Quantitative Real-Time PCR Analysis of YKL-40 and Its Comparison with Mammalian Chitinase mRNAs in Normal Human Tissues Using a Single Standard DNA
Abstract
:1. Introduction
2. Results
2.1. Establishment of the Real-Time PCR System for Detection of YKL-40 mRNA in Human Tissues
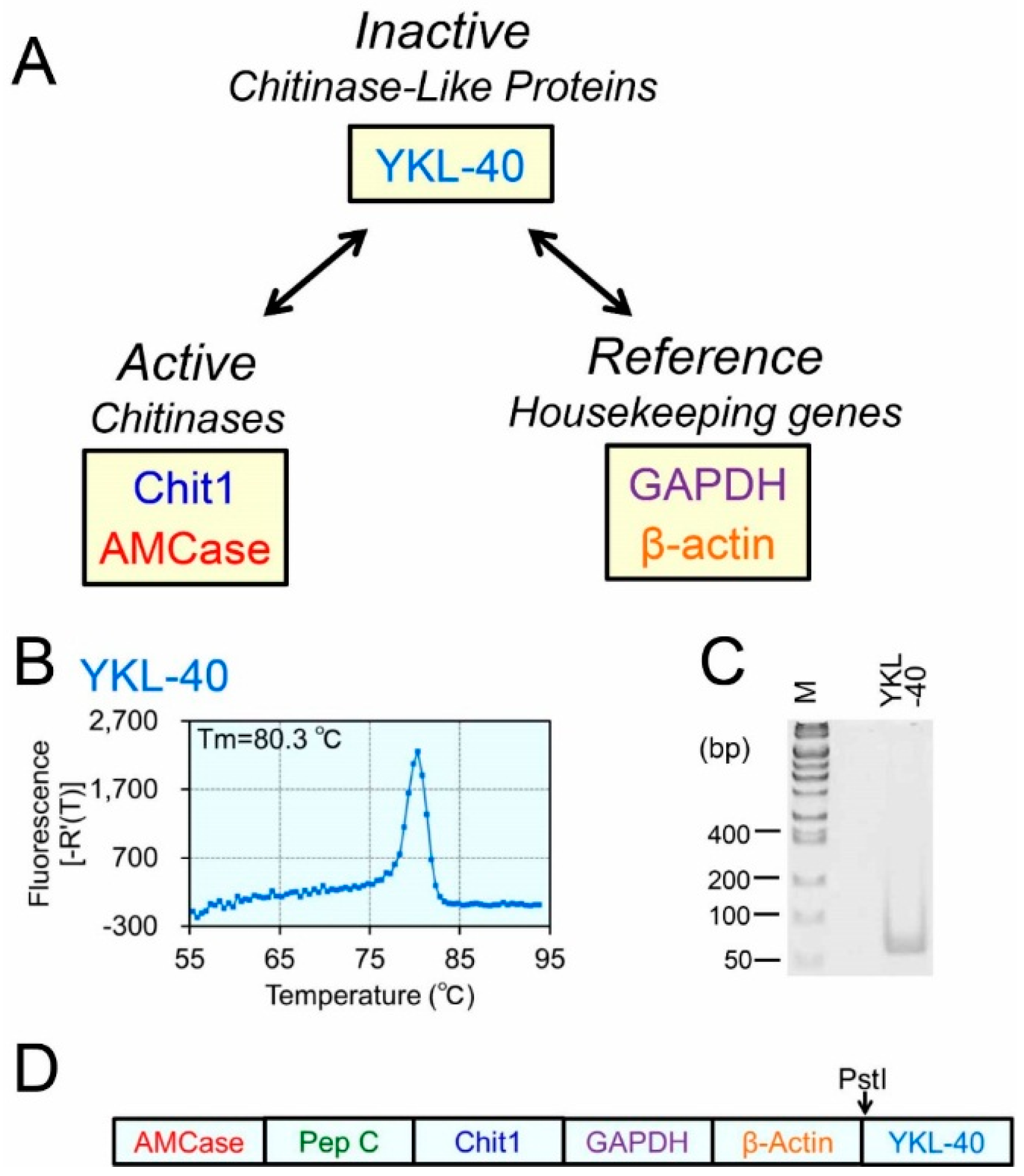
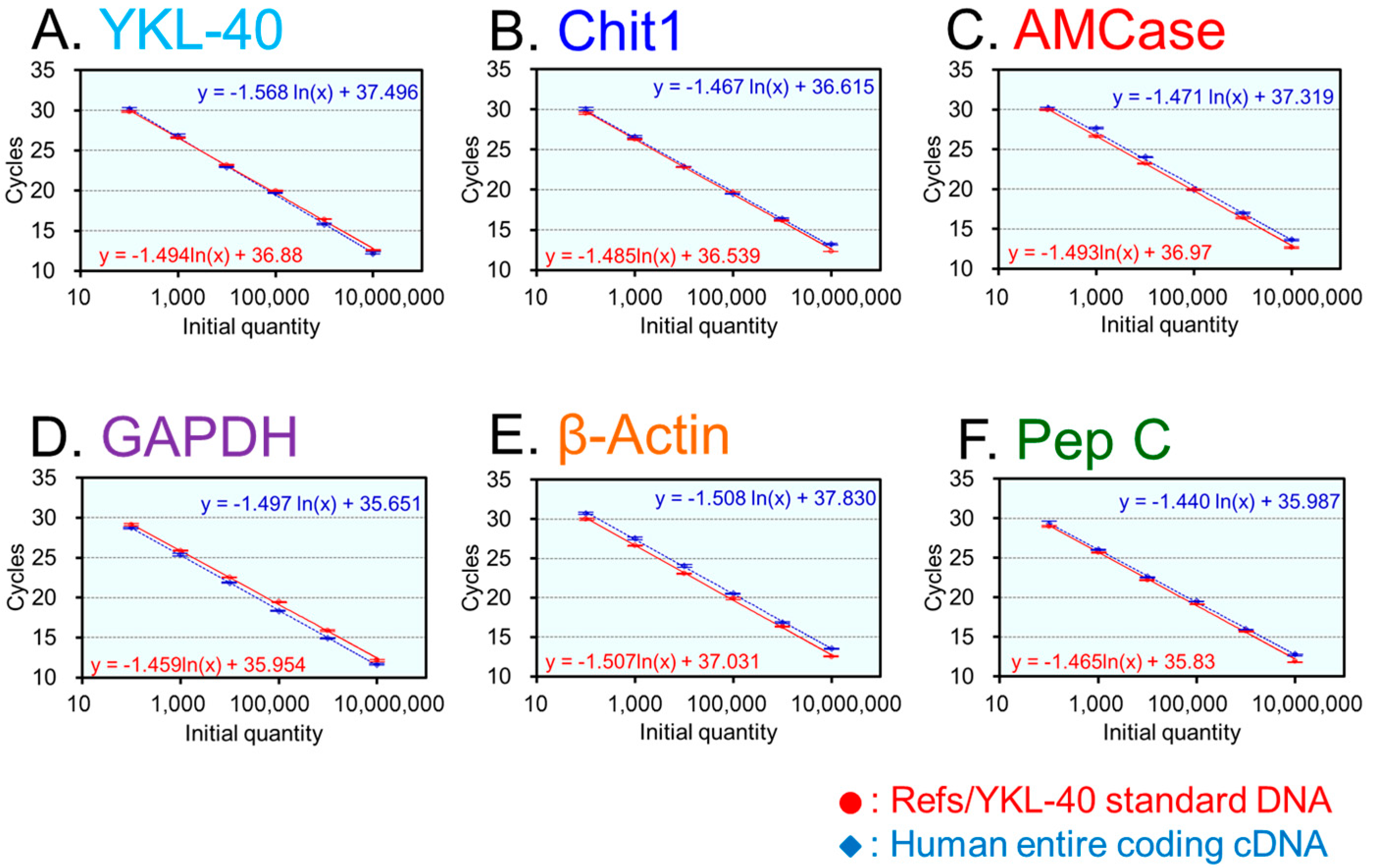
2.2. Construction of the Human Refs/YKL-40 Standard DNA and Validation of Our qPCR System
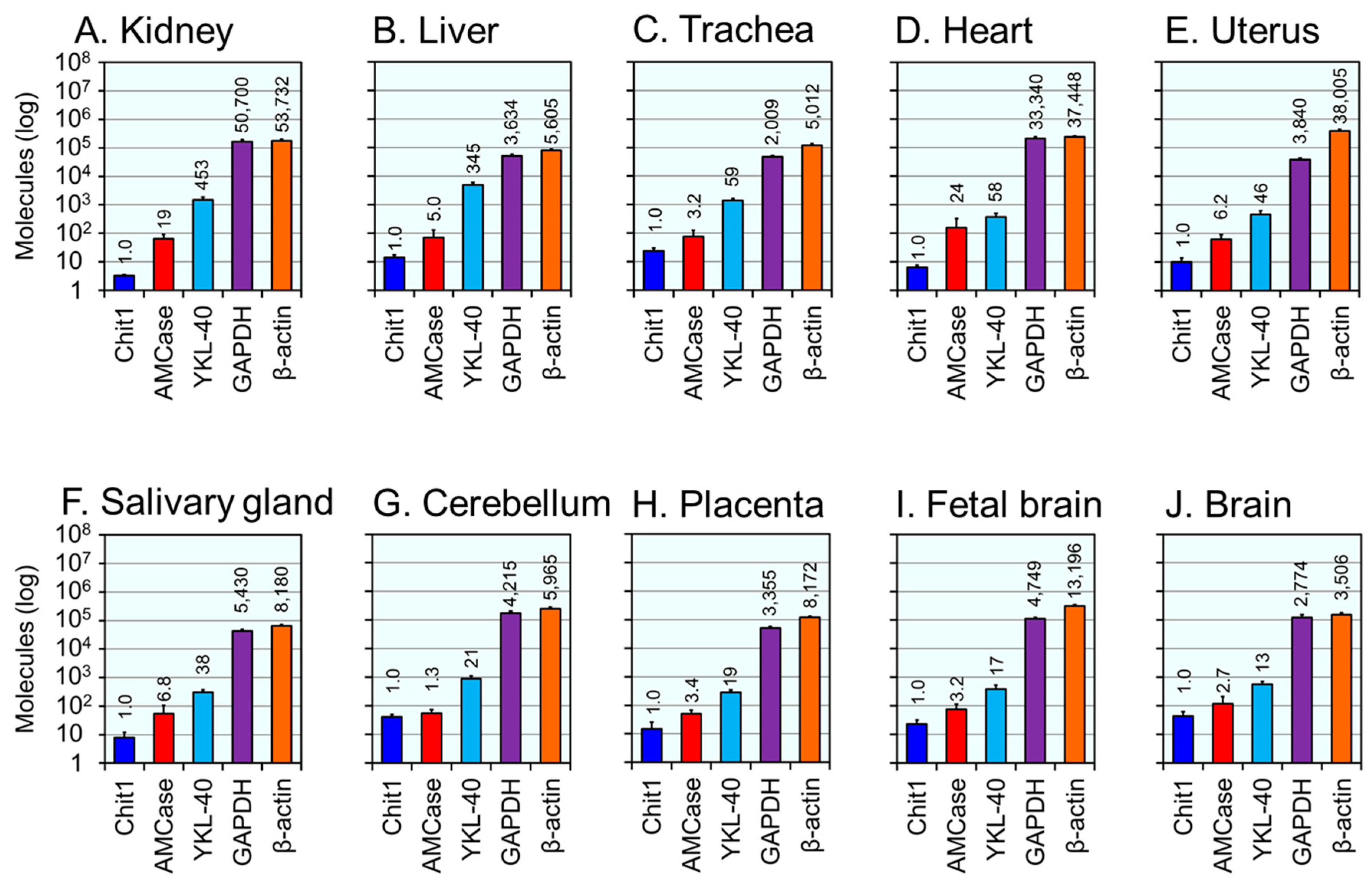
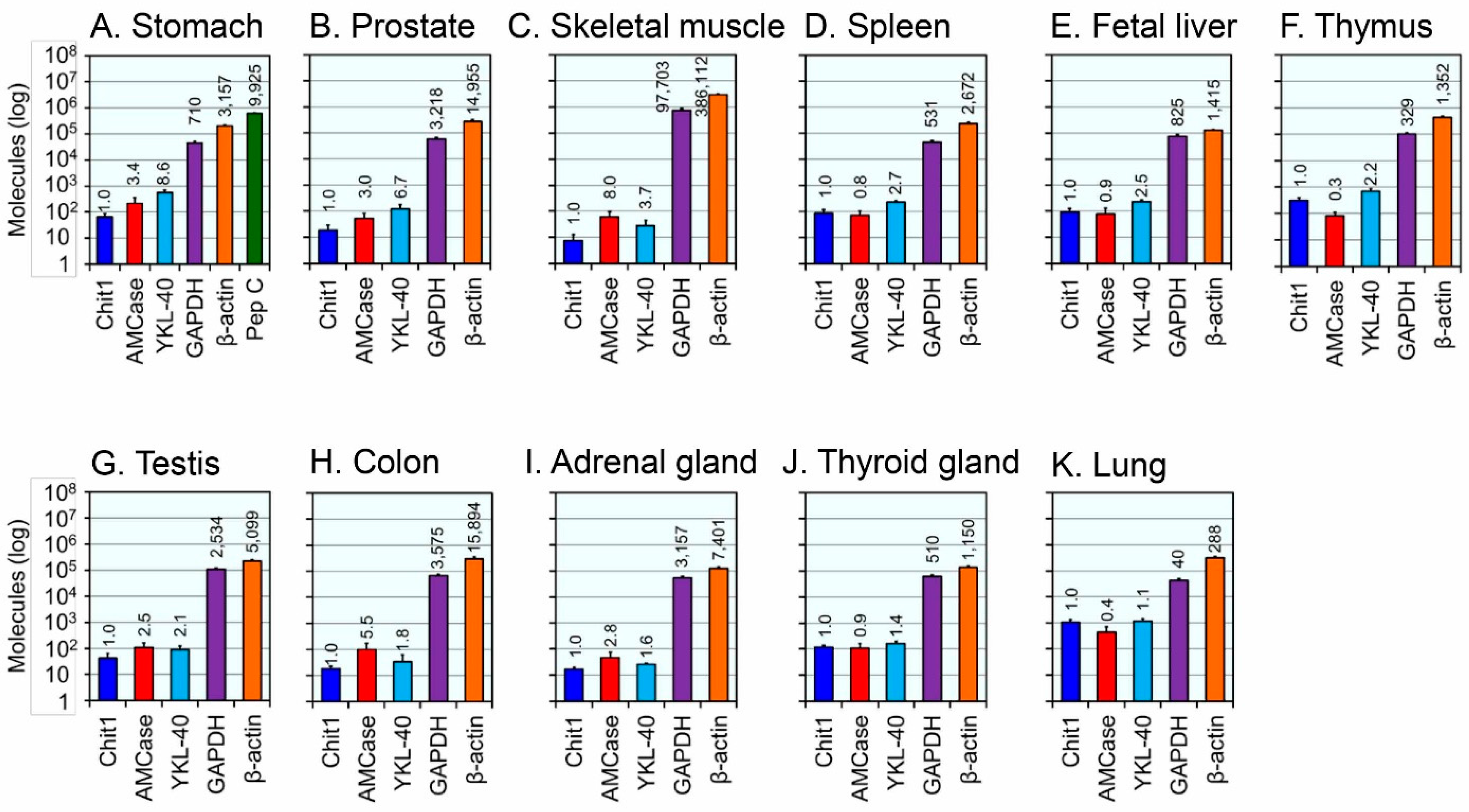
2.3. Expression Levels of the YKL-40 mRNA in Normal Human Tissues

2.4. Comparison of YKL-40, Mammalian Chitinases and Housekeeping Genes mRNA Levels in Healthy Human Tissues
3. Discussion
4. Experimental Section
4.1. RNA and cDNA Preparation
4.2. Selection of Primer Pairs for qPCR
4.3. Construction of the Human Refs/YKL-40 Standard DNA
4.4. Preparation of YKL-40 cDNAs Covering the Entire Coding Region
4.5. Standard Curves and mRNA Quantification Using Real-Time PCR
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Khoushab, F.; Yamabhai, M. Chitin research revisited. Mar. Drugs 2010, 8, 1988–2012. [Google Scholar] [CrossRef] [PubMed]
- Bueter, C.L.; Specht, C.A.; Levitz, S.M. Innate sensing of chitin and chitosan. PLoS Pathog. 2013, 9, e1003080. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.G.; Da Silva, C.A.; Dela Cruz, C.S.; Ahangari, F.; Ma, B.; Kang, M.J.; He, C.H.; Takyar, S.; Elias, J.A. Role of chitin and chitinase/chitinase-like proteins in inflammation, tissue remodeling, and injury. Annu. Rev. Physiol. 2011, 73, 479–501. [Google Scholar] [CrossRef] [PubMed]
- Bussink, A.P.; Speijer, D.; Aerts, J.M.; Boot, R.G. Evolution of mammalian chitinase(-like) members of family 18 glycosyl hydrolases. Genetics 2007, 177, 959–970. [Google Scholar] [CrossRef] [PubMed]
- Hollak, C.E.; van Weely, S.; van Oers, M.H.; Aerts, J.M. Marked elevation of plasma chitotriosidase activity. A novel hallmark of gaucher disease. J. Clin. Investig. 1994, 93, 1288–1292. [Google Scholar] [CrossRef] [PubMed]
