The Impact of SMAD4 Loss on Outcome in Patients with Advanced Pancreatic Cancer Treated with Systemic Chemotherapy
Abstract
:1. Introduction
2. Results
2.1. Patient Characteristics and Clinicopathological Variables
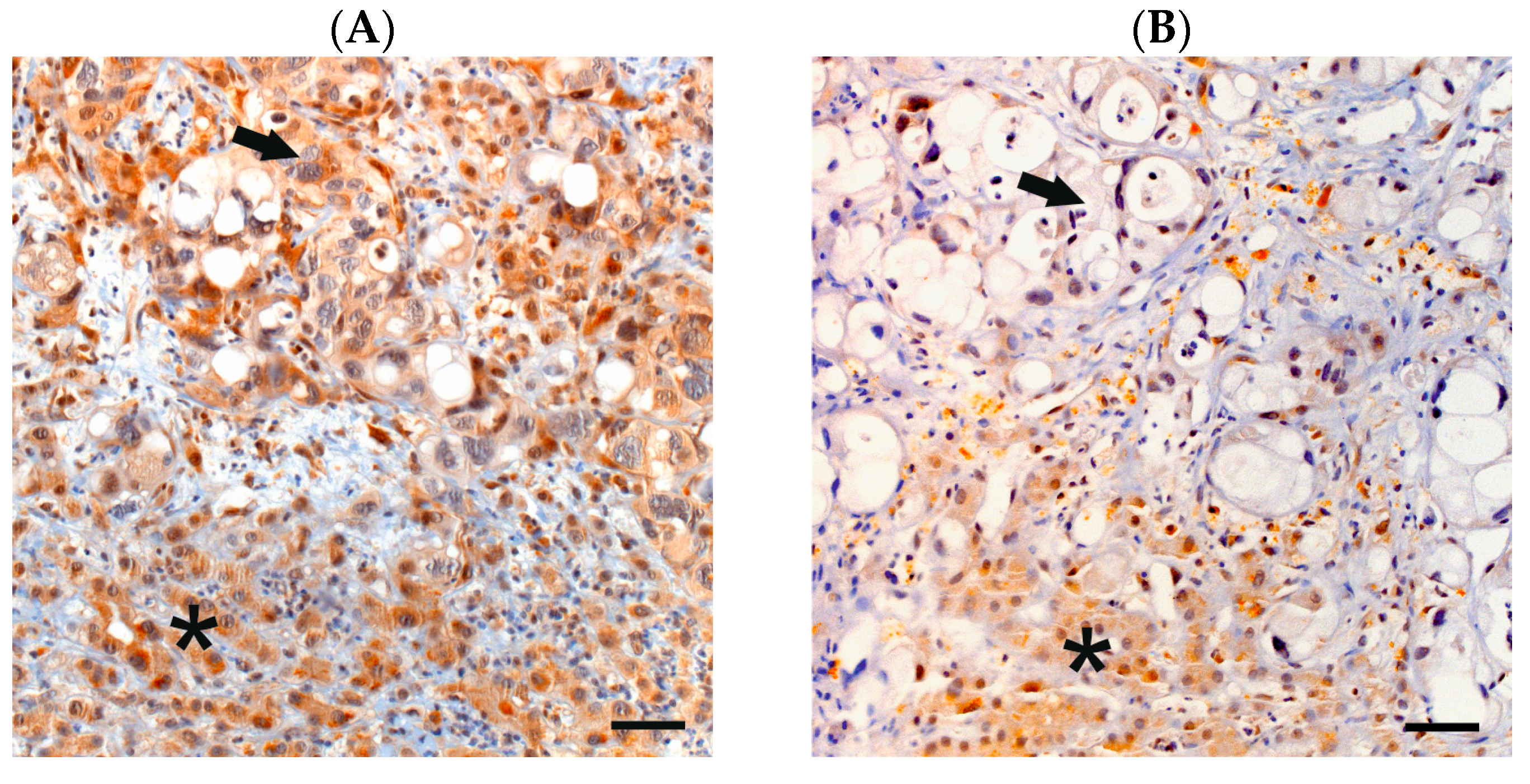
2.2. Mothers against Decapentaplegic Homolog 4 (SMAD4) Expression, Clinicopathological Variables and Survival Analyses
3. Discussion
4. Materials and Methods
4.1. Patient Population
4.2. Tumor Samples, SMAD4 Immunohistochemistry and Scoring Algorithm
4.3. Statistical Analyses
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CI | Confidence interval |
| DPC4 | Deleted in pancreatic cancer 4 |
| FFPE | Formalin fixed paraffin embedded |
| FOLFIRINOX | 5-FU, folinic acid, irinotecan, oxaliplatin |
| HR | Hazard ratio |
| IHC | Immunohistochemistry |
| KPS | Karnofsky performance status |
| LAPC | Locally advanced pancreatic cancer |
| µm | Micrometer |
| nab-paclitaxel | Nanoparticle albumin-bound paclitaxel |
| OS | Overall survival |
| PC | Pancreatic cancer |
| PFS | Progression-free survival |
| pts | patients |
| RUNX3 | Runt-related transcription factor 3 |
| SMAD4 | Mothers against decapentaplegic homolog 4 |
| US | United States |
References
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting cancer incidence and deaths to 2030: The unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014, 74, 2913–2921. [Google Scholar] [CrossRef] [PubMed]
- Kamisawa, T.; Wood, L.D.; Itoi, T.; Takaori, K. Pancreatic cancer. Lancet 2016, 388, 71–85. [Google Scholar] [CrossRef]
- Gourgou-Bourgade, S.; Bascoul-Mollevi, C.; Desseigne, F.; Ychou, M.; Bouché, O.; Guimbaud, R.; Bécouarn, Y.; Adenis, A.; Raoul, J.-L.; Boige, V.; et al. Impact of FOLFIRINOX compared with gemcitabine on quality of life in patients with metastatic pancreatic cancer: Results from the PRODIGE 4/ACCORD 11 randomized trial. J. Clin. Oncol. 2012, 31, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Kruger, S.; Haas, M.; Ormanns, S.; Bächmann, S.; Siveke, J.T.; Kirchner, T.; Heinemann, V.; Boeck, S. Translational research in pancreatic ductal adenocarcinoma: Current evidence and future concepts. World J. Gastroenterol. 2014, 20, 10769. [Google Scholar] [CrossRef] [PubMed]
- Hahn, S.A.; Schutte, M.; Hoque, A.; Moskaluk, C.A. DCP4, A candidate tumor supressor gene at human chromosome 18q21.1. Science 1996, 271, 350. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Wu, W.; Huang, C.; Cen, G.; Jiang, T.; Cao, J.; Huang, K.; Qiu, Z. SMAD4 and its role in pancreatic cancer. Tumor Biol. 2015, 36, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Iacobuzio-Donahue, C.A.; Fu, B.; Yachida, S.; Luo, M.; Abe, H.; Henderson, C.M.; Vilardell, F.; Wang, Z.; Keller, J.W.; Banerjee, P.; et al. DPC4 gene status of the primary carcinoma correlates with patterns of failure in patients with pancreatic cancer. J. Clin. Oncol. 2009, 27, 1801–1813. [Google Scholar] [CrossRef] [PubMed]
