The Role of HMGB1 Signaling Pathway in the Development and Progression of Hepatocellular Carcinoma: A Review
Abstract
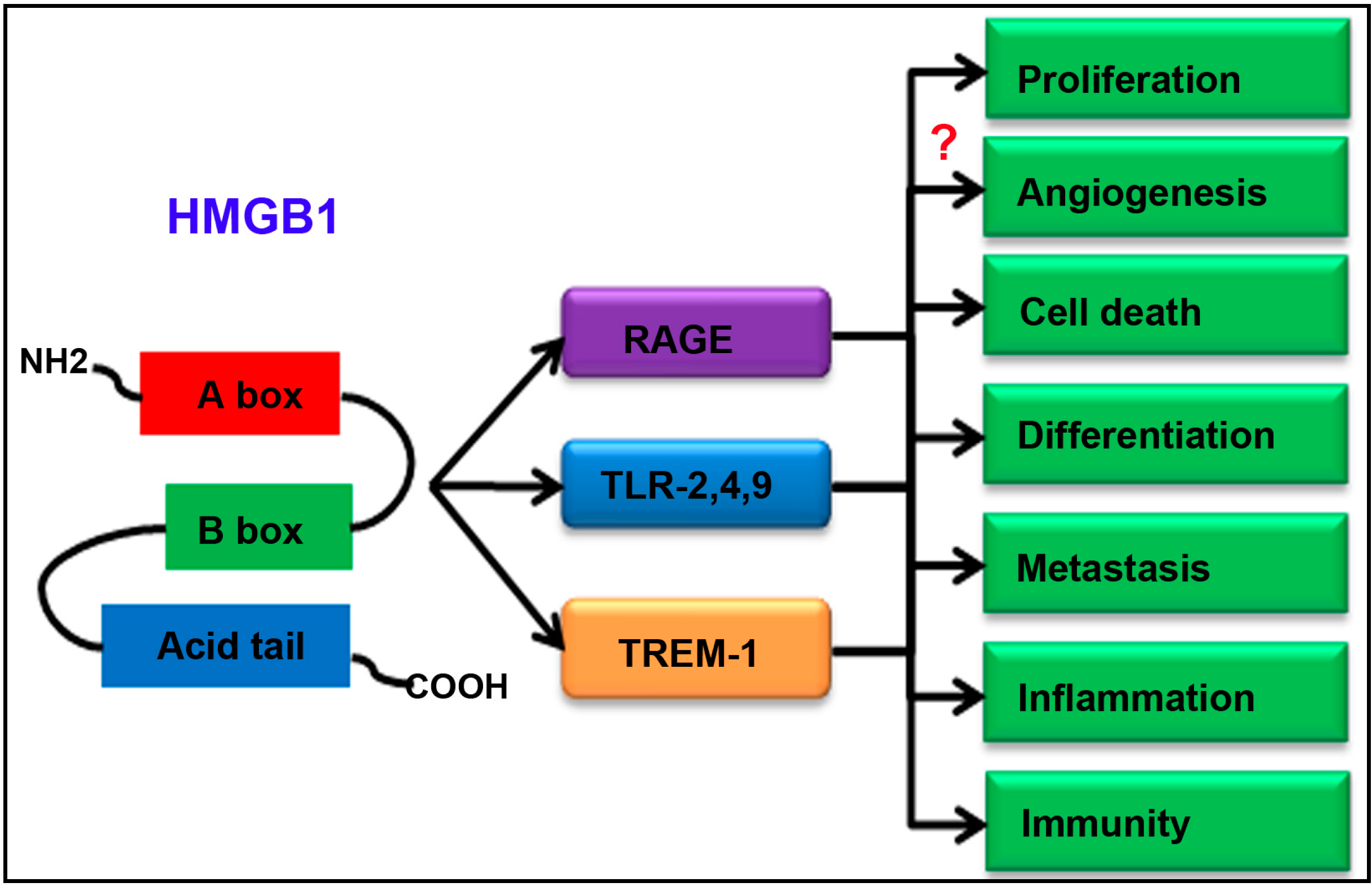
:1. Introduction
2. HMGB1 and Cell Proliferation in HCC
3. HMGB1 and Angiogenesis in HCC
4. HMGB1 and Cell Death in HCC
5. HMGB1 and Cell Differentiation in HCC
6. HMGB1 and Metastasis in HCC
7. HMGB1 and Inflammatory Response in HCC
8. HMGB1 and Immune Function in HCC
9. Discussion
| Location of HMGB1 | Function | Receptor | References |
|---|---|---|---|
| recombinant | induce proliferation | NA | [47] |
| NA | promote progression | NA | [48] |
| NA | promote metastasis | NA | [49] |
| NA | induce proliferation and metastasis, block apoptosis | RAGE | [50] |
| NA | induce proliferation | RAGE | [51] |
| NA | induce proliferation and invasion | RAGE | [52] |
| cytosol | induce proliferation | TLR-9 | [53] |
| NA | induce angiogenesis | RAGE | [56] |
| cytosol | induce autophagic cell death | NA | [57] |
| NA | induce proliferation and reduce apoptosis | RAGE | [32] |
| NA | induce apoptosis | NA | [60] |
| perinuclear | induce differentiation | NA | [63] |
| nucleus | block differentiation | NA | [63] |
| NA | induce differentiation | RAGE | [64] |
| NA | induce metastasis | NA | [66] |
| NA | induce metastasis | NA | [67] |
| NA | induce metastasis | RAGE | [68] |
| NA | induce metastasis | RAGE | [69] |
| NA | induce metastasis | TLR-4 and RAGE | [36] |
| NA | induce metastasis | NA | [67] |
| NA | induce inflammation | NA | [71] |
| NA | induce inflammation | TREM-1 | [45] |
| NA | induce inflammation | RAGE | [72] |
| NA | induce inflammation | TREM-1 and RAGE | [73] |
| NA | induce immunity | RAGE | [33] |
| NA | induce proliferation and metastasis | NA | [82] |
| serum | induce carcinogenesis | NA | [83] |
| NA | induce carcinogenesis | NA | [84] |
| NA | induce proliferation and metastasis | NA | [85] |
| serum | induce metastasis | TLR-4 | [86] |
| recombinant | induce proliferation and metastasis, reduce apoptosis | TLR-2 | [87] |
10. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, N.; Cheung, F.; Lao, L.; Li, C.; Feng, Y. Chinese medicines for prevention and treatment of human hepatocellular carcinoma: Current progress on pharmacological actions and mechanisms. J. Integr. Med. 2015, 13, 142–164. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Kang, R.; Chen, R.; Zhang, Q.; Hou, W.; Wu, S.; Cao, L.; Huang, J.; Yu, Y.; Fan, X.G.; Yan, Z.; et al. Hmgb1 in health and disease. Mol. Asp. Med. 2014, 40, 1–116. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, M.E.; Manfredi, A.A. High-mobility group box 1 (hmgb1) protein at the crossroads between innate and adaptive immunity. Immunol. Rev. 2007, 220, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Nace, G.W.; McDonald, K.A.; Tai, S.; Klune, J.R.; Rosborough, B.R.; Ding, Q.; Loughran, P.; Zhu, X.; Beer-Stolz, D.; et al. Hepatocyte-specific high-mobility group box 1 deletion worsens the injury in liver ischemia/reperfusion: A role for intracellular high-mobility group box 1 in cellular protection. Hepatology 2014, 59, 1984–1997. [Google Scholar] [CrossRef] [PubMed]
