Broad-Spectrum Drugs Against Viral Agents
Abstract
:1. Introduction
2. TLR3 agonists: dsRNA
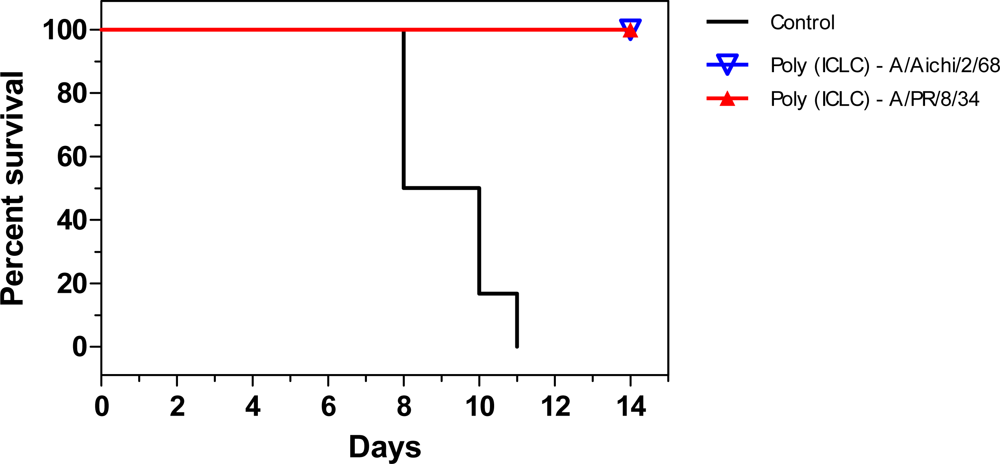
2.1. Poly (ICLC) as prophylaxis
2.2. Poly (ICLC) as an adjuvant
2.3. Poly (ICLC) safety
3. TLR9 agonists: CpG ODNs
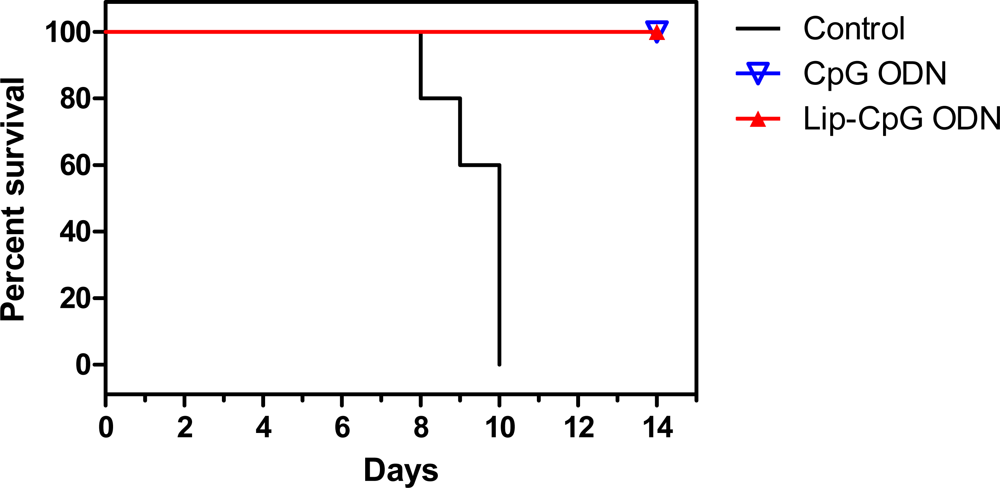
3.1. CpG ODN as prophylaxis
3.2. CpG ODN as an adjuvant
3.3 CpG ODN safety
4. TLR7 agonists
5. Influenza A H5N1: cytokine response
6. Influenza A H5N1: use of non-specific immune stimulators
6. Conclusions
References
- Wong, SS; Yuen, KY. Avian influenza virus infections in humans. Chest 2006, 129, 156–168. [Google Scholar]
- Li, S; Perdue, ML; Patzer, E. Seed viruses containing novel avian HA and NA antigens for prevention against potential influenza pandemic. Dev. Biol. (Basel) 2002, 110, 135–141. [Google Scholar]
- Brooks, MJ; Sasadeusz, JJ; Tannock, GA. Antiviral chemotherapeutic agents against respiratory viruses: where are we now and what’s in the pipeline? Curr. Opin. Pulm. Med 2004, 10, 197–203. [Google Scholar]
- Matrosovich, MN; Matrosovich, TY; Gray, T; Roberts, NA; Klenk, HD. Neuraminidase is important for the initiation of influenza virus infection in human airway epithelium. J. Virol 2004, 78, 12665–12667. [Google Scholar]
- WHO. http://www.who.int/csr/disease/avian_influenza/en/, accessed 25 Feb 2008.
- de Jong, MD; Tran, TT; Truong, HK; Vo, MH; Smith, GJ; Nguyen, VC; Bach, VC; Phan, TQ; Do, QH; Guan, Y; et al. Oseltamivir resistance during treatment of influenza A (H5N1) infection. N. Engl. J. Med 2005, 353, 2667–2672. [Google Scholar]
- Le, QM; Kiso, M; Someya, K; Sakai, YT; Nguyen, TH; Nguyen, KH; Pham, ND; Ngyen, HH; Yamada, S; Muramoto, Y; et al. Avian flu: isolation of drug–resistant H5N1 virus. Nature 2005, 437, 1108. [Google Scholar]
- Bright, RA; Medina, MJ; Xu, X; Perez-Oronoz, G; Wallis, TR; Davis, XM; Povinelli, L; Cox, NJ; Klimov, AI. Incidence of adamantane resistance among influenza A (H3N2) viruses isolated worldwide from 1994 to 2005: a cause for concern. Lancet 2005, 366, 1175–1181. [Google Scholar]
- Ashkar, AA; Bauer, S; Mitchell, WJ; Vieira, J; Rosenthal, KL. Local delivery of CpG oligodeoxynucleotides induces rapid changes in the genital mucosa and inhibits replication, but not entry, of herpes simplex virus type 2. J. Virol 2003, 77, 8948–8956. [Google Scholar]
- Hemmi, H; Takeuchi, O; Kawai, T; Kaisho, T; Sato, S; Sanjo, H; Matsumoto, M; Hoshino, K; Wagner, H; Takeda, K; et al. A Toll-like receptor recognizes bacterial DNA. Nature 2000, 408, 740–745. [Google Scholar]
- McCluskie, MJ; Weeratna, RD. Novel adjuvant systems. Curr. Drug Targets Infect. Disord 2001, 1, 263–271. [Google Scholar]
- Krieg, AM. CpG motifs in bacterial DNA and their immune effects. Annu. Rev. Immunol 2002, 20, 709–760. [Google Scholar]
- Vollmer, J; Weeratna, R; Payette, P; Jurk, M; Schetter, C; Laucht, M; Wader, T; Tluk, S; Liu, M; Davis, HL; et al. Characterization of three CpG oligodeoxynucleotide classes with distinct immunostimulatory activities. Eur. J. Immunol 2004, 34, 251–262. [Google Scholar]
- Lund, JM; Alexopoulou, L; Sato, A; Karow, M; Adams, NC; Gale, NW; Iwasaki, A; Flavell, RA. Recognition of single-stranded RNA viruses by Toll-like receptor 7. Proc. Natl. Acad. Sci. USA 2004, 101, 5598–5603. [Google Scholar]
- Kambayashi, T; Assarsson, E; Lukacher, AE; Ljunggren, HG; Jensen, PE. Memory CD8+ T cells provide an early source of IFN-gamma. J. Immunol 2003, 170, 2399–2408. [Google Scholar]
- Weber, F; Wagner, V; Rasmussen, SB; Hartmann, R; Paludan, SR. Double-stranded RNA is produced by positive-strand RNA viruses and DNA viruses but not in detectable amounts by negative-strand RNA viruses. J. Virol 2006, 80, 5059–5064. [Google Scholar]
- Guillot, L; Le Goffic, R; Bloch, S; Escriou, N; Akira, S; Chignard, M; Si-Tahar, M. Involvement of toll-like receptor 3 in the immune response of lung epithelial cells to double-stranded RNA and influenza A virus. J. Biol. Chem 2005, 280, 5571–5580. [Google Scholar]
- Chan, MC; Cheung, CY; Chui, WH; Tsao, SW; Nicholls, JM; Chan, YO; Chan, RW; Long, HT; Poon, LL; Guan, Y; et al. Proinflammatory cytokine responses induced by influenza A (H5N1) viruses in primary human alveolar and bronchial epithelial cells. Respir. Res 2005, 6, 135–147. [Google Scholar]
- Sivori, S; Falco, M; Della Chiesa, M; Carlomagno, S; Vitale, M; Moretta, L; Moretta, A. CpG and double-stranded RNA trigger human NK cells by Toll-like receptors: induction of cytokine release and cytotoxicity against tumors and dendritic cells. Proc. Natl. Acad. Sci. USA 2004, 101, 10116–10121. [Google Scholar]
- Miller, JL; Anders, EM. Virus-cell interactions in the induction of type 1 interferon by influenza virus in mouse spleen cells. J. Gen. Virol 2003, 84, (Pt 1). 193–202. [Google Scholar]
- Kulka, M; Alexopoulou, L; Flavell, RA; Metcalfe, DD. Activation of mast cells by double-stranded RNA: evidence for activation through Toll-like receptor 3. J. Allergy Clin. Immunol 2004, 114, 174–182. [Google Scholar]
- Fujita, H; Asahina, A; Mitsui, H; Tamaki, K. Langerhans cells exhibit low responsiveness to double-stranded RNA. Biochem. Biophys. Res. Commun 2004, 319, 832–839. [Google Scholar]
- Coccia, EM; Severa, M; Giacomini, E; Monneron, D; Remoli, ME; Julkunen, I; Cella, M; Lande, R; Uze, G. Viral infection and Toll-like receptor agonists induce a differential expression of type I and lambda interferons in human plasmacytoid and monocyte-derived dendritic cells. Eur. J. Immunol 2004, 34, 796–805. [Google Scholar]
