Effects of Co-Culture Media on Hepatic Differentiation of hiPSC with or without HUVEC Co-Culture
Abstract
:1. Introduction
2. Results
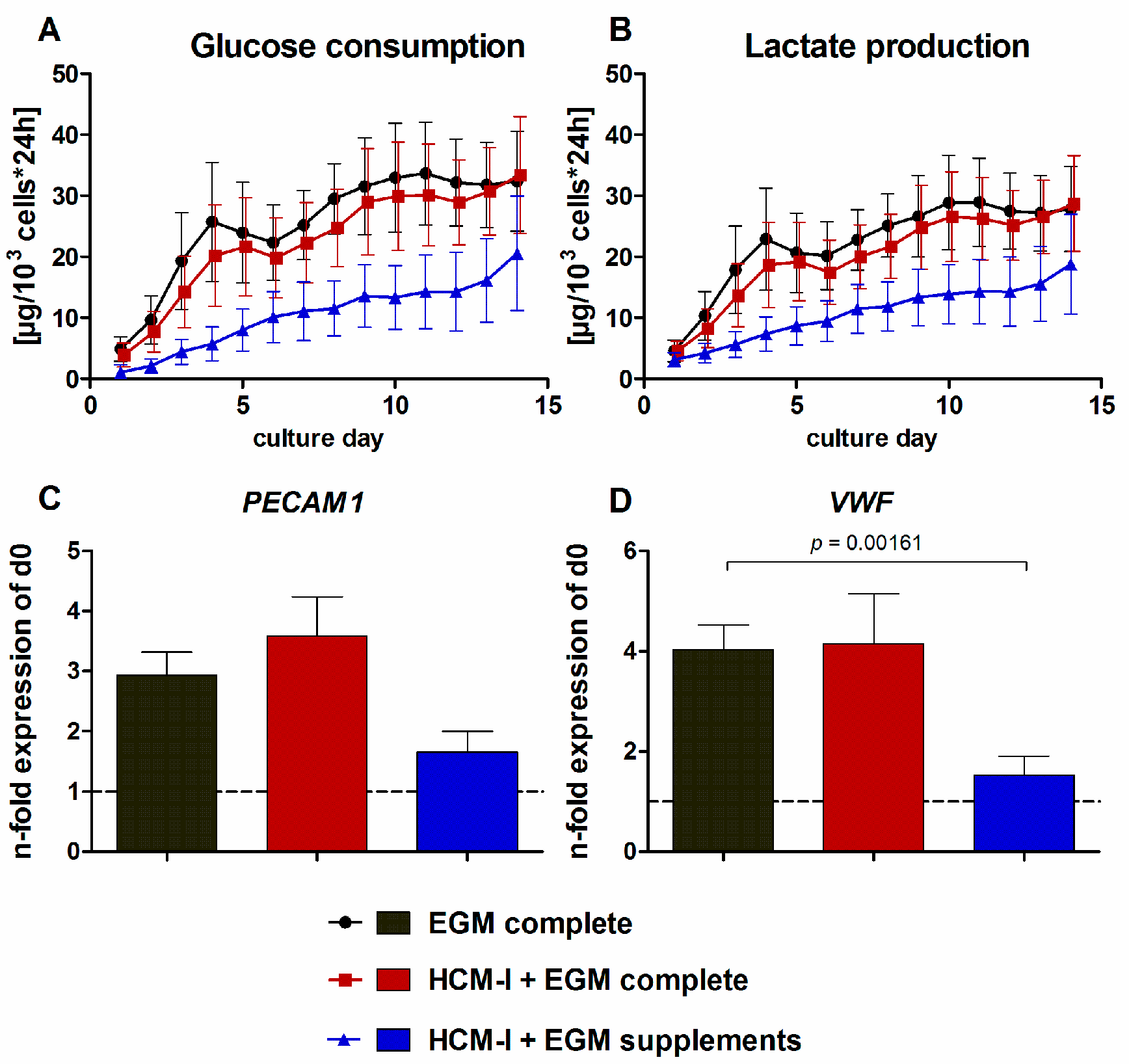
2.1. Effect of Culture Media Variations on Mono-Cultures of Human Umbilical Vein Endothelial Cells (HUVEC)
2.2. Hepatic Differentiation of hiPSC-Derived Definitive Endoderm (DE) Cells with or without HUVEC Co-Cultivation Using Different Co-Culture Media
2.2.1. Morphological Characteristics of hiPSC-Derived Hepatocyte-like Cells (HLC) and Co-Cultured HUVEC
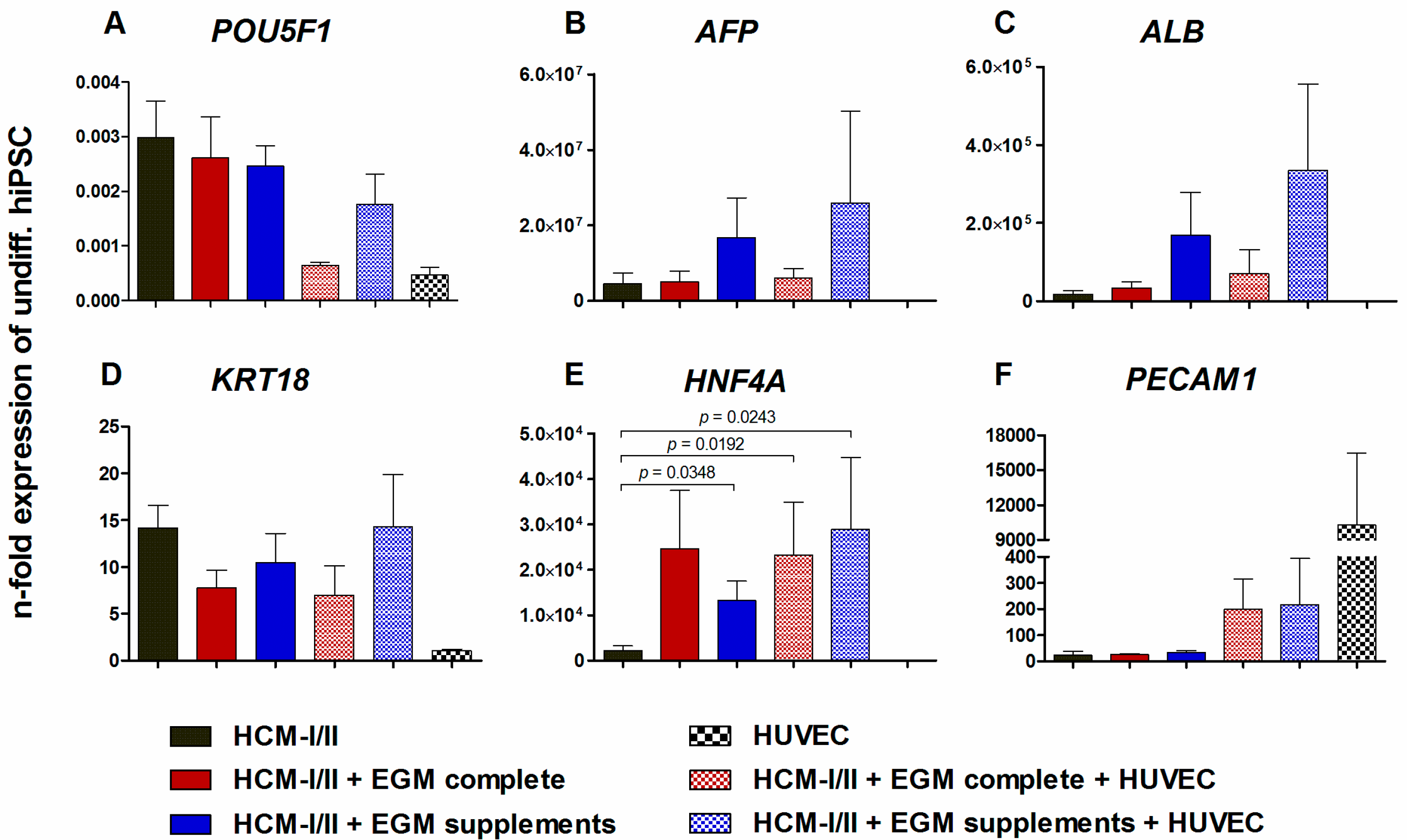
2.2.2. Gene Expression of Stage-Specific and Endothelial Cell Markers
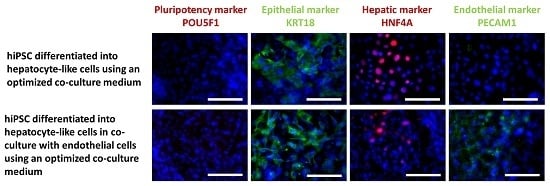
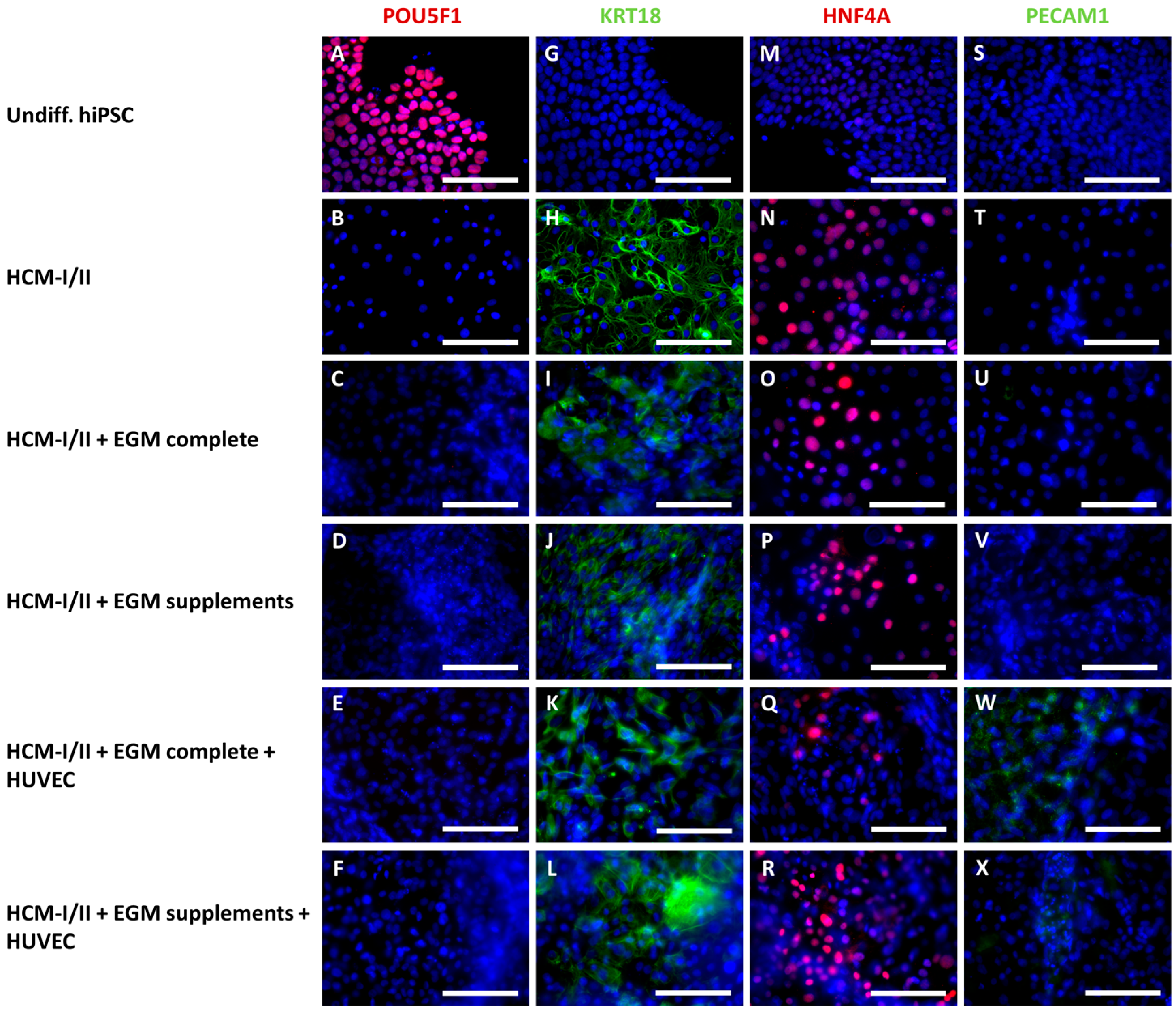
2.2.3. Immunocytochemical Analysis of Stage-Specific and Endothelial Cell Markers
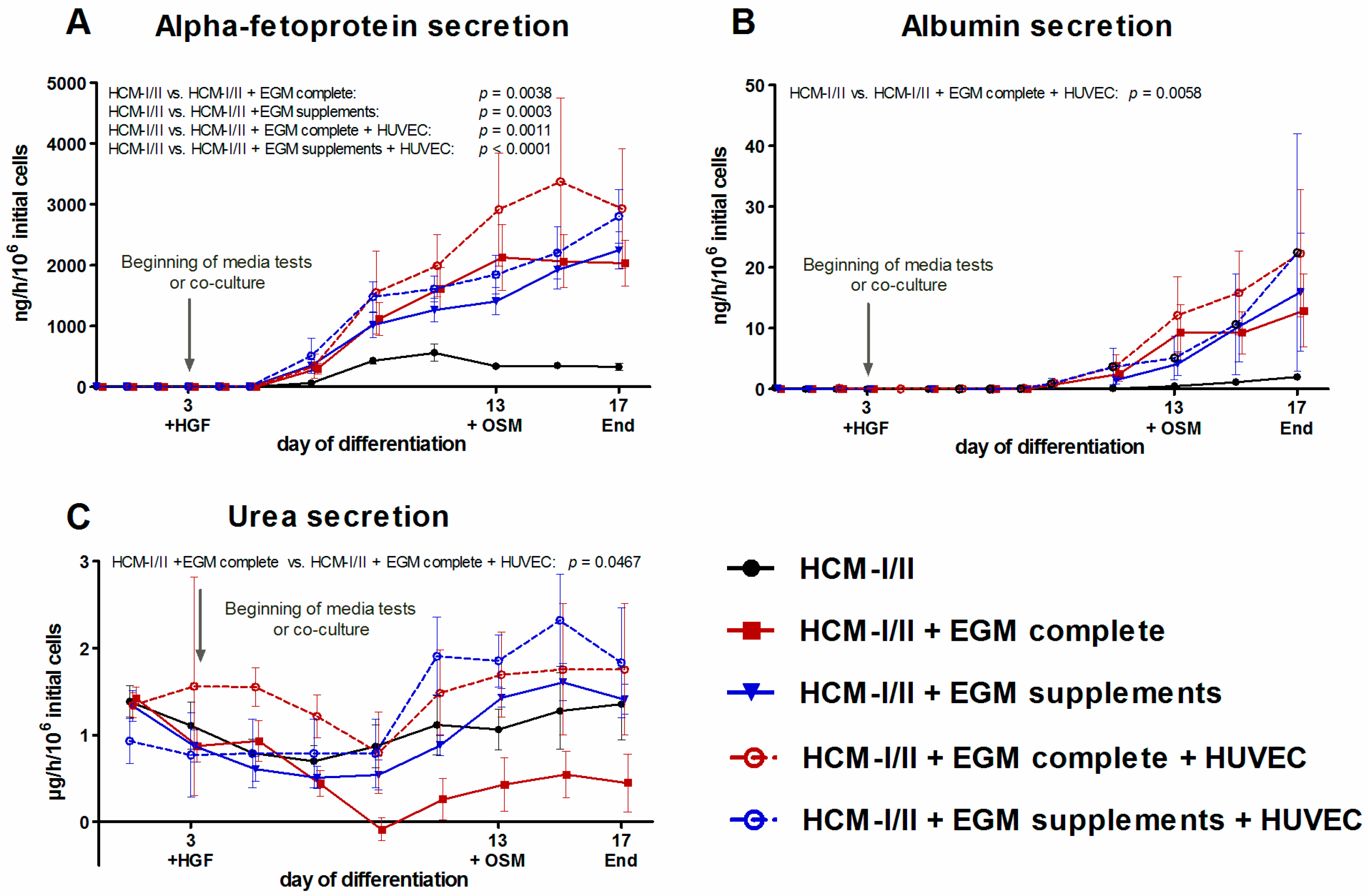
2.2.4. Secretion of α-Fetoprotein (AFP), Albumin and Urea
2.2.5. Functional Analysis of Different Cytochrome P450 (CYP) Isoenzymes
3. Discussion
4. Materials and Methods
4.1. Culture of HUVEC
4.2. Culture and Hepatic Differentiation of hiPSC
4.3. Culture Medium Testing in Mono-Cultures of HUVEC
4.4. Co-Culture of hiPSC-Derived DE cells with HUVEC
4.5. Analyses of Biochemical Parameters
4.6. Gene Expression Analysis
4.7. Immunocytochemical Staining
4.8. Measurement of Cytochrome P450 (CYP) Isoenzyme Activities
4.9. Statistical Evaluation
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AFP | α-fetoprotein |
| ALB | Albumin |
| bFGF | Basic fibroblast growth factor |
| BMP | Bone morphogenetic proteins |
| CYP | Cytochrome P450 |
| DE | Definitive endoderm |
| DMEM | Dulbecco’s modified eagle’s medium |
| DMSO | Dimethyl sulfoxide |
| ECGF | Endothelial cell growth factor |
| EGF | Epidermal growth factor |
| EGM | Endothelial cell growth medium |
| FBS | Fetal bovine serum |
| FGF | Fibroblast growth factor |
| GAPDH | Glyceraldehyde-3-phosphate dehydrogenase |
| HCM | Hepatocyte culture medium |
| hESC | Human embryonic stem cells |
| HGF | hepatocyte growth factor |
| hiPSC | Human induced pluripotent stem cells |
| HLC | Hepatocyte-like cells |
| HNF4A | Hepatocyte nuclear factor 4 α |
| HUVEC | Human umbilical vein endothelial cells |
| KRT18 | Cytokeratin 18 |
| L15 | Leibovitz’s |
| LDH | Lactate dehydrogenase |
| OSM | Oncostatin M |
| PECAM1 | Platelet and endothelial cell adhesion molecule 1 |
| POU5F1 | POU domain, class 5, transcription factor 1 |
| RPMI | Roswell Park Memorial Institute |
| VWF | Von Willebrand factor |
References
- Yi, F.; Liu, G.H.; Izpisua Belmonte, J.C. Human induced pluripotent stem cells derived hepatocytes: Rising promise for disease modeling, drug development and cell therapy. Protein Cell 2012, 4, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Passier, R.; Orlova, V.; Mummery, C. Complex tissue and disease modeling using hiPSCs. Cell Stem Cell 2016, 18, 309–321. [Google Scholar] [CrossRef] [PubMed]
- Suter-Dick, L.; Alves, P.M.; Blaauboer, B.J.; Bremm, K.D.; Brito, C.; Coecke, S.; Flick, B.; Fowler, P.; Hescheler, J.; Ingelman-Sundberg, M.; et al. Stem cell-derived systems in toxicology assessment. Stem Cells Dev. 2015, 24, 1284–1296. [Google Scholar] [CrossRef] [PubMed]
