Investigating the Quantitative Structure-Activity Relationships for Antibody Recognition of Two Immunoassays for Polycyclic Aromatic Hydrocarbons by Multiple Regression Methods
Abstract
: Polycyclic aromatic hydrocarbons (PAHs) are ubiquitous contaminants found in the environment. Immunoassays represent useful analytical methods to complement traditional analytical procedures for PAHs. Cross-reactivity (CR) is a very useful character to evaluate the extent of cross-reaction of a cross-reactant in immunoreactions and immunoassays. The quantitative relationships between the molecular properties and the CR of PAHs were established by stepwise multiple linear regression, principal component regression and partial least square regression, using the data of two commercial enzyme-linked immunosorbent assay (ELISA) kits. The objective is to find the most important molecular properties that affect the CR, and predict the CR by multiple regression methods. The results show that the physicochemical, electronic and topological properties of the PAH molecules have an integrated effect on the CR properties for the two ELISAs, among which molar solubility (Sm) and valence molecular connectivity index (3χv) are the most important factors. The obtained regression equations for RisC kit are all statistically significant (p < 0.005) and show satisfactory ability for predicting CR values, while equations for RaPID kit are all not significant (p > 0.05) and not suitable for predicting. It is probably because that the RisC immunoassay employs a monoclonal antibody, while the RaPID kit is based on polyclonal antibody. Considering the important effect of solubility on the CR values, cross-reaction potential (CRP) is calculated and used as a complement of CR for evaluation of cross-reactions in immunoassays. Only the compounds with both high CR and high CRP can cause intense cross-reactions in immunoassays.1. Introduction
Polycyclic aromatic hydrocarbons (PAHs) are ubiquitous contaminants found in air, water, sediment and soil. They are a large and diverse class of compounds consisting of two or more fused aromatic rings produced by both natural and anthropogenic processes. Since many PAHs and the metabolites are mutagens and carcinogens, PAHs have been listed as priority pollutants in many countries. PAHs rarely occur as individual compounds in the environment, but always as a complex mixture of various compounds. Conventional analytical methods for PAHs are gas chromatography (GC) and high-performance liquid chromatography (HPLC) which require time-consuming pretreatment extraction procedures.
In recent years, immunoassays have represented a fast, sensitive, inexpensive and field-portable analytical method to complement traditional chromatographic procedures for PAHs [1]. Some immunoassay techniques, such as enzyme-linked immunosorbent assay (ELISA) [2,3], fluorescence polarization immunoassay [4], chemiluminescent immunoassay [5], real-time immuno-polymerase chain reaction assay [6] and immunosensor [7], were developed for PAHs analysis. Several ELISA test kits for PAHs have been produced and are commercially available, among which RaPID and RisC are the most widely studied and used [8]. They have been used for determination of PAHs in water [9,10], soil [11–15], biological [16,17] and some other samples [18]. The RisC kit is used in US EPA method 4035 for rapid screening test of PAHs in soil samples.
Cross-reactions are common phenomena in immunoassays, i.e., the antibody responds with compounds structurally related to the analyte. Cross-reactivity (CR) is an essential character to evaluate the extent of cross-reactions in immunoreactions and immunoassays. Although CR affects the specificity of the immunoassay and possibly results in bias in the test result, sometimes it can be explored to identify metabolites or structurally similar compounds of the analyte for class-specific immunoassays. It is believed that the CR of a cross-reactant is determined by the physicochemical and structural properties of the compound.
In our previous work, we found that CR values of PAHs are significantly correlated with the data of logarithm of octanol-water partition coefficient (log Kow), but it seemed that CR cannot be solely explained by log Kow [19]. In this article, in addition to log Kow, other six representative physicochemical, electronic and topological descriptors are selected to investigate the correlation between CR and the molecular properties of PAHs. Three multiple regression methods, including stepwise multiple linear regression (MLR), principal component regression (PCR) and partial least square regression (PLSR), were employed to develop the quantitative structure-activity relationship (QSAR) models using the data of two commercial PAHs ELISA kits. The objective of this work is to find the most important molecular properties that affect the CR of PAHs in immunoassays, and if possible, to find the feasibility of predicting CR by multiple regression analysis. Moreover, considering the important effect of water solubility on the CR, we calculated cross-reaction potential (CRP) and used it to complement CR for evaluation of cross-reactions in immunoassays.
