Pterins as Sensors of Response to the Application of Fe3+-Dextran in Piglets
Abstract
:1. Introduction
2. Results and Discussion
2.1. Iron Concentrations
2.2. Leukocyte Counts
2.3. Cortisol Concentrations
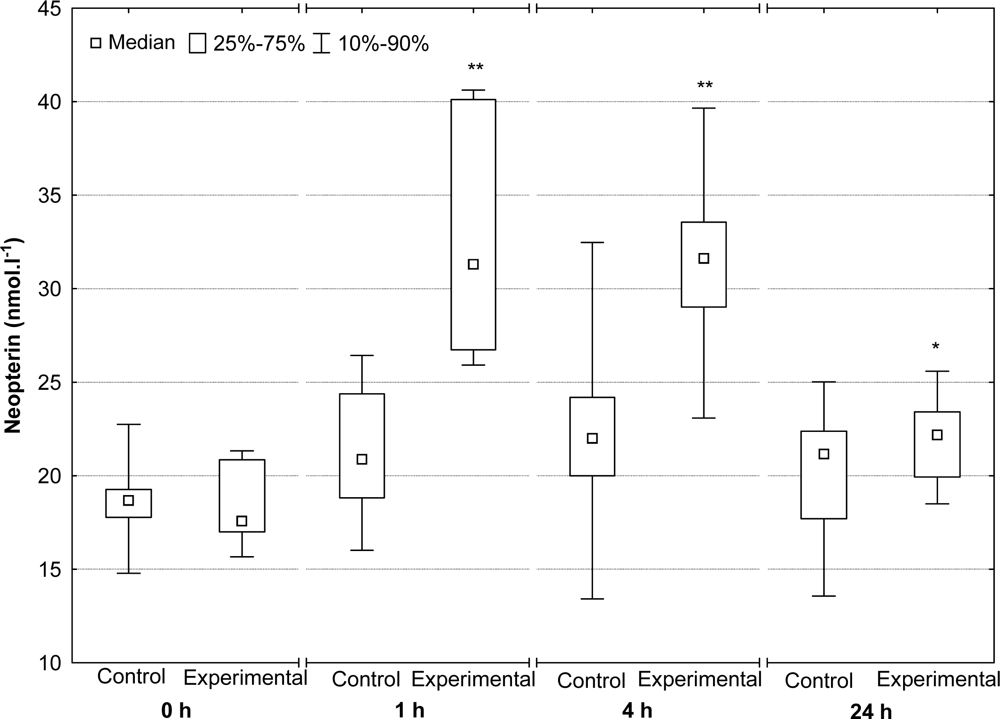
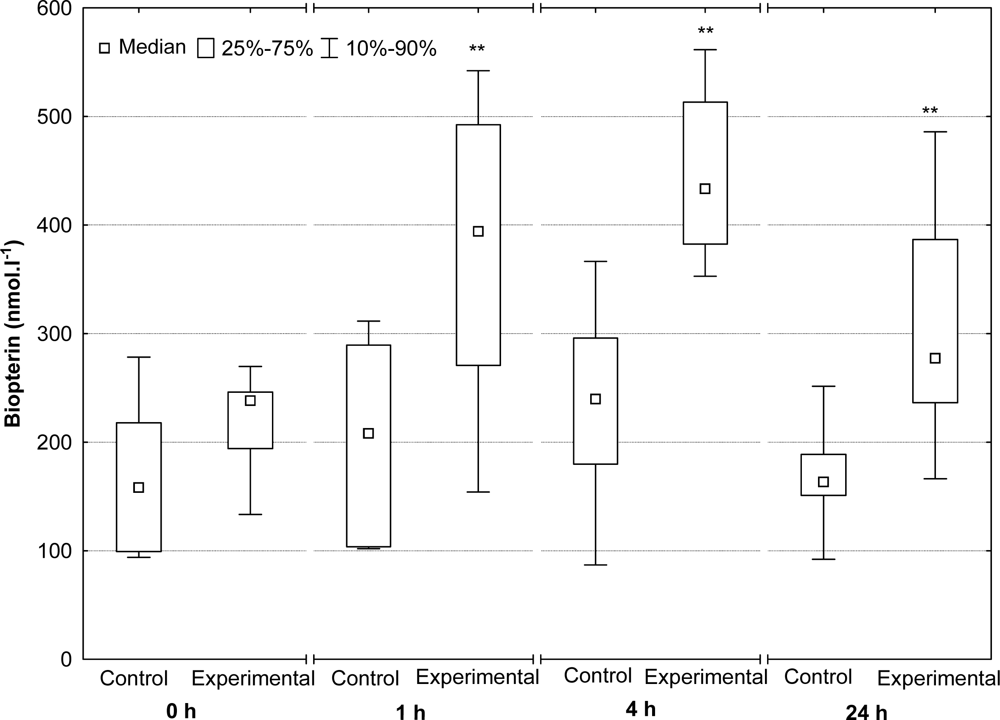
2.4. Serum Neopterin and Biopterin Concentrations
3. Instruments and Methods
3.1. Experimental Design
3.2. Haematological Examination
3.3. Determination of Iron Concentration
3.4. Sample Treatment before Pterine Determination
3.5. HPLC Analysis
3.6. Sample Treatment before Cortisol Determination
3.7. HPLC Analysis of Cortisol
3.8. Validation Prameters
3.9. Statistical Evaluation
4. Conclusions
Acknowledgments
References and Notes
- Berdovska, A.; Zwirska-Korczala, K. Neopterin measurement in clinical diagnosis. J. Clin. Pharm. Ther 2001, 26, 319–329. [Google Scholar]
- Brown, G.M. The biosynthesis of pteridines. Adv. Enzymol 1971, 35, 5–77. [Google Scholar]
- Zimmerman, A.W.; Jyonouchi, H.; Comi, A.M.; Connors, S.L.; Milstien, S.; Varsou, A. Cerebrospinal fluid and serum markers of inflammation in autism. Pediatr. Neurol 2005, 33, 195–201. [Google Scholar]
- Ziegler, I.; Fink, M.; Wilmanns, W. Biopterin level in peripheral blood cells as marker for hemopoietic cell proliferation during leukemia and polycythemia vera. Blut 1982, 44, 231–240. [Google Scholar]
- Hashimoto, R.; Nagatsu, T.; Ohta, T.; Mizutani, M.; Omura, I. Changes in the concentrations of tetrahydrobiopterin, the cofactor of tyrosine hydroxylase, in blood under physical stress and depression. Ann. N.Y. Acad. Sci 2004, 1018, 378–386. [Google Scholar]
- Murr, C.; Fuith, L.C.; Winder, B.; Wirleitner, B.; Baier-Bitterlich, G.; Fuchs, D. Increased neopterin concentrations in patients with cancer: indicator of oxidative stress? Anticancer Res 1999, 19, 1721–1728. [Google Scholar]
- Forrest, C.M.; Youd, P.; Kennedy, A.; Gould, S.R.; Darlington, L.G.; Stone, T.W. Purine, kynurenine, neopterin and lipid peroxidation levels in inflammatory bowel disease. J. Biomed. Sci 2002, 9, 436–442. [Google Scholar]