- Kang, R.; Zhang, Q.; Hou, W.; Yan, Z.; Chen, R.; Bonaroti, J.; Bansal, P.; Billiar, T.R.; Tsung, A.; Wang, Q.; et al. Intracellular hmgb1 inhibits inflammatory nucleosome release and limits acute pancreatitis in mice. Gastroenterology 2014, 146, 1097–1107. [Google Scholar] [CrossRef] [PubMed]
- Yanai, H.; Matsuda, A.; An, J.; Koshiba, R.; Nishio, J.; Negishi, H.; Ikushima, H.; Onoe, T.; Ohdan, H.; Yoshida, N.; et al. Conditional ablation of hmgb1 in mice reveals its protective function against endotoxemia and bacterial infection. Proc. Natl. Acad. Sci. USA 2013, 110, 20699–20704. [Google Scholar] [CrossRef] [PubMed]
- Huebener, P.; Pradere, J.P.; Hernandez, C.; Gwak, G.Y.; Caviglia, J.M.; Mu, X.; Loike, J.D.; Jenkins, R.E.; Antoine, D.J.; Schwabe, R.F. The hmgb1/rage axis triggers neutrophil-mediated injury amplification following necrosis. J. Clin. Investig. 2015, 125, 539–550. [Google Scholar] [CrossRef] [PubMed]
- Moriwaka, Y.; Luo, Y.; Ohmori, H.; Fujii, K.; Tatsumoto, N.; Sasahira, T.; Kuniyasu, H. Hmgb1 attenuates anti-metastatic defense of the lymph nodes in colorectal cancer. Pathobiology 2010, 77, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Stoetzer, O.J.; Fersching, D.M.I.; Salat, C.; Steinkohl, O.; Gabka, C.J.; Hamann, U.; Braun, M.; Feller, A.M.; Heinemann, V.; Siegele, B.; et al. Circulating immunogenic cell death biomarkers hmgb1 and rage in breast cancer patients during neoadjuvant chemotherapy. Tumor Biol. 2013, 34, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Fersching, D.M.I.; Stotzer, O.J.; Siegele, B.; Nagel, D.; Holdenrieder, S. Nucleosomes, rage and hmgb1 in predicting response to neoadjuvant chemotherapy in breast cancer patients. Tumor Biol. 2010, 31, 99. [Google Scholar]
- Wang, C.H.; Fei, G.R.; Liu, Z.M.; Li, Q.C.; Xu, Z.G.; Ren, T. Hmgb1 was a pivotal synergistic effecor for cpg oligonucleotide to enhance the progression of human lung cancer cells. Cancer Biol. Ther. 2012, 13, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.L.; Tsai, J.R.; Hwang, J.J.; Chou, S.H.; Cheng, Y.J.; Lin, F.Y.; Chen, Y.L.; Hung, C.Y.; Chen, W.C.; Chen, Y.H.; et al. High-mobility group box 1-mediated matrix metalloproteinase-9 expression in non-small cell lung cancer contributes to tumor cell invasiveness. Am. J. Respir. Cell Mol. Biol. 2010, 43, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.H.; Wang, H.M.; Wang, J. Expression of hmgb1 and nf-kappa b p65 and its significance in non-small cell lung cancer. Wspolczesna Onkol. 2013, 17, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Shang, G.H.; Jia, C.Q.; Tian, H.; Xiao, W.; Li, Y.; Wang, A.H.; Dong, L.; Lin, D.J. Serum high mobility group box protein 1 as a clinical marker for non-small cell lung cancer. Respir. Med. 2009, 103, 1949–1953. [Google Scholar] [CrossRef] [PubMed]
- Gnanasekar, M.; Thirugnanam, S.; Ramaswamy, K. Short hairpin RNA (shRNA) constructs targeting high mobility group box-1 (HMGB1) expression leads to inhibition of prostate cancer cell survival and apoptosis. Int. J. Oncol. 2009, 34, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Pang, X.A.; Zhang, Y.; Wei, H.; Zhang, J.; Luo, Q.S.; Huang, C.L.; Zhang, S.L. Expression and effects of high-mobility group box 1 in cervical cancer. Int. J. Mol. Sci. 2014, 15, 8699–8712. [Google Scholar] [CrossRef] [PubMed]