2. Experimental Section
2.1. Molecular Descriptor Data Set
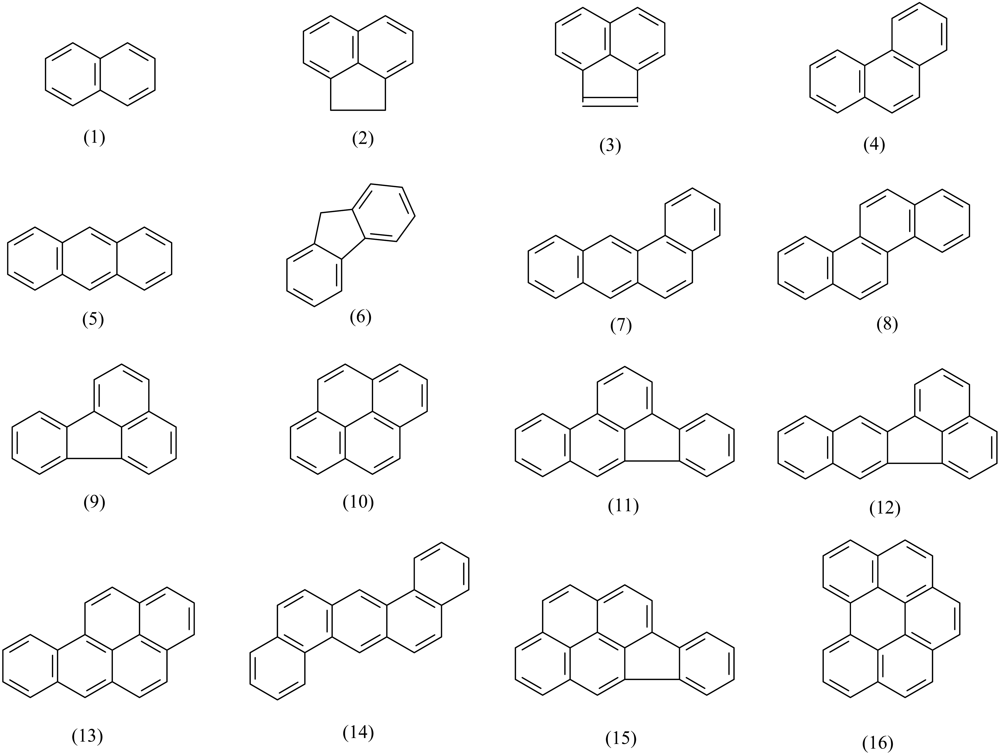
The data of 16 representative PAHs (Figure 1) analyzed by the two ELISA kits were used for modeling. Seven typical physicochemical, electronic and topological descriptors are selected for developing the QSAR models. They are molar solubility (Sm), the logarithm of octanol-water partition coefficient (log Kow), the gap between the highest occupied molecular orbital energy and the lowest unoccupied molecular orbital energy (EHOMO − ELUMO), and four valence molecular connectivity indices (0χv, 1χv, 2χv, 3χv). The data of solubility (S) and log Kow were obtained from Mackay et al. [20]. Sm was calculated by dividing S with the molecular weight. The data of EHOMO − ELUMO were from de Lima Ribeiro and Ferreira [21]. The data of 0χv, 1χv, 2χv and 3χv were cited from Govers and Aiking [22].
2.2. Data Set of Cross-Reactivity
The experimental data of cross-reactivity (CR) for the two commercial PAHs ELISA kits, RaPID and RisC, were obtained from Krämer [8]. Since activity data used for QSARs should be in molar dimensions, CR values were converted to molar cross-reactivity (MCR), i.e., the ratio of the molar IC50 of target analyte and the cross-reactant, for QSAR modeling [19]. Then, the predicted MCR was calculated by the QSAR models and converted to CR for comparison with the experimental CR value. We assumed that the CR values “<0.5%” and “<1.6%” were low enough to describe low levels of cross-reactions in the two ELISA kit tests, and reasonably considered the CR value “<” to be “=” for regression modeling [19].
2.3. Regression Analysis
The data of 14 compounds of the 16 PAHs were submitted as training set for regression analysis, and anthracene and benzo[a]pyrene were used as the test set. In order to reduce the colinearity and the number of the molecular descriptors, the analysis of the quantitative relationship between log MCR and the molecular descriptors was performed by stepwise MLR, PCR and PLSR employing SAS 8.1 software. In the stepwise MLR procedure, the data of the seven descriptors of the 16 PAHs were collected in a single data matrix, and the key descriptors were selected by adding descriptors one by one to perform a multivariable regression calculation. The variables significant at the 0.15 level were left in the model. In PCR analysis, the original descriptors were subjected to principal component analysis, and the subset of principal components explaining more than 90% of the variance was extracted. Then, the principal components extracted were subjected to multiple linear regression analysis. The PLSR method reduced large volume of descriptors to several components that were most correlative with the CR. These components were the linear combinations of the descriptors and used as new variables for regression analysis. The optimum number of components for regression analysis was obtained by the leave-one-out cross-validation procedure.