- Murr, C.; Winder, B.; Wirleitner, B.; Fuchs, D. Neopterin as a marker for immune system activation. Curr. Drug Metab 2002, 3, 175–187. [Google Scholar]
- Fuchs, D.; Wachter, H. Neopterin. In International Clinical Laboratory Diagnostics, 1st ed; TH Books Verlagsgesellschaft mbH: Frankfurt, Germany, 1998. [Google Scholar]
- Jerin, A.; Pozar-Lukanovic, N.; Sojar, V.; Stanisavljevic, D.; Pavern-Erzen, V.; Osredkar, J. Neopterin: an early marker of surgical stress and hypoxic reperfusion damage during liver surgery. Clin. Chem. Lab. Med 2002, 40, 663–666. [Google Scholar]
- Svoboda, M. Metabolické poruchy u prasat (Metabolic disorders in pigs). Farmář 2004, 12, 35–36. [Google Scholar]
- Guo, D.; Jaber, B.L.; Lee, S.; Perianaygam, M.C.; King, A.J.; Periba, B.J.G.; Balakrishnan, V.S. Impact of iron dextran on polymorphonuclear cell function among hemodialysis patients. Clin. Nephrol 2002, 58, 134–142. [Google Scholar]
- Brohee, D.; Vanhaeverbeek, M.; Kennes, B.; Neve, P. Leukocyte and lymphocyte subsets after a short pharmacological stress by intravenous epinephrine and hydrocortisone in healthy humans. Int. J. Neurosci 1990, 53, 53–62. [Google Scholar]
- Brock, J.H. Iron and immunity. J. Nutr. Immunol 1993, 2, 47–106. [Google Scholar]
- Toman, M. ‘Veterinární Imunologie (Veterinary Immunology), 1st ed; Grada Publishing: Prague, Czech Republic, 2000; p. 350. [Google Scholar]
- Walker, E.M., Jr.; Walker, S.M. Effects of iron overload on the immune system. Ann. Clin. Lab. Sci 2000, 30, 354–365. [Google Scholar]
- Cardier, J.E.; Romano, E.; Soyano, A. T lymphocytes subsets in experimental iron overload. Immunopharmacol. Immunotoxicol 1997, 19, 75–87. [Google Scholar]
- Spear, A.T.; Sherman, A.B. Iron deficiency alters DMBA-induced tumor burden and natural killer cell cytotoxicity rats. J. Nutr 1992, 122, 46–55. [Google Scholar]
- Kuvibidila, S.R.; Kitchens, D.; Baliga, B.S. In vivo and in vitro iron deficiency reduces protein kinase C activity and translocation in murine splenic and purified T cells. J. Cell. Biochem 1999, 74, 468–478. [Google Scholar]