- Mittal, D.; Saccheri, F.; Venereau, E.; Pusterla, T.; Bianchi, M.E.; Rescigno, M. Tlr4-mediated skin carcinogenesis is dependent on immune and radioresistant cells. Embo J. 2010, 29, 2242–2252. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, T.; Sakazume, K.; Tonooka, A.; Zaitsu, M.; Takeshima, Y.; Mikami, K.; Uekusa, T. Cytosolic hmgb1 expression in human renal clear cell cancer indicates higher pathological T classifications and tumor grades. Urol. J. 2013, 10, 960–965. [Google Scholar] [PubMed]
- Wu, F.; Zhao, Z.H.; Ding, S.T.; Wu, H.H.; Lu, J.J. High mobility group box 1 protein is methylated and transported to cytoplasm in clear cell renal cell carcinoma. Asian J. Cancer Prev. 2013, 14, 5789–5795. [Google Scholar] [CrossRef]
- Song, B.; Song, W.G.; Li, Z.J.; Xu, Z.F.; Wang, X.W.; Wang, C.X.; Liu, J. Effect of hmgb1 silencing on cell proliferation, invasion and apoptosis of mgc-803 gastric cancer cells. Cell Biochem. Funct. 2012, 30, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhu, J.S.; Yang, Y.C.; Zhou, Z.; Chen, W.X.; Chen, N.W. Enhancing the therapeutic effects of ethyl pyruvate on gastric cancer through knockdown of yap1 expression. Eur. J. Inflamm. 2013, 11, 123–132. [Google Scholar]
- Akaike, H.; Kono, K.; Sugai, H.; Takahashi, A.; Mimura, K.; Kawaguchi, Y.; Fujii, H. Expression of high mobility group box chromosomal protein-1 (hmgb-1) in gastric cancer. Anticancer Res. 2007, 27, 449–457. [Google Scholar] [PubMed]
- Zhang, J.; Kou, Y.B.; Zhu, J.S.; Chen, W.X.; Li, S. Knockdown of hmgb1 inhibits growth and invasion of gastric cancer cells through the nf-.B pathway in vitro and in vivo. Int. J. Oncol. 2014, 44, 1268–1276. [Google Scholar] [PubMed]
- Chung, H.W.; Lee, S.G.; Kim, H.; Hong, D.J.; Chung, J.B.; Stroncek, D.; Lim, J.B. Serum high mobility group box-1 (hmgb1) is closely associated with the clinical and pathologic features of gastric cancer. J. Transl. Med. 2009, 7, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Soreide, K.; Sund, M. Epidemiological-molecular evidence of metabolic reprogramming on proliferation, autophagy and cell signaling in pancreas cancer. Cancer Lett. 2015, 356, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Wittwer, C.; Boeck, S.; Heinemann, V.; Haas, M.; Stieber, P.; Nagel, D.; Holdenrieder, S. Circulating nucleosomes and immunogenic cell death markers hmgb1, srage and dnase in patients with advanced pancreatic cancer undergoing chemotherapy. Int. J. Cancer 2013, 133, 2619–2630. [Google Scholar] [CrossRef] [PubMed]
- Kang, R.; Tang, D.L. Autophagy in pancreatic cancer pathogenesis and treatment. Am. J. Cancer Res. 2012, 2, 383–396. [Google Scholar] [PubMed]
- Li, B.; Jiang, S.D.; Zheng, X.F.; Ni, B.B.; Yang, Y.H.; Chen, J.W.; Chen, K.; Jiang, L.S. Expression of the inflammatory molecule hmgb1 in human osteosarcoma and its clinical relevance. Eur. J. Inflamm. 2013, 11, 61–73. [Google Scholar]