- Larrey, D. Epidemiology and individual susceptibility to adverse drug reactions affecting the liver. Semin. Liver Dis. 2002, 22, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Sgro, C.; Clinard, F.; Ouazir, K.; Chanay, H.; Allard, C.; Guilleminet, C.; Lenoir, C.; Lemoine, A.; Hillon, P. Incidence of drug-induced hepatic injuries: A French population-based study. Hepatology 2002, 36, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Takayama, K.; Kawabata, K.; Nagamoto, Y.; Kishimoto, K.; Tashiro, K.; Sakurai, F.; Tachibana, M.; Kanda, K.; Hayakawa, T.; Furue, M.K.; et al. 3D spheroid culture of hESC/hiPSC-derived hepatocyte-like cells for drug toxicity testing. Biomaterials 2013, 34, 1781–1789. [Google Scholar] [CrossRef] [PubMed]
- Ware, B.R.; Berger, D.R.; Khetani, S.R. Prediction of drug-induced liver injury in Micropatterned Co-cultures Containing iPSC-Derived Human Hepatocytes. Toxicol. Sci. 2015, 145, 252–262. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Liu, H.; Ikeda, Y.; Amiot, B.P.; Rinaldo, P.; Duncan, S.A.; Nyberg, S.L. Hepatocyte-like cells differentiated from human induced pluripotent stem cells: Relevance to cellular therapies. Stem Cell Res. 2012, 9, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Cai, J.; Liu, Y.; Zhao, D.; Yong, J.; Duo, S.; Song, X.; Guo, Y.; Zhao, Y.; Qin, H.; et al. Efficient generation of hepatocyte-like cells from human induced pluripotent stem cells. Cell Res. 2009, 19, 1233–1242. [Google Scholar] [CrossRef] [PubMed]
- Si-Tayeb, K.; Noto, F.K.; Nagaoka, M.; Li, J.; Battle, M.A.; Duris, C.; North, P.E.; Dalton, S.; Duncan, S.A. Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells. Hepatology 2010, 51, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Baxter, M.; Withey, S.; Harrison, S.; Segeritz, C.P.; Zhang, F.; Atkinson-Dell, R.; Rowe, C.; Gerrard, D.T.; Sison-Young, R.; Jenkins, R.; et al. Phenotypic and functional analyses show stem cell-derived hepatocyte-like cells better mimic fetal rather than adult hepatocytes. J. Hepatol. 2015, 62, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Takayama, K.; Mizuguchi, H. Generation of human pluripotent stem cell-derived hepatocyte-like cells for drug toxicity screening. Drug Metab. Pharmacokinet. 2017, 32, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, G.J.; Hay, D.C.; Park, I.H.; Fletcher, J.; Hannoun, Z.; Payne, C.M.; Dalgetty, D.; Black, J.R.; Ross, J.A.; Samuel, K.; et al. Generation of functional human hepatic endoderm from human induced pluripotent stem cells. Hepatology 2010, 51, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Vosough, M.; Omidinia, E.; Kadivar, M.; Shokrgozar, M.A.; Pournasr, B.; Aghdami, N.; Baharvand, H. Generation of functional hepatocyte-like cells from human pluripotent stem cells in a scalable suspension culture. Stem Cells Dev. 2013, 22, 2693–2705. [Google Scholar] [CrossRef] [PubMed]
- Hay, D.C.; Fletcher, J.; Payne, C.; Terrace, J.D.; Gallagher, R.C.; Snoeys, J.; Black, J.R.; Wojtacha, D.; Samuel, K.; Hannoun, Z.; et al. Highly efficient differentiation of hESCs to functional hepatic endoderm requires ActivinA and Wnt3a signaling. Proc. Natl. Acad. Sci. USA 2008, 105, 12301–12306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cai, J.; Zhao, Y.; Liu, Y.; Ye, F.; Song, Z.; Qin, H.; Meng, S.; Chen, Y.; Zhou, R.; Song, X.; et al. Directed differentiation of human embryonic stem cells into functional hepatic cells. Hepatology 2007, 45, 1229–1239. [Google Scholar] [CrossRef] [PubMed]