- Werner-Felmayer, G.; Golderer, G.; Werner, E.R. Tetrahydrobiopterin biosynthesis, utilization and pharmacological effects. Curr. Drug Metab 2002, 3, 159–173. [Google Scholar]
- Razumovitch, J.A.; Semenkova, G.N.; Fuchs, D.; Cherenkevich, S.N. Influence of neopterin on the generation of reactive oxygen species in human neutrophils. FEBS Lett 2003, 549, 83–86. [Google Scholar]
- Baier-Bitterlich, G.; Fuchs, D.; Murr, C.H.; Reibnegger, G.; Werner-Felmayer, G.; Sgonc, R.; Bőck, G.; Dierich, M.P.; Wachter, H. Effect of neopterin and 7,8-dihydroneopterin on tumor necrosis factor-α induced programmed cell death. FEBS Lett 1995, 364, 234–238. [Google Scholar]
- Oettl, K.; Wirleitner, B.; Baier-Bitterlich, G.; Grammer, T.; Fuchs, D.; Reibnegger, G. Formation of oxygen radicals in solutions of 7,8-dihydroneopterin. Biochem. Biophys. Res. Commun 1999, 264, 262–267. [Google Scholar]
- Schrodl, W.; Kunze, R.; Kruger, M. Determination of C-reactive protein and neopterin in serum diseased and bacterially infected swine. Berl. Műnch. Tierärzth. Wschr 1998, 111, 321–325. [Google Scholar]
- Amann, A.; Winder, B.; Rieder, J.; Antretter, H.; Hoffmann, G.; Mayr, V.; Strohmenger, H.U.; Fuchs, D. Monitoring of immune activation using biochemical changes in a porcine model of cardiac arrest. Mediat. Inflamm 2001, 10, 343–346. [Google Scholar]
- Fujioka, H.; Shintaku, H.; Nakanishi, H.; Kim, T.J.; Kusuda, S.; Yamano, T. Biopterin in the acute phase of hypoxia-ischemia in a neonatal pig model. Brain Develop 2008, 30, 1–6. [Google Scholar]
- Carru, C.; Zinellu, A.; Sotgia, S.; Serra, R.; Usai, M.F.; Pintus, G.F.; Pes, G.M.; Deiana, L. A new HPLC methods for serum neopterin measurement and relationship with plasma thiols in healthy subjects. Biomed. Chromatogr 2004, 18, 360–366. [Google Scholar]
| Fe (ng·mL−1) | ||||
|---|---|---|---|---|
| 0 h | 1 h | 4 h | 24 h | |
| Control | 26.02 ± 7.21 | 25.38 ± 9.10 | 28.15 ± 8.40 | 22.92 ± 6.31 |
| Fe3+-dextran group | 29.93 ± 6.26 | 144.30 ± 44.2 ** | 324.95 ± 59.80 ** | 331.03 ± 44.82 ** |