- Levy, HB; Baer, G; Baron, S; Buckler, CE; Gibbs, CJ; Iadarola, MJ; London, WT; Rice, J. A modified polyriboinosinic-polyribocytidylic acid complex that induces interferon in primates. J. Infect. Dis 1975, 132, 434–439. [Google Scholar]
- Kende, M. Prophylactic and therapeutic efficacy of poly(I,C)-LC against Rift Valley fever virus infection in mice. J. Biol. Response Mod 1985, 4, 503–511. [Google Scholar]
- Wang, J; Matsukura, S; Watanabe, S; Adachi, M; Suzaki, H. Involvement of Toll-like receptors in the immune response of nasal polyp epithelial cells. Clin. Immunol 2007, 124, 345–352. [Google Scholar]
- Ieki, K; Matsukura, S; Kokubu, F; Kimura, T; Kuga, H; Kawaguchi, M; Odaka, M; Suzuki, S; Watanabe, S; Takeuchi, H; et al. Double-stranded RNA activates RANTES gene transcription through co-operation of nuclear factor-kappaB and interferon regulatory factors in human airway epithelial cells. Clin. Exp. Allergy 2004, 34, 745–752. [Google Scholar]
- Matsukura, S; Kokubu, F; Kurokawa, M; Kawaguchi, M; Ieki, K; Kuga, H; Odaka, M; Suzuki, S; Watanabe, S; Takeuchi, H; et al. Synthetic double-stranded RNA induces multiple genes related to inflammation through Toll-like receptor 3 depending on NF-kappaB and/or IRF-3 in airway epithelial cells. Clin. Exp. Allergy 2006, 36, 1049–1062. [Google Scholar]
- Auch, CJ; Saha, RN; Sheikh, FG; Liu, X; Jacobs, BL; Pahan, K. Role of protein kinase R in double-stranded RNA-induced expression of nitric oxide synthase in human astroglia. FEBS Lett 2004, 563, 223–228. [Google Scholar]
- Chirigos, MA; Welker, R; Schlick, E; Saito, T; Ruffmann, R. Vaccine adjuvant effects, and immune response, to synthetic polymers MVE and poly ICLC. Prog. Clin. Biol. Res 1984, 161, 467–479. [Google Scholar]
- Twilley, TA; Mason, L; Talmadge, JE; Wiltrout, RH. Increase in liver-associated natural killer activity by polyribonucleotides. Nat. Immun. Cell Growth Regul 1987, 6, 279–290. [Google Scholar]
- Wong, JP; Saravolac, EG; Sabuda, D; Levy, HB; Kende, M. Prophylactic and therapeutic efficacies of poly(IC.LC) against respiratory influenza A virus infection in mice. Antimicrob. Agents Chemother 1995, 39, 2574–2576. [Google Scholar]
- Matsukura, S; Kokubu, F; Kurokawa, M; Kawaguchi, M; Ieki, K; Kuga, H; Odaka, M; Suzuki, S; Watanabe, S; Homma, T; et al. Role of RIG-I, MDA-5, and PKR on the expression of inflammatory chemokines induced by synthetic dsRNA in airway epithelial cells. Int. Arch. Allergy Immunol 2007, 143(Suppl 1), 80–83. [Google Scholar]
- Marshall-Clarke, S; Downes, JE; Haga, IR; Bowie, AG; Borrow, P; Pennock, JL; Grencis, RK; Rothwell, P. Polyinosinic acid is a ligand for toll-like receptor 3. J. Biol. Chem 2007, 282, 24759–24766. [Google Scholar]
- Sakurai, M; Iigo, M; Sasaki, Y; Nakagawa, K; Fujiwara, Y; Tamura, T; Ohe, Y; Bungo, M; Saijo, N. Lack of correlation between interferon levels induced by polyribonucleotides and their antimetastatic effect. Oncology 1990, 47, 251–256. [Google Scholar]
- Bever, CT, Jr; McFarlin, DE; Levy, HB. A comparison of interferon responses to poly ICLC in males and females. J. Interferon Res 1985, 5, 423–428. [Google Scholar]
- Bever, CT, Jr; McFarlin, DE; Levy, HB. A comparison of interferon responses to poly ICLC in males and females. J. Interferon Res 1992, Spec No. 85–90. [Google Scholar]
- Sugiyama, T; Hoshino, K; Saito, M; Yano, T; Sasaki, I; Yamazaki, C; Akira, S; Kaisho, T. Immunoadjuvant effects of polyadenylic:polyuridylic acids through TLR3 and TLR7. Int. Immunol 2008, 20, 1–9. [Google Scholar]
- Baer, GM; Shaddock, JH; Moore, SA; Yager, PA; Baron, SS; Levy, HB. Successful prophylaxis against rabies in mice and Rhesus monkeys: the interferon system and vaccine. J. Infect. Dis 1977, 136, 286–291. [Google Scholar]
- Sidwell, RW; Huffman, JH; Barnard, DL; Smee, DF; Warren, RP; Chirigos, MA; Kende, M; Huggins, J. Antiviral and immunomodulating inhibitors of experimentally-induced Punta Toro virus infections. Antiviral Res 1994, 25, 105–122. [Google Scholar]
- Wong, JP; Nagata, LP; Christopher, ME; Salazar, AM; Dale, RM. Prophylaxis of acute respiratory virus infections using nucleic acid-based drugs. Vaccine 2005, 23, 2266–2268. [Google Scholar]
- Robalino, J; Browdy, CL; Prior, S; Metz, A; Parnell, P; Gross, P; Warr, G. Induction of antiviral immunity by double-stranded RNA in a marine invertebrate. J. Virol 2004, 78, 10442–10448. [Google Scholar]
- Stephen, EL; Sammons, ML; Pannier, WL; Baron, S; Spertzel, RO; Levy, HB. Effect of a nuclease-resistant derivative of polyriboinosinic-polyribocytidylic acid complex on yellow fever in rhesus monkeys (Macaca mulatta). J. Infect. Dis 1977, 136, 122–126. [Google Scholar]
- Stephen, EL; Hilmas, DE; Levy, HB; Spertzel, RO. Protective and toxic effects of a nuclease-resistant derivative of polyriboinosinic-polyribocytidylic acid on Venezuelan equine encephalomyelitis virus in rhesus monkeys. J. Infect. Dis 1979, 139, 267–272. [Google Scholar]
- Peres, A; Seemayer, TA; Lapp, WS. The effects of polyinosinic: polycytidylic acid (pI:C) on the GVH reaction: immunopathological observations. Clin. Immunol. Immunopathol 1986, 39, 102–111. [Google Scholar]
- Carter, WA; Strayer, D. U.S. Patent Application # 20,050,137,154, 2004.