- Brolén, G.; Sivertsson, L.; Björquist, P.; Eriksson, G.; Ek, M.; Semb, H.; Johansson, I.; Andersson, T.B.; Ingelman-Sundberg, M.; Heins, N. Hepatocyte-like cells derived from human embryonic stem cells specifically via definitive endoderm and a progenitor stage. J. Biotechnol. 2010, 145, 284–294. [Google Scholar] [CrossRef] [PubMed]
- Hay, D.C.; Zhao, D.; Fletcher, J.; Hewitt, Z.A.; McLean, D.; Urruticoechea-Uriguen, A.; Black, J.R.; Elcombe, C.; Ross, J.A.; Wolf, R.; et al. Efficient differentiation of hepatocytes from human embryonic stem cells exhibiting markers recapitulating liver development in vivo. Stem Cells 2008, 26, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Takayama, K.; Inamura, M.; Kawabata, K.; Katayama, K.; Higuchi, M.; Tashiro, K.; Nonaka, A.; Sakurai, F.; Hayakawa, T.; Furue, M.K.; et al. Efficient generation of functional hepatocytes from human embryonic stem cells and induced pluripotent stem cells by HNF4α transduction. Mol. Ther. 2012, 20, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Doddapaneni, R.; Chawla, Y.K.; Das, A.; Kalra, J.K.; Ghosh, S.; Chakraborti, A. Overexpression of microRNA-122 enhances in vitro hepatic differentiation of fetal liver-derived stem/progenitor cells. J. Cell Biochem. 2013, 114, 1575–1583. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.G.; Qiu, R.L.; Wu, Y.H.; Li, Z.X.; Xie, P.; Zhang, J.; Zhou, J.J.; Zeng, L.X.; Tang, J.; Maharjan, A.; et al. Overexpression of miR-122 promotes the hepatic differentiation and maturation of mouse ESCs through a miR-122/FoxA1/HNF4a-positive feedback loop. Liver Int. 2014, 34, 281–295. [Google Scholar] [CrossRef] [PubMed]
- Gieseck, R.L., III; Hannan, N.R.; Bort, R.; Hanley, N.A.; Drake, R.A.; Cameron, G.W.; Wynn, T.A.; Vallier, L. Maturation of induced pluripotent stem cell derived hepatocytes by 3D-culture. PLoS ONE 2014, 9, e86372. [Google Scholar] [CrossRef] [PubMed]
- Freyer, N.; Knöspel, F.; Strahl, N.; Amini, L.; Schrade, P.; Bachmann, S.; Damm, G.; Seehofer, D.; Jacobs, F.; Monshouwer, M.; et al. Hepatic differentiation of human induced pluripotent stem cells in a perfused three-dimensional multicompartment bioreactor. BioRes. Open Access 2016, 5, 235–248. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Yoshitomi, H.; Rossant, J.; Zaret, K.S. Liver organogenesis promoted by endothelial cells prior to vascular function. Science 2001, 294, 559–563. [Google Scholar] [CrossRef] [PubMed]
- Takebe, T.; Sekine, K.; Enomura, M.; Koike, H.; Kimura, M.; Ogaeri, T.; Zhang, R.R.; Ueno, Y.; Zheng, Y.W.; Koike, N.; et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 2013, 499, 481–484. [Google Scholar] [CrossRef] [PubMed]
- Godoy, P.; Schmidt-Heck, W.; Natarajan, K.; Lucendo-Villarin, B.; Szkolnicka, D.; Asplund, A.; Björquist, P.; Widera, A.; Stöber, R.; Campos, G.; et al. Gene networks and transcription factor motifs defining the differentiation of stem cells into hepatocyte-like cells. J. Hepatol. 2015, 63, 934–942. [Google Scholar] [CrossRef] [PubMed]