2.4. Cross-Reaction Potential
CR is the ratio of the IC50 (the 50% inhibition concentration) of the target analyte and the IC50 of the cross-reactant. Considering the effect of water solubility on the CR value and the immunoassay results, we defined cross-reaction potential (or cross-reaction probability, CRP), i.e., 100-fold the ratio between the solubility (S) of a cross-reactant and the IC50 value [Equation (1)], and used it as a complement of CR to evaluate the extent of cross-reaction. CRP reflects the relative extent of cross-reaction of a non-target cross-reactant compared with the water solubility. The data set of S was from Mackay et al. [20], and IC50 data were from Krämer [8]. We assumed that the IC50 value “>1,000 μg·L−1” was high enough to be considered as “=1,000 μg·L−1” for CRP calculation:
3. Results and Discussion
3.1. Effect of Molecular Properties on Cross-Reactivity
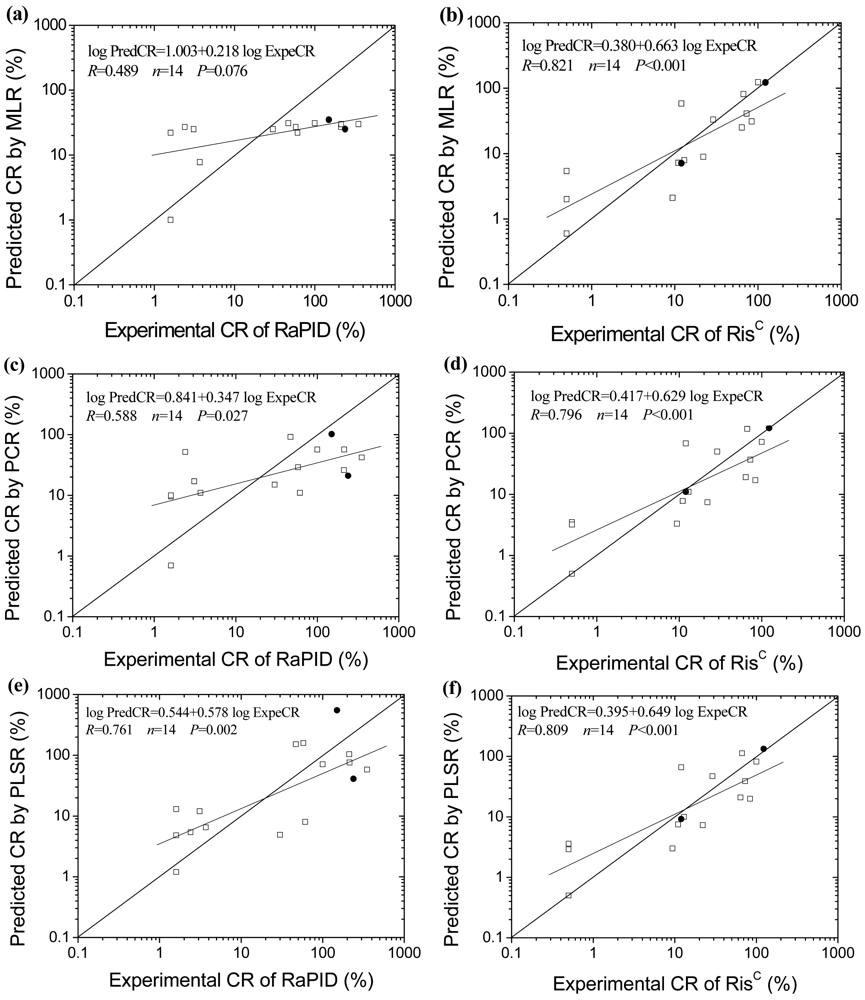
The molecular structures of the 16 PAHs analyzed by the two ELISAs are shown in Figure 1. Since antibodies and antigens in immunoreactions are not mass-equivalent but rather molar-equivalent, molar cross-reactivity (MCR) rather than mass cross-reactivity (CR) is applied to investigate the quantitative structure and cross-reactivity relationships. The obtained stepwise MLR, PCR and PLSR equations and statistical parameters are illustrated in Table 1.