- Wong, JP; Yang, H; Nagata, L; Kende, M; Levy, H; Schnell, G; Blasetti, K. Liposome-mediated immunotherapy against respiratory influenza virus infection using double-stranded RNA poly ICLC. Vaccine 1999, 17, 1788–1795. [Google Scholar]
- Kende, M. Prophylactic and therapeutic efficacy of poly(I,C)-LC against Rift Valley fever virus infection in mice. J. Biol. Response Mod 1985, 4, 503–511. [Google Scholar]
- Kende, M; Lupton, HW; Rill, WL; Gibbs, P; Levy, HB; Canonico, PG. Ranking of prophylactic efficacy of poly(ICLC) against Rift Valley fever virus infection in mice by incremental relative risk of death. Antimicrob. Agents Chemother 1987, 31, 1194–1198. [Google Scholar]
- Olsen, AL; Morrey, JD; Smee, DF; Sidwell, RW. Correlation between breakdown of the blood-brain barrier and disease outcome of viral encephalitis in mice. Antiviral Res 2007, 75, 104–112. [Google Scholar]
- Barnard, DL; Day, CW; Bailey, K; Heiner, M; Montgomery, R; Lauridsen, L; Chan, PK; Sidwell, RW. Evaluation of immunomodulators, interferons and known in vitro SARS-coV inhibitors for inhibition of SARS-coV replication in BALB/c mice. Antivir. Chem. Chemother 2006, 17, 275–284. [Google Scholar]
- Morrey, JD; Day, CW; Julander, JG; Blatt, LM; Smee, DF; Sidwell, RW. Effect of interferon-alpha and interferon-inducers on West Nile virus in mouse and hamster animal models. Antivir. Chem. Chemother 2004, 15, 101–109. [Google Scholar]
- Kende, M; Lupton, HW; Rill, WL; Levy, HB; Canonico, PG. Enhanced therapeutic efficacy of poly(ICLC) and ribavirin combinations against Rift Valley fever virus infection in mice. Antimicrob. Agents Chemother 1987, 31, 986–990. [Google Scholar]
- Rettenmaier, MA; Berman, ML; DiSaia, PJ. Treatment of advanced ovarian cancer with polyinosinic-polycytidylic lysine carboxymethylcellulose (poly(ICLC]. Gynecol. Oncol 1986, 24, 359–361. [Google Scholar]
- Ichinohe, T; Watanabe, I; Ito, S; Fujii, H; Moriyama, M; Tamura, S; Takahashi, H; Sawa, H; Chiba, J; Kurata, T; et al. Synthetic double-stranded RNA poly(I:C) combined with mucosal vaccine protects against influenza virus infection. J. Virol 2005, 79, 2910–2919. [Google Scholar]
- Ichinohe, T; Tamura, S; Kawaguchi, A; Ninomiya, A; Imai, M; Itamura, S; Odagiri, T; Tashiro, M; Takahashi, H; Sawa, H; et al. Cross-Protection against H5N1 Influenza Virus Infection Is Afforded by Intranasal Inoculation with Seasonal Trivalent Inactivated Influenza Vaccine. J. Infect. Dis 2007, 196, 1313–1320. [Google Scholar]
- Ichinohe, T; Kawaguchi, A; Tamura, S; Takahashi, H; Sawa, H; Ninomiya, A; Imai, M; Itamura, S; Odagiri, T; Tashiro, M; et al. Intranasal immunization with H5N1 vaccine plus Poly I:Poly C(12)U, a Toll-like receptor agonist, protects mice against homologous and heterologous virus challenge. Microbes Infect 2007, 9, 1333–1340. [Google Scholar]
- DeCicco, KL; Youngdahl, JD; Ross, AC. All-trans-retinoic acid and polyriboinosinic : Polyribocytidylic acid in combination potentiate specific antibody production and cell-mediated immunity. Immunology 2001, 104, 341–348. [Google Scholar]
- Harrington, DG; Crabbs, CL; Hilmas, DE; Brown, JR; Higbee, GA; Cole, FEJ; Levy, HB. Adjuvant effects of low doses of a nuclease-resistant derivative of polyinosinic acid . polycytidylic acid on antibody responses of monkeys to inactivated Venezuelan equine encephalomyelitis virus vaccine. Infect. Immun 1979, 24, 160–166. [Google Scholar]
- Houston, WE; Crabbs, CL; Stephen, EL; Levy, HB. Modified polyriboinosinic-polyribocytidylic acid, an immunological adjuvant. Infect. Immun 1976, 14, 318–319. [Google Scholar]
- Siddiqi, NJ; Puri, SK; Dutta, GP; Maheshwari, RK; Pandey, VC. Studies on hepatic oxidative stress and antioxidant defence system during chloroquine/poly ICLC treatment of Plasmodium yoelii nigeriensis infected mice. Mol. Cell. Biochem 1999, 194, 179–183. [Google Scholar]
- Montefiori, DC; Robinson, WEJ; Mitchell, WM. In vitro evaluation of mismatched double-stranded RNA (ampligen) for combination therapy in the treatment of acquired immunodeficiency syndrome. AIDS Res. Hum. Retroviruses 1989, 5, 193–203. [Google Scholar]
- Pantelic, L; Sivakumaran, H; Urosevic, N. Differential induction of antiviral effects against West Nile virus in primary mouse macrophages derived from flavivirus-susceptible and congenic resistant mice by alpha/beta interferon and poly(I-C). J. Virol 2005, 79, 1753–1764. [Google Scholar]
- Coppenhaver, DH; Singh, IP; Sarzotti, M; Levy, HB; Baron, S. Treatment of intracranial alphavirus infections in mice by a combination of specific antibodies and an interferon inducer. Am. J. Trop. Med. Hyg 1995, 52, 34–40. [Google Scholar]
- Ewel, CH; Urba, WJ; Kopp, WC; Smith, JW, 2nd; Steis, RG; Rossio, JL; Longo, DL; Jones, MJ; Alvord, WG; Pinsky, CM; et al. Polyinosinic-polycytidylic acid complexed with poly-L-lysine and carboxymethylcellulose in combination with interleukin 2 in patients with cancer: clinical and immunological effects. Cancer Res 1992, 52, 3005–3010. [Google Scholar]
- Gatmaitan, BG; Legaspi, RC; Levy, HB; Lerner, AM. Modified polyriboinosinic-polyribocytidylic acid complex: induction of serum interferon, fever, and hypotension in rabbits. Antimicrob. Agents Chemother 1980, 17, 49–54. [Google Scholar]
- Traynor, TR; Majde, JA; Bohnet, SG; Krueger, JM. Intratracheal double-stranded RNA plus interferon-gamma: a model for analysis of the acute phase response to respiratory viral infections. Life Sci 2004, 74, 2563–2576. [Google Scholar]
- Robinson, RA; DeVita, VT; Levy, HB; Baron, S; Hubbard, SP; Levine, AS. A phase I-II trial of multiple-dose polyriboinosic-polyribocytidylic acid in patieonts with leukemia or solid tumors. J. Natl. Cancer Inst 1976, 57, 599–602. [Google Scholar]
- Krown, SE; Kerr, D; Stewart, WE, 2nd; Field, AK; Oettgen, HF. Phase I trials of poly(I,C) complexes in advanced cancer. J. Biol. Response Mod 1985, 4, 640–649. [Google Scholar]
- Lampkin, BC; Levine, AS; Levy, H; Krivit, W; Hammond, D. Phase II trial of poly(I,C)-LC, an interferon inducer, in the treatment of children with acute leukemia and neuroblastoma: A report from the Children’s Cancer Study Group. J. Biol. Response Mod 1985, 4, 531–537. [Google Scholar]
- Lampkin, BC; Levine, AS; Levy, H; Krivit, W; Hammond, D. Phase II trial of a complex polyriboinosinic-polyribocytidylic acid with poly-L-lysine and carboxymethyl cellulose in the treatment of children with acute leukemia and neuroblastoma: a report from the Children’s Cancer Study Group. Cancer Res 1985, 45, (11 Pt 2). 5904–5909. [Google Scholar]
- Durie, BG; Levy, HB; Voakes, J; Jett, JR; Levine, AS. Poly(I,C)-LC as an interferon inducer in refractory multiple myeloma. J. Biol. Response Mod 1985, 4, 518–524. [Google Scholar]
- McFarlin, DE; Bever, CT; Salazar, AM; Levy, HB. A preliminary trial of poly(I,C)-LC in multiple sclerosis. J. Biol. Response Mod 1985, 4, 544–548. [Google Scholar]
- Stevenson, HC; Abrams, PG; Schoenberger, CS; Smalley, RB; Herberman, RB; Foon, KA. A phase I evaluation of poly(I,C)-LC in cancer patients. J. Biol. Response Mod 1985, 4, 650–655. [Google Scholar]
- Hawkins, MJ; Levin, M; Borden, EC. An Eastern Cooperative Oncology Group phase I-II pilot study of polyriboinosinic-polyribocytidylic acid poly-L-lysine complex in patients with metastatic malignant melanoma. J. Biol. Response Mod 1985, 4, 664–668. [Google Scholar]