| 0 h | 1 h | 4 h | 24 h | p | ||
|---|---|---|---|---|---|---|
| Leukocytes (109/L) | Control | 18.6 ± 2.9 | 21.1 ± 4.9 | 21.2 ± 4.0 | 18.8 ± 4.4 | 0.05 |
| Fe3+-dextran | 17.9 ± 2.9 | 20.9 ± 5.5 | 21.3 ± 7.6 | 22.8 ± 8.9 | 0.05 | |
| Neutrophils (%) | Control | 38.9 ± 19.9 | 59.8 ± 14.4 | 56.2 ± 16.8 | 46.8 ± 9.9 | 0.05 |
| Fe3+-dextran | 42.1 ± 11.8 | 60.7 ± 9.0 | 64.0 ± 12.1 | 56.7 ± 13.0 | 0.05 | |
| Lymphocytes (%) | Control | 55.2 ± 13.6 | 35.8 ± 13.2 | 41.6 ± 16.8 | 46.7 ± 11.1 | 0.05 |
| Fe3+-dextran | 52.4 ± 13.2 | 35.7 ± 9.1 | 33.3 ± 14.6 | 38.9 ± 15.0 | 0.05 | |
| Monocytes (%) | Control | 4.5 ± 3.3 | 4.3 ± 3.8 | 1.9 ± 2.2 | 4.8 ± 5.1 | 0.05 |
| Fe3+-dextran | 4.5 ± 2.4 | 2.8 ± 1.8 | 2.4 ± 2.6 | 3.7 ± 2.7 | 0.05 | |
| Eosinophils (%) | Control | 1.1 ± 0.8 | 0.1 ± 0.3 | 0.3 ± 0.6 | 1.8 ± 1.7 | 0.05 |
| Fe3+-dextran | 0.9 ± 0.9 | 0.6 ± 1.0 | 0.2 ± 0.6 | 0.5 ± 1.0 | 0.05 | |
| N/L ratio | Control | 0.70 | 1.67 | 1.35 | 1.00 | 0.05 |
| Fe3+-dextran | 0.80 | 1.70 | 1.92 | 1.46 | 0.05 | |
| Sampling | Cortisol (nmol·L−1) | |
|---|---|---|
| Control group | Fe3+-dextran group | |
| 0 h | 164. 29 ± 62.00 | 218.14 ± 80.59 |
| 24 h | 502.39 ± 89.35 ** | 501.56 ± 157.77 ** |
©2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/)
Share and Cite
Smutna, M.; Svoboda, M.; Breinekova, K. Pterins as Sensors of Response to the Application of Fe3+-Dextran in Piglets. Sensors 2010, 10, 890-900. https://doi.org/10.3390/s100100890
Smutna M, Svoboda M, Breinekova K. Pterins as Sensors of Response to the Application of Fe3+-Dextran in Piglets. Sensors. 2010; 10(1):890-900. https://doi.org/10.3390/s100100890
Chicago/Turabian StyleSmutna, Miriam, Martin Svoboda, and Klara Breinekova. 2010. "Pterins as Sensors of Response to the Application of Fe3+-Dextran in Piglets" Sensors 10, no. 1: 890-900. https://doi.org/10.3390/s100100890
APA StyleSmutna, M., Svoboda, M., & Breinekova, K. (2010). Pterins as Sensors of Response to the Application of Fe3+-Dextran in Piglets. Sensors, 10(1), 890-900. https://doi.org/10.3390/s100100890