It shows that the regression models for RisC kit are all significant (p < 0.005), while the models for RaPID kit are all not significant (p > 0.05). The probable reason is that RisC immunoassay employs a monoclonal antibody, while RaPID kit is based on polyclonal antibody. In the stepwise MLR procedure for RaPID, only Sm enters the regression model, and the other six molecular descriptors are excluded from the regression equation. As for RisC, Sm and 3χv are left in the model. In the PCR procedure, the two most significant principal components (PC1 and PC2) describe respectively 85.7% and 8.3%, and totally 94.0% of the variance. Eigenvectors of the principal components indicate that PC1 demonstrates the integrated character of the seven descriptors, while PC2 mainly represents the character of Sm. The regression equations for RaPID and RisC are Equation (2) and Equation (3) respectively. In the PLSR procedure, the models are optimized by leave-one-out cross-validation, and the optimum numbers of components are found to be 4 and 2 for RaPID and RisC, respectively. The parameter estimates for centered and scaled data (marked by *) are shown in Equation (4) and Equation (5). The results of stepwise MLR, PCR and PLSR imply that Sm plays an important role in affecting the CR property of the PAHs for the two ELISA kits, and 3χv also affects the CR for RisC kit to some extent:
3.2. Predicting Cross-Reactivity
CR is one of the most important characteristics of an ELISA test, and influences the extent of cross-reaction and the results of ELISAs significantly. However, due to the difficulty and expense in term of cost and time, not all of the CR data of the cross-reactants are available. Moreover, it is impractical to directly measure the CR of the cross-reactants which are not commercially available, so predicted CR values of the PAHs for the two ELISA kits were calculated using the obtained MLR, PCR and PLSR models, and compared with the experimental data (Table 2, Figure 2). The predicted CR values for RisC agree very well with the experimental data, while the predicted and experimental data for RaPID do not agree well with each other. The models were further external validated using the data of three-ringed anthracene and five-ringed benzo[a]pyrene as test set. The range of predicting error for anthracene is from −1.6% to +8.1%, and for benzo[a]pyrene is from −40.8% to −8.3%. It appears that the obtained models can successfully predict the CR for RisC kit, but present poor predicting ability for RaPID kit.
3.3. Cross-Reaction Potential
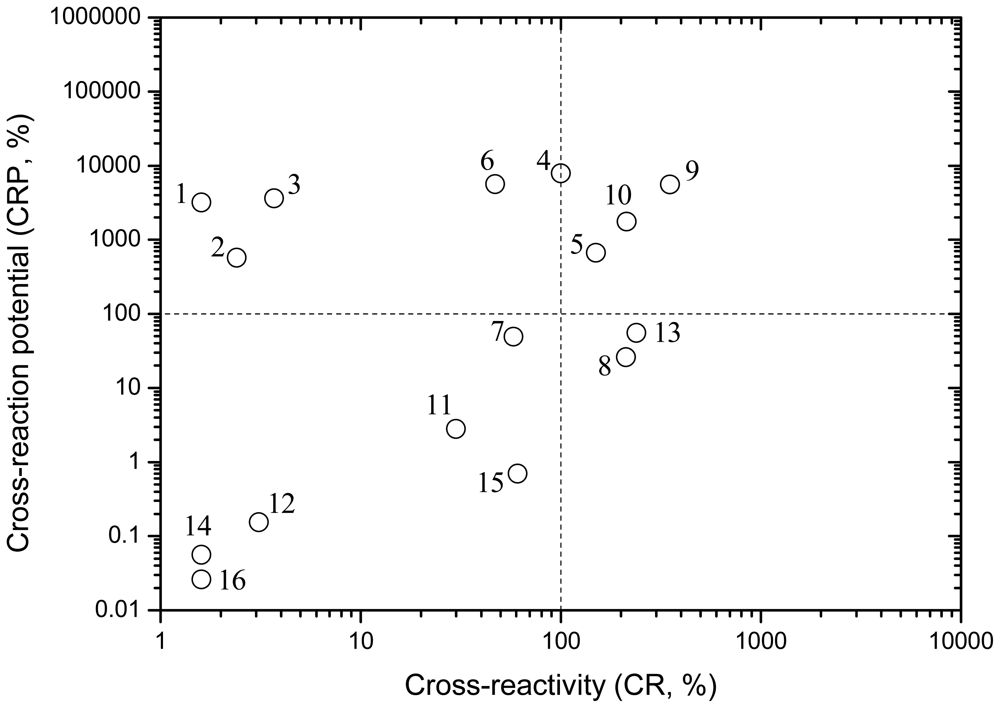
Generally speaking, higher CR values imply higher levels of immunoreactions. However, the antigen-antibody reactions in immunoassays are carried out in water or buffer solutions, so if the solubility of a cross-reactant is much lower than the IC50 value, it cannot possibly cause intense cross-reactions in the immunoassays. That is to say, the concentration of this compound in real water samples cannot be high enough to evoke high extent cross-reaction in immunoassays, even though the CR is very high. Some of the 16 PAHs are very hydrophobic compounds, and the solubility is much lower than the tested IC50 value. For example, the IC50 referring to water analysis for benzo[a]pyrene for the RaPID ELISA kit is 6.9 μg·L−1, while the solubility of benzo[a]pyrene is 3.8 μg·L−1 (Table 3), so although benzo[a]pyrene has a high CR of 239% in the RaPID ELISA, the concentration of benzo[a]pyrene in water samples cannot be possibly high enough to evoke a high level of cross-reaction. Hence, considering the important effect of water solubility on immunoreactions and immunoassays, cross-reaction potential (or cross-reaction probability, CRP), i.e., the relative IC50 of a non-target cross-reactant compared with its water solubility, was defined and used as a complement of CR to evaluate the potential and probability that a cross-reaction would occur.
