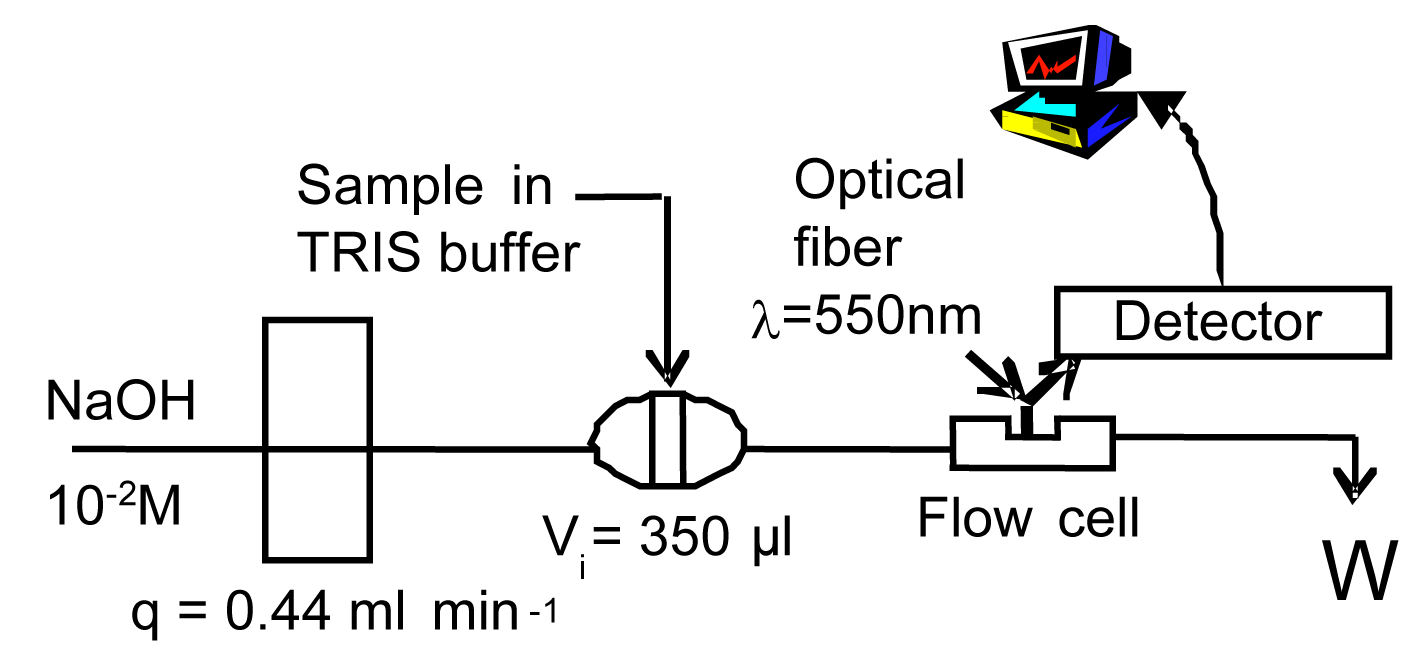
3.4 Manifold and Calibration Procedure
3.5 Procedure for determination of thiocyanate in water
3.6 Procedure for determination of thiocyanate in saliva
4. Conclusions
Acknowledgments
References and Notes
- Morf, W.E.; Seiler, K.; Ruterholz, B.; Simon, W. Desing of a calcium-selective optode membrane based on neutral ionophores. Anal. Chem. 1990, 62, 738–742. [Google Scholar]
- Valcarcel, M.; Luque de Castro, M.D. Flow-through (Bio)Chemical Sensors; Elsevier Science: Amsterdam, 1994. [Google Scholar]
- Janata, J.; Josowicz, M.; Vanysek, P.; DeVaney, D.M. Chemical Sensors. Anal.Chem. 1998, 70, 179R–208R. [Google Scholar]
- Wolfbeis, O.S. Fiber-Optic Chemical Sensors and Biosensors. Anal. Chem. 2000, 72, 81–90. [Google Scholar]
- Oehme, I.; Wolfbeis, O.S. Optical sensors for determination of heavy metal ions. Mikrochim. Acta 1997, 126, 177–192. [Google Scholar]
- Bakker, E.; Buhlmann, P.; Pretsch, E. Carrier-based ion-selective electrodes and bulk optodes. 1. General characteristics. Chem. Rev. 1997, 97, 3083–3132. [Google Scholar]
- Buhlmann, P.; Pretsch, E.; Bakker, E. Carrier-based ion-selective electrodes and bulk optodes. 2. Ionophores for potentiometric and optical sensors. Chem. Rev. 1998, 98, 1593–1687. [Google Scholar]
- Seitz, W.R. Fiber Optic Chemical Sensors and Biosensors; Wolfbeis, O.S., Ed.; CRC Press: Boca Raton, 1991; Volume 2, pp. 1–177. [Google Scholar]
- Seiler, K.; Simon, W. Theorical aspects of bulk optode membranes. Anal. Chim. Acta 1992, 266, 73–87. [Google Scholar]
- Hong, Y.K.; Cha, G.S.; Shin, D.S.; Nam, H. Studies of polymer matrix effect for coextraction type anion-selective optode and determination of thiocyanate in human saliva. Bull. Korean Chem. Soc. 1994, 15, 836–841. [Google Scholar]
- Zumbado, M.; Jimenez, M.; Schosinsky, N.; Perez, A. Determination of thiocyanate in saliva and urine as a marker for smoking. Acta Bioquim. Clin. Latinoam. 1998, 32, 39–48. [Google Scholar]
- Brocco, G.; Rossato, R.; Fraccaroli, M.; Lippi, G. An automated method based on plasma thiocyanate determination for the identification of exposure to tobacco smoke. Eur. J. Lab. Med. 1999, 7, 106–110. [Google Scholar]
- Torano, J.S.; van Kan, H. Simultaneous determination of tobacco smoke uptake parameters nicotine, cotinine and thiocyanate in urine, saliva and hair, using gas chromatography-mass spectrometry for characterization of smoking status of recently exposed subjects. Analyst 2003, 128, 838–843. [Google Scholar]
- Olea, M.R.F.; Ruiz, M.D.; Palomares, H.J. Determination of thiocyanate in vegetables by gas chromatography in relation to endemic goiter. J. Anal.Toxicol. 1988, 12, 307–309. [Google Scholar]
