Shear Stress Transmission Model for the Flagellar Rotary Motor
Abstract
:1. Introduction
2. Objects to be discussed
2.1. Structure of the flagellar rotary motor
2.2. Major experimental results to be explained by the model
- (1)
- One revolution of the flagellar rotation consists of a constant number of steps at low speed [1].
- (2)
- The flagellar rotation velocity ω is proportional to the transmembrane potential difference at low speed [1].
- (3a)
- (3b)
- (3c)
- (3d)
- (4)
- There are experimental observations that the flagellar rotate in the same direction when the direction of the proton passage is reversed (for references, cf. Section 5.2).
- (5)
- The cell produces constant torque to rotate a flagellum even when the cell is rotated relative to the flagellum by external forces (for references, cf. Section 5.3).
- (6)
- The cell has a switch that reverses the sense of the flagelllar rotation with the same absolute value of torque for chemotaxis (for references, cf. Section 5.4).
3. Basic ideas of the new model
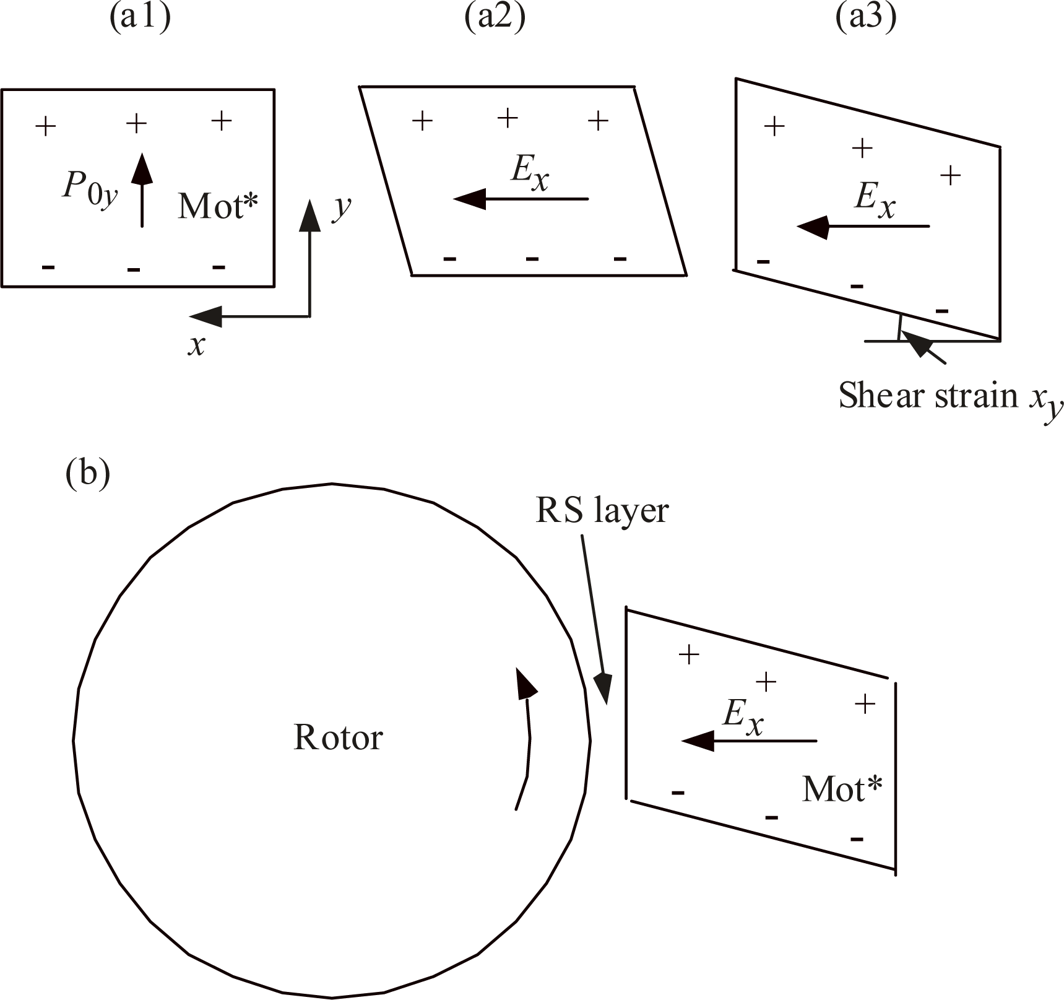
3.1. Mot* is assumed to be a shear force generator
3.2. The RS layer is viscoelastic
3.3. Expression for the shear force transmitted into the RS layer
4. Theoretical predictions on flagellar rotation
4.1. Flagellar rotation velocity and torque
4.1.a. Step size of Rotor rotation
4.1.b. Rotation velocity as a function of the transmembrane potential difference
4.1.c. Energy balance and critical rotation velocity
4.1.d. Torque as a function of rotation velocity
4.1.e. On the effect of temperature
- (3b) ωη does not depend upon cRS according to (32), (34).