- Renkema, G.H.; Boot, R.G.; Muijsers, A.O.; Donker-Koopman, W.E.; Aerts, J.M. Purification and characterization of human chitotriosidase, a novel member of the chitinase family of proteins. J. Biol. Chem. 1995, 270, 2198–2202. [Google Scholar] [CrossRef] [PubMed]
- Boot, R.G.; Renkema, G.H.; Strijland, A.; van Zonneveld, A.J.; Aerts, J.M. Cloning of a cDNA encoding chitotriosidase, a human chitinase produced by macrophages. J. Biol. Chem. 1995, 270, 26252–26256. [Google Scholar] [CrossRef] [PubMed]
- Boot, R.G.; Blommaart, E.F.; Swart, E.; Ghauharali-van der Vlugt, K.; Bijl, N.; Moe, C.; Place, A.; Aerts, J.M. Identification of a novel acidic mammalian chitinase distinct from chitotriosidase. J. Biol. Chem. 2001, 276, 6770–6778. [Google Scholar] [CrossRef] [PubMed]
- Boot, R.G.; Bussink, A.P.; Verhoek, M.; de Boer, P.A.; Moorman, A.F.; Aerts, J.M. Marked differences in tissue-specific expression of chitinases in mouse and man. J. Histochem. Cytochem. 2005, 53, 1283–1292. [Google Scholar] [CrossRef] [PubMed]
- Kawada, M.; Hachiya, Y.; Arihiro, A.; Mizoguchi, E. Role of mammalian chitinases in inflammatory conditions. Keio J. Med. 2007, 56, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Henrissat, B. A classification of glycosyl hydrolases based on amino acid sequence similarities. Biochem. J. 1991, 280, 309–316. [Google Scholar] [PubMed]
- Cantarel, B.L.; Coutinho, P.M.; Rancurel, C.; Bernard, T.; Lombard, V.; Henrissat, B. The carbohydrate-active enzymes database (cazy): An expert resource for glycogenomics. Nucleic Acids Res. 2009, 37, D233–D238. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, T.; Kobori, K.; Miyashita, K.; Fujii, T.; Sakai, H.; Uchida, M.; Tanaka, H. Identification of glutamic acid 204 and aspartic acid 200 in chitinase a1 of bacillus circulans WL-12 as essential residues for chitinase activity. J. Biol. Chem. 1993, 268, 18567–18572. [Google Scholar] [PubMed]
- Hakala, B.E.; White, C.; Recklies, A.D. Human cartilage gp-39, a major secretory product of articular chondrocytes and synovial cells, is a mammalian member of a chitinase protein family. J. Biol. Chem. 1993, 268, 25803–25810. [Google Scholar] [PubMed]
