A Study of Complexation-ability of Neutral Schiff Bases to Some Metal Cations
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental
3.1 Extraction Procedure
3.2 Extraction Equilibria
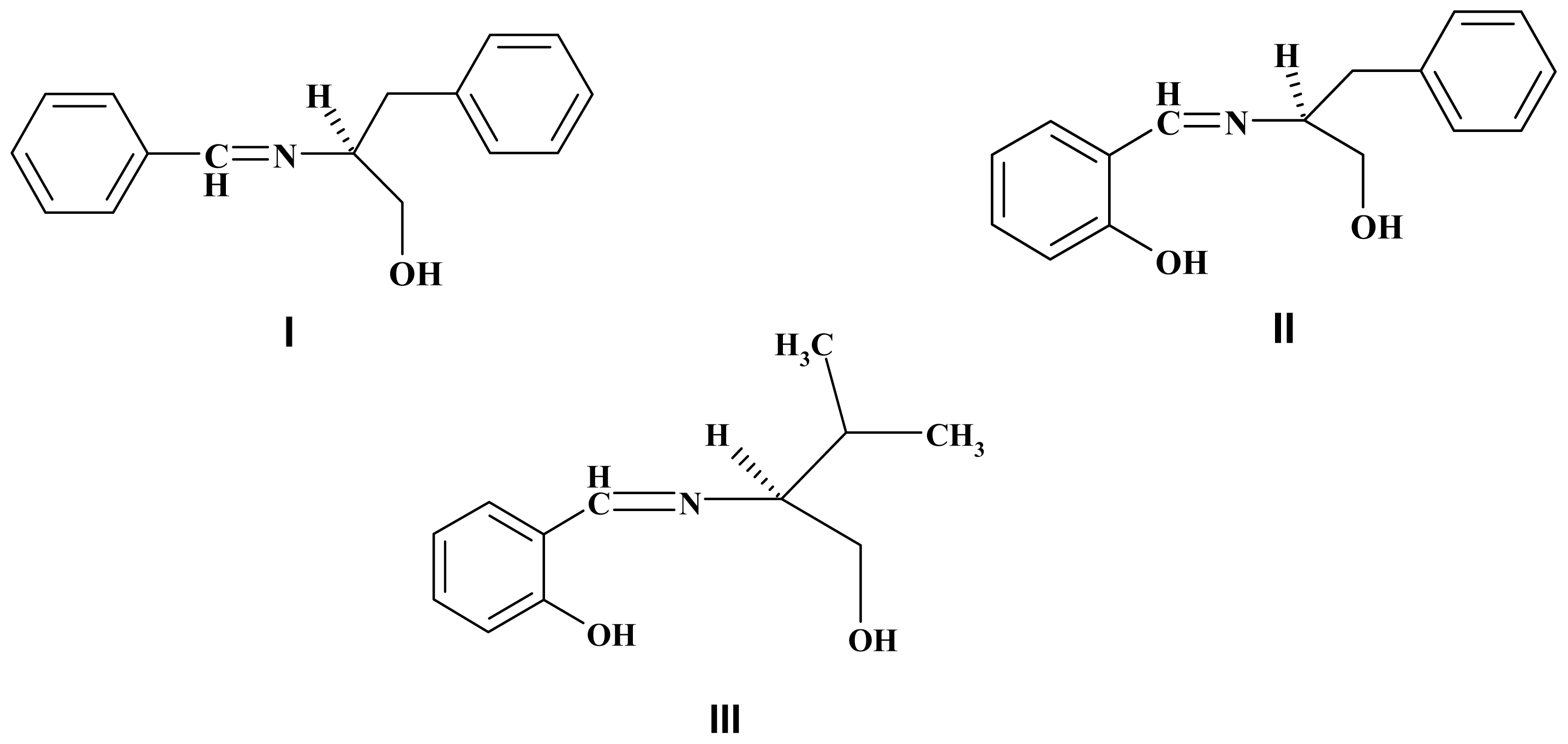

| K′D | VALUE | Chiral Schiff Bases | |||
|---|---|---|---|---|---|
| I | II | III | |||
| NaPar | 1.12* | Log Kext | 9.38 | 10.54 | 11.02 |
| % Ext | 49.62 | 76.00 | 83.01 | ||
| KD,L(a) | 0.98 | 3.17 | 4.88 | ||
| NaPic | 0.02* | Log Kext | — | — | — |
| % Ext | — | — | — | ||
| KD,L(a) | — | — | — | ||
| KPic | 0.09* | Log Kext | — | — | — |
| % Ext | — | — | — | ||
| KD,L(a) | — | — | — | ||
| Cation | I | II | III | |||
|---|---|---|---|---|---|---|
| KD | logKext | KD | logKext | KD | logKext | |
| Ca2+ | 0,20 | 8,24 | - | - | 0,04 | 7,44 |
| Mg2+ | - | - | - | - | - | - |
| Cr2+ | 0,08 | 7,79 | 0,03 | 7,37 | 0,20 | 8,24 |
| Fe2+ | 0,36 | 8,62 | 0,23 | 8,35 | 0,40 | 8,68 |
| Zn2+ | 12,48 | 12,15 | 15,15 | 12,39 | 14,76 | 12,36 |
| Cd2+ | - | - | - | - | 0,04 | 7,44 |
| Ni2+ | 0,82 | 9,23 | 0,73 | 9,13 | 0,88 | 9,29 |
| Mn2+ | - | - | - | - | - | - |
| Co2+ | 0,35 | 8,60 | 0,29 | 8,48 | 0,41 | 8,71 |
References
- Larrow, J.F.; Jacobsen, E.N. Asymmetric processes catalyzed by chiral (salen)metal complexes. Top. Organomet. Chem 2004, 6, 123–152. [Google Scholar]Cozzi, P.G. Metal–Salen Schiff base complexes in catalysis: Practical aspects. Chem. Soc. Rev 2004, 33, 410–421. [Google Scholar]Curini, A.; Epifano, F.; Maltese, F.; Marcotullio, M.C. Novel chiral Schiff base ligands from amino acid amides and salicylaldehyde. Tetrahedron Lett 2002, 43, 3821–3823. [Google Scholar]Mukherjee, P.; Biswas, C.; Drew, M.G.B.; Ghosh, A. Structural variations in Ni(II) complexes of salen type di-Schiff base ligands. Polyhedron 2007, 26, 3121–3128. [Google Scholar]
- Ambroziak, K.; Szypa, M. A synthesis of unsymmetrical chiral salen ligands derived from 2- hydroxynaphthaldehyde and substituted salicylaldehydes. Tetrahedron Lett 2007, 48, 3331–3335. [Google Scholar]Campbell, E.J.; Nguyen, S.T. Unsymmetrical salen-type ligands: High yield synthesis of salentype Schiff bases containing two different benzaldehyde moieties. Tetrahedron Lett 2001, 42, 1221–1225. [Google Scholar]Asadi, M.; Jamshid, K.A.; Kyanfar, A.H. Synthesis, characterization and equilibrium study of the dinuclear adducts formation between nickel(II) Salen-type complexes with diorganotin(IV) dichlorides in chloroform. Inorg. Chim. Acta 2007, 360, 1725–1730. [Google Scholar]Vogt, A.; Wolowiec, S.; Prasad, R.L.; Gupta, A.; Skarlewski, J. Synthesis and characterization of nickel(II), copper(II), manganese(III) and iron(III) complexes with new chiral salen-type ligand N,N′-bis(3,5-di-tert-butylsalicylidene)-(1R,3S)-1,3-diamine-1,2,2-trimethylcyclopentane. Polyhedron 1998, 17, 1231–1240. [Google Scholar]