- Maciag, T.; Cerundolo, J.; Ilsley, S.; Kelley, P.R.; Forand, R. An endothelial cell growth factor from bovine hypothalamus: Identification and partial characterization. Proc. Natl. Acad. Sci. USA 1979, 76, 5674–5678. [Google Scholar] [CrossRef] [PubMed]
- Thornton, S.C.; Mueller, S.N.; Levine, E.M. Human endothelial cells: Use of heparin in cloning and long-term serial cultivation. Science 1983, 222, 623–625. [Google Scholar] [CrossRef] [PubMed]
- Spivak-Kroizman, T.; Lemmon, M.A.; Dikic, I.; Ladbury, J.E.; Pinchasi, D.; Huang, J.; Jaye, M.; Crumley, G.; Schlessinger, J.; Lax, I. Heparin-induced oligomerization of FGF molecules is responsible for FGF receptor dimerization, activation, and cell proliferation. Cell 1994, 79, 1015–1024. [Google Scholar] [CrossRef]
- Kang, S.S.; Gosselin, C.; Ren, D.; Greisler, H.P. Selective stimulation of endothelial cell proliferation with inhibition of smooth muscle cell proliferation by fibroblast growth factor-1 plus heparin delivered from fibrin glue suspensions. Surgery 1995, 118, 280–286. [Google Scholar] [CrossRef]
- Montagnani, M.; Golovchenko, I.; Kim, I.; Koh, G.Y.; Goalstone, M.L.; Mundhekar, A.N.; Johansen, M.; Kucik, D.F.; Quon, M.J.; Draznin, B. Inhibition of phosphatidylinositol 3-kinase enhances mitogenic actions of insulin in endothelial cells. J. Biol. Chem. 2002, 277, 1794–1799. [Google Scholar] [CrossRef] [PubMed]
- Carlevaro, M.F.; Albini, A.; Ribatti, D.; Gentili, C.; Benelli, R.; Cermelli, S.; Cancedda, R.; Cancedda, F.D. Transferrin promotes endothelial cell migration and invasion: implication in cartilage neovascularization. J. Cell. Biol. 1997, 136, 1375–1384. [Google Scholar] [CrossRef] [PubMed]
- Kotamraju, S.; Chitambar, C.R.; Kalivendi, S.V.; Joseph, J.; Kalyanaraman, B. Transferrin receptor-dependent iron uptake is responsible for doxorubicin-mediated apoptosis in endothelial cells: Role of oxidant-induced iron signaling in apoptosis. J. Biol. Chem. 2002, 277, 17179–17187. [Google Scholar] [CrossRef] [PubMed]
- Dhar-Mascareño, M.; Cárcamo, J.M.; Golde, D.W. Hypoxia-reoxygenation-induced mitochondrial damage and apoptosis in human endothelial cells are inhibited by vitamin C. Free Radic. Biol. Med. 2005, 38, 1311–1322. [Google Scholar] [CrossRef] [PubMed]
- Piconi, L.; Quagliaro, L.; Assaloni, R.; Da Ros, R.; Maier, A.; Zuodar, G.; Ceriello, A. Constant and intermittent high glucose enhances endothelial cell apoptosis through mitochondrial superoxide overproduction. Diabetes Metab. Res. Rev. 2006, 22, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Gong, J.; Zhang, J.; Yamada, Y.; Borgeld, H.J.; Yagi, K. Mitochondrial genotype associated with longevity and its inhibitory effect on mutagenesis. Mech. Ageing Dev. 2000, 116, 65–76. [Google Scholar] [CrossRef]
- Ameri, J.; Ståhlberg, A.; Pedersen, J.; Johansson, J.K.; Johannesson, M.M.; Artner, I.; Semb, H. FGF2 specifies hESC-derived definitive endoderm into foregut/midgut cell lineages in a concentration-dependent manner. Stem Cells 2010, 28, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Touboul, T.; Hannan, N.R.; Corbineau, S.; Martinez, A.; Martinet, C.; Branchereau, S.; Mainot, S.; Strick-Marchand, H.; Pedersen, R.; Di Santo, J.; et al. Generation of functional hepatocytes from human embryonic stem cells under chemically defined conditions that recapitulate liver development. Hepatology 2010, 51, 1754–1765. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Jang, Y.J.; An, S.Y.; Son, J.; Lee, J.; Lee, G.; Park, J.Y.; Park, H.J.; Hwang, D.Y.; Kim, J.H.; et al. Enhanced Metabolizing Activity of Human ES Cell-Derived Hepatocytes Using a 3D Culture System with Repeated Exposures to Xenobiotics. Toxicol. Sci. 2015, 147, 190–206. [Google Scholar] [CrossRef] [PubMed]