- Bever, CT, Jr; McFarland, HF; Levy, HB; McFarlin, DE. Cortisol induction by poly ICLC: implications for clinical trials of interferon. Ann. Neurol 1988, 23, 196–199. [Google Scholar]
- Giantonio, BJ; Hochster, H; Blum, R; Wiernik, PH; Hudes, GR; Kirkwood, J; Trump, D; Oken, MM. Toxicity and response evaluation of the interferon inducer poly ICLC administered at low dose in advanced renal carcinoma and relapsed or refractory lymphoma: a report of two clinical trials of the Eastern Cooperative Oncology Group. Invest. New Drugs 2001, 19, 89–92. [Google Scholar]
- Zare, F; Bokarewa, M; Nenonen, N; Bergstrom, T; Alexopoulou, L; Flavell, RA; Tarkowski, A. Arthritogenic properties of double-stranded (viral) RNA. J. Immunol 2004, 172, 5656–5663. [Google Scholar]
- Levy, HB; Riley, FL. A comparison of immune modulating effects of interferon and interferon inducers. In Lymphokines; Openheim, J, Pick, A, Eds.; Academic Press: New York, 1983; Volume 8, pp. 303–322. [Google Scholar]
- Fortier, ME; Kent, S; Ashdown, H; Poole, S; Boksa, P; Luheshi, GN. The viral mimic, polyinosinic:polycytidylic acid, induces fever in rats via an interleukin-1-dependent mechanism. Am. J. Physiol. Regul. Integr. Comp. Physiol 2004, 287, R759–766. [Google Scholar]
- Chadha, KC; Dembinski, WE; Dunn, CB; Aradi, J; Bardos, TJ; Dunn, JA; Ambrus, JL, Sr. Effect of increasing thiolation of the polycytidylic acid strand of poly I:poly C on the alpha, beta and gamma interferon-inducing properties, antiviral and antiproliferative activities. Antiviral Res 2004, 64, 171–177. [Google Scholar]
- Marshall, JD; Hessel, EM; Gregorio, J; Abbate, C; Yee, P; Chu, M; Van Nest, G; Coffman, RL; Fearon, KL. Novel chimeric immunomodulatory compounds containing short CpG oligodeoxyribonucleotides have differential activities in human cells. Nucleic Acids Res 2003, 31, 5122–5133. [Google Scholar]
- Rankin, R; Pontarollo, R; Ioannou, X; Krieg, AM; Hecker, R; Babiuk, LA; van Drunen Littel-van den Hurk, S. CpG motif identification for veterinary and laboratory species demonstrates that sequence recognition is highly conserved. Antisense Nucleic Acid Drug Dev 2001, 11, 333–340. [Google Scholar]
- Kurata, K; Iwata, A; Masuda, K; Sakaguchi, M; Ohno, K; Tsujimoto, H. Identification of CpG oligodeoxynucleotide sequences that induce IFN-gamma production in canine peripheral blood mononuclear cells. Vet. Immunol. Immunopathol 2004, 102, 441–450. [Google Scholar]
- Hartmann, G; Weeratna, RD; Ballas, ZK; Payette, P; Blackwell, S; Suparto, I; Rasmussen, WL; Waldschmidt, M; Sajuthi, D; Purcell, RH; et al. Delineation of a CpG phosphorothioate oligodeoxynucleotide for activating primate immune responses in vitro and in vivo. J. Immunol 2000, 164, 1617–1624. [Google Scholar]
- Fearon, K; Marshall, JD; Abbate, C; Subramanian, S; Yee, P; Gregorio, J; Teshima, G; Ott, G; Tuck, S; Van Nest, G; et al. A minimal human immunostimulatory CpG motif that potently induces IFN-gamma and IFN-alpha production. Eur. J. Immunol 2003, 33, 2114–2122. [Google Scholar]
- Cornelie, S; Hoebeke, J; Schacht, AM; Bertin, B; Vicogne, J; Capron, M; Riveau, G. Direct evidence that toll-like receptor 9 (TLR9) functionally binds plasmid DNA by specific cytosine-phosphate-guanine motif recognition. J. Biol. Chem 2004, 279, 15124–15129. [Google Scholar]
- Klinman, DM; Currie, D; Gursel, I; Verthelyi, D. Use of CpG oligodeoxynucleotides as immune adjuvants. Immunol. Rev 2004, 199, 201–216. [Google Scholar]
- Ishii, KJ; Gursel, I; Gursel, M; Klinman, DM. Immunotherapeutic utility of stimulatory and suppressive oligodeoxynucleotides. Curr. Opin. Mol. Ther 2004, 6, 166–174. [Google Scholar]
- Yi, AK; Peckham, DW; Ashman, RF; Krieg, AM. CpG DNA rescues B cells from apoptosis by activating NFkappaB and preventing mitochondrial membrane potential disruption via a chloroquine-sensitive pathway. Int. Immunol 1999, 11, 2015–2024. [Google Scholar]
- Hartmann, G; Weiner, GJ; Krieg, AM. CpG DNA: a potent signal for growth, activation, and maturation of human dendritic cells. Proc. Natl. Acad. Sci. USA 1999, 96, 9305–9310. [Google Scholar]
- Bernasconi, NL; Onai, N; Lanzavecchia, A. A role for Toll-like receptors in acquired immunity: up-regulation of TLR9 by BCR triggering in naive B cells and constitutive expression in memory B cells. Blood 2003, 101, 4500–4504. [Google Scholar]
- Kerkmann, M; Rothenfusser, S; Hornung, V; Towarowski, A; Wagner, M; Sarris, A; Giese, T; Endres, S; Hartmann, G. Activation with CpG-A and CpG-B oligonucleotides reveals two distinct regulatory pathways of type I IFN synthesis in human plasmacytoid dendritic cells. J. Immunol 2003, 170, 4465–4474. [Google Scholar]
- Hornung, V; Rothenfusser, S; Britsch, S; Krug, A; Jahrsdorfer, B; Giese, T; Endres, S; Hartmann, G. Quantitative expression of toll-like receptor 1-10 mRNA in cellular subsets of human peripheral blood mononuclear cells and sensitivity to CpG oligodeoxynucleotides. J. Immunol 2002, 168, 4531–4537. [Google Scholar]
- Amcheslavsky, A; Zou, W; Bar-Shavit, Z. Toll-like receptor 9 regulates tumor necrosis factor-alpha expression by different mechanisms. Implications for osteoclastogenesis. J. Biol. Chem 2004, 279, 54039–54045. [Google Scholar]
- Zhao, Q; Matson, S; Herrera, CJ; Fisher, E; Yu, H; Krieg, AM. Comparison of cellular binding and uptake of antisense phosphodiester, phosphorothioate, and mixed phosphorothioate and methylphosphonate oligonucleotides. Antisense Res. Dev 1993, 3, 53–66. [Google Scholar]
- Agrawal, S; Temsamani, J; Galbraith, W; Tang, J. Pharmacokinetics of antisense oligonucleotides. Clin. Pharmacokinet 1995, 28, 7–16. [Google Scholar]
- Iversen, PL; Zhu, S; Meyer, A; Zon, G. Cellular uptake and subcellular distribution of phosphorothioate oligonucleotides into cultured cells. Antisense Res. Dev 1992, 2, 211–222. [Google Scholar]
- Klinman, DM; Currie, D. Hierarchical recognition of CpG motifs expressed by immunostimulatory oligodeoxynucleotides. Clin. Exp. Immunol 2003, 133, 227–232. [Google Scholar]
- Verthelyi, D; Gursel, M; Kenney, RT; Lifson, JD; Liu, S; Mican, J; Klinman, DM. CpG oligodeoxynucleotides protect normal and SIV-infected macaques from Leishmania infection. J. Immunol 2003, 170, 4717–4723. [Google Scholar]
- Krieg, AM; Love-Homan, L; Yi, AK; Harty, JT. CpG DNA induces sustained IL-12 expression in vivo and resistance to Listeria monocytogenes challenge. J. Immunol 1998, 161, 2428–2434. [Google Scholar]
- Krieg, AM; Efler, SM; Wittpoth, M; Al Adhami, MJ; Davis, HL. Induction of systemic TH1-like innate immunity in normal volunteers following subcutaneous but not intravenous administration of CPG 7909, a synthetic B-class CpG oligodeoxynucleotide TLR9 agonist. J. Immunother 2004, 27, 460–471. [Google Scholar]
- Christopher, ME; Wong, JP. Broad-spectrum antiviral defence against influenza viruses using nucleic acid-based immunomodulators. In Recent Developments on the Avian Influenza (H5N1) Crisis; Wong, JP, Ed.; Transworld Research Network: Trivandrum, 2006; pp. 85–118. [Google Scholar]