- Jia, L.; Clear, A.; Liu, F.T.; Matthews, J.; Uddin, N.; McCarthy, A.; Hoxha, E.; Durance, C.; Iqbal, S.; Gribben, J.G. Extracellular hmgb1 promotes differentiation of nurse-like cells in chronic lymphocytic leukemia. Blood 2014, 123, 1709–1719. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.; Dai, W.; Wang, F.; Lu, J.; Shen, M.; Chen, K.; Li, J.; Zhang, Y.; Wang, C.; Yang, J.; et al. Ethyl pyruvate inhibits proliferation and induces apoptosis of hepatocellular carcinoma via regulation of the hmgb1-rage and akt pathways. Biochem. Biophys. Res. Commun. 2014, 443, 1162–1168. [Google Scholar] [CrossRef] [PubMed]
- Kohles, N.; Nagel, D.; Jungst, D.; Stieber, P.; Holdenrieder, S. Predictive value of immunogenic cell death biomarkers hmgb1, srage, and dnase in liver cancer patients receiving transarterial chemoembolization therapy. Tumour Biol. 2012, 33, 2401–2409. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.B. Role of high mobility group box 1 and its signaling pathways in renal diseases. J. Recept. Signal Transduct. 2014, 34, 348–350. [Google Scholar] [CrossRef] [PubMed]
- Roh, Y.S.; Seki, E. Toll-like receptors in alcoholic liver disease, non-alcoholic steatohepatitis and carcinogenesis. J. Gastroenterol. Hepatol. 2013, 28, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Chang, Y.; Liang, X.; Cardinal, J.S.; Huang, H.; Thorne, S.H.; Monga, S.P.; Geller, D.A.; Lotze, M.T.; Tsung, A. High-mobility group box 1 activates caspase-1 and promotes hepatocellular carcinoma invasiveness and metastases. Hepatology 2012, 55, 1863–1875. [Google Scholar] [CrossRef] [PubMed]
- Conti, L.; Lanzardo, S.; Arigoni, M.; Antonazzo, R.; Radaelli, E.; Cantarella, D.; Calogero, R.A.; Cavallo, F. The noninflammatory role of high mobility group box 1/toll-like receptor 2 axis in the self-renewal of mammary cancer stem cells. FASEB J. 2013, 27, 4731–4744. [Google Scholar] [CrossRef] [PubMed]
- Friggeri, A.; Yang, Y.; Banerjee, S.; Park, Y.J.; Liu, G.; Abraham, E. Hmgb1 inhibits macrophage activity in efferocytosis through binding to the alphavbeta3-integrin. Am. J. Physiol. Cell Physiol. 2010, 299, 8. [Google Scholar] [CrossRef] [PubMed]
- Song, J.X.; Lu, J.H.; Liu, L.F.; Chen, L.L.; Durairajan, S.S.; Yue, Z.; Zhang, H.Q.; Li, M. Hmgb1 is involved in autophagy inhibition caused by snca/alpha-synuclein overexpression: A process modulated by the natural autophagy inducer corynoxine b. Autophagy 2014, 10, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Young, J.; Song, D.; Esko, J.D. Heparan sulfate is essential for high mobility group protein 1 (hmgb1) signaling by the receptor for advanced glycation end products (rage). J. Biol. Chem. 2011, 286, 41736–41744. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.Y.; Tang, J.; Zheng, P.; Liu, Y. Cd24 and siglec-10 selectively repress tissue damage-induced immune responses. Science 2009, 323, 1722–1725. [Google Scholar] [CrossRef] [PubMed]
- Chiba, S.; Baghdadi, M.; Akiba, H.; Yoshiyama, H.; Kinoshita, I.; Dosaka-Akita, H.; Fujioka, Y.; Ohba, Y.; Gorman, J.V.; Colgan, J.D.; et al. Tumor-infiltrating dcs suppress nucleic acid-mediated innate immune responses through interactions between the receptor tim-3 and the alarmin hmgb1. Nat. Immunol. 2012, 13, 832–842. [Google Scholar] [CrossRef] [PubMed]