- Rehli, M.; Krause, S.W.; Andreesen, R. Molecular characterization of the gene for human cartilage gp-39 (CHI3L1), a member of the chitinase protein family and marker for late stages of macrophage differentiation. Genomics 1997, 43, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.M.; Copeland, N.G.; Gilbert, D.J.; Jenkins, N.A.; Kirkpatrick, R.B.; Rosenberg, M. Genetic characterization of the murine Ym1 gene and identification of a cluster of highly homologous genes. Genomics 1998, 54, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Webb, D.C.; McKenzie, A.N.; Foster, P.S. Expression of the Ym2 lectin-binding protein is dependent on interleukin (IL)-4 and IL-13 signal transduction: Identification of a novel allergy-associated protein. J. Biol. Chem. 2001, 276, 41969–41976. [Google Scholar] [CrossRef] [PubMed]
- Shackelton, L.M.; Mann, D.M.; Millis, A.J. Identification of a 38-kDa heparin-binding glycoprotein (gp38k) in differentiating vascular smooth muscle cells as a member of a group of proteins associated with tissue remodeling. J. Biol. Chem. 1995, 270, 13076–13083. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Trinh, K.; Figueira, W.F.; Price, P.A. Isolation and sequence of a novel human chondrocyte protein related to mammalian members of the chitinase protein family. J. Biol. Chem. 1996, 271, 19415–19420. [Google Scholar] [CrossRef] [PubMed]
- Arias, E.B.; Verhage, H.G.; Jaffe, R.C. Complementary deoxyribonucleic acid cloning and molecular characterization of an estrogen-dependent human oviductal glycoprotein. Biol. Reprod. 1994, 51, 685–694. [Google Scholar] [CrossRef] [PubMed]
- Sendai, Y.; Komiya, H.; Suzuki, K.; Onuma, T.; Kikuchi, M.; Hoshi, H.; Araki, Y. Molecular cloning and characterization of a mouse oviduct-specific glycoprotein. Biol. Reprod. 1995, 53, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Kzhyshkowska, J.; Mamidi, S.; Gratchev, A.; Kremmer, E.; Schmuttermaier, C.; Krusell, L.; Haus, G.; Utikal, J.; Schledzewski, K.; Scholtze, J.; et al. Novel stabilin-1 interacting chitinase-like protein (SI-CLP) is up-regulated in alternatively activated macrophages and secreted via lysosomal pathway. Blood 2006, 107, 3221–3228. [Google Scholar] [CrossRef] [PubMed]