- Dong, L; Mori, I; Hossain, MJ; Liu, B; Kimura, Y. An immunostimulatory oligodeoxynucleotide containing a cytidine-guanosine motif protects senescence-accelerated mice from lethal influenza virus by augmenting the T helper type 1 response. J. Gen. Virol 2003, 84, (Pt 6). 1623–1628. [Google Scholar]
- Jahrsdorfer, B; Muhlenhoff, L; Blackwell, SE; Wagner, M; Poeck, H; Hartmann, E; Jox, R; Giese, T; Emmerich, B; Endres, S; et al. B-cell lymphomas differ in their responsiveness to CpG oligodeoxynucleotides. Clin. Cancer Res 2005, 11, 1490–1499. [Google Scholar]
- Gursel, I; Gursel, M; Ishii, KJ; Klinman, DM. Sterically stabilized cationic liposomes improve the uptake and immunostimulatory activity of CpG oligonucleotides. J. Immunol 2001, 167, 3324–3328. [Google Scholar]
- Elkins, KL; Rhinehart-Jones, TR; Stibitz, S; Conover, JS; Klinman, DM. Bacterial DNA containing CpG motifs stimulates lymphocyte-dependent protection of mice against lethal infection with intracellular bacteria. J. Immunol 1999, 162, 2291–2298. [Google Scholar]
- Klinman, DM. Therapeutic applications of CpG-containing oligodeoxynucleotides. Antisense Nucleic Acid Drug Dev 1998, 8, 181–184. [Google Scholar]
- Klinman, DM; Conover, J; Coban, C. Repeated administration of synthetic oligodeoxynucleotides expressing CpG motifs provides long-term protection against bacterial infection. Infect. Immun 1999, 67, 5658–5663. [Google Scholar]
- Gomis, S; Babiuk, L; Godson, DL; Allan, B; Thrush, T; Townsend, H; Willson, P; Waters, E; Hecker, R; Potter, A. Protection of chickens against Escherichia coli infections by DNA containing CpG motifs. Infect. Immun 2003, 71, 857–863. [Google Scholar]
- Verthelyi, D; Ishii, KJ; Gursel, M; Takeshita, F; Klinman, DM. Human peripheral blood cells differentially recognize and respond to two distinct CPG motifs. J. Immunol 2001, 166, 2372–2377. [Google Scholar]
- Zimmermann, S; Egeter, O; Hausmann, S; Lipford, GB; Rocken, M; Wagner, H; Heeg, K. CpG oligodeoxynucleotides trigger protective and curative Th1 responses in lethal murine leishmaniasis. J. Immunol 1998, 160, 3627–3630. [Google Scholar]
- Juffermans, NP; Leemans, JC; Florquin, S; Verbon, A; Kolk, AH; Speelman, P; van Deventer, SJ; van der Poll, T. CpG oligodeoxynucleotides enhance host defense during murine tuberculosis. Infect. Immun 2002, 70, 147–152. [Google Scholar]
- Klinman, DM; Verthelyi, D; Takeshita, F; Ishii, KJ. Immune recognition of foreign DNA: A cure for bioterrorism? Immunity 1999, 11, 123–129. [Google Scholar]
- Gramzinski, RA; Doolan, DL; Sedegah, M; Davis, HL; Krieg, AM; Hoffman, SL. Interleukin-12- and gamma interferon-dependent protection against malaria conferred by CpG oligodeoxynucleotide in mice. Infect. Immun 2001, 69, 1643–1649. [Google Scholar]
- Olbrich, AR; Schimmer, S; Heeg, K; Schepers, K; Schumacher, TN; Dittmer, U. Effective postexposure treatment of retrovirus-induced disease with immunostimulatory DNA containing CpG motifs. J. Virol 2002, 76, 11397–11404. [Google Scholar]
- Pyles, RB; Higgins, D; Chalk, C; Zalar, A; Eiden, J; Brown, C; Van Nest, G; Stanberry, LR. Use of immunostimulatory sequence-containing oligonucleotides as topical therapy for genital herpes simplex virus type 2 infection. J. Virol 2002, 76, 11387–11396. [Google Scholar]
- Sethi, S; Lipford, G; Wagner, H; Kretzschmar, H. Postexposure prophylaxis against prion disease with a stimulator of innate immunity. Lancet 2002, 360, 229–230. [Google Scholar]
- Dalloul, RA; Lillehoj, HS; Okamura, M; Xie, H; Min, W; Ding, X; Heckert, RA. In vivo effects of CpG oligodeoxynucleotide on Eimeria infection in chickens. Avian Dis 2004, 48, 783–790. [Google Scholar]
- Moldoveanu, Z; Love-Homan, L; Huang, WQ; Krieg, AM. CpG DNA, a novel immune enhancer for systemic and mucosal immunization with influenza virus. Vaccine 1998, 16, 1216–1224. [Google Scholar]
- McCluskie, MJ; Davis, HL. Oral, intrarectal and intranasal immunizations using CpG and non-CpG oligodeoxynucleotides as adjuvants. Vaccine 2000, 19, 413–422. [Google Scholar]
- Hayashi, M; Satou, E; Ueki, R; Yano, M; Miyano-Kurosaki, N; Fujii, M; Takaku, H. Resistance to influenza A virus infection by antigen-conjugated CpG oligonucleotides, a novel antigen-specific immunomodulator. Biochem. Biophys. Res. Commun 2005, 329, 230–236. [Google Scholar]
- Joseph, A; Louria-Hayon, I; Plis-Finarov, A; Zeira, E; Zakay-Rones, Z; Raz, E; Hayashi, T; Takabayashi, K; Barenholz, Y; Kedar, E. Liposomal immunostimulatory DNA sequence (ISS-ODN): an efficient parenteral and mucosal adjuvant for influenza and hepatitis B vaccines. Vaccine 2002, 20, 3342–3354. [Google Scholar]
- Klinman, DM; Ishii, KJ; Gursel, M; Gursel, I; Takeshita, S; Takeshita, F. Immunotherapeutic applications of CpG-containing oligodeoxynucleotides. Drug News Perspect 2000, 13, 289–296. [Google Scholar]
- Cooper, CL; Davis, HL; Morris, ML; Efler, SM; Krieg, AM; Li, Y; Laframboise, C; Al Adhami, MJ; Khaliq, Y; Seguin, I; et al. Safety and immunogenicity of CPG 7909 injection as an adjuvant to Fluarix influenza vaccine. Vaccine 2004, 22, 3136–3143. [Google Scholar]
- Halperin, SA; Van Nest, G; Smith, B; Abtahi, S; Whiley, H; Eiden, JJ. A phase I study of the safety and immunogenicity of recombinant hepatitis B surface antigen co-administered with an immunostimulatory phosphorothioate oligonucleotide adjuvant. Vaccine 2003, 21, 2461–2467. [Google Scholar]
- Friedberg, JW; Kim, H; McCauley, M; Hessel, EM; Sims, P; Fisher, DC; Nadler, LM; Coffman, RL; Freedman, AS. Combination immunotherapy with a CpG oligonucleotide (1018 ISS) and rituximab in patients with non-Hodgkin lymphoma: increased interferon-alpha/beta-inducible gene expression, without significant toxicity. Blood 2005, 105, 489–495. [Google Scholar]
- Siegrist, CA; Pihlgren, M; Tougne, C; Efler, SM; Morris, ML; AlAdhami, MJ; Cameron, DW; Cooper, CL; Heathcote, J; Davis, HL; et al. Co-administration of CpG oligonucleotides enhances the late affinity maturation process of human anti-hepatitis B vaccine response. Vaccine 2004, 23, 615–622. [Google Scholar]
- Speiser, DE; Lienard, D; Rufer, N; Rubio-Godoy, V; Rimoldi, D; Lejeune, F; Krieg, AM; Cerottini, JC; Romero, P. Rapid and strong human CD8+ T cell responses to vaccination with peptide, IFA, and CpG oligodeoxynucleotide 7909. J. Clin. Invest 2005, 115, 739–746. [Google Scholar]
- Paul, S. Technology evaluation: CpG-7909, Coley. Curr. Opin. Mol. Ther 2003, 5, 553–559. [Google Scholar]
- Cooper, CL; Davis, HL; Morris, ML; Efler, SM; Adhami, MA; Krieg, AM; Cameron, DW; Heathcote, J. CPG 7909, an immunostimulatory TLR9 agonist oligodeoxynucleotide, as adjuvant to Engerix-B HBV vaccine in healthy adults: a double-blind phase I/II study. J. Clin. Immunol 2004, 24, 693–701. [Google Scholar]
- Babiuk, S; Baca-Estrada, ME; Middleton, DM; Hecker, R; Babiuk, LA; Foldvari, M. Biphasic lipid vesicles (Biphasix) enhance the adjuvanticity of CpG oligonucleotides following systemic and mucosal administration. Curr. Drug. Deliv 2004, 1, 9–15. [Google Scholar]
- Zakhartchouk, AN; Sharon, C; Satkunarajah, M; Auperin, T; Viswanathan, S; Mutwiri, G; Petric, M; See, RH; Brunham, RC; Finlay, BB; et al. Immunogenicity of a receptor-binding domain of SARS coronavirus spike protein in mice: implications for a subunit vaccine. Vaccine 2007, 25, 136–143. [Google Scholar]