The IC50 and CRP data for the 16 PAHs for RaPID kit are shown in Table 3. The CRP values for RisC kit are not calculated because the IC50 values are not available. In addition to the target analyte of phenanthrene, the 15 cross-reactants in RaPID ELISA can be divided into four groups according to CR and CRP (Figure 3): (I) CR > 100%, CRP > 100%; (II) CR > 100%, CRP < 100%; (III) CR < 100%, CRP > 100%; and (IV) CR < 100%, CRP < 100%. The compounds of group (I) might cause intense cross-reactions and affect the determination of phenanthrene, while the group (IV) compounds have little cross-reaction effect on the analysis results. As for the group (II) compounds, the CR is high, while the CPR is low because of the relatively low solubility. The group (III) compounds are two-ringed and three-ringed PAHs, and less cross-reactive but more water soluble. The extent of the cross-reactions of group (II) and (III) compounds depends on both the CR and the CRP properties.
It should be pointed out that the RaPID PAHs ELISA kit is applied not only for water samples [9,10], but more often for soil samples [11,14,15]. In the pretreatment procedure, PAHs were usually extracted from the soil samples by methanol and diluted by buffer. PAHs are very hydrophobic molecules and can be adsorbed to soils at very high concentration. In the immunoassays of PAHs, much attention should be paid to the solubility of the compounds during the procedures of solvent extraction and buffer dilution.
3.4. Comparison of the Two Kits
The comparison of RaPID and RisC PAHs ELISA kits based on the character and the applicability are illustrated in Table 4. It seems that RisC ELISA is more specific, while RaPID ELISA is applied for more kinds of environmental samples. The selection of appropriate ELISAs for PAHs depends on the objective and request of the analysis.
4. Conclusions
RaPID and RisC are two widely studied ELISA kits used for analysis of PAHs. Three regression methods, including stepwise MLR, PCR and PLSR, were successfully applied to investigate the correlation between the molecular properties and the CR properties of PAHs for the two ELISA kits. It seems that the physicochemical, electronic and topological properties of the PAH molecules have an integrated effect on the CR properties for the two kits. Sm and 3χv show especially strong effects on CR, which implies the important role of hydrophobic interactions and molecular shape in the PAH-antibody reactions. The obtained regression equations for RisC kit are all statistically significant (p < 0.005) and show satisfactory ability for predicting CR values, while equations for RaPID kit are all not significant (p > 0.05) and not suitable for prediction. It is probably because that the RisC immunoassay employs a monoclonal antibody, while the RaPID kit is based on polyclonal antibody. Considering the important effect of solubility on CR for the two PAHs ELISAs, cross-reaction potential (CRP) is defined and used as a complement of CR to evaluate the extent of cross-reaction in immunoassays. We believe that only the compounds with both high CR and high CRP can cause intense cross-reactions in immunoassays. This work demonstrated the feasibility of multiple regression methods in investigating the quantitative structure-CR relationships and predicting CR in immunoassays.
Acknowledgments
The authors thank Hui-Xuan Gao of the School of Mathematical Sciences of Peking University, China, for her help in statistical analysis.