- Tasnim, F.; Phan, D.; Toh, Y.C.; Yu, H. Cost-effective differentiation of hepatocyte-like cells from human pluripotent stem cells using small molecules. Biomaterials 2015, 70, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Stevens, B.; Chang, J.; Milbrandt, J.; Barres, B.A.; Hell, J.W. NS21: Re-defined and modified supplement B27 for neuronal cultures. J. Neurosci. Methods. 2008, 171, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; DeLaForest, A.; Fisher, J.; Urick, A.; Wagner, T.; Twaroski, K.; Cayo, M.; Nagaoka, M.; Duncan, S.A. Protocol for Directed Differentiation of Human Pluripotent Stem Cells toward a Hepatocyte Fate. StemBook. 2012. Available online: http://www.ncbi.nlm.nih.gov/books/NBK133278/PubMed (accessed on 2 August 2017).
- DeLaForest, A.; Nagaoka, M.; Si-Tayeb, K.; Noto, F.K.; Konopka, G.; Battle, M.A.; Duncan, S.A. HNF4A is essential for specification of hepatic progenitors from human pluripotent stem cells. Development 2011, 138, 4143–4153. [Google Scholar] [CrossRef] [PubMed]
- Dean, S.; Tang, J.I.; Seckl, J.R.; Nyirenda, M.J. Developmental and tissue-specific regulation of hepatocyte nuclear factor 4-alpha (HNF4-alpha) isoforms in rodents. Gene Expr. 2010, 14, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, A.; Katsura, S.; Ito, R.; Hashiba, W.; Sekine, H.; Fujiki, R.; Kato, S. Multiple post-translational modifications in hepatocyte nuclear factor 4α. Biochem. Biophys. Res. Commun. 2011, 410, 749–753. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Jiménez, C.P.; Castell, J.V.; Gómez-Lechón, M.J.; Jover, R. Transcriptional activation of CYP2C9, CYP1A1, and CYP1A2 by hepatocyte nuclear factor 4alpha requires coactivators peroxisomal proliferator activated receptor-gamma coactivator 1alpha and steroid receptor coactivator 1. Mol. Pharmacol. 2006, 70, 1681–1692. [Google Scholar] [CrossRef] [PubMed]
- Tirona, R.G.; Lee, W.; Leake, B.F.; Lan, L.B.; Cline, C.B.; Lamba, V.; Parviz, F.; Duncan, S.A.; Inoue, Y.; Gonzalez, F.J.; et al. The orphan nuclear receptor HNF4alpha determines PXR- and CAR-mediated xenobiotic induction of CYP3A4. Nat. Med. 2003, 9, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Pascussi, J.M.; Gerbal-Chaloin, S.; Drocourt, L.; Maurel, P.; Vilarem, M.J. The expression of CYP2B6, CYP2C9 and CYP3A4 genes: A tangle of networks of nuclear and steroid receptors. Biochim. Biophys. Acta 2003, 1619, 243–253. [Google Scholar] [CrossRef]
- Matsumoto, K.; Nakamura, T. Heparin functions as a hepatotrophic factor by inducing production of hepatocyte growth factor. Biochem. Biophys. Res. Commun. 1996, 227, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Behbahan, I.S.; Duan, Y.; Lam, A.; Khoobyari, S.; Ma, X.; Ahuja, T.P.; Zern, M.A. New approaches in the differentiation of human embryonic stem cells and induced pluripotent stem cells toward hepatocytes. Stem Cell Rev. 2011, 7, 748–759. [Google Scholar] [CrossRef] [PubMed]