- Crane, C.H.; Varadhachary, G.R.; Yordy, J.S.; Staerkel, G.A.; Javle, M.M.; Safran, H.; Haque, W.; Hobbs, B.D.; Krishnan, S.; Fleming, J.B.; et al. Phase II trial of cetuximab, gemcitabine, and oxaliplatin followed by chemoradiation with cetuximab for locally advanced (T4) pancreatic adenocarcinoma: Correlation of SMAD4 (DPC4) immunostaining with pattern of disease progression. J. Clin. Oncol. 2011, 29, 3031–3043. [Google Scholar] [CrossRef] [PubMed]
- Bachet, J.-B.; Marechal, R.; Demetter, P.; Bonnetain, F.; Couvelard, A.; Svrcek, M.; Bardier-Dupas, A.; Hammel, P.; Sauvanet, A.; Louvet, C.; et al. Contribution of CXCR4 and SMAD4 in predicting disease progression pattern and benefit from adjuvant chemotherapy in resected pancreatic adenocarcinoma. Ann. Oncol. 2012, 23, 2321–2335. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Fujii, T.; Shimoyama, Y.; Kanda, M.; Nakayama, G.; Sugimoto, H.; Koike, M.; Nomoto, S.; Fujiwara, M.; Nakao, A.; et al. SMAD4 expression predicts local spread and treatment failure in resected pancreatic cancer. Pancreas 2015, 44, 661–664. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.; Kim, H.; Hwang, D.; Lee, J.; Song, K.; Jun, E.; Shim, I.; Hong, S.; Kim, H.; Park, K.; et al. The DPC4/SMAD4 genetic status determines recurrence patterns and treatment outcomes in resected pancreatic ductal adenocarcinoma: A prospective cohort study. Oncotarget 2017, 8, 17945–17959. [Google Scholar] [PubMed]
- Winter, J.M.; Tang, L.H.; Klimstra, D.S.; Liu, W.; Linkov, I.; Brennan, M.F.; D’Angelica, M.I.; deMatteo, R.P.; Fong, Y.; Jarnagin, W.R.; et al. Failure patterns in resected pancreas adenocarcinoma: Lack of predicted benefit to SMAD4 expression. Ann. Surg. 2013, 258, 331. [Google Scholar] [CrossRef] [PubMed]
- Xing, S.; Yang, H.; Liu, J.; Zheng, X.; Feng, J.; Li, X.; Li, W. Prognostic value of SMAD4 in pancreatic cancer: A meta-analysis. Transl. Oncol. 2016, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jin, K.; Chen, X.; Lv, J.; Ji, K. Clinicopathological significance of SMAD4 loss in pancreatic ductal adenocarcinomas: A systematic review and meta-analysis. Oncotarget 2017, 8, 16704–16711. [Google Scholar] [CrossRef] [PubMed]
- Oshima, M.; Okano, K.; Muraki, S.; Haba, R.; Maeba, T.; Suzuki, Y.; Yachida, S. Immunohistochemically detected expression of 3 major genes (CDKN2A/p16, TP53, and SMAD4/DPC4) strongly predicts survival in patients with resectable pancreatic cancer. Ann. Surg. 2013, 258, 331–346. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.H.; Kim, S.C.; Hong, S.-M.; Kim, Y.H.; Song, K.-B.; Park, K.-M.; Lee, Y.-J. Genetic alterations of K-ras, p53, c-erbB-2, and DPC4 in pancreatic ductal adenocarcinoma and their correlation with patient survival. Pancreas 2013, 42, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; He, C.; Geng, S.; Sheng, H.; Shen, X.; Zhang, X.; Li, H.; Zhu, S.; Chen, X.; Yang, C.; et al. RhoT1 and Smad4 are correlated with lymph node metastasis and overall survival in pancreatic cancer. PLoS ONE 2012, 7, e42234. [Google Scholar] [CrossRef] [PubMed]