References
- Fähnrich, K.A.; Pravda, M.; Guilbault, G.G. Immunochemical determination of polycyclic aromatic hydrocarbons (PAHs). Anal. Lett. 2002, 35, 1269–1300. [Google Scholar]
- Matschulat, D.; Deng, A.; Niessner, R.; Knopp, D. Development of a highly sensitive monoclonal antibody based ELISA for detection of benzo[a]pyrene in potable water. Analyst 2005, 130, 1078–1086. [Google Scholar]
- Spier, C.R.; Bromage, E.S.; Harris, T.M.; Unger, M.A.; Kaattari, S.L. The development and evaluation of monoclonal antibodies for the detection of polycyclic aromatic hydrocarbons. Anal. Biochem. 2009, 387, 287–293. [Google Scholar]
- Goryacheva, I.Y.; Eremin, S.A.; Shutaleva, E.A.; Suchanek, M.; Niessner, R.; Knopp, D. Development of a fluorescence polarization immunoassay for polycyclic aromatic hydrocarbons. Anal. Lett. 2007, 40, 1445–1460. [Google Scholar]
- Roda, A.; Simoni, P.; Mirasoli, M.; Baraldini, M.; Violante, F.S. Development of a chemiluminescent enzyme immunoassay for urinary 1-hydroxypyrene. Anal. Bioanal. Chem. 2002, 372, 751–758. [Google Scholar]
- Zhuang, H.S.; Zhou, C. Determination of anthracene by real-time immuno-polymerase chain reaction assay. Anal. Chim. Acta 2009, 633, 278–282. [Google Scholar]
- Wei, M.Y.; Wen, S.D.; Yang, X.Q.; Guo, L.H. Development of redox-labeled electrochemical immunoassay for polycyclic aromatic hydrocarbons with controlled surface modification and catalytic voltammetric detection. Biosens. Bioelectron. 2009, 24, 2909–2914. [Google Scholar]
- Krämer, P.M. A strategy to validate immunoassay test kits for TNT and PAHs as a field screening method for contaminated sites in Germany. Anal. Chim. Acta 1998, 376, 3–11. [Google Scholar]
- Castillo, M.; Oubina, A.; Barceló, D. Evalution of ELISA kits followed by liquid chromatography-atmospheric pressure chemical ionization-mass spectrometry for the determination of organic pollutants in industrial effluents. Environ. Sci. Technol. 1998, 32, 2180–2184. [Google Scholar]
- Barceló, D.; Oubina, A.; Salau, J.S.; Perez, S. Determination of PAHs in river water samples by ELISA. Anal. Chim. Acta 1998, 376, 49–53. [Google Scholar]
- Chuang, J.C.; van Emon, J.M.; Chou, Y.L.; Junod, N.; Finegold, J.K.; Wilson, N.K. Comparison of immunoassay and gas chromatography-mass spectrometry for measurement of polycyclic aromatic hydrocarbons in contaminated soil. Anal. Chim. Acta 2003, 486, 31–39. [Google Scholar]
- Nording, M.; Haglund, P. Evaluation of the structure/cross-reactivity relationship of polycyclic aromatic compounds using an enzyme-linked immunosorbent assay kit. Anal. Chim. Acta 2003, 487, 43–50. [Google Scholar]
- Nording, M.; Frech, K.; Persson, Y.; Forsman, M.; Haglund, P. On the semi-quantification of polycyclic aromatic hydrocarbons in contaminated soil by an enzyme-linked immunosorbent assay kit. Anal. Chim. Acta 2006, 555, 107–113. [Google Scholar]
- Fillmann, G.; Bicego, M.C.; Zamboni, A.; Fileman, T.W.; Depledge, M.H.; Readman, J.W. Validation of immunoassay methods to determine hydrocarbon contamination in estuarine sediments. J. Braz. Soc. 2007, 18, 774–781. [Google Scholar]
- Rigou, P.; Saini, S.; Setford, S.J. Field-based supercritical fluid extraction and immunoassay for determination of PAHs in soils. Int. J. Environ. Anal. Chem. 2004, 84, 979–994. [Google Scholar]
- Troisi, G.M.; Borjesson, L. Development of an immunoassay for the determination of polycyclic aromatic hydrocarbons in plasma samples from oiled seabirds. Environ. Sci. Technol. 2005, 39, 3748–3755. [Google Scholar]
- Fillmann, G.; Watson, G.M.; Howsam, M.; Francioni, E.; Depledge, M.H.; Readman, J.W. Urinary PAH metabolites biomarkers of exposure in aquatic environments. Environ. Sci. Technol. 2004, 38, 2649–2656. [Google Scholar]
- Kim, I.S.; Ritchie, L.; Setford, S.; Taylor, J.; Allen, M.; Wilson, G.; Heywood, R.; Pahlavanpour, B.; Saini, S. Quantitative immunoassay for determining polyaromatic hydrocarbons in electrical insulating oils. Anal. Chim. Acta 2001, 450, 13–25. [Google Scholar]
- Zhang, Y.F.; Ma, Y.; Gao, Z.X.; Dai, S.G. Predicting the cross-reactivities of polycyclic aromatic hydrocarbons in ELISA by regression analysis and CoMFA methods. Anal. Bioanal. Chem. 2010, 397, 2551–2557. [Google Scholar]