- Morrison, B.W.; Leder, P. Neu and ras initiate murine mammary tumors that share genetic markers generally absent in c-myc and int-2-initiated tumors. Oncogene 1994, 9, 3417–3426. [Google Scholar] [PubMed]
- Johansen, J.S. Studies on serum YKL-40 as a biomarker in diseases with inflammation, tissue remodelling, fibroses and cancer. Dan. Med. Bull. 2006, 53, 172–209. [Google Scholar] [PubMed]
- Lee, C.G.; Hartl, D.; Lee, G.R.; Koller, B.; Matsuura, H.; Da Silva, C.A.; Sohn, M.H.; Cohn, L.; Homer, R.J.; Kozhich, A.A.; et al. Role of breast regression protein 39 (BRP-39)/chitinase 3-like-1 in Th2 and IL-13-induced tissue responses and apoptosis. J. Exp. Med. 2009, 206, 1149–1166. [Google Scholar] [CrossRef] [PubMed]
- Sohn, M.H.; Kang, M.J.; Matsuura, H.; Bhandari, V.; Chen, N.Y.; Lee, C.G.; Elias, J.A. The chitinase-like proteins breast regression protein-39 and YKL-40 regulate hyperoxia-induced acute lung injury. Am. J. Respir. Crit. Care Med. 2010, 182, 918–928. [Google Scholar] [CrossRef] [PubMed]
- Letuve, S.; Kozhich, A.; Arouche, N.; Grandsaigne, M.; Reed, J.; Dombret, M.C.; Kiener, P.A.; Aubier, M.; Coyle, A.J.; Pretolani, M. YKL-40 is elevated in patients with chronic obstructive pulmonary disease and activates alveolar macrophages. J. Immunol. 2008, 181, 5167–5173. [Google Scholar] [CrossRef] [PubMed]
- Hector, A.; Kormann, M.S.; Mack, I.; Latzin, P.; Casaulta, C.; Kieninger, E.; Zhou, Z.; Yildirim, A.O.; Bohla, A.; Rieber, N.; et al. The chitinase-like protein YKL-40 modulates cystic fibrosis lung disease. PLoS ONE 2011, 6, e24399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johansen, J.S.; Stoltenberg, M.; Hansen, M.; Florescu, A.; Horslev-Petersen, K.; Lorenzen, I.; Price, P.A. Serum YKL-40 concentrations in patients with rheumatoid arthritis: Relation to disease activity. Rheumatology 1999, 38, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Bernardi, D.; Podswiadek, M.; Zaninotto, M.; Punzi, L.; Plebani, M. YKL-40 as a marker of joint involvement in inflammatory bowel disease. Clin. Chem. 2003, 49, 1685–1688. [Google Scholar] [CrossRef] [PubMed]