- Tascilar, M.; Skinner, H.G.; Rosty, C.; Sohn, T.; Wilentz, R.E.; Offerhaus, G.J.A.; Adsay, V.; Abrams, R.A.; Cameron, J.L.; Kern, S.E.; et al. The SMAD4 protein and prognosis of pancreatic ductal adenocarcinoma. Clin. Cancer Res. 2001, 7, 4111–4121. [Google Scholar]
- Infante, J.R.; Matsubayashi, H.; Sato, N.; Tonascia, J.; Klein, A.P.; Riall, T.A.; Yeo, C.; Iacobuzio-Donahue, C.; Goggins, M. Peritumoral fibroblast SPARC expression and patient outcome with resectable pancreatic adenocarcinoma. J. Clin. Oncol. 2007, 25, 311–325. [Google Scholar] [CrossRef] [PubMed]
- Biankin, A.; Biankin, S.; Kench, J.; Morey, A.; Lee, C.; Head, D.; Eckstein, R.; Hugh, T.; Henshall, S.; Sutherland, R.; et al. Aberrant p16INK4A and DPC4/Smad4 expression in intraductal papillary mucinous tumours of the pancreas is associated with invasive ductal adenocarcinoma. Gut 2002, 50, 861–868. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Srinivasan, R.; Wig, J.D. SMAD4 genetic alterations predict a worse prognosis in patients with pancreatic ductal adenocarcinoma. Pancreas 2012, 41, 541–546. [Google Scholar] [CrossRef] [PubMed]
- Whittle, M.C.; Izeradjene, K.; Rani, P.G.; Feng, L.; Carlson, M.A.; DelGiorno, K.E.; Wood, L.D.; Goggins, M.; Hruban, R.H.; Chang, A.E.; et al. RUNX3 controls a metastatic switch in pancreatic ductal adenocarcinoma. Cell 2015, 161, 1341–1360. [Google Scholar] [CrossRef] [PubMed]
- Bardeesy, N.; Cheng, K.-h.; Berger, J.H.; Chu, G.C.; Pahler, J.; Olson, P.; Hezel, A.F.; Horner, J.; Lauwers, G.Y.; Hanahan, D.; et al. Smad4 is dispensable for normal pancreas development yet critical in progression and tumor biology of pancreas cancer. Genes Dev. 2006, 20, 3131–3146. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Brosnan, J.A.; Blackford, A.L.; Sur, S.; Hruban, R.H.; Kinzler, K.W.; Vogelstein, B.; Maitra, A.; Diaz, L.A.; Iacobuzio-Donahue, C.A.; et al. Genetically defined subsets of human pancreatic cancer show unique in vitro chemosensitivity. Clin. Cancer Res. 2012, 18, 6511–6530. [Google Scholar] [CrossRef] [PubMed]
- Ormanns, S.; Haas, M.; Baechmann, S.; Altendorf-Hofmann, A.; Remold, A.; Quietzsch, D.; Clemens, M.R.; Bentz, M.; Geissler, M.; Lambertz, H.; et al. Impact of SPARC expression on outcome in patients with advanced pancreatic cancer not receiving nab-paclitaxel: A pooled analysis from prospective clinical and translational trials. Br. J. Cancer 2016, 115, 1520–1529. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Group | n | OS (Months) | p (Log-Rank) | HR | 95% CI | PFS (Months) | p (Log-Rank) | HR | 95% CI |
|---|---|---|---|---|---|---|---|---|---|---|