- Kang, L.I.; Mars, W.M.; Michalopoulos, G.K. Signals and cells involved in regulating liver regeneration. Cells 2012, 1, 1261–1292. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Qu, X.; Zhu, W.; Li, Y.S.; Yuan, S.; Zhang, H.; Liu, J.; Wang, P.; Lai, C.S.; Zanella, F.; et al. Deterministically patterned biomimetic human iPSC-derived hepatic model via rapid 3D bioprinting. Proc. Natl. Acad. Sci. USA 2016, 113, 2206–2211. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Nishida, T. Differential effects of epidermal growth factor and interleukin 6 on corneal epithelial cells and vascular endothelial cells. Cornea 1999, 18, 452–458. [Google Scholar] [CrossRef] [PubMed]
- Gentilini, G.; Kirschbaum, N.E.; Augustine, J.A.; Aster, R.H.; Visentin, G.P. Inhibition of human umbilical vein endothelial cell proliferation by the CXC chemokine, platelet factor 4 (PF4), is associated with impaired downregulation of p21(Cip1/WAF1). Blood 1999, 93, 25–33. [Google Scholar] [PubMed]
- Du, C.; Narayanan, K.; Leong, M.F.; Wan, A.C. Induced pluripotent stem cell-derived hepatocytes and endothelial cells in multi-component hydrogel fibers for liver tissue engineering. Biomaterials 2014, 35, 6006–6014. [Google Scholar] [CrossRef] [PubMed]
- Javed, M.S.; Yaqoob, N.; Iwamuro, M.; Kobayashi, N.; Fujiwara, T. Generation of hepatocyte-like cells from human induced pluripotent stem (iPS) cells by co-culturing embryoid body cells with liver non-parenchymal cell line TWNT-1. J. Coll. Physicians Surg. Pak. 2014, 24, 91–96. [Google Scholar] [PubMed]
- Berger, D.R.; Ware, B.R.; Davidson, M.D.; Allsup, S.R.; Khetani, S.R. Enhancing the functional maturity of induced pluripotent stem cell-derived human hepatocytes by controlled presentation of cell-cell interactions in vitro. Hepatology 2015, 61, 1370–1381. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Yasuchika, K.; Fukumitsu, K.; Kawamoto, T.; Kawamura-Saitoh, M.; Amagai, Y.; Ikai, I.; Uemoto, S.; Kawase, E.; Suemori, H.; et al. In vitro hepatic maturation of human embryonic stem cells by using a mesenchymal cell line derived from murine fetal livers. Cell Tissue Res. 2010, 339, 505–512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, W.; Lu, Y.C.; Frankel, A.S.; An, D.; Schwartz, R.E.; Ma, M. Engraftment of human induced pluripotent stem cell-derived hepatocytes in immunocompetent mice via 3D co-aggregation and encapsulation. Sci. Rep. 2015, 5, 16884. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.D.; Kim, K.H.; Lee, S.G.; Choi, S.Y.; Kim, Y.C.; Byun, K.S.; Cha, I.H.; Park, K.Y.; Cho, C.H.; Choi, D.H. Hepatic differentiation from human embryonic stem cells using stromal cells. J. Surg. Res. 2011, 170, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Nagamoto, Y.; Tashiro, K.; Takayama, K.; Ohashi, K.; Kawabata, K.; Sakurai, F.; Tachibana, M.; Hayakawa, T.; Furue, M.K.; Mizuguchi, H. The promotion of hepatic maturation of human pluripotent stem cells in 3D co-culture using type I collagen and Swiss 3T3 cell sheets. Biomaterials 2012, 33, 4526–4534. [Google Scholar] [CrossRef] [PubMed]
- Collardeau-Frachon, S.; Scoazec, J.Y. Vascular development and differentiation during human liver organogenesis. Anat. Rec. 2008, 291, 614–627. [Google Scholar] [CrossRef] [PubMed]