- Koutroubakis, I.E.; Petinaki, E.; Dimoulios, P.; Vardas, E.; Roussomoustakaki, M.; Maniatis, A.N.; Kouroumalis, E.A. Increased serum levels of YKL-40 in patients with inflammatory bowel disease. Int. J. Colorectal Dis. 2003, 18, 254–259. [Google Scholar] [PubMed]
- Vind, I.; Johansen, J.S.; Price, P.A.; Munkholm, P. Serum YKL-40, a potential new marker of disease activity in patients with inflammatory bowel disease. Scand. J. Gastroenterol. 2003, 38, 599–605. [Google Scholar] [CrossRef] [PubMed]
- Chupp, G.L.; Lee, C.G.; Jarjour, N.; Shim, Y.M.; Holm, C.T.; He, S.; Dziura, J.D.; Reed, J.; Coyle, A.J.; Kiener, P.; et al. A chitinase-like protein in the lung and circulation of patients with severe asthma. N. Engl. J. Med. 2007, 357, 2016–2027. [Google Scholar] [CrossRef] [PubMed]
- Ober, C.; Tan, Z.; Sun, Y.; Possick, J.D.; Pan, L.; Nicolae, R.; Radford, S.; Parry, R.R.; Heinzmann, A.; Deichmann, K.A.; et al. Effect of variation in CHI3L1 on serum YKL-40 level, risk of asthma, and lung function. N. Engl. J. Med. 2008, 358, 1682–1691. [Google Scholar] [CrossRef] [PubMed]
- Vos, K.; Steenbakkers, P.; Miltenburg, A.M.; Bos, E.; van Den Heuvel, M.W.; van Hogezand, R.A.; de Vries, R.R.; Breedveld, F.C.; Boots, A.M. Raised human cartilage glycoprotein-39 plasma levels in patients with rheumatoid arthritis and other inflammatory conditions. Ann. Rheum. Dis. 2000, 59, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Johansen, J.S.; Moller, S.; Price, P.A.; Bendtsen, F.; Junge, J.; Garbarsch, C.; Henriksen, J.H. Plasma YKL-40: A new potential marker of fibrosis in patients with alcoholic cirrhosis? Scand. J. Gastroenterol. 1997, 32, 582–590. [Google Scholar] [CrossRef] [PubMed]