- Penzo, M.; Molteni, R.; Suda, T.; Samaniego, S.; Raucci, A.; Habiel, D.M.; Miller, F.; Jiang, H.P.; Li, J.; Pardi, R.; et al. Inhibitor of nf-kappa b kinases alpha and beta are both essential for high mobility group box 1-mediated chemotaxis. J. Immunol. 2010, 184, 4497–4509. [Google Scholar] [CrossRef] [PubMed]
- Pedrazzi, M.; Averna, M.; Sparatore, B.; Patrone, M.; Salamino, F.; Marcoli, M.; Maura, G.; Cervetto, C.; Frattaroli, D.; Pontremoli, S.; et al. Potentiation of nmda receptor-dependent cell responses by extracellular high mobility group box 1 protein. PLoS ONE 2012, 7, 31. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Li, J.; Salcedo, R.; Mivechi, N.F.; Trinchieri, G.; Horuzsko, A. The proinflammatory myeloid cell receptor trem-1 controls kupffer cell activation and development of hepatocellular carcinoma. Cancer Res. 2012, 72, 3977–3986. [Google Scholar] [CrossRef] [PubMed]
- Dumitriu, I.E.; Baruah, P.; Manfredi, A.A.; Bianchi, M.E.; Rovere-Querini, P. Hmgb1: Guiding immunity from within. Trends Immunol. 2005, 26, 381–387. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Fan, X.; Zhou, R.; Wang, H. Effect of hmgb1 on human hepatoma cell line-hepg2 proliferation. J. Cent. South Univ. Med. Sci. 2010, 35, 451–457. [Google Scholar]
- Bi, M.R.; Zhu, L.Y.; Yan, B.Z.; Chen, L.Y.; Wang, F.X.; Ma, Y.J.; Yang, B.S. Association of upregulated hmgb1 and c-iap2 proteins with hepatocellular carcinoma development and progression. Hepat. Mon. 2014, 14, 23552. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Ding, Y.; Huang, J.; Li, Q.; Liu, Y.; Ni, W.; Zhang, Y.; Zhu, Y.; Chen, L.; Chen, B. The association of hmgb1 gene with the prognosis of hcc. PLoS ONE 2014, 9, 89097. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhao, L.H.; Huang, B.; Wang, R.Y.; Yuan, S.X.; Tao, Q.F.; Xu, Y.; Sun, H.Y.; Lin, C.; Zhou, W.P. Pioglitazone, a ppargamma agonist, inhibits growth and invasion of human hepatocellular carcinoma via blockade of the rage signaling. Mol. Carcinog. 2014, 12, 22231. [Google Scholar]
- Yaser, A.M.; Huang, Y.; Zhou, R.R.; Hu, G.S.; Xiao, M.F.; Huang, Z.B.; Duan, C.J.; Tian, W.; Tang, D.L.; Fan, X.G. The role of receptor for advanced glycation end products (rage) in the proliferation of hepatocellular carcinoma. Int. J. Mol. Sci. 2012, 13, 5982–5997. [Google Scholar] [CrossRef] [PubMed]
- Takada, M.; Ku, Y.; Toyama, H.; Suzuki, Y.; Kuroda, Y. Suppressive effects of tea polyphenol and conformational changes with receptor for advanced glycation end products (rage) expression in human hepatoma cells. Hepato Gastroenterol. 2002, 49, 928–931. [Google Scholar]
- Liu, Y.; Yan, W.; Tohme, S.; Chen, M.; Fu, Y.; Tian, D.; Lotze, M.; Tang, D.; Tsung, A. Hypoxia induced hmgb1 and mitochondrial DNA interactions mediate tumor growth in hepatocellular carcinoma through toll-like receptor 9. J. Hepatol. 2015, 63, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Van Beijnum, J.R.; Dings, R.P.; van der Linden, E.; Zwaans, B.M.; Ramaekers, F.C.; Mayo, K.H.; Griffioen, A.W. Gene expression of tumor angiogenesis dissected: Specific targeting of colon cancer angiogenic vasculature. Blood 2006, 108, 2339–2348. [Google Scholar] [CrossRef] [PubMed]