- Si-Tayeb, K.; Lemaigre, F.P.; Duncan, S.A. Organogenesis and development of the liver. Dev. Cell 2010, 18, 175–189. [Google Scholar] [CrossRef] [PubMed]
- Rafii, S.; Butler, J.M.; Ding, B.S. Angiocrine functions of organ-specific endothelial cells. Nature 2016, 529, 316–325. [Google Scholar] [CrossRef] [PubMed]
- Ding, B.S.; Nolan, D.J.; Butler, J.M.; James, D.; Babazadeh, A.O.; Rosenwaks, Z.; Mittal, V.; Kobayashi, H.; Shido, K.; Lyden, D.; et al. Inductive angiocrine signals from sinusoidal endothelium are required for liver regeneration. Nature 2010, 468, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Haque, A.; Gheibi, P.; Stybayeva, G.; Gao, Y.; Torok, N.; Revzin, A. Ductular reaction-on-a-chip: Microfluidic co-cultures to study stem cell fate selection during liver injury. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Takebe, T.; Zhang, R.R.; Koike, H.; Kimura, M.; Yoshizawa, E.; Enomura, M.; Koike, N.; Sekine, K.; Taniguchi, H. Generation of a vascularized and functional human liver from an iPSC-derived organ bud transplant. Nat. Protoc. 2014, 9, 396–409. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Hu, K.; Smuga-Otto, K.; Tian, S.; Stewart, R.; Slukvin, I.I.; Thomson, J.A. Human induced pluripotent stem cells free of vector and transgene sequences. Science 2009, 324, 797–801. [Google Scholar] [CrossRef] [PubMed]
- Hay, D.C.; Zhao, D.; Ross, A.; Mandalam, R.; Lebkowski, J.; Cui, W. Direct differentiation of human embryonic stem cells to hepatocyte-like cells exhibiting functional activities. Cloning Stem Cells 2007, 9, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Knöspel, F.; Freyer, N.; Stecklum, M.; Gerlach, J.C.; Zeilinger, K. Periodic harvesting of embryonic stem cells from a hollow-fiber membrane based four-compartment bioreactor. Biotechnol. Prog. 2016, 32, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]



| Cell Type Used for Co-Culture with hiPSC/hESC | Ratio (hiPSC/hESC-Derived Cells:Co-Cultured Cell Type(s)) | Co-Culture Method | Culture Medium Applied | Ref. |
|---|---|---|---|---|
| Murine hepatic stromal cell line MLSgt20 | 1:1 | Self-aggregation in microwells | DMEM + FBS + differentiation factors | [58] |
| Murine embryonic fibroblasts (3T3-J2) | 0.04:1 | Seeding of DE cells onto mitomycin-treated 3T3-J2 feeder cells | Hepatocyte culture medium + FBS | [60] |
| Murine embryonic fibroblasts (swiss 3T3) | Not specified | Hepatoblast monolayer covered with 3T3 cell sheet | L15 medium + differentiation factors | [61] |
| HUVEC and mesenchymal stem cells | 10:7:2 | Spontaneous formation of 3D liver buds | HCM (without EGF) + EGM, 1:1 | [25,67] |
| hiPSC-derived endothelial cells | 2 : 1 | Multicomponent hydrogel fibers containing galactose for HLC and collagen for endothelial cells | Not specified | [55] |
| Hepatic stellate cell line TWNT-1 | Not specified | Cell inserts | DMEM-F12 + knockout serum replacer + DMSO | [56] |
| Murine embryonic fibroblasts (3T3-J2) | 2:1 or 2.5:1 for cryopreserved HLC | Micropatterned co-culture containing collagen coating and matrigel overlay | RPMI + B27 supplement + differentiation factors | [57] |
| Murine embryonic fibroblasts (3T3-J2) | 2:1 | Self-aggregation in microwells | RPMI + B27 supplement + differentiation factors | [59] |