- Johansen, J.S.; Cintin, C.; Jorgensen, M.; Kamby, C.; Price, P.A. Serum YKL-40: A new potential marker of prognosis and location of metastases of patients with recurrent breast cancer. Eur. J. Cancer 1995, 31A, 1437–1442. [Google Scholar] [CrossRef]
- Cintin, C.; Johansen, J.S.; Christensen, I.J.; Price, P.A.; Sorensen, S.; Nielsen, H.J. Serum YKL-40 and colorectal cancer. Br. J. Cancer 1999, 79, 1494–1499. [Google Scholar] [CrossRef] [PubMed]
- Prakash, M.; Bodas, M.; Prakash, D.; Nawani, N.; Khetmalas, M.; Mandal, A.; Eriksson, C. Diverse pathological implications of YKL-40: Answers may lie in “outside-in” signaling. Cell Signal. 2013, 25, 1567–1573. [Google Scholar] [CrossRef] [PubMed]
- Letuve, S.; Kozhich, A.; Humbles, A.; Brewah, Y.; Dombret, M.C.; Grandsaigne, M.; Adle, H.; Kolbeck, R.; Aubier, M.; Coyle, A.J.; et al. Lung chitinolytic activity and chitotriosidase are elevated in chronic obstructive pulmonary disease and contribute to lung inflammation. Am. J. Pathol. 2010, 176, 638–649. [Google Scholar] [CrossRef] [PubMed]
- Watabe-Rudolph, M.; Song, Z.; Lausser, L.; Schnack, C.; Begus-Nahrmann, Y.; Scheithauer, M.O.; Rettinger, G.; Otto, M.; Tumani, H.; Thal, D.R.; et al. Chitinase enzyme activity in csf is a powerful biomarker of alzheimer disease. Neurology 2012, 78, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Bierbaum, S.; Nickel, R.; Koch, A.; Lau, S.; Deichmann, K.A.; Wahn, U.; Superti-Furga, A.; Heinzmann, A. Polymorphisms and haplotypes of acid mammalian chitinase are associated with bronchial asthma. Am. J. Respir. Crit. Care Med. 2005, 172, 1505–1509. [Google Scholar] [CrossRef] [PubMed]
- Seibold, M.A.; Donnelly, S.; Solon, M.; Innes, A.; Woodruff, P.G.; Boot, R.G.; Burchard, E.G.; Fahy, J.V. Chitotriosidase is the primary active chitinase in the human lung and is modulated by genotype and smoking habit. J. Allergy Clin. Immunol. 2008, 122, 944–950. [Google Scholar] [CrossRef] [PubMed]
- Seibold, M.A.; Reese, T.A.; Choudhry, S.; Salam, M.T.; Beckman, K.; Eng, C.; Atakilit, A.; Meade, K.; Lenoir, M.; Watson, H.G.; et al. Differential enzymatic activity of common haplotypic versions of the human acidic mammalian chitinase protein. J. Biol. Chem. 2009, 284, 19650–19658. [Google Scholar] [CrossRef] [PubMed]