- Zhang, W.; Jacobsen, E.N. Asymmetric olefin epoxidation with sodium hypochlorite catalyzed by easily prepared chiral Mn(III) salen complex. J. Org. Chem 1991, 56, 2296–2298. [Google Scholar]
- Spero, D.M.; Kapadia, S.R. A Novel method for the asymmetric synthesis of, -disubstituted alkylamines via Grignard additions to ketimines. J. Org. Chem 1997, 62, 5537–5541. [Google Scholar]
- Vetter, A.H.; Berkessel, A. Schiff-base ligands carrying two elements of chirality: Matchedmismatched effects in the vanadium-catalyzed sulfoxidation of thioethers with hydrogen peroxide. Tetrahedron Lett 1998, 39, 1741–1744. [Google Scholar]
- Guo-Fu, Z.; Cheng-Lie, Y. Asymmetric trimethylsilylcyanation of benzaldehyde catalyzed by novel Ti(IV)-chiral Schiff base complexes. J. Mol. Catal. A: Chem 1998, 132, L1–L4. [Google Scholar]Hayashi, M.; Miyamoto, Y.; Inoue, T.; Oguni, N. Enantioselective trimethylsilylcyanation of some aldehydes by chiral titanium Schiff’s base complexes. J. Chem. Soc., Chem. Commun 1991, 1752–1753. [Google Scholar]Hayashi, M.; Miyamoto, Y.; Inoue, T.; Oguni, N. Enantioselective trimethylsilylcyanation of some aldehydes catalyzed by chiral Schiff base-titanium alkoxide complexes. J. Org. Chem 1993, 58, 1515–1522. [Google Scholar]
- Shi, M.; Wang, C.J. Axially dissymmetric binaphthyldiimine chiral Salen-type ligands for catalytic asymmetric addition of diethylzinc to aldehyde. Tetrahedron: Asymmetry 2002, 13, 2161–2166. [Google Scholar]Kureshy, R.I.; Khan, N.H.; Abdi, S.H.R.; Patel, S.T.; Iyer, P.; Suresh, E.; Dastidar, P.I. Chiral Ni(II) Schiff base complex-catalysed enantioselective epoxidation of prochiral non-functionalised alkenes. J. Mol. Catal. A: Chem 2000, 160, 217–227. [Google Scholar]Yang, Z.-H.; Wang, L.; Zhou, Z.; Zhou, Q.; Tang, C. Synthesis of new chiral Schiff bases and their application in the asymmetric trimethylsilylcyanation of aromatic aldehydes. Tetrahedron: Asymmetry 2001, 12, 1579–1582. [Google Scholar]
- Temel, H.; Hosgoren, H. New Cu(II), Mn(III), Ni(II) and Zn(II) complexes with chiral quadridentate Schiff base. Transition Metal Chemistry 2002, 27, 609–612. [Google Scholar]Temel, H.; Ilhan, S.; Sekerci, M.; Ziyadanogullari, R. Synthesis and characterization of new Cu(II), Ni(II), Co(II) and Zn(II) complexes with Schiff base. Spectrosc. Letters 2002, 35, 219–228. [Google Scholar]Temel, H.; Sekerci, M. Novel complexes of Manganese(II), Cobalt(II), Copper(II) and Zinc(II) with Schiff base derived from 1,2-bis-(p-amino-phenoxy)ethane and salicylaldehyde. Synth. React. Inorg. Met.-Org. Chem 2001, 31, 849–857. [Google Scholar]Temel, H.; Cakir, U.; Otludil, B.; Ugras, H.I. Synthesis, spectral and biological studies of Mn(II), Ni(II), Cu(II) and Zn(II) complexes with tetradentate Schiff base ligand. Complexation studies and the determination of stability constant (Ke). Synth. React. Inorg. Met.-Org. Chem 2001, 31, 1323–1337. [Google Scholar]Temel, H.; Cakir, U.; Ugras, H.I. Synthesis and characterization of Zn(II), Cu(II) and Ni(II) complexes with bidentate Schiff base ligands. Complexation studies and the determination of stability constant (Ke). Russ. J. Inorg. Chem 2001, 46, 2022–2026. [Google Scholar]
