Ethyl (E)-4-(2,4-Dimethoxyphenyl)-6-(2,4-dimethoxystyryl)-2-oxo-1,2,3,4-tetrahydropyrimidine-5-carboxylate
Abstract
:1. Introduction
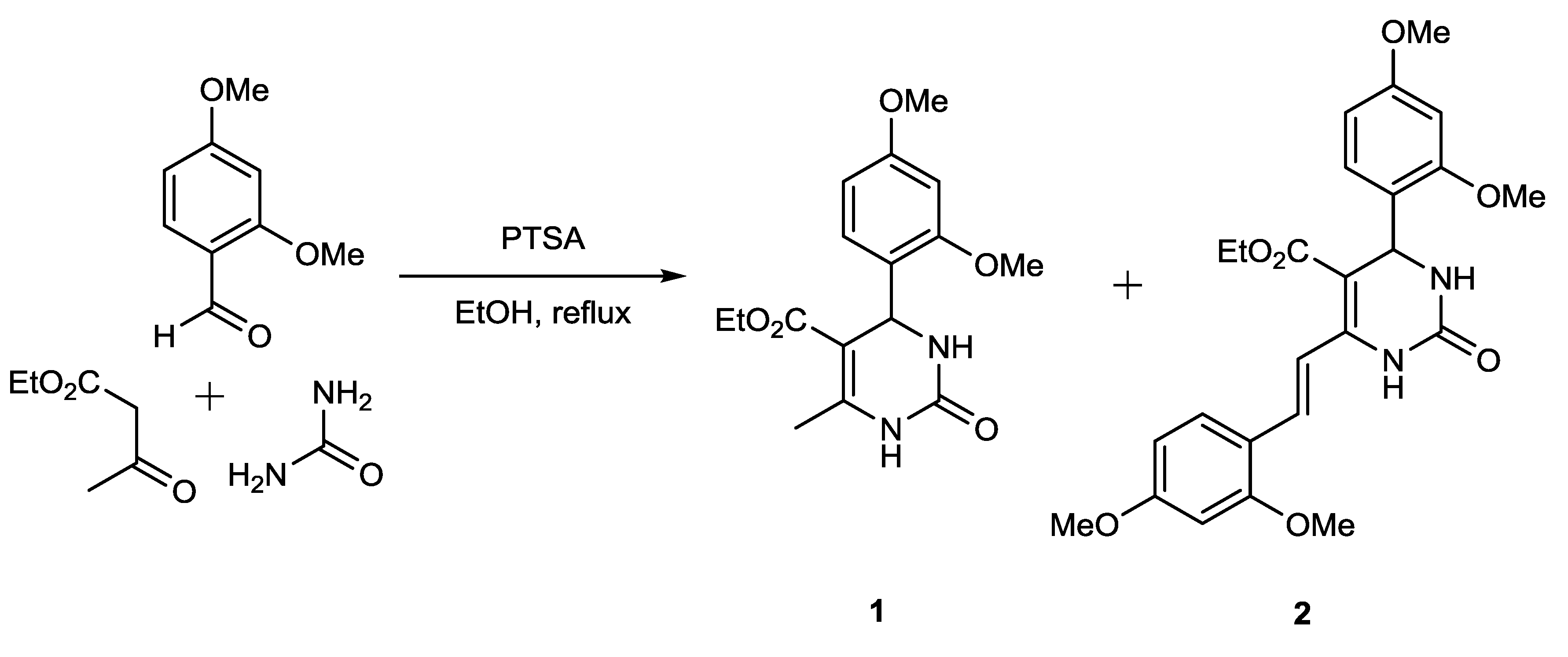
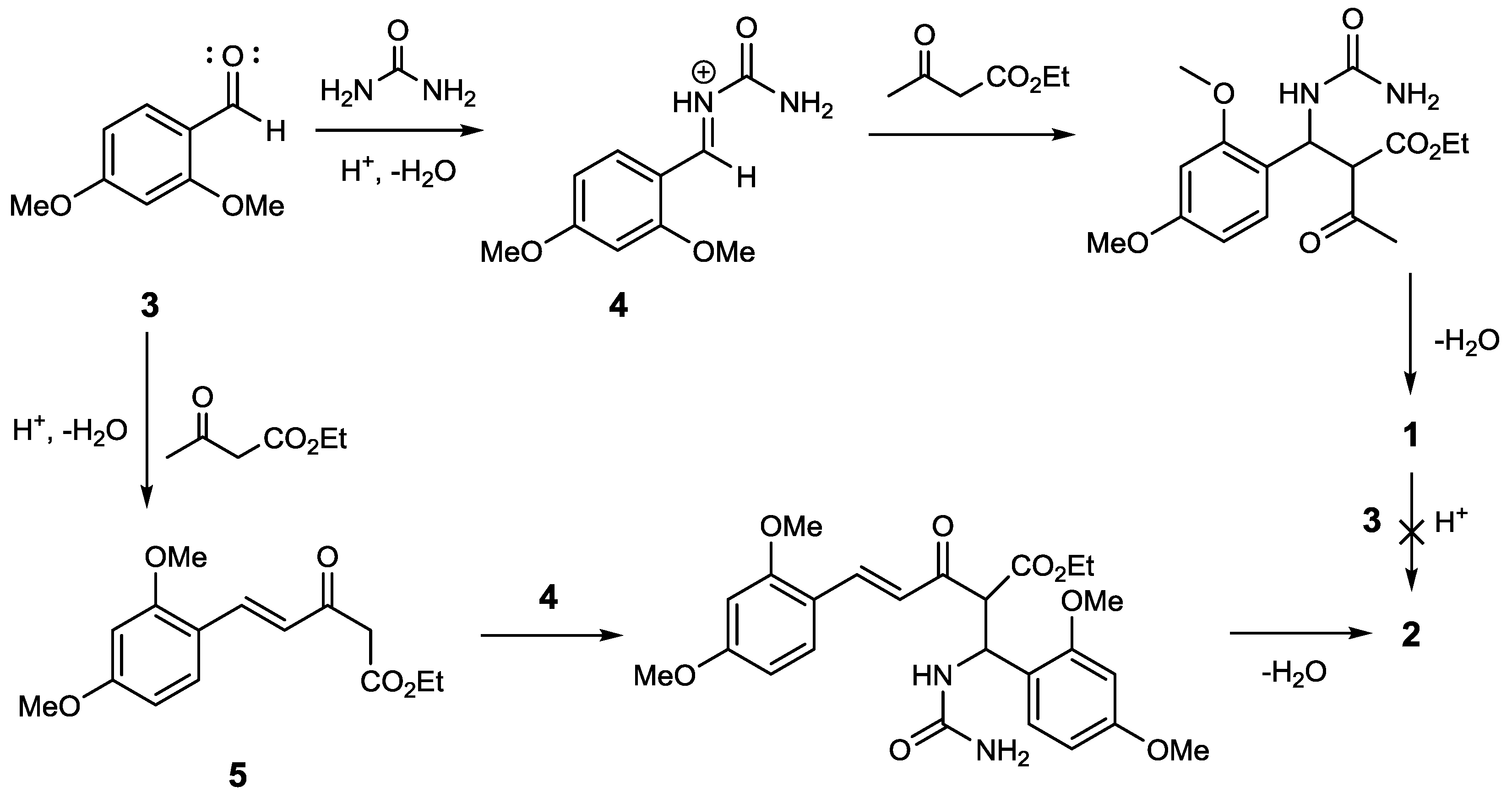
2. Results and Discussion
3. Materials and Methods
3.1. General
3.2. Synthesis of Compound 2
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kappe, C.O.; Stadler, A. The Biginelli dihydropyrimidine synthesis. Org. React. 2014, 63, 1–116. [Google Scholar]
- Kappe, C.O. Recent advances in the Biginelli dihydropyrimidine synthesis. New tricks from an old dog. Acc. Chem. Res. 2000, 33, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Byk, G.; Gottlieb, H.E.; Herscovici, J.; Mirkin, F. New regioselective multicomponent reaction: One pot synthesis of spiro heterobicyclic aliphatic rings. J. Comb. Chem. 2000, 2, 732–735. [Google Scholar] [CrossRef] [PubMed]
- Abelman, M.M.; Smith, S.C.; James, D.R. Cyclic ketones and substituted α-keto acids as alternative substrates for novel Biginelli-like scaffold syntheses. Tetrahedron Lett. 2003, 44, 4559–4562. [Google Scholar] [CrossRef]
- Lei, M.; Ma, L.; Hu, L. An efficient and environmentally friendly procedure for synthesis of pyrimidinone derivatives by use of a Biginelli-type reaction. Monatshefte fur Chemie 2010, 141, 1005–1008. [Google Scholar] [CrossRef]