- Ohno, M.; Tsuda, K.; Sakaguchi, M.; Sugahara, Y.; Oyama, F. Chitinase mRNA levels by quantitative PCR using the single standard DNA: Acidic mammalian chitinase is a major transcript in the mouse stomach. PLoS ONE 2012, 7, e50381. [Google Scholar] [CrossRef] [PubMed]
- Ohno, M.; Togashi, Y.; Tsuda, K.; Okawa, K.; Kamaya, M.; Sakaguchi, M.; Sugahara, Y.; Oyama, F. Quantification of chitinase mRNA levels in human and mouse tissues by real-time PCR: Species-specific expression of acidic mammalian chitinase in stomach tissues. PLoS ONE 2013, 8, e67399. [Google Scholar] [CrossRef] [PubMed]
- Ohno, M.; Kida, Y.; Sakaguchi, M.; Sugahara, Y.; Oyama, F. Establishment of a quantitative PCR system for discriminating chitinase-like proteins: Catalytically inactive breast regression protein-39 and Ym1 are constitutive genes in mouse lung. BMC Mol. Biol. 2014, 15, 23. [Google Scholar] [CrossRef] [PubMed]
- Kouadjo, K.E.; Nishida, Y.; Cadrin-Girard, J.F.; Yoshioka, M.; St-Amand, J. Housekeeping and tissue-specific genes in mouse tissues. BMC Genomics 2007, 8, 127. [Google Scholar] [CrossRef] [PubMed]
- Dabek, J.; Wilczok, J.; Kulach, A.; Gasior, Z. Altered transcriptional activity of gene encoding GAPDH in peripheral blood mononuclear cells from patients with cardiac syndrome X—An important part in pathology of microvascular angina? Arch. Med. Sci. 2010, 6, 709–712. [Google Scholar] [CrossRef] [PubMed]
- Zainuddin, A.; Chua, K.H.; Abdul Rahim, N.; Makpol, S. Effect of experimental treatment on GAPDH mRNA expression as a housekeeping gene in human diploid fibroblasts. BMC Mol. Biol. 2010, 11, 59. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Zheng, T.; Homer, R.J.; Kim, Y.K.; Chen, N.Y.; Cohn, L.; Hamid, Q.; Elias, J.A. Acidic mammalian chitinase in asthmatic Th2 inflammation and IL-13 pathway activation. Science 2004, 304, 1678–1682. [Google Scholar] [CrossRef] [PubMed]