- (3c) ωcr is inversely proportional to cRS according to (34).
- (3d) ω* and ω*η are proportional to 1/cRS1/2 and hence decreases with increasing cRS according to (32), (44).
4.1.f. On the energy efficiency
4.2. Effect of reversal of proton passage direction
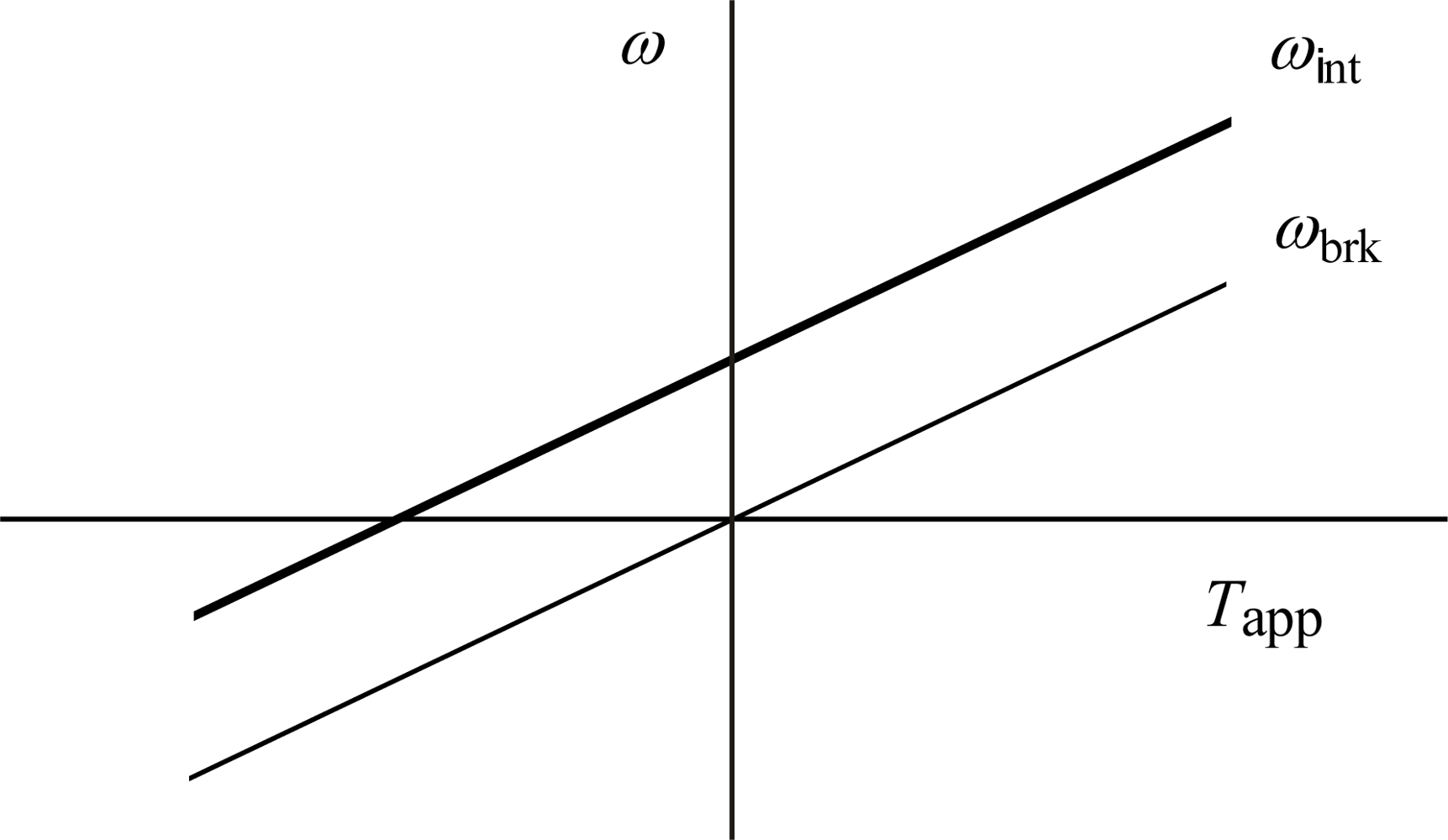
4.3. Effect of externally applied torque on rotor
4.4. Switch mechanism for changing rotation direction
5. Comparison of the theoretical predictions with experimental observations
5.1. The flagellar rotation velocity and torque
5.1.a. Number of rotation steps per one revolution
5.1.b. Rotation velocity as a function of the transmembrane potential difference
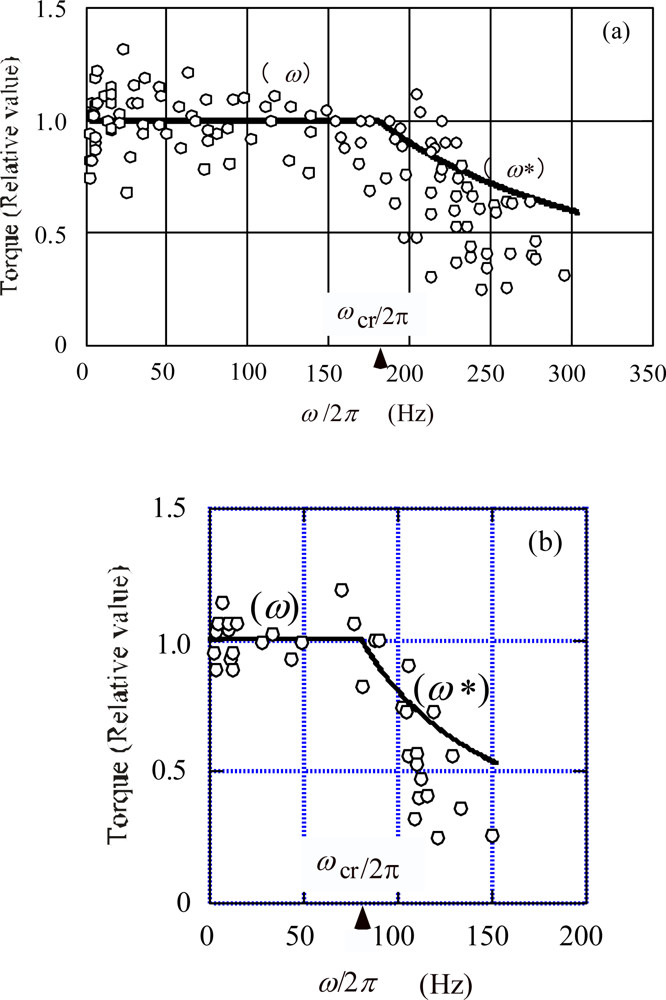
5.1.c. Torque as a function of rotation velocity
5.1.d. On the effect of temperature
- (3b) When ω <ωcr, the torque varies little with temperature because ωη does not depend upon cRS, in agreement with the observation (3b) cited in Section 2.2.
- (3c) The critical velocity ωcr decreases at lower temperatures because ωcr is inversely proportional to cRS, in agreement with the experimental results cited in Figure 8 (a) and (b).
- (3d) When ω <ωcr, the torque declines more steeply at lower temperature because ω*η is proportional to 1/cRS1/2, in agreement with the experimental results cited in Figures 8 (a) and (b).
5.2. Effect of reversal of proton passage direction
5.3. Effect of externally applied torque on rotor
5.4. Switch mechanism for changing rotation direction
6. Discussion
6.1. Summary and discussion
- (1)
- When the flagellar rotation velocity ω is smaller than the critical velocity ωcr , one revolution of the flagellar rotation consists of a constant number of rotation steps as proved at the end of Section 5.1a.
- (2)
- (3a)
- As indicated by the lines and curves in Figure 8, the torque exerted on the flagella by the cell is independent of the flagellar rotation velocity ω and remains constant when ω < ωcr, and then sharply decreases above ωcr, in agreement with the experimental data.
- (3b)
- When ω< ωcr, the torque is expected to vary little with temperature, as discussed in Section 5.1c, in agreement with experimental observations.
- (3c)
- The critical velocity ωcr shifts to lower speeds at lower temperatures as discussed in Section 5.1c (cf. Figure 8(a) and (b)).