- Bao, M; Zhang, Y; Wan, M; Dai, L; Hu, X; Wu, X; Wang, L; Deng, P; Wang, J; Chen, J; et al. Anti-SARS-CoV immunity induced by a novel CpG oligodeoxynucleotide. Clin. Immunol 2006, 118, 180–187. [Google Scholar]
- Rankin, R; Pontarollo, R; Gomis, S; Karvonen, B; Willson, P; Loehr, BI; Godson, DL; Babiuk, LA; Hecker, R; van Drunen Littel-van den Hurk, S. CpG-containing oligodeoxynucleotides augment and switch the immune responses of cattle to bovine herpesvirus-1 glycoprotein D. Vaccine 2002, 20, 3014–3022. [Google Scholar]
- Kwissa, M; Amara, RR; Robinson, HL; Moss, B; Alkan, S; Jabbar, A; Villinger, F; Pulendran, B. Adjuvanting a DNA vaccine with a TLR9 ligand plus Flt3 ligand results in enhanced cellular immunity against the simian immunodeficiency virus. J. Exp. Med 2007, 204, 2733–2746. [Google Scholar]
- Kumar, S; Jones, TR; Oakley, MS; Zheng, H; Kuppusamy, SP; Taye, A; Krieg, AM; Stowers, AW; Kaslow, DC; Hoffman, SL. CpG oligodeoxynucleotide and Montanide ISA 51 adjuvant combination enhanced the protective efficacy of a subunit malaria vaccine. Infect. Immun 2004, 72, 949–957. [Google Scholar]
- Wooldridge, JE; Ballas, Z; Krieg, AM; Weiner, GJ. Immunostimulatory oligodeoxynucleotides containing CpG motifs enhance the efficacy of monoclonal antibody therapy of lymphoma. Blood 1997, 89, 2994–2998. [Google Scholar]
- Klinman, DM. CpG DNA as a vaccine adjuvant. Expert Rev. Vaccines 2003, 2, 305–315. [Google Scholar]
- Katsumi, A; Emi, N; Abe, A; Hasegawa, Y; Ito, M; Saito, H. Humoral and cellular immunity to an encoded protein induced by direct DNA injection. Hum. Gene Ther 1994, 5, 1335–1339. [Google Scholar]
- Mor, G; Singla, M; Steinberg, AD; Hoffman, SL; Okuda, K; Klinman, DM. Do DNA vaccines induce autoimmune disease? Hum. Gene Ther 1997, 8, 293–300. [Google Scholar]
- Gilkeson, GS; Conover, J; Halpern, M; Pisetsky, DS; Feagin, A; Klinman, DM. Effects of bacterial DNA on cytokine production by (NZB/NZW)F1 mice. J. Immunol 1998, 161, 3890–3895. [Google Scholar]
- Segal, BM; Klinman, DM; Shevach, EM. Microbial products induce autoimmune disease by an IL-12-dependent pathway. J. Immunol 1997, 158, 5087–5090. [Google Scholar]
- Segal, BM; Chang, JT; Shevach, EM. CpG oligonucleotides are potent adjuvants for the activation of autoreactive encephalitogenic T cells in vivo. J. Immunol 2000, 164, 5683–5688. [Google Scholar]
- Bachmaier, K; Neu, N; de la Maza, LM; Pal, S; Hessel, A; Penninger, JM. Chlamydia infections and heart disease linked through antigenic mimicry. Science 1999, 283, 1335–1339. [Google Scholar]
- Zeuner, RA; Verthelyi, D; Gursel, M; Ishii, KJ; Klinman, DM. Influence of stimulatory and suppressive DNA motifs on host susceptibility to inflammatory arthritis. Arthritis Rheum 2003, 48, 1701–1707. [Google Scholar]
- Cowdery, JS; Chace, JH; Yi, AK; Krieg, AM. Bacterial DNA induces NK cells to produce IFN-gamma in vivo and increases the toxicity of lipopolysaccharides. J. Immunol 1996, 156, 4570–4575. [Google Scholar]
- Hartmann, G; Krug, A; Waller-Fontaine, K; Endres, S. Oligodeoxynucleotides enhance lipopolysaccharide-stimulated synthesis of tumor necrosis factor: dependence on phosphorothioate modification and reversal by heparin. Mol. Med 1996, 2, 429–438. [Google Scholar]
- Sparwasser, T; Miethke, T; Lipford, G; Borschert, K; Hacker, H; Heeg, K; Wagner, H. Bacterial DNA causes septic shock. Nature 1997, 386, 336–337. [Google Scholar]
- Heikenwalder, M; Polymenidou, M; Junt, T; Sigurdson, C; Wagner, H; Akira, S; Zinkernagel, R; Aguzzi, A. Lymphoid follicle destruction and immunosuppression after repeated CpG oligodeoxynucleotide administration. Nat. Med 2004, 10, 187–192. [Google Scholar]
- Jiao, X; Wang, RY; Qiu, Q; Alter, HJ; Shih, JW. Enhanced hepatitis C virus NS3 specific Th1 immune responses induced by co-delivery of protein antigen and CpG with cationic liposomes. J. Gen. Virol 2004, 85, (Pt 6). 1545–1553. [Google Scholar]
- Mui, B; Raney, SG; Semple, SC; Hope, MJ. Immune stimulation by a CpG-containing oligodeoxynucleotide is enhanced when encapsulated and delivered in lipid particles. J. Pharmacol. Exp. Ther 2001, 298, 1185–1192. [Google Scholar]
- Benimetskaya, L; Wittenberger, T; Stein, CA; Hofmann, HP; Weller, C; Lai, JC; Miller, P; Gekeler, V. Changes in gene expression induced by phosphorothioate oligodeoxynucleotides (including G3139) in PC3 prostate carcinoma cells are recapitulated at least in part by treatment with interferon-beta and -gamma. Clin. Cancer Res 2004, 10, 3678–3688. [Google Scholar]
- Diwan, M; Tafaghodi, M; Samuel, J. Enhancement of immune responses by co-delivery of a CpG oligodeoxynucleotide and tetanus toxoid in biodegradable nanospheres. J. Control. Release 2002, 85, 247–262. [Google Scholar]
- Hayashi, M; Kuwahara, M; Ogata, M; Miyao-Kurosaki, N; Abel, T; Ueki, R; Yano, M; Fujii, M; Hartmann, G; Takaku, H. Evaluating the immune responses stimulated by CpG oligodeoxynucleotides. Nucleic Acids Res. Suppl 2003, 323–324. [Google Scholar]
- Kandimalla, ER; Bhagat, L; Yu, D; Cong, Y; Tang, J; Agrawal, S. Conjugation of ligands at the 5′-end of CpG DNA affects immunostimulatory activity. Bioconjug. Chem 2002, 13, 966–974. [Google Scholar]
- Kandimalla, ER; Bhagat, L; Zhu, FG; Yu, D; Cong, YP; Wang, D; Tang, JX; Tang, JY; Knetter, CF; Lien, E; et al. A dinucleotide motif in oligonucleotides shows potent immunomodulatory activity and overrides species-specific recognition observed with CpG motif. Proc. Natl. Acad. Sci. USA 2003, 100, 14303–14308. [Google Scholar]
- Diebold, SS; Massacrier, C; Akira, S; Paturel, C; Morel, Y; Reis e Sousa, C. Nucleic acid agonists for Toll-like receptor 7 are defined by the presence of uridine ribonucleotides. Eur. J. Immunol 2006, 36, 3256–3267. [Google Scholar]
- Wang, JP; Liu, P; Latz, E; Golenbock, DT; Finberg, RW; Libraty, DH. Flavivirus activation of plasmacytoid dendritic cells delineates key elements of TLR7 signaling beyond endosomal recognition. J. Immunol 2006, 177, 7114–7121. [Google Scholar]
- Hornung, V; Barchet, W; Schlee, M; Hartmann, G. RNA recognition via TLR7 and TLR8. Handb. Exp. Pharmacol 2008, 183, 71–86. [Google Scholar]
- Novak, NY; Yu, CF; Bieber, T; Allam, JP. Toll-like receptor 7 agonists and skin. Drug News Perspect 2008, 21(3), 158–165. [Google Scholar]
- Hanten, JA; Vasilakos, JP; Riter, CL; Neys, L; Lipson, KE; Alkan, SS; Birmachu, W. Comparison of human B cell activation by TLR7 and TLR9 agonists. BMC Immunol 2008, 9, 39. [Google Scholar]
- Butchi, NB; Pourciau, S; Du, M; Morgan, TW; Peterson, KE. Analysis of the neuroinflammatory response to TLR7 stimulation in the brain: comparison of multiple TLR7 and/or TLR8 agonists. J. Immunol 2008, 180, 7604–7612. [Google Scholar]
- Birmachu, W; Gleason, RM; Bulbulian, BJ; Riter, CL; Vasilakos, JP; Lipson, KE; Nikolsky, Y. Transcriptional networks in plasmacytoid dendritic cells stimulated with synthetic TLR 7 agonists. BMC Immunol 2007, 8, 26. [Google Scholar]
- Gantier, MP; Tong, S; Behlke, MA; Xu, D; Phipps, S; Foster, PS; Williams, BR. TLR7 is involved in sequence-specific sensing of single-stranded RNAs in human macrophages. J. Immunol 2008, 180, 2117–2124. [Google Scholar]
- Delgado, MA; Elmaoued, RA; Davis, AS; Kyei, G; Deretic, V. Toll-like receptors control autophagy. E.M.B.O. J 2008, 27, 1110–1121. [Google Scholar]