- Zhang, H.; Zhou, Z.; Yao, Z.; Xu, F.; Shen, Q. Efficient synthesis of pyrimidinone derivatives by ytterbium chloride catalyzed Biginelli-type reaction under solvent-free conditions. Tetrahedron Lett. 2009, 50, 1622–1624. [Google Scholar] [CrossRef]
- Hajipour, A.R.; Ghayeb, Y.; Sheikhan, N.; Ruoho, A.E. Brønsted acidic ionic liquid as an efficient and reusable catalyst for one-pot, three-component synthesis of pyrimidinone derivatives via Biginelli-type reaction under solvent-free conditions. Synth. Commun. 2011, 41, 2226–2233. [Google Scholar] [CrossRef]
- Amoozadeh, A.; Rahmani, S.; Nemati, F. Poly(ethylene)glycol/AlCl3 as a new and efficient system for multicomponent Biginelli-type synthesis of pyrimidinone derivatives. Heterocycl. Commun. 2013, 19, 69–73. [Google Scholar] [CrossRef]
- Zhu, Y.; Huang, S.; Pan, Y. Highly chemoselective multicomponent Biginelli-type condensations of cycloalkanones, urea or thiourea and aldehydes. Eur. J. Org. Chem. 2005, 2005, 2354–2367. [Google Scholar] [CrossRef]
- Wan, Y.; Yuan, R.; Xu, H.; Wang, C.; Qi, J.; Wu, H. A regioselective Biginelli-like reaction controlled by the size of alicyclic mono-ketones. J. Heterocycl. Chem. 2014, 51, E123–E128. [Google Scholar] [CrossRef]
- Ghashang, M.; Mansoor, S.S.; Aswin, K. An efficient and environmentally friendly procedure for the synthesis of some novel 8-benzylidene-4-phenyl-3,4,5,6,7,8-hexahydro-1H-quinazolin-2-ones/thiones using tetrabutylammonium hexatungstate as a reusable heterogeneous catalyst under solvent-free conditions. Bull. Korean Chem. Soc. 2013, 34, 3289–3294. [Google Scholar]