- (3d)
- When ω>ωcr, the torque declines more steeply at lower temperatures as discussed in Section 5.1c (cf. Figure 8(a) and (b)).
- (4)
- The model predicts that the flagella rotate in the same direction when direction of the proton passage is reversed, as discussed in Section 4.2 and 5.2 (cf. Figure 9).
- (5)
- The cell produces constant torque for the flagellum even when the cell is rotated relative to the flagellum by external forces, as discussed in Sects. 4.3 and 5.3 (cf. Figure 10).
- (6)
- It is possible that the cell reverses the sense of the flagelllar rotation with the same absolute value of velocity if the direction of P0y is changeable by chemical modification of Mot*, as discussed in Sects. 4. 4 and 5. 4.
6.2. Comparison of the flagellar motor with the FOF1-ATPase motor
Appendix
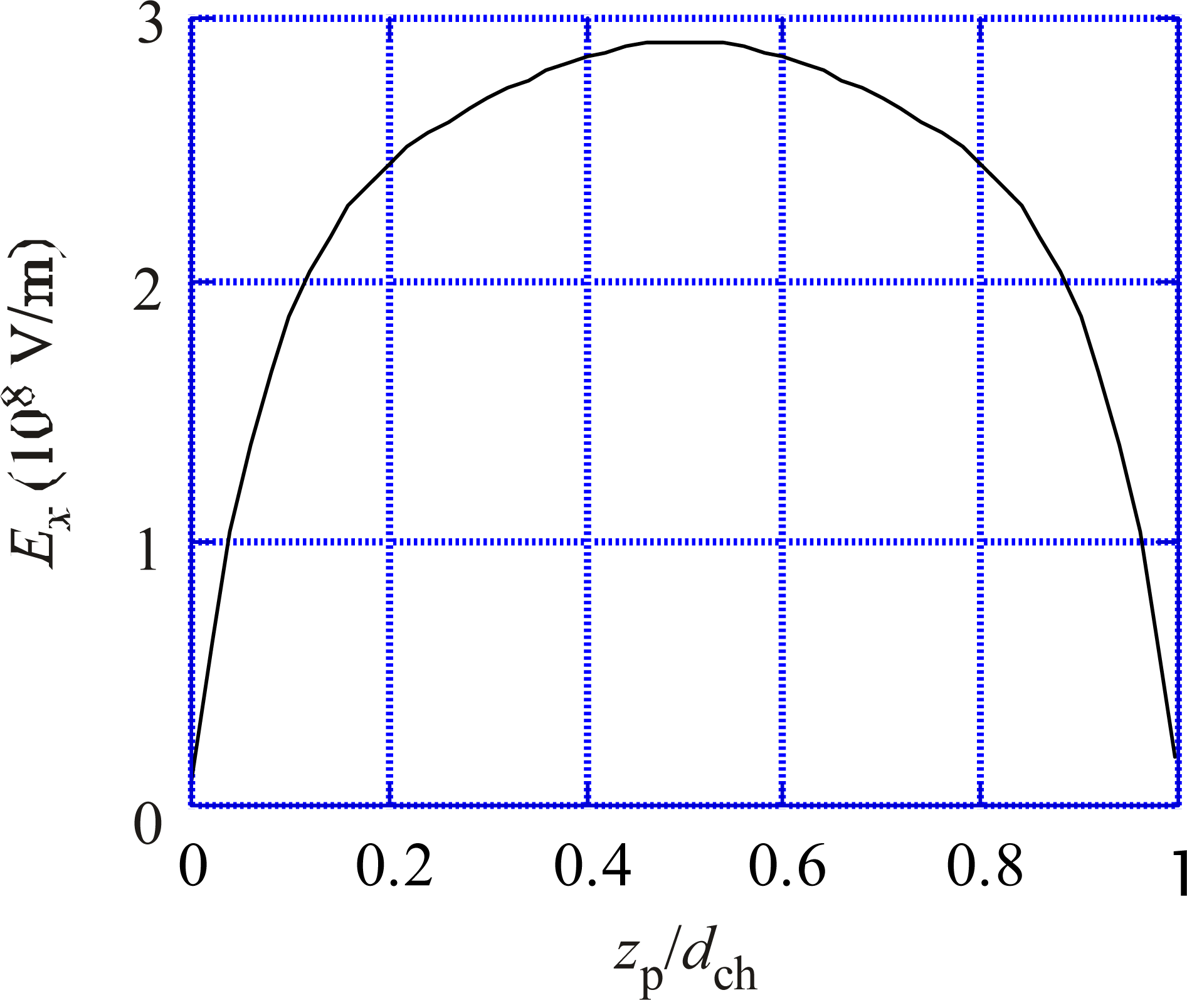
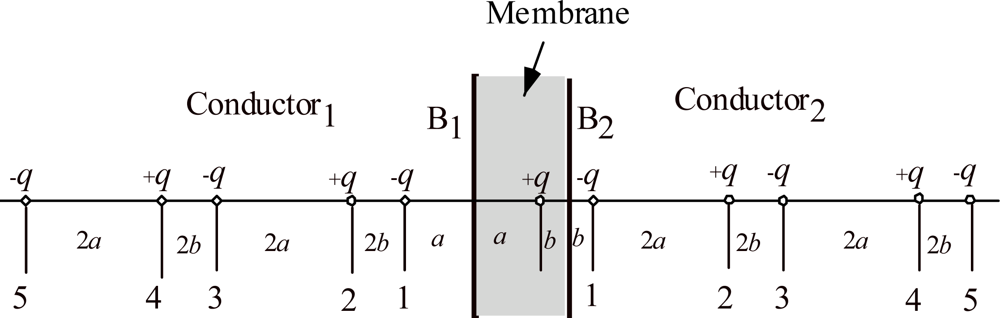
Calculation of the electric field produced by the proton pair in dielectric membrane
References
- Berg, HC. Constraints on models for the flagellar rotary motor. Phil. Trans. R. Soc. Lond. B 2000, 355, 491–501. [Google Scholar]
- Mitsui, T; Ohshima, H. Proposed model for the flagellar rotary motor. Colloids Surfaces B 2005, 46, 32–44. [Google Scholar]
- Mitsui, T; Tatsuzaki, I; Nakamura, E. An Introduction to the Physics of Ferroelectrics; Translated from Japanese by; Ishibashi, Y; Tatsuzaki, I; Nakamura, E; Burfoot, JC, Translators; Gordon & Breach: New York, 1976. [Google Scholar]
- Xing, J; Bai, F; Berry, R; Oster, G. Torque-speed ralationship of the bacterial flagellar motor. Proc. Nat. Acad. Sci 2006, 103, 1260–1265. [Google Scholar]
- Blair, DF; Berg, HC. Restoration of torque in defective flagellar motor. Science 1988, 242, 1678–1681. [Google Scholar]
- Braun, TF; Blair, DF. Targeted disulfide cross-linking of the MotB protein of Escherichia coli: Evidense for two H+ channels in stator complex. Biochem 2001, 40, 13051–13059. [Google Scholar]
- Mitsui, T. Ferroelectrics and antiferroelectrics. In Springer Handbook of Condensed Matter and Material Data; Martienssen, W, Warlimont, H, Eds.; Springer-Verlag: Berlin, 2005; Chapter 4.5. [Google Scholar]
- Tanford, C. Physical Chemistry of Macromolecules; John Wiley & Sons: New York, 1961; p. 111. [Google Scholar]