- Hayashi, M.; Inoue, T.; Miyamoto, Y.; Oguni, N. Asymmetric carbon---carbon bond forming reactions catalyzed by chiral Schiff base—titanium alkoxide complexes. Tetrahedron 1994, 50, 4385–4398. [Google Scholar]Hayashi, M.; Inoue, T.; Oguni, N. Novel enantioselective reaction of diketene with aldehydes promoted by chiral Schiff base–titanium alkoxide complex. J. Chem. Soc., Chem. Commun 1994, 341–342. [Google Scholar]
- Bermejo, M.R.; Sousa, A.; Garcia-Deibe, A.; Maneiro, M.; Sanmartin, M.; Fondo, J. Synthesis and characterization of new manganese(III) complexes with asymmetrical onsn Schiff bases. Polyhedron 1999, 18, 511–518. [Google Scholar]
- Cakir, U.; Cicek, B. Extraction-ability and -selectivity of tetra-aza-crown ethers for transition metal cations. Transition Metal Chemistry 2004, 29, 263–268. [Google Scholar]Cakir, U.; Ozer, M.; Icen, M. A.; Ugras, H.I.; Bulut, M. Synthesis of some 3-phenyl chromenone-crown ethers and equilibrium studies on complexation with ion-pair extraction of sodium and potassium dyes. Dyes and Pigments 2004, 60, 177–185. [Google Scholar]Ziyadanogullari, B.; Topal, G.; Erdogan, S.; Hamamci, C.; Hosgoren, H. Effect of structural modifications of diaza-18-crown-6 on the extractability and selectivity of univalent metal picrates. Talanta 2001, 53, 1083–1087. [Google Scholar]
- Takeda, Y.; Endo, K.; Katsuta, S.; Ouichi, M. Extraction of sodium and potassium picrates with 15-(2,5-dioxahexyl)-15-methyl-16-crown-5 (lariat 16C5) into various organic solvents. Elucidation of fundamental equilibria which determine the extraction ability for Na+ and K+ and the selectivity. Talanta 2001, 54, 575–584. [Google Scholar]
- Katsuta, S.; Tsuchiya, F.; Takeda, Y. Equilibrium studies on complexation in water and solvent extraction of zinc(II) and cadmium(II) with benzo-18-crown-6. Talanta 2000, 51, 637–644. [Google Scholar]
- Takeda, Y.; Kawarabayashi, A.; Takahashi, K.; Kudo, Y. Extraction of alkali metal picrates with benzo-18-crown-6 into various organic solvents. Fundamental equilibria which govern the highest extraction selectivity for K+ among alkali metal ions. Bull. Chem Soc Jpn 1995, 68, 1309–1314. [Google Scholar]
- Takeda, Y.; Kawarabayashi, A.; Endo, K.; Yahata, T.; Kudo, Y.; Katsuta, S. Solvent extraction of alkali metal (Li-Cs) picrates with 18-crown-6 into various diluents. Elucidation of fundamental equilibria which govern the extraction-ability and –selectivity. Analytical Science 1998, 14, 215– 224. [Google Scholar]