- Schlueter, C.; Weber, H.; Meyer, B.; Rogalla, P.; Roser, K.; Hauke, S.; Bullerdiek, J. Angiogenetic signaling through hypoxia: Hmgb1: An angiogenetic switch molecule. Am. J. Pathol. 2005, 166, 1259–1263. [Google Scholar] [CrossRef]
- Takino, J.; Yamagishi, S.; Takeuchi, M. Glycer-ages-rage signaling enhances the angiogenic potential of hepatocellular carcinoma by upregulating vegf expression. World J. Gastroenterol. 2012, 18, 1781–1788. [Google Scholar] [CrossRef] [PubMed]
- Cheng, P.; Ni, Z.; Dai, X.; Wang, B.; Ding, W.; Rae Smith, A.; Xu, L.; Wu, D.; He, F.; Lian, J. The novel bh-3 mimetic apogossypolone induces beclin-1- and ros-mediated autophagy in human hepatocellular carcinoma cells. Cell Death Dis. 2013, 7, 17. [Google Scholar] [CrossRef]
- Huebener, P.; Gwak, G.Y.; Pradere, J.P.; Quinzii, C.M.; Friedman, R.; Lin, C.S.; Trent, C.M.; Mederacke, I.; Zhao, E.; Dapito, D.H.; et al. High-mobility group box 1 is dispensable for autophagy, mitochondrial quality control, and organ function in vivo. Cell Metab. 2014, 19, 539–547. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Tang, D. Hmgb1-dependent and -independent autophagy. Autophagy 2014, 10, 1873–1876. [Google Scholar] [CrossRef] [PubMed]
- Gwak, G.Y.; Moon, T.G.; Lee, D.H.; Yoo, B.C. Glycyrrhizin attenuates hmgb1-induced hepatocyte apoptosis by inhibiting the p38-dependent mitochondrial pathway. World J. Gastroenterol. 2012, 18, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Nikoletopoulou, V.; Markaki, M.; Palikaras, K.; Tavernarakis, N. Crosstalk between apoptosis, necrosis and autophagy. Biochim. Biophys. Acta 2013, 12, 13. [Google Scholar] [CrossRef] [PubMed]
- Huebener, P.; Gwak, G.Y.; Schwabe, R.F. Comment on: Hmgb1-dependent and -independent autophagy. Autophagy 2015, 11, 1187–1188. [Google Scholar] [CrossRef] [PubMed]
- Kostova, N.; Zlateva, S.; Ugrinova, I.; Pasheva, E. The expression of hmgb1 protein and its receptor rage in human malignant tumors. Mol. Cell. Biochem. 2010, 337, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Hiwatashi, K.; Ueno, S.; Abeyama, K.; Kubo, F.; Sakoda, M.; Maruyama, I.; Hamanoue, M.; Natsugoe, S.; Aikou, T. A novel function of the receptor for advanced glycation end-products (rage) in association with tumorigenesis and tumor differentiation of hcc. Ann. Surg. Oncol. 2008, 15, 923–933. [Google Scholar] [CrossRef] [PubMed]
- Ashtari, S.; Pourhoseingholi, M.A.; Sharifian, A.; Zali, M.R. Hepatocellular carcinoma in asia: Prevention strategy and planning. World J. Hepatol. 2015, 7, 1708–1717. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Wang, Z.; Li, X.; Li, J.; Huang, Y.; Fan, X.; Duan, Y. Reduced high-mobility group box 1 expression induced by rna interference inhibits the bioactivity of hepatocellular carcinoma cell line hcclm3. Dig. Dis. Sci. 2012, 57, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Liu, Y.; Varley, P.; Chang, Y.; He, X.X.; Huang, H.; Tang, D.; Lotze, M.T.; Lin, J.; Tsung, A. High-mobility group box 1 promotes hepatocellular carcinoma progression through mir-21-mediated matrix metalloproteinase activity. Cancer Res. 2015, 75, 1645–1656. [Google Scholar] [CrossRef] [PubMed]