- Oosawa, F; Asakura, S. Thermodynamics of the Polymerization of Protein. Academic Press: London, 1975; pp. 66–67. [Google Scholar]
- Blair, DF. Flagellar movement driven by proton translocation. FEBS Letters 2003, 545, 86–95. [Google Scholar]
- Landau, LD; Lifshitz, EM. Theory of Elasticity, Volume 7 of Course of Theoretical Physics; Translated from Russian by; Sykes, JB; Reid, WH, Translators; Pergamon Press: London, 1959. [Google Scholar]
- Mitsui, T; Nakamura, E; Gesi, K; Ikeda, T; Makita, Y; Marutake, M; Nomura, S; Sawaguchi, E; Shigenari, T; Shiozaki, Y; Tatsuzaki, I; Toyoda, K; Adachi, M; Harada, J; Shiosaki, T; Wakino, K; Yamada, T. Hellwege, K-H, Hellwege, AM, Eds.; Landolt-Boernstein Tables III/16b. In Ferroelectrics and Related Substances; Springer-Verlag: Berlin, 1982; p. 78. [Google Scholar]
- Langevin, D. Viscoelasticity of monolayers. In Encyclopedia of Surface and Colloid Science; Dekker: New York, 2002; pp. 5584–5599. [Google Scholar]
- Nakayama, H; Mitsui, T; Nishihara, M; Kito, M. Relation between growth temperature of E. coli and phase transition temperatures of its cytoplasmic and outer membranes. Biochim. Biophys. Acta 1980, 601, 1–10. [Google Scholar]
- Landau, LD; Lifshitz, EM. Fluid Mechanics. Volume 6 Course of Theoretical Physics. Translated from Russian by; Sykes, JB; Reid, WH, Translators; Pergamon Press: London, 1966. [Google Scholar]
- Ferry, JD. Viscoelastic Properties of Polymers; John Wiley & Sons, Inc: New York, 1960. [Google Scholar]
- Saito, N. The Physics of Polymers (in Japanese); Shokabou: Tokyo, 1976. [Google Scholar]
- Berg, HC; Turner, L. Torque generated by the flagellar motor of Escherichia coli. Biophys. J 1993, 65, 2201–2216. [Google Scholar]
- Berry, RM; Berg, HC. Torque generated by the bacterial flagellar motor close to stall. Biophys. J 1996, 71, 3501–3510. [Google Scholar]
- Berry, RM; Berg, HC. Absence of a barrier to backwards rotation of the bacterial flagellar motor demonstrated with optical tweezers. Proc. Natl. Acad. Sci. USA 1997, 94, 14433–14437. [Google Scholar]
- Jones, CJ; Aizawa, S. The bacterial flagellum and flagellar motor: Structure, assembly and function. Adv. Microb. Physiol 1991, 32, 109–172. [Google Scholar]