| Sex | male | 84 | 8.3 | 0.673 | 0.926 | 0.649–1.323 | 6.8 | 0.578 | 1.123 | 0.745–1.692 |
| female | 59 | 8.3 | 5.3 | |||||||
| Age group | <60 years | 55 | 8.3 | 0.414 | 1163 | 0.810–1.670 | 6.4 | 0.859 | 1.038 | 0.686–1.571 |
| ≥60 years | 88 | 8.4 | 6.7 | |||||||
| KPS | ≤80 | 57 | 6.3 | 0.017 | 1566 | 1.081–2.280 | 4.1 | 0.056 | 1.511 | 0.987–2.314 |
| >80 | 76 | 11.1 | 7.6 | |||||||
| Stage at start of palliative CTX | locally advanced | 15 | 13.6 | 0.064 | 1782 | 0.957–3.317 | 10.0 | 0.054 | 2.015 | 0.972–4.177 |
| metastatic | 128 | 8.1 | 6.3 | |||||||
| CTX type | gemcitabine-based | 99 | 8.3 | 0.250 | 1091 | 0.863–1.379 | 7.6 | 0.017 | 1.336 | 1.040–1.715 |
| fluoropyrimidine-based | 25 | 9.1 | 4.0 | |||||||
| gem/fluo—based | 19 | 11.0 | 4.7 | |||||||
| Tumor grade | G1–G2 | 61 | 11.5 | 0.171 | 1280 | 0.897–1.827 | 8.6 | 0.135 | 1.367 | 0.905–2.066 |
| G3–G4 | 82 | 7.8 | 5.5 | |||||||
| Metastatic sites | 0 | 15 | 13.6 | 0.180 | 1208 | 0.908–1.608 | 10.0 | 0.097 | 1,439 | 1.022–2.025 |
| 1 | 90 | 8.3 | 6.6 | |||||||
| >1 | 38 | 7.8 | 4.7 |
| Chemotherapy Subgroup | SMAD4 | n | % | OS (Months) | p (Log-Rank) | HR | 95% CI | PFS (Months) | p (Log-Rank) | HR | 95% CI |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Total | expressed | 51 | 35.7 | 7.8 | 0.656 | 1088 | 0.751–1.576 | 5.8 | 0.038 | 1565 | 1.020–2.399 |
| lost | 92 | 64.3 | 8.5 | 7.0 | |||||||
| Gemcitabine | expressed | 35 | 35.4 | 8.3 | 0.722 | 1069 | 0.680–1.679 | 6.8 | 0.037 | 1790 | 1.028–3.116 |
| lost | 64 | 64.6 | 8.3 | 8.9 | |||||||
| Fluopyrimidine | expressed | 10 | 40.0 | 11.5 | 0.104 | 0.471 | 0.187–1.190 | 3.6 | 0.185 | 0.524 | 0.198–1.387 |
| lost | 15 | 60.0 | 7.2 | 6.7 | |||||||
| Gemcitabine + fluopyrimidine | expressed | 6 | 31.6 | 4.3 | 0.017 | 4277 | 1.185–15.431 | 2.5 | 0.018 | 3489 | 1.148–10.603 |
| lost | 13 | 68.4 | 12.5 | 6.2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ormanns, S.; Haas, M.; Remold, A.; Kruger, S.; Holdenrieder, S.; Kirchner, T.; Heinemann, V.; Boeck, S. The Impact of SMAD4 Loss on Outcome in Patients with Advanced Pancreatic Cancer Treated with Systemic Chemotherapy. Int. J. Mol. Sci. 2017, 18, 1094. https://doi.org/10.3390/ijms18051094
Ormanns S, Haas M, Remold A, Kruger S, Holdenrieder S, Kirchner T, Heinemann V, Boeck S. The Impact of SMAD4 Loss on Outcome in Patients with Advanced Pancreatic Cancer Treated with Systemic Chemotherapy. International Journal of Molecular Sciences. 2017; 18(5):1094. https://doi.org/10.3390/ijms18051094
Chicago/Turabian StyleOrmanns, Steffen, Michael Haas, Anna Remold, Stephan Kruger, Stefan Holdenrieder, Thomas Kirchner, Volker Heinemann, and Stefan Boeck. 2017. "The Impact of SMAD4 Loss on Outcome in Patients with Advanced Pancreatic Cancer Treated with Systemic Chemotherapy" International Journal of Molecular Sciences 18, no. 5: 1094. https://doi.org/10.3390/ijms18051094