- Wang, JP; Bowen, GN; Padden, C; Cerny, A; Finberg, RW; Newburger, PE; Kurt-Jones, EA. Toll-like receptor-mediated activation of neutrophils by influenza A virus. Blood 2008, 112, 2028–2034. [Google Scholar]
- Wu, CC; Hayashi, T; Takabayashi, K; Sabet, M; Smee, DF; Guiney, DD; Cottam, HB; Carson, DA. Immunotherapeutic activity of a conjugate of a Toll-like receptor 7 ligand. Proc. Natl. Acad. Sci. USA 2007, 104, 3990–3995. [Google Scholar]
- Hammerbeck, DM; Burleson, GR; Schuller, CJ; Vasilakos, JP; Tomai, M; Egging, E; Cochran, FR; Woulfe, S; Miller, RL. Administration of a dual toll-like receptor 7 and toll-like receptor 8 agonist protects against influenza in rats. Antiviral Res 2007, 73, 1–11. [Google Scholar]
- Krug, A. Nucleic acid recognition receptors in autoimmunity. Handb. Exp. Pharmacol 2008, 183, 129–151. [Google Scholar]
- Deane, JA; Pisitkun, P; Barrett, RS; Feigenbaum, L; Town, T; Ward, JM; Flavell, RA; Bolland, S. Control of toll-like receptor 7 expression is essential to restrict autoimmunity and dendritic cell proliferation. Immunity 2007, 27, 801–810. [Google Scholar]
- Barrat, FJ; Meeker, T; Chan, JH; Guiducci, C; Coffman, RL. Treatment of lupus-prone mice with a dual inhibitor of TLR7 and TLR9 leads to reduction of autoantibody production and amelioration of disease symptoms. Eur. J. Immunol 2007, 37, 3582–3586. [Google Scholar]
- Berghöfer, B; Frommer, T; Haley, G; Fink, L; Bein, G; Hackstein, H. TLR7 ligands induce higher IFN-alpha production in females. J. Immunol 2006, 177, 2088–2096. [Google Scholar]
- Matrosovich, MN; Matrosovich, TY; Gray, T; Roberts, NA; Klenk, HD. Human and avian influenza viruses target different cell types in cultures of human airway epithelium. Proc. Natl. Acad. Sci. USA 2004, 101, 4620–4624. [Google Scholar]
- Olofsson, S; Kumlin, U; Dimock, K; Arnberg, N. Avian influenza and sialic acid receptors: more than meets the eye? Lancet Infect. Dis 2005, 5, 184–188. [Google Scholar]
- Sidwell, RW; Huffman, JH; Bailey, KW; Wong, MH; Nimrod, A; Panet, A. Inhibitory effects of recombinant manganese superoxide dismutase on influenza virus infections in mice. Antimicrob. Agents Chemother 1996, 40, 2626–2631. [Google Scholar]
- Oda, T; Akaike, T; Hamamoto, T; Suzuki, F; Hirano, T; Maeda, H. Oxygen radicals in influenza-induced pathogenesis and treatment with pyran polymer-conjugated SOD. Science 1989, 244, 974–976. [Google Scholar]
- Kawaguchi, M; Kokubu, F; Kuga, H; Tomita, T; Matsukura, S; Suzaki, H; Huang, SK; Adachi, M. Influenza virus A stimulates expression of eotaxin by nasal epithelial cells. Clin. Exp. Allergy 2001, 31, 873–880. [Google Scholar]
- Matsukura, S; Kokubu, F; Kubo, H; Tomita, T; Tokunaga, H; Kadokura, M; Yamamoto, T; Kuroiwa, Y; Ohno, T; Suzaki, H; et al. Expression of RANTES by normal airway epithelial cells after influenza virus A infection. Am. J. Respir. Cell. Mol. Biol 1998, 18, 255–264. [Google Scholar]
- Schultz-Cherry, S; Hinshaw, VS. Influenza virus neuraminidase activates latent transforming growth factor beta. J. Virol 1996, 70, 8624–8629. [Google Scholar]
- Ohyama, K; Sano, T; Toyoda, H. Predominant contribution of IFN-beta expression to apoptosis induction in human uterine cervical fibroblast cells by influenza-virus infection. Biol. Pharm. Bull 2004, 27, 1750–1757. [Google Scholar]
- Liu, B; Mori, I; Hossain, MJ; Dong, L; Chen, Z; Kimura, Y. Local immune responses to influenza virus infection in mice with a targeted disruption of perforin gene. Microb. Pathog 2003, 34, 161–167. [Google Scholar]
- He, XS; Draghi, M; Mahmood, K; Holmes, TH; Kemble, GW; Dekker, CL; Arvin, AM; Parham, P; Greenberg, HB. T cell-dependent production of IFN-gamma by NK cells in response to influenza A virus. J. Clin. Invest 2004, 114, 1812–1819. [Google Scholar]
- Seo, SH; Peiris, M; Webster, RG. Protective cross-reactive cellular immunity to lethal A/Goose/Guangdong/1/96-like H5N1 influenza virus is correlated with the proportion of pulmonary CD8(+) T cells expressing gamma interferon. J. Virol 2002, 76, 4886–4890. [Google Scholar]
- Cheung, CY; Poon, LL; Lau, AS; Luk, W; Lau, YL; Shortridge, KF; Gordon, S; Guan, Y; Peiris, JS. Induction of proinflammatory cytokines in human macrophages by influenza A (H5N1) viruses: a mechanism for the unusual severity of human disease? Lancet 2002, 360, 1831–1837. [Google Scholar]
- Dybing, JK; Schultz-Cherry, S; Swayne, DE; Suarez, DL; Perdue, ML. Distinct pathogenesis of Hong Kong-origin H5N1 viruses in mice compared to that of other highly pathogenic H5 avian influenza viruses. J. Virol 2000, 74, 1443–1450. [Google Scholar]
- Klenk, HD. Infection of the endothelium by influenza viruses. Thromb. Haemost 2005, 94, 262–265. [Google Scholar]
- Rimmelzwaan, GF; Kuiken, T; van Amerongen, G; Bestebroer, TM; Fouchier, RA; Osterhaus, AD. A primate model to study the pathogenesis of influenza A (H5N1) virus infection. Avian Dis 2003, 47(3 Suppl), 931–933. [Google Scholar]
- Uiprasertkul, M; Puthavathana, P; Sangsiriwut, K; Pooruk, P; Srisook, K; Peiris, M; Nicholls, JM; Chokephaibulkit, K; Vanprapar, N; Auewarakul, P. Influenza A H5N1 replication sites in humans. Emerg. Infect. Dis 2005, 11, 1036–1041. [Google Scholar]
- Koyama, S; Sato, E; Tsukadaira, A; Haniuda, M; Numanami, H; Kurai, M; Nagai, S; Izumi, T. Vascular endothelial growth factor mRNA and protein expression in airway epithelial cell lines in vitro. Eur. Respir. J 2002, 20, 1449–1456. [Google Scholar]
- Iwasaki, T; Itamura, S; Nishimura, H; Sato, Y; Tashiro, M; Hashikawa, T; Kurata, T. Productive infection in the murine central nervous system with avian influenza virus A (H5N1) after intranasal inoculation. Acta. Neuropathol 2004, 108, 485–492. [Google Scholar]
- Lipatov, AS; Andreansky, S; Webby, RJ; Hulse, DJ; Rehg, JE; Krauss, S; Perez, DR; Doherty, PC; Webster, RG; Sangster, MY. Pathogenesis of Hong Kong H5N1 influenza virus NS gene reassortants in mice: the role of cytokines and B- and T-cell responses. J. Gen. Virol 2005, 86, (Pt 4). 1121–1130. [Google Scholar]
- Seo, SH; Hoffmann, E; Webster, RG. The NS1 gene of H5N1 influenza viruses circumvents the host anti-viral cytokine responses. Virus Res 2004, 103, 107–113. [Google Scholar]
- Tumpey, TM; Lu, X; Morken, T; Zaki, SR; Katz, JM. Depletion of lymphocytes and diminished cytokine production in mice infected with a highly virulent influenza A (H5N1) virus isolated from humans. J. Virol 2000, 74, 6105–6116. [Google Scholar]
- Szretter, KJ; Gangappa, S; Lu, X; Smith, C; Shieh, WJ; Zaki, SR; Sambhara, S; Tumpey, TM; Katz, JM. Role of host cytokine responses in the pathogenesis of avian H5N1 influenza viruses in mice. J. Virol 2007, 81, 2736–2744. [Google Scholar]
- Wolpe, SD; Davatelis, G; Sherry, B; Beutler, B; Hesse, DG; Nguyen, HT; Moldawer, LL; Nathan, CF; Lowry, SF; Cerami, A. Macrophages secrete a novel heparin-binding protein with inflammatory and neutrophil chemokinetic properties. J. Exp. Med 1988, 167, 570–581. [Google Scholar]
- Wolpe, SD; Cerami, A. Macrophage inflammatory proteins 1 and 2: members of a novel superfamily of cytokines. FASEB J 1989, 3, 2565–2573. [Google Scholar]
- Rothwell, NJ. Annual review prize lecture cytokines - killers in the brain? J. Physiol 1999, 514, (Pt 1). 3–17. [Google Scholar]