- Kudo, Y.; Usami, J.; Katsuta, S.; Takeda, Y. Solvent extraction of permanganates (Na, K) by 18-crown-6 ether from water into 1,2-dichloroethane: Elucidation of an extraction equilibrium based on component equilibria. Talanta 2003, 59, 1213–1218. [Google Scholar]
- Cakir, U.; Cicek, B.; Yildiz, Y.K.; Alkan, M. Solvent effect upon ion–pair extraction of different sodium dyes using some crown ethers. J. Inc. Phenom 1999, 34, 153–165. [Google Scholar]
- Gokel, G.W. Lariat ethers: From simple sidearms to supramolecular systems. Chem. Soc. Rev 1992, 21, 39–47. [Google Scholar]
- Inoue, Y.; Gokel, G.W. (Eds.) Cation Binding by Macrocycles; Marcel Dekker: C.J. Basel, 1990.
- Sachleben, R.A.; Deng, Y.P.; Bailey, D.R.; Moyer, B.A. Ring-size and substituent effects in the solvent extraction of alkali metal nitrates by crown ethers in 1,2-dichloroethane and 1–octanol. Solvent Ext Ion Exchange 1996, 14, 995–1015. [Google Scholar]
- Yoshio, M.; Noguchi, H. Crown ethers for chemical analysis: A Review. Anal. Lett 1982, 15, 1197–1276. [Google Scholar]
- Tumerdem, R.; Topal, G.; Turgut, Y. Asymmetric reduction of acetophenone using lithium aluminium hydride modified with some novel amino alcohol Schiff bases. Tetrahedron: Asymmetry 2005, 16, 865–868. [Google Scholar]
- Takeda, Y.; Kato, H. The solvent extraction of bivalent metal picrates by 15-crown-5, 18-crown-6, and dibenzo-18-crown-6. Bull. Chem. Soc. Jpn 1979, 52, 1027–1030. [Google Scholar]
- Takeda, Y.; Takagi, C. Solvent effects on extraction of sodium picrate with 15-crown-5. The determination of ion-pair formation constant of crown ether–metal salt 1:1:1 complex in water. Bull. Chem. Soc. Jpn 1994, 67, 56–60. [Google Scholar]
- Kolthoff, I.M.; Chantooni, M.K. Crown ether complexed alkali metal picrate ion pairs in watersaturated dichloromethane as studied by electrolytic conductance and by partitioning into water. Effect of lithium chloride on partitioning. J. Chem. Eng. Data 1997, 42, 49–53. [Google Scholar]
© 2007 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Topal, G.; Tümerdem, R.; Basaran, I.; Gümüş, A.; Cakir, U. A Study of Complexation-ability of Neutral Schiff Bases to Some Metal Cations. Int. J. Mol. Sci. 2007, 8, 933-942. https://doi.org/10.3390/i8090933
Topal G, Tümerdem R, Basaran I, Gümüş A, Cakir U. A Study of Complexation-ability of Neutral Schiff Bases to Some Metal Cations. International Journal of Molecular Sciences. 2007; 8(9):933-942. https://doi.org/10.3390/i8090933
Chicago/Turabian StyleTopal, Giray, Recep Tümerdem, Ismet Basaran, Arzu Gümüş, and Umit Cakir. 2007. "A Study of Complexation-ability of Neutral Schiff Bases to Some Metal Cations" International Journal of Molecular Sciences 8, no. 9: 933-942. https://doi.org/10.3390/i8090933