- Sweileh, J.A. Determination of cyanide and thiocyanate by a spectrophotometric flow-injection method. Anal. Chim. Acta 1989, 220, 65–74. [Google Scholar]
- Brown, P.D.; Morra, M. J. Ion chromatographic determination of thiocyanate ion in soil. J. Agric. Food Chem. 1991, 39, 1226–1228. [Google Scholar]
- Olea, F.; Pilar, P. Determination of serum levels of dietary thiocyanate. J.Anal. Toxicol. 1992, 16, 258–260. [Google Scholar]
- van Staden, J.F.; Botha, A. Spectrophotometric determination of thiocyanate by sequential injection análisis. Anal.Chim. Acta 2000, 403, 279–286. [Google Scholar]
- Gong, B.; Gong, G. Fluorimetric method for the determination of thiocyanate with 2′,7' –dichlorofluorescein and iodine. Anal. Chim. Acta 1999, 394, 171–175. [Google Scholar]
- Abbaspour, A.; Kamyabi, M.A.; Esmaeilbeig, A.R.; Kia, R. Thiocyanate-selective electrode based on unsymmetrical benzoN4 nickel(II) macrocyclic complexes. Talanta 2002, 57, 859–867. [Google Scholar]
- Ardakani, M.M.; Ensafi, A.A.; Niasari, M.S.; Chahooki, S.M. Selective thiocyanate poly(vinyl chloride) membrane based on a 1,8-dibenzyl-1,3,6,8,10,13-hexaazacyclotetradecane-Ni(II) perchlorate. Anal. Chim. Acta 2002, 462, 25–30. [Google Scholar]
- Yuan, R.; Wang, X.L.; Xu, L.; Chai, Y. Q.; Sun, Z. Y.; Huang, X. Q.; Li, Q. F.; Zhao, Q.; Zhou, L. Highly selective thiocyanate electrode based on bis-bebzoinsemitriethylenetetraamine binuclear copper(II) complex as neutral carrier. Electrochem. Communications 2003, 5, 717–721. [Google Scholar]
- Navratil, T.; Kopanica, M. Indirect voltammetric determination of thiocyanate ions in electroplating solutions. Chemia Analityczna 2003, 48, 127–138. [Google Scholar]
- Sánchez-Pedreño, C.; Ortuño, J.A.; Albero, M. I.; García, M. S.; Valero, M. V. Development of a new bulk optode membrana for the determination of mercury(II). Anal. Chim. Acta 2000, 414, 195–203. [Google Scholar]
- Sánchez-Pedreño, C.; Ortuño, J.A.; Albero, M. I.; García, M. S.; García de las Bayonas, J. C. A new procedure for the construction of flow-through optodes. Application to the determination of copper(II). Fresenius J. Anal. Chem. 2000, 366, 811–815. [Google Scholar]
- García, M.S.; Sánchez-Pedreño, C.; Ortuño, J. A.; Albero, M. I.; Expósito, R. Kinetic methods for the determination of cadmium (II) based on a flow-through bulk optode. Talanta 2002, 56, 481–489. [Google Scholar]
- García, M.S.; Albero, M. I.; Ortuño, J. A.; Sánchez-Pedreño, C.; Expósito, R. Flow-through bulk optode for the spectrophotometric determination of perchlorate. Microchim. Acta 2003, 143, 59–63. [Google Scholar]