- Beşoluk, Ş.; Kucukislamoglu, M.; Zengin, M.; Arslan, M.; Nebioǧlu, M. An efficient one-pot synthesis of dihydropyrimidinones catalyzed by zirconium hydrogen phosphate under solvent-free conditions. Turk. J. Chem. 2010, 34, 411–416. [Google Scholar]
- Jin, T.; Zhang, S.; Li, T. p-Toluenesulfonic acid-catalyzed efficient synthesis of dihydropyrimidines: Improved high yielding protocol for the Biginelli reaction. Synth. Commun. 2002, 32, 1847–1851. [Google Scholar] [CrossRef]
- Bose, A.K.; Pednekar, S.; Ganguly, S.N.; Manhas, M.S. A simplified green chemistry approach to the Biginelli reaction using “Grindstone Chemistry”. Tetrahedron Lett. 2004, 45, 8351–8353. [Google Scholar] [CrossRef]
- Chen, Q.; Liu, Q.; Wang, H. Methyl 6-methyl-1-(4-methylphenyl)-2-oxo-4-phenyl-1,2,3,4-tetrahydropyrimidine-5-carboxylate. Molbank 2012, 2012, M752. [Google Scholar] [CrossRef]
- An, L.; Han, L.; Wang, Z.; Huang, T.; Zhu, H. Calix[8]arene sulfonic acid catalyzed three-component reaction for convenient synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones under ultrasonic irradiation. Biol. Pharm. Bull. 2016, 39, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, L.; Duan, X.; Yan, X.; Yan, Y.; Liu, Q.; Liu, T.; Zhang, G. Iron-catalyzed four-member multicomponent reaction for assembly of (E)-6-arylvinyl-3,4-dihydropyrimidin-2(1H)-ones. Tetrahedron 2015, 71, 7745–7751. [Google Scholar] [CrossRef]