- Reese, T.A.; Liang, H.E.; Tager, A.M.; Luster, A.D.; Van Rooijen, N.; Voehringer, D.; Locksley, R.M. Chitin induces accumulation in tissue of innate immune cells associated with allergy. Nature 2007, 447, 92–96. [Google Scholar] [CrossRef] [PubMed]
- He, C.H.; Lee, C.G.; Dela Cruz, C.S.; Lee, C.M.; Zhou, Y.; Ahangari, F.; Ma, B.; Herzog, E.L.; Rosenberg, S.A.; Li, Y.; et al. Chitinase 3-like 1 regulates cellular and tissue responses via IL-13 receptor α2. Cell Rep. 2013, 4, 830–841. [Google Scholar] [CrossRef] [PubMed]
- Dela Cruz, C.S.; Liu, W.; He, C.H.; Jacoby, A.; Gornitzky, A.; Ma, B.; Flavell, R.; Lee, C.G.; Elias, J.A. Chitinase 3-like-1 promotes streptococcus pneumoniae killing and augments host tolerance to lung antibacterial responses. Cell Host Microbe 2012, 12, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A.; Beaulieu, J.F.; Huggett, J.; Jaggi, R.; Kibenge, F.S.; Olsvik, P.A.; Penning, L.C.; Toegel, S. MIQE precis: Practical implementation of minimum standard guidelines for fluorescence-based quantitative real-time pcr experiments. BMC Mol. Biol. 2010, 11, 74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ohno, M.; Bauer, P.O.; Kida, Y.; Sakaguchi, M.; Sugahara, Y.; Oyama, F. Quantitative Real-Time PCR Analysis of YKL-40 and Its Comparison with Mammalian Chitinase mRNAs in Normal Human Tissues Using a Single Standard DNA. Int. J. Mol. Sci. 2015, 16, 9922-9935. https://doi.org/10.3390/ijms16059922
Ohno M, Bauer PO, Kida Y, Sakaguchi M, Sugahara Y, Oyama F. Quantitative Real-Time PCR Analysis of YKL-40 and Its Comparison with Mammalian Chitinase mRNAs in Normal Human Tissues Using a Single Standard DNA. International Journal of Molecular Sciences. 2015; 16(5):9922-9935. https://doi.org/10.3390/ijms16059922
Chicago/Turabian StyleOhno, Misa, Peter O. Bauer, Yuta Kida, Masayoshi Sakaguchi, Yasusato Sugahara, and Fumitaka Oyama. 2015. "Quantitative Real-Time PCR Analysis of YKL-40 and Its Comparison with Mammalian Chitinase mRNAs in Normal Human Tissues Using a Single Standard DNA" International Journal of Molecular Sciences 16, no. 5: 9922-9935. https://doi.org/10.3390/ijms16059922