- Mackay, D.; Shiu, W.Y.; Ma, K.C.; Lee, S.C. Handbook of Physical-Chemical Properties and Environmental Fate for Organic Chemicals, 2nd ed.; Taylor & Francis: Boca Raton, FL, USA, 2006. [Google Scholar]
- De Lima Ribeiro, F.A.; Ferreira, M.M.C. QSPR models of boiling point, octanol-water partition coefficient and retention time index of polycyclic aromatic hydrocarbons. J. Mol. Struct. Theochem 2003, 663, 109–126. [Google Scholar]
- Govers, H.; Ruepert, C.; Aiking, H. Quantitative structure-activity relationships for polycyclic aromatic hydrocarbons: Correlation between molecular connectivity, physico-chemical properties, bioconcentration and toxicity in Daphnia pulex. Chemosphere 1984, 13, 227–236. [Google Scholar]
- Xu, Z.L.; Xie, G.M.; Li, Y.X.; Wang, B.F.; Beier, R.C.; Lei, H.T.; Wang, H.; Shen, Y.D.; Sun, Y.M. Production and characterization of a broad-specificity polyclonal antibody for O,O-diethyl organophosphorus pesticides and a quantitative structure-activity relationship study of antibody recognition. Anal. Chim. Acta 2009, 647, 90–96. [Google Scholar]
- Xu, Z.L.; Shen, Y.D.; Zheng, W.X.; Beier, R.C.; Xie, G.M.; Dong, J.X.; Yang, J.Y.; Wang, H.; Lei, H.T.; She, Z.G.; et al. Broad-specific immunoassay for O,O-diethyl organophosphorus pesticides: Application of molecular modeling to improve assay sensitivity and study antibody recognition. Anal. Chem. 2010, 82, 9314–9321. [Google Scholar]
- Yuan, M.; Liu, B.; Liu, E.; Sheng, W.; Zhang, Y.; Crossan, A.; Kennedy, I.; Wang, S. Immunoassay for phenylurea herbicides: Application of molecular modeling and quantitative structure-activity relationship analysis on an antigen-antibody interaction study. Anal. Chem. 2011, 83, 4767–4774. [Google Scholar]
| ELISA kit. | Regression method | Regression Equation | Statistics | ||||
|---|---|---|---|---|---|---|---|
| F | P | R2 | n | PRESS | |||
| RaPID | MLR | log MCR = 1.542 − 0.00676 × Sm | 4.30 | 0.0603 | 0.2638 | 14 | 11.30 |
| PCR | log MCR = 4.907 − 0.01004 × Sm − 0.07337 × log Kow − 0.2108 × (EHOMO − ELUMO) − 0.04482 × 0χv − 0.06256 × 1χv − 0.06708 × χv − 0.4319 × 3χv | 3.17 | 0.0819 | 0.3656 | 14 | 16.68 | |
| PLSR | log MCR = 10.49 − 0.008925 × Sm + 1.015 × log Kow − 1.132 × (EHOMO − ELUMO) + 0.1073 × 0χv − 0.04788 × 1χv − 0.4657 × 2χv − 8.925 × 3χv | 3.30 | 0.0634 | 0.5943 | 14 | 10.88 | |
| RisC | MLR | log MCR = 3.439 − 0.01270 × Sm − 4.041 × 3χv | 12.19 | 0.0050 | 0.6603 | 14 | 4.62 |
| PCR | log MCR = 5.800 − 0.01231 × Sm − 0.1162 × log Kow − 0.2169 × (EHOMO − ELUMO) − 0.06868 × 0χv − 0.09743 × 1χv − 0.1065 × 2χv − 0.7045 × 3χv | 8.95 | 0.0049 | 0.6193 | 14 | 5.13 | |
| PLSR | log MCR = 5.485 − 0.01245 × Sm − 0.05071 × log Kow − 0.1704 × (EHOMO − ELUMO) − 0.06290 × 0χv − 0.1027 × 1χv − 0.1375 × 2χv − 1.255 × 3χv | 9.71 | 0.0037 | 0.6383 | 14 | 4.92 | |
| Number | Compound | CR (%) of RaPID | CR (%) of RisC | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Experimental a | Predicted by MLR | Predicted by PCR | Predicted by PLSR | Experimental a | Predicted by MLR | Predicted by PCR | Predicted by PLSR | ||
| 1 | Naphthalene | <1.6 | 1.0 | 0.7 | 1.2 | 0.5 | 0.6 | 0.5 | 0.5 |
| 2 | Acenaphthene | 2.4 | 27 | 52 | 5.4 | 12 | 58 | 68 | 66 |
| 3 | Acenaphthylene | 3.7 | 7.8 | 11 | 6.5 | 13 | 7.9 | 11 | 10 |
| 4 | Phenanthrene | 100 | 31 | 57 | 71 | 100 | 123 | 72 | 82 |
| 5 | Anthracene | 150 | 35 | 102 | 550 | 123 | 122 | 121 | 133 |
| 6 | Fluorene | 47 | 31 | 92 | 152 | 67 | 81 | 118 | 113 |
| 7 | Benzo[a]anthracene | 58 | 27 | 29 | 158 | 64 | 25 | 19 | 21 |
| 8 | Chrysene | 212 | 27 | 26 | 104 | 84 | 31 | 17 | 20 |
| 9 | Fluoranthene | 351 | 30 | 42 | 58 | 73 | 41 | 37 | 39 |
| 10 | Pyrene | 214 | 30 | 57 | 76 | 29 | 33 | 50 | 47 |
| 11 | Benzo[b]fluoranthene | 30 | 25 | 15 | 4.9 | 22 | 8.9 | 7.4 | 7.3 |
| 12 | Benzo[k]fluoranthene | 3.1 | 25 | 17 | 12 | 11 | 7.2 | 7.8 | 7.5 |
| 13 | Benzo[a]pyrene | 239 | 25 | 21 | 41 | 12 | 7.1 | 11 | 9.2 |
| 14 | Dibenzo[a,h]anthracene | <1.6 | 22 | 9.6 | 13 | <0.5 | 5.4 | 3.5 | 3.6 |
| 15 | Indeno[1,2,3-cd]pyrene | 61 | 22 | 11 | 8.0 | 9.4 | 2.1 | 3.3 | 3.0 |
| 16 | Benzo[g,h,i]perylene | <1.6 | 22 | 10 | 4.8 | <0.5 | 2.0 | 3.2 | 2.9 |
aData are from [8].