- Gong, W.; Wang, Z.Y.; Chen, G.X.; Liu, Y.Q.; Gu, X.Y.; Liu, W.W. Invasion potential of h22 hepatocarcinoma cells is increased by hmgb1-induced tumor nf-kappab signaling via initiation of hsp70. Oncol. Rep. 2013, 30, 1249–1256. [Google Scholar] [PubMed]
- Chen, R.C.; Yi, P.P.; Zhou, R.R.; Xiao, M.F.; Huang, Z.B.; Tang, D.L.; Huang, Y.; Fan, X.G. The role of hmgb1-rage axis in migration and invasion of hepatocellular carcinoma cell lines. Mol. Cell. Biochem. 2014, 390, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Sitia, G.; Iannacone, M.; Muller, S.; Bianchi, M.E.; Guidotti, L.G. Treatment with hmgb1 inhibitors diminishes ctl-induced liver disease in hbv transgenic mice. J. Leukoc. Biol. 2007, 81, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.X.; Wu, H.P.; Zhang, H.L.; Ashton, C.; Tong, C.; Wu, H.; Qian, Q.J.; Wang, H.Y.; Ying, Q.L. P53 promotes inflammation-associated hepatocarcinogenesis by inducing hmgb1 release. J. Hepatol. 2013, 59, 762–768. [Google Scholar] [CrossRef] [PubMed]
- Pusterla, T.; Nemeth, J.; Stein, I.; Wiechert, L.; Knigin, D.; Marhenke, S.; Longerich, T.; Kumar, V.; Arnold, B.; Vogel, A.; et al. Receptor for advanced glycation endproducts (rage) is a key regulator of oval cell activation and inflammation-associated liver carcinogenesis in mice. Hepatology 2013, 58, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Catanzaro, R.; Celep, G.; Illuzzi, N.; Milazzo, M.; Rastmanesh, R.; Yaduvanshi, S.K.; He, F.; Trushin, M.; Sapienza, C.; Srivastava, N.; et al. Anti-inflammatory and anti-mutagenic effect of the yhk phytocompound in hepatocytes: In view of an age-management liver-protecting approach. Rejuvenation Res. 2014, 17, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Tsung, A.; Zheng, N.; Jeyabalan, G.; Izuishi, K.; Klune, J.R.; Geller, D.A.; Lotze, M.T.; Lu, L.; Billiar, T.R. Increasing numbers of hepatic dendritic cells promote hmgb1-mediated ischemia-reperfusion injury. J. Leukoc. Biol. 2007, 81, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Ito, N.; DeMarco, R.A.; Mailliard, R.B.; Han, J.; Rabinowich, H.; Kalinski, P.; Stolz, D.B.; Zeh, H.J., 3rd; Lotze, M.T. Cytolytic cells induce hmgb1 release from melanoma cell lines. J. Leukoc. Biol. 2007, 81, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Scaffidi, P.; Misteli, T.; Bianchi, M.E. Release of chromatin protein hmgb1 by necrotic cells triggers inflammation. Nature 2002, 418, 191–195. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, L.X.; Pang, P.; Cui, Z.; Aung, S.; Haley, D.; Fox, B.A.; Urba, W.J.; Hu, H.M. Tumor-derived autophagosome vaccine: Mechanism of cross-presentation and therapeutic efficacy. Clin. Cancer Res. 2011, 17, 7047–7057. [Google Scholar] [CrossRef] [PubMed]
- Uramoto, H.; Izumi, H.; Nagatani, G.; Ohmori, H.; Nagasue, N.; Ise, T.; Yoshida, T.; Yasumoto, K.; Kohno, K. Physical interaction of tumour suppressor p53/p73 with ccaat-binding transcription factor 2 (ctf2) and differential regulation of human high-mobility group 1 (hmg1) gene expression. Biochem. J. 2003, 371, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Y.; Wang, H.C.; Fan, S.J. Growth suppression and radiosensitivity increase by hmgb1 in breast cancer. Acta Pharmacol. Sin. 2007, 28, 1957–1967. [Google Scholar] [CrossRef] [PubMed]