- Bonne, D.; Coquerel, Y.; Constantieux, T.; Rodriguez, J. 1,3-Dicarbonyl compounds in stereoselective domino and multicomponent reactions. Tetrahedron Asymmetry 2010, 21, 1085–1109. [Google Scholar] [CrossRef]
- Jin, Y.; Roberts, F.G.; Coates, R.M. Stereoselective isoprenoid chain extension with acetoacetate dianion: (E,E,E)-geranylgeraniol from (E,E)-farnesol. Org. Synth. 2007, 84, 43–57. [Google Scholar]
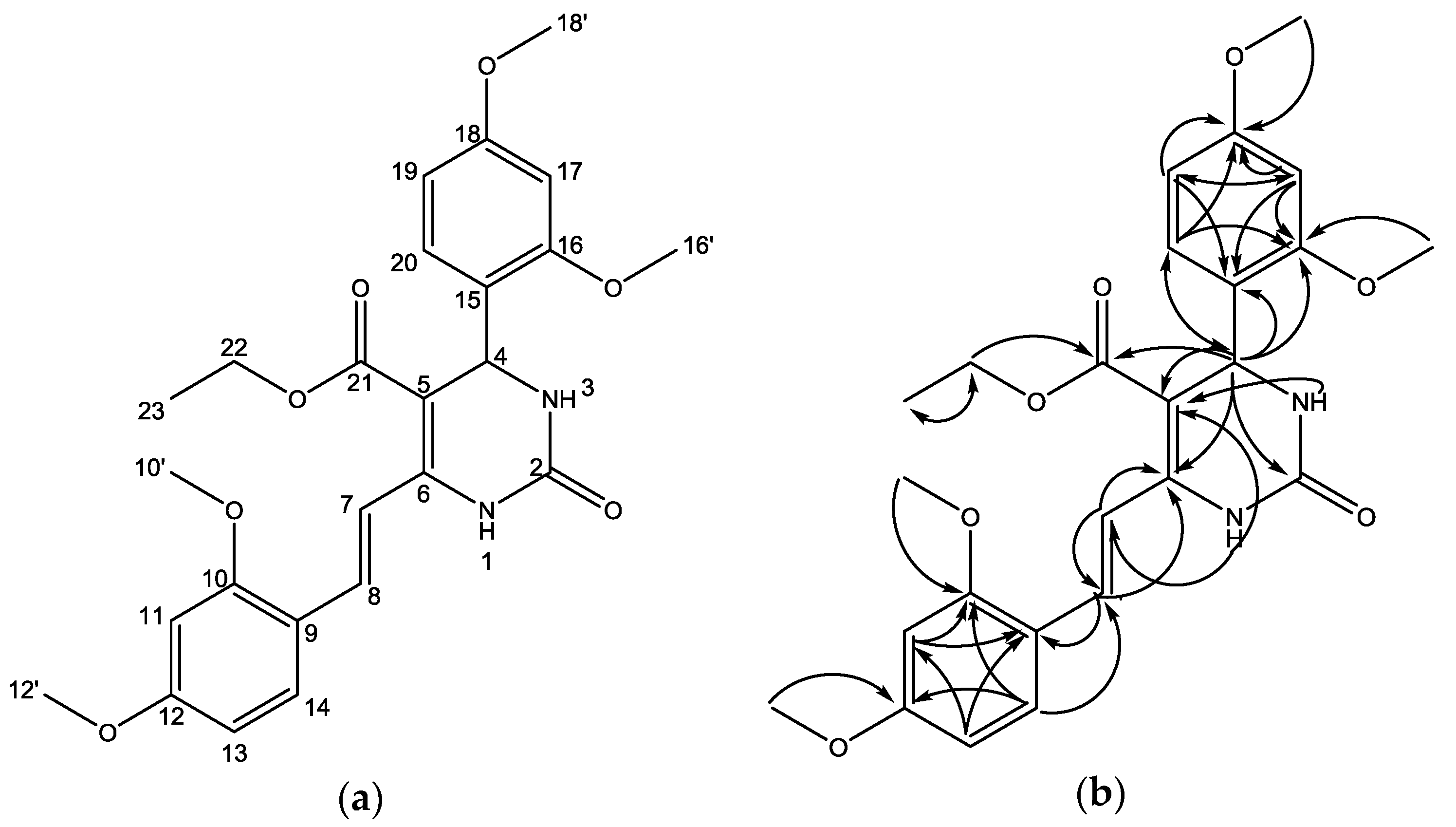
| No. Atom | δH (mult, J Hz) | δC (ppm) | HMBC |
|---|---|---|---|
| 1 | 6.83 (s, 1H) | C-5, C-7 | |
| 2 | 153.1 | ||
| 3 | 5.67 (s, 1H) | C-5 | |
| 4 | 5.73 (d, J = 2.8 Hz, 1H) | 50.0 | C-2, C-5, C-6, C-15, C-16, C-20, C-21 |
| 5 | 99.9 | ||
| 6 | 145.2 | ||
| 7 | 7.30 (d, J = 17.0 Hz, 1H) | 117.4 | C-6, C-8, C-9 |
| 8 | 8.09 (d, J = 17.0 Hz, 1H) | 127.7 | C-5, C-6, C-9 |
| 9 | 117.6 | ||
| 10 | 162.2 | ||
| 10′ | 3.83 (s, 3H) | 55.5–55.6 | C-10 |
| 11 | 6.44 (d, J = 2.3 Hz, 1H) | 98.9 | C-9, C-10 |
| 12 | 158.9 | ||
| 12′ | 3.85 (s, 3H) | 55.5–55.6 | C-12 |
| 13 | 6.51 (dd, J = 8.6, 2.3 Hz, 1H) | 105.6 | C-9, C-11 |
| 14 | 7.59 (d, J = 8.6 Hz, 1H) | 128.2 | C-8, C-10, C-12 |
| 15 | 122.4 | ||
| 16 | 157.9 | ||
| 16′ | 3.85 (s, 3H) | 55.5–55.6 | C-16 |
| 17 | 5.46 (d, 1H) | 98.4 | C-15, C-16, C-18, C-19 |
| 18 | 160.7 | ||
| 18′ | 3.77 (s, 3H) | 55.5–55.6 | C-18 |