| Number | Compound | S (mg·L−1) a | IC50 (μg·L−1) b | CRP (%) |
|---|---|---|---|---|
| 1 | Naphthalene | 31.7 | >1,000 | 3,170 |
| 2 | Acenaphthene | 3.93 | 688 | 571 |
| 3 | Acenaphthylene | 16.1 | 447 | 3,602 |
| 4 | Phenanthrene | 1.29 | 16.5 | 7,818 |
| 5 | Anthracene | 0.073 | 11 | 664 |
| 6 | Fluorene | 1.98 | 35.2 | 5,625 |
| 7 | Benzo[a]anthracene | 0.014 | 28.4 | 49 |
| 8 | Chrysene | 0.002 | 7.8 | 26 |
| 9 | Fluoranthene | 0.26 | 4.7 | 5,532 |
| 10 | Pyrene | 0.135 | 7.7 | 1,753 |
| 11 | Benzo[b]fluoranthene | 1.5 × 10−3 | 54.2 | 2.8 |
| 12 | Benzo[k]fluoranthene | 8.1 × 10−4 | 524 | 0.155 |
| 13 | Benzo[a]pyrene | 3.8 × 10−3 | 6.9 | 55 |
| 14 | Dibenzo[a,h]anthracene | 5.6 × 10−4 | >1,000 | 0.056 |
| 15 | Indeno[1,2,3-cd]pyrene | 1.9 × 10−4 | 27.2 | 0.699 |
| 16 | Benzo[g,h,i]perylene | 2.6 × 10−4 | >1,000 | 0.026 |
aData are from [20];bData are referring to water analysis and from [8].
| RaPID | RisC | |
|---|---|---|
| Reference compound | Phenanthrene | Phenanthrene |
| Analysis mode | Competitive heterogeneous ELISA, antibody is coated on tubes | Competitive heterogeneous ELISA, antibody is immobilized to magnetic particles |
| Cross-reactivity | 1.6%∼351% | 0.5%∼123% |
| Specificity | Not so specific | Relatively specific |
| Antibody | Polyclonal | Monoclonal |
| Cross-reactant | Anthracene, chrysene, fluoranthene, pyrene and benzo[a]pyrene | Anthracene |
| Samples | Water, soil, biological and some other samples | Mostly soil samples till now |
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, Y.-F.; Zhang, L.; Gao, Z.-X.; Dai, S.-G. Investigating the Quantitative Structure-Activity Relationships for Antibody Recognition of Two Immunoassays for Polycyclic Aromatic Hydrocarbons by Multiple Regression Methods. Sensors 2012, 12, 9363-9374. https://doi.org/10.3390/s120709363
Zhang Y-F, Zhang L, Gao Z-X, Dai S-G. Investigating the Quantitative Structure-Activity Relationships for Antibody Recognition of Two Immunoassays for Polycyclic Aromatic Hydrocarbons by Multiple Regression Methods. Sensors. 2012; 12(7):9363-9374. https://doi.org/10.3390/s120709363
Chicago/Turabian StyleZhang, Yan-Feng, Li Zhang, Zhi-Xian Gao, and Shu-Gui Dai. 2012. "Investigating the Quantitative Structure-Activity Relationships for Antibody Recognition of Two Immunoassays for Polycyclic Aromatic Hydrocarbons by Multiple Regression Methods" Sensors 12, no. 7: 9363-9374. https://doi.org/10.3390/s120709363