- Yuen, KY; Wong, SS. Human infection by avian influenza A H5N1. Hong Kong Med. J 2005, 11, 189–199. [Google Scholar]
- Guan, Y; Poon, LL; Cheung, CY; Ellis, TM; Lim, W; Lipatov, AS; Chan, KH; Sturm-Ramirez, KM; Cheung, CL; Leung, YH; et al. H5N1 influenza: a protean pandemic threat. Proc. Natl. Acad. Sci. USA 2004, 101, 8156–8161. [Google Scholar]
- Salomon, R; Hoffmann, E; Webster, RG. Inhibition of the cytokine response does not protect against lethal H5N1 influenza infection. Proc. Natl. Acad. Sci. USA 2007, 104, 12479–12481. [Google Scholar]
- Marshall-Clarke, S; Tasker, L; Buchatska, O; Downes, J; Pennock, J; Wharton, S; Borrow, P; Wiseman, DZ. Influenza H2 haemagglutinin activates B cells via a MyD88-dependent pathway. Eur. J. Immunol 2006, 36, 95–106. [Google Scholar]
- Heer, AK; Shamshiev, A; Donda, A; Uematsu, S; Akira, S; Kopf, M; Marsland, BJ. TLR signaling fine-tunes anti-influenza B cell responses without regulating effector T cell responses. J. Immunol 2007, 178, 2182–2191. [Google Scholar]
- Ghosh, TK; Mickelson, DJ; Solberg, JC; Lipson, KE; Inglefield, JR; Alkan, SS. TLR-TLR cross talk in human PBMC resulting in synergistic and antagonistic regulation of type-1 and 2 interferons, IL-12 and TNF-alpha. Int. Immunopharmacol 2007, 7, 1111–1121. [Google Scholar]
- Marshall, JD; Heeke, DS; Gesner, ML; Livingston, B; Van Nest, G. Negative regulation of TLR9-mediated IFN-alpha induction by a small-molecule, synthetic TLR7 ligand. J. Leukoc. Biol 2007, 82, 497–508. [Google Scholar]
- Berghöfer, B; Haley, G; Frommer, T; Bein, G; Hackstein, H. Natural and synthetic TLR7 ligands inhibit CpG-A- and CpG-C-oligodeoxynucleotide-induced IFN-alpha production. J. Immunol 2007, 178, 4072–4079. [Google Scholar]
- Wang, J; Shao, Y; Bennett, TA; Shankar, RA; Wightman, PD; Reddy, LG. The functional effects of physical interactions among Toll-like receptors 7, 8, and 9. J. Biol. Chem 2006, 281, 37427–37434. [Google Scholar]
- Guerrero-Plata, A; Baron, S; Poast, JS; Adegboyega, PA; Casola, A; Garofalo, RP. Activity and regulation of alpha interferon in respiratory syncytial virus and human metapneumovirus experimental infections. J. Virol 2005, 79, 10190–10199. [Google Scholar]
- Schlender, J; Hornung, V; Finke, S; Gunthner-Biller, M; Marozin, S; Brzozka, K; Moghim, S; Endres, S; Hartmann, G; Conzelmann, KK. Inhibition of toll-like receptor 7- and 9-mediated alpha/beta interferon production in human plasmacytoid dendritic cells by respiratory syncytial virus and measles virus. J. Virol 2005, 79, 5507–5515. [Google Scholar]
- Renshaw, M; Rockwell, J; Engleman, C; Gewirtz, A; Katz, J; Sambhara, S. Cutting edge: impaired Toll-like receptor expression and function in aging. J. Immunol 2002, 169, 4697–4701. [Google Scholar]
- Kunder, SC; Wu, L; Morahan, PS. Protection against murine cytomegalovirus infection in aged mice and mice with severe combined immunodeficiency disease with the biological response modifiers polyribosinic-polycytidylic acid stabilized with L-lysine and carboxymethylcellulose, maleic anhydride divinyl ether and colony stimulating factor 1. Antiviral Res 1993, 21, 233–245. [Google Scholar]
- Chu, RS; Targoni, OS; Krieg, AM; Lehmann, PV; Harding, CV. CpG oligodeoxynucleotides act as adjuvants that switch on T helper 1 (Th1) immunity. J. Exp. Med 1997, 186, 1623–1631. [Google Scholar]
- Manning, BM; Enioutina, EY; Visic, DM; Knudson, AD; Daynes, RA. CpG DNA functions as an effective adjuvant for the induction of immune responses in aged mice. Exp. Gerontol 2001, 37, 107–126. [Google Scholar]
- Maletto, B; Ropolo, A; Moron, V; Pistoresi-Palencia, MC. CpG-DNA stimulates cellular and humoral immunity and promotes Th1 differentiation in aged BALB/c mice. J. Leukoc. Biol 2002, 72, 447–454. [Google Scholar]
- Wong, JP; Christopher, ME; Salazar, AM; Dale, RM; Sun, LQ; Wang, M. Nucleic acid-based antiviral drugs against seasonal and avian influenza viruses. Vaccine 2007, 25, 3175–3178. [Google Scholar]
- Whitmore, MM; DeVeer, MJ; Edling, A; Oates, RK; Simons, B; Lindner, D; Williams, BR. Synergistic activation of innate immunity by double-stranded RNA and CpG DNA promotes enhanced antitumor activity. Cancer Res 2004, 64, 5850–5860. [Google Scholar]
- Werling, D; Hope, JC; Howard, CJ; Jungi, TW. Differential production of cytokines, reactive oxygen and nitrogen by bovine macrophages and dendritic cells stimulated with Toll-like receptor agonists. Immunology 2004, 111, 41–52. [Google Scholar]

| Species | Motif | Reference |
|---|---|---|
| Rodents | 5′ PuPuCGPyPy 3′ | [82, 83] |
| Canines | 5′ ATCGAT 3′ | [84] |
| Primates | 5′ GTCGTT 3′ | [82, 85, 86] |
| Type | Features | Comments |
|---|---|---|
| D-type (CpG- A)
| - mixed phosphodiester / phosphorothioate backbone
- 1–2 CpG dinucleotides in a central phosphodiester region flanked on both the 5′ and 3′ ends with phosphorothioate nucleotides - CpG motif is located within a palindromic sequence - run of G’s at 3′ end [88, 93] | - stimulate NK cells to produce IFN-γ [13, 88, 93]
- stimulate pDCs to produce large amounts of IFN-α, IFN-β and TNF-α [13, 88, 93, 94] - stimulate IFN-α and IFN-γ secretion and maturation of human DCs in vitro [100] - induce IFN-α within the first 12 h with considerable amounts still produced at 24–48 h [93] - indirectly activate monocytes to differentiate into myeloid DCs and produce chemokines (IL-10) [88] - trigger the maturation of APCs [88] - do not stimulate B-cells or other subsets of DCs [89] - active in mice [89], nonhuman primates [100] - best activity in humans [13, 88, 93] |
| K-type (CpG- B)
| - phosphorothioate backbone
- multiple TCGT/A motifs[88, 89] | - stimulate strong B-cell and NK cell activation [13]
- activate pDCs to produce IFN-α, -β, IL-6, -8, TNF-α and IP-10 but very little IFN-γ [89, 93, 101] - IFN-α induction is relatively low and limited to the first 12 h [93] - short-lived induction of IFN-γ, IL-6 and TNF-α in BALB/c mice but IL-12 remains elevated for at least 8 d [101] - stimulate B-cells to secrete IL-6, IL-10 and IgM [88, 89, 102] - stimulate cytokine production [13] - induce cell proliferation and IL-6 production from human PBMCs [100] - active in mice [89], human PBMC in vitro [100] - poorly active in primates [89] |
| C-type (CpG- C) | - phosphorothioate backbone
- palindromic sequence - no poly(G) stretch - TCGTCG at the 5′ end - frequently contains a K-type motif (GTCGTT) [13, 88] | - stimulate strong B-cell and NK cell activation [13]
- stimulate pDCs to produce IFN-α [13, 88] - potent Th1 adjuvant [13] - stimulate B-cells to secrete IL-6, IL-10 and IgM [88, 89, 102] |
© 2008 by MDPI This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Christopher, M.E.; Wong, J.P. Broad-Spectrum Drugs Against Viral Agents. Int. J. Mol. Sci. 2008, 9, 1561-1594. https://doi.org/10.3390/ijms9091561
Christopher ME, Wong JP. Broad-Spectrum Drugs Against Viral Agents. International Journal of Molecular Sciences. 2008; 9(9):1561-1594. https://doi.org/10.3390/ijms9091561
Chicago/Turabian StyleChristopher, Mary E., and Jonathan P. Wong. 2008. "Broad-Spectrum Drugs Against Viral Agents" International Journal of Molecular Sciences 9, no. 9: 1561-1594. https://doi.org/10.3390/ijms9091561