- Fung, DC; Berg, HC. Powering the flagellar motor of Escherichia coli with an external voltage source. Nature 1995, 375, 809–812. [Google Scholar]
- Chen, X; Berg, HC. Torque-speed relationship of the flagallar rotary motor of Escherichia coli. Biophys. J 2000, 78, 1036–1041. [Google Scholar]
- Manson, MD; Tedesco, PM; Berg, HC. Energetics of flagellar rotation in bacteria. J. Mol. Biol 1980, 138, 541–561. [Google Scholar]
- Berg, HC; Manson, MD; Conley, MP. Dynamics and energetics of flagellar rotation in bacteria. Symp. Soc. exp. Biol 1982, 35, 1–31. [Google Scholar]
- Mitsui, T; Ohshima, H. Remarks on muscle contraction mechanism. Int. J. Mol. Sci 2008, 9, 872–904. [Google Scholar]
- Sowa, Y; Rowe, AD; Leak, MC; Yasushi, T; Homma, M; Ishijima, A; Berry, RM. Direct observation of steps in rotation of the bacterial flagella motor. Nature 2005, 437, 916–919. [Google Scholar]
- Yoshida, M; Muneyuki, E; Hisabori, T. ATP synthase - A marvelous rotary engine of the cell. Nature Reviews 2001, 2, 1–9. [Google Scholar]
- Abrahams, JP; Leslie, AGW; Lutter, R; Walker, JE. Structure at 2.8 Ǻ resolution of F1-ATPase from bovine heart mitochondria. Nature 1994, 370, 621–628. [Google Scholar]
- Stock, D; Leslie, AGW; Walker, JE. Molecular architecture of the rotary motor in ATP synthase. Science 1999, 286, 1700–1705. [Google Scholar]
- Weber, J; Senior, AE. ATP synthesis driven by proton transport in F1F0-ATP synthase. FEBS Letters 2003, 545, 61–70. [Google Scholar]
- Jackson, JD. Classical Electrodynamics, 1st Edtion ed; John Wiley & Sons: New York, 1962. [Google Scholar]






© 2008 by MDPI This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mitsui, T.; Ohshima, H. Shear Stress Transmission Model for the Flagellar Rotary Motor. Int. J. Mol. Sci. 2008, 9, 1595-1620. https://doi.org/10.3390/ijms9091595
Mitsui T, Ohshima H. Shear Stress Transmission Model for the Flagellar Rotary Motor. International Journal of Molecular Sciences. 2008; 9(9):1595-1620. https://doi.org/10.3390/ijms9091595
Chicago/Turabian StyleMitsui, Toshio, and Hiroyuki Ohshima. 2008. "Shear Stress Transmission Model for the Flagellar Rotary Motor" International Journal of Molecular Sciences 9, no. 9: 1595-1620. https://doi.org/10.3390/ijms9091595