- Ortuño, J. A.; Albero, M. I.; García, M. S.; Sánchez-Pedreño, C.; García, M. I.; Expósito, R. Flow-through bulk optode for the fluorimetric determination of perchlorate. Talanta 2003, 60, 563–569. [Google Scholar]
- IUPAC Nomenclature, symbols, unit and their usage in spectrochemical analysis. II. Data interpretation. Pure Appl. Chem. 1976, 45, 24.
- Glazier, S.A.; Arnold, M.A. Selectivity of membrane electrodes based on derivatives of dibenzyltin dichloride. Anal. Chem. 1991, 63, 754–759. [Google Scholar]
- Greenberg, A.E.; Rhodes, R.; Clesceri, L.S. Standard methods for the examination of water and wastewater, 16 Ed. ed; American Public Health Association: Washington, 1985; p. p. 348. [Google Scholar]

| Anion | log KSCN-, X- | [X-] / M assayed |
|---|---|---|
| SO4 2-, HCO3-, Cl-, PO43-, Ac-, Br- | No interference up to 1×10-1 | |
| NO3- | -2.5 | 1×10-1 |
| I - | -2.1 | 1×10-2 |
| ClO4- | 0.1 | 1×10-3 |
| Sample | SCN- content / mg l-1 | |||
|---|---|---|---|---|
| Added | Proposed Method * | Recovery % | Reference Method * | |
| Spring water | 29.0 | 28.8 ± 0.4 | 99.3 | 29.2 ± 0.1 |
| 58.1 | 58.3 ± 0.8 | 100.3 | 58.7 ± 0.2 | |
| Tap water | 29.0 | 29.1 ± 0.1 | 100.3 | 29.4 ± 0.1 |
| 58.1 | 59.3 ± 0.4 | 102.1 | 58.7 ± 0.2 | |
| Reservoir water | 29.0 | 29.1 ± 0.1 | 100.3 | 29.4 ± 0.1 |
| 58.1 | 60.4 ± 0.9 | 104.0 | 57.0 ± 0.2 | |
© 2006 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
García, S., Mª; Ortuño, J.A.; Sánchez-Pedreño, C.; Albero, I., Mª; Fernández, J., Mª. Flow-through Bulk Optode for Spectrophotometric Determination of Thiocyanate and Its Application to Water and Saliva Analysis. Sensors 2006, 6, 1224-1233. https://doi.org/10.3390/s6101224
García S Mª, Ortuño JA, Sánchez-Pedreño C, Albero I Mª, Fernández J Mª. Flow-through Bulk Optode for Spectrophotometric Determination of Thiocyanate and Its Application to Water and Saliva Analysis. Sensors. 2006; 6(10):1224-1233. https://doi.org/10.3390/s6101224
Chicago/Turabian StyleGarcía, Soledad, Mª, Joaquín A. Ortuño, Concepción Sánchez-Pedreño, Isabel Albero, Mª, and José Fernández, Mª. 2006. "Flow-through Bulk Optode for Spectrophotometric Determination of Thiocyanate and Its Application to Water and Saliva Analysis" Sensors 6, no. 10: 1224-1233. https://doi.org/10.3390/s6101224
APA StyleGarcía, S., Mª, Ortuño, J. A., Sánchez-Pedreño, C., Albero, I., Mª, & Fernández, J., Mª. (2006). Flow-through Bulk Optode for Spectrophotometric Determination of Thiocyanate and Its Application to Water and Saliva Analysis. Sensors, 6(10), 1224-1233. https://doi.org/10.3390/s6101224