- Kang, R.; Zhang, Q.; Zeh, H.J., 3rd; Lotze, M.T.; Tang, D. Hmgb1 in cancer: Good, bad, or both? Clin. Cancer Res. 2013, 19, 4046–4057. [Google Scholar] [CrossRef] [PubMed]
- Kang, R.; Tang, D.; Schapiro, N.E.; Loux, T.; Livesey, K.M.; Billiar, T.R.; Wang, H.; van Houten, B.; Lotze, M.T.; Zeh, H.J. The hmgb1/rage inflammatory pathway promotes pancreatic tumor growth by regulating mitochondrial bioenergetics. Oncogene 2014, 33, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.D.; Cui, L.; Peng, C.H.; Cheng, D.F.; Han, B.S.; Huang, F. Expression and clinical significance of hmgb1 in human liver cancer: Knockdown inhibits tumor growth and metastasis in vitro and in vivo. Oncol. Rep. 2013, 29, 87–94. [Google Scholar] [PubMed]
- Ganz, M.; Bukong, T.N.; Csak, T.; Saha, B.; Park, J.K.; Ambade, A.; Kodys, K.; Szabo, G. Progression of non-alcoholic steatosis to steatohepatitis and fibrosis parallels cumulative accumulation of danger signals that promote inflammation and liver tumors in a high fat-cholesterol-sugar diet model in mice. J. Transl. Med. 2015, 13. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.L.; Yu, L.X.; Yang, W.; Tang, L.; Lin, Y.; Wu, H.; Zhai, B.; Tan, Y.X.; Shan, L.; Liu, Q.; et al. Profound impact of gut homeostasis on chemically-induced pro-tumorigenic inflammation and hepatocarcinogenesis in rats. J. Hepatol. 2012, 57, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Dou, C.; Wang, Y.; Jia, Y.; Li, Q.; Zheng, X.; Yao, Y.; Liu, Q.; Song, T. Highmobility group box 1 has a prognostic role and contributes to epithelial mesenchymal transition in human hepatocellular carcinoma. Mol. Med. Rep. 2015, 12, 5997–6004. [Google Scholar]
- Yu, L.X.; Yan, L.; Yang, W.; Wu, F.Q.; Ling, Y.; Chen, S.Z.; Tang, L.; Tan, Y.X.; Cao, D.; Wu, M.C.; et al. Platelets promote tumour metastasis via interaction between tlr4 and tumour cell-released high-mobility group box1 protein. Nat. Commun. 2014, 5, 5256. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Su, L.; Li, Q.; Sun, L.; Lv, J.; Li, J.; Cheng, B. Suppression of toll-like receptor 2 expression inhibits the bioactivity of human hepatocellular carcinoma. Tumour Biol. 2014, 35, 9627–9637. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Xiang, L.; Li, H.; Chen, P.; Feng, Y.; Zhang, J.; Yang, N.; Li, F.; Wang, Y.; Zhang, Q.; et al. The Role of HMGB1 Signaling Pathway in the Development and Progression of Hepatocellular Carcinoma: A Review. Int. J. Mol. Sci. 2015, 16, 22527-22540. https://doi.org/10.3390/ijms160922527
Wang X, Xiang L, Li H, Chen P, Feng Y, Zhang J, Yang N, Li F, Wang Y, Zhang Q, et al. The Role of HMGB1 Signaling Pathway in the Development and Progression of Hepatocellular Carcinoma: A Review. International Journal of Molecular Sciences. 2015; 16(9):22527-22540. https://doi.org/10.3390/ijms160922527
Chicago/Turabian StyleWang, Xuanbin, Longchao Xiang, Hongliang Li, Ping Chen, Yibin Feng, Jingxuan Zhang, Nian Yang, Fei Li, Ye Wang, Quifang Zhang, and et al. 2015. "The Role of HMGB1 Signaling Pathway in the Development and Progression of Hepatocellular Carcinoma: A Review" International Journal of Molecular Sciences 16, no. 9: 22527-22540. https://doi.org/10.3390/ijms160922527