| Primary rat hepatocytes | 1:2 | Microfluidic co-culture | DMEM + FBS + maintenance factors and IMDM + FBS + DMSO + differentiation factors | [66] |
| HUVEC and adipose derived stem cells | 1:1:0.02 | 3D bioprinting of in vivo like liver lobule structures | HCM (without EGF) + EGM-2, 1:1 | [52] |
| HUVEC | 2:1 | HUVEC grow in free spaces of hiPSC-derived DE cell monolayers | HCM + EGM complete, 1:1 and HCM + EGM Supplements | Present study |
| Component | EGM complete | HCM-I | HCM-II | HCM-I/II + EGM Complete | HCM-I/II + EGM Supplements |
|---|---|---|---|---|---|
| Hepatocyte culture medium (HCM) Bullet Kit | - | 100% (v/v) | 50% (v/v) | 97.5% (v/v) | |
| Endothelial cell growth medium (EGM) | 97.5% (v/v) | - | 48.75% (v/v) | - | |
| EGM Supplements | 2.5% (v/v) | - | 1.25% (v/v) | 2.5% (v/v) | |
| Human hepatocyte growth factor, recombinant | - | 10 ng/mL | 10 ng/mL | 10 ng/mL | 10 ng/mL |
| Human oncostatin M, recombinant | - | - | 10 ng/mL | 10 ng/mL 1 | 10 ng/mL 1 |
| Gentamycin | 0.05 mg/mL | 0.05 mg/mL | 0.05 mg/mL | 0.05 mg/mL | 0.05 mg/mL |
| Gene Symbol | Gene Name | Assay ID |
|---|---|---|
| AFP | α fetoprotein | HS00173490_m1 |
| ALB | albumin | HS00910225_m1 |
| GAPDH | glyceraldehyde-3-phosphate dehydrogenase | HS03929097_g1 |
| HNF4A | Hepatocyte nuclear factor 4, α | Hs00230853_m1 |
| KRT18 | Keratin 18 | Hs02827483_g1 |
| PECAM1 | platelet and endothelial cell adhesion molecule 1 | Hs00169777_m1 |
| POU5F1 | POU domain, class 5, transcription factor 1 | HS00999632_g1 |
| VWF | von Willebrand factor | Hs00169795_m1 |
| Antibody Type and Specifity | Protein Symbol | Species | Manufacturer | Article-No. | Final Conc. (µg/mL) |
|---|---|---|---|---|---|
| Primary Antibody | |||||
| Cytokeratin 18 | CK18 | mouse | Santa Cruz | Sc-6259 | 2 |
| Hepatocyte nuclear factor 4 α | HNF4A | rabbit | Santa Cruz | Sc-8987 | 4 |
| Platelet endothelial cell adhesion molecule 1 | PECAM1 | mouse | Abcam | ab24590 | 5 |
| POU domain, class 5, transcription factor 1 | OCT3 | rabbit | Santa Cruz | Sc-9081 | 2 |
| Von Willebrand factor | VWF | rabbit | Abcam | ab6994 | 35.5 |
| Secondary antibody | |||||
| Alexa Fluor® 488 anti-mouse | goat | Life Technologies | A-11029 | 2 | |
| Alexa Fluor® 594 anti-rabbit | goat | Life Technologies | A-11037 | 2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freyer, N.; Greuel, S.; Knöspel, F.; Strahl, N.; Amini, L.; Jacobs, F.; Monshouwer, M.; Zeilinger, K. Effects of Co-Culture Media on Hepatic Differentiation of hiPSC with or without HUVEC Co-Culture. Int. J. Mol. Sci. 2017, 18, 1724. https://doi.org/10.3390/ijms18081724
Freyer N, Greuel S, Knöspel F, Strahl N, Amini L, Jacobs F, Monshouwer M, Zeilinger K. Effects of Co-Culture Media on Hepatic Differentiation of hiPSC with or without HUVEC Co-Culture. International Journal of Molecular Sciences. 2017; 18(8):1724. https://doi.org/10.3390/ijms18081724
Chicago/Turabian StyleFreyer, Nora, Selina Greuel, Fanny Knöspel, Nadja Strahl, Leila Amini, Frank Jacobs, Mario Monshouwer, and Katrin Zeilinger. 2017. "Effects of Co-Culture Media on Hepatic Differentiation of hiPSC with or without HUVEC Co-Culture" International Journal of Molecular Sciences 18, no. 8: 1724. https://doi.org/10.3390/ijms18081724