| 19 | 6.36 (dd, J = 8.4, 2.2 Hz, 1H) | 103.9 | C-15, C-17, C-18 |
| 20 | 7.01 (d, J = 8.4 Hz, 1H) | 127.5 | C-4, C-16, C-18 |
| 21 | 165.8 | ||
| 22 | 4.09 (m, 2H) | 60.2 | C-21, C-23 |
| 23 | 1.14 (t, J = 7.1 Hz, 3H) | 14.3 | C-22 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suwito, H.; Zulianto, L.; Ul Haq, K.; Erwanto, E.; Abdulloh, A.; Novi Kristanti, A.; Indriani, I. Ethyl (E)-4-(2,4-Dimethoxyphenyl)-6-(2,4-dimethoxystyryl)-2-oxo-1,2,3,4-tetrahydropyrimidine-5-carboxylate. Molbank 2017, 2017, M946. https://doi.org/10.3390/M946
Suwito H, Zulianto L, Ul Haq K, Erwanto E, Abdulloh A, Novi Kristanti A, Indriani I. Ethyl (E)-4-(2,4-Dimethoxyphenyl)-6-(2,4-dimethoxystyryl)-2-oxo-1,2,3,4-tetrahydropyrimidine-5-carboxylate. Molbank. 2017; 2017(3):M946. https://doi.org/10.3390/M946
Chicago/Turabian StyleSuwito, Hery, Lutfan Zulianto, Kautsar Ul Haq, Erwanto Erwanto, Abdulloh Abdulloh, Alfinda Novi Kristanti, and Indriani Indriani. 2017. "Ethyl (E)-4-(2,4-Dimethoxyphenyl)-6-(2,4-dimethoxystyryl)-2-oxo-1,2,3,4-tetrahydropyrimidine-5-carboxylate" Molbank 2017, no. 3: M946. https://doi.org/10.3390/M946
APA StyleSuwito, H., Zulianto, L., Ul Haq, K., Erwanto, E., Abdulloh, A., Novi Kristanti, A., & Indriani, I. (2017). Ethyl (E)-4-(2,4-Dimethoxyphenyl)-6-(2,4-dimethoxystyryl)-2-oxo-1,2,3,4-tetrahydropyrimidine-5-carboxylate. Molbank, 2017(3), M946. https://doi.org/10.3390/M946




