Rapid and Efficient Functionalized Ionic Liquid-Catalyzed Aldol Condensation Reactions Associated with Microwave Irradiation
Abstract
:1. Introduction
2. Results and Discussion
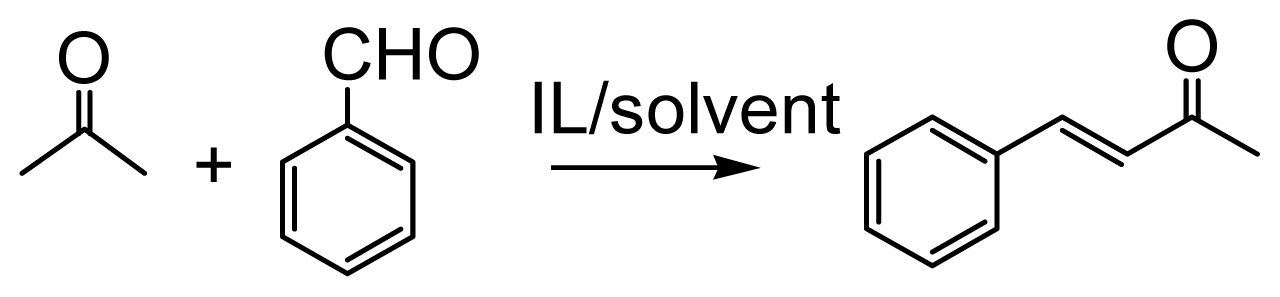
2.1. Effect of Solvents on the Aldol Condensation Reaction
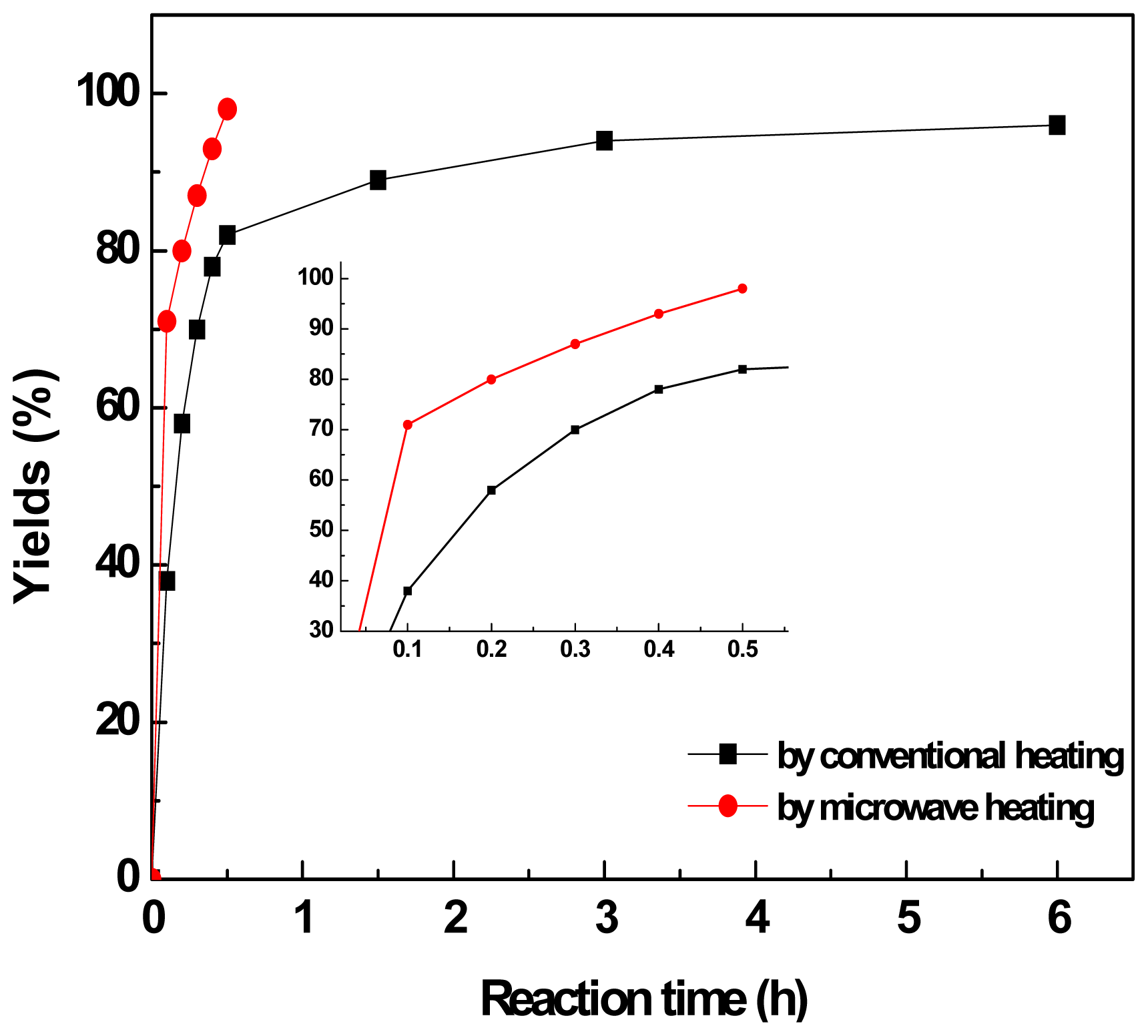
2.2. Comparison of the Aldol Condensation Reactions under Different Heating Conditions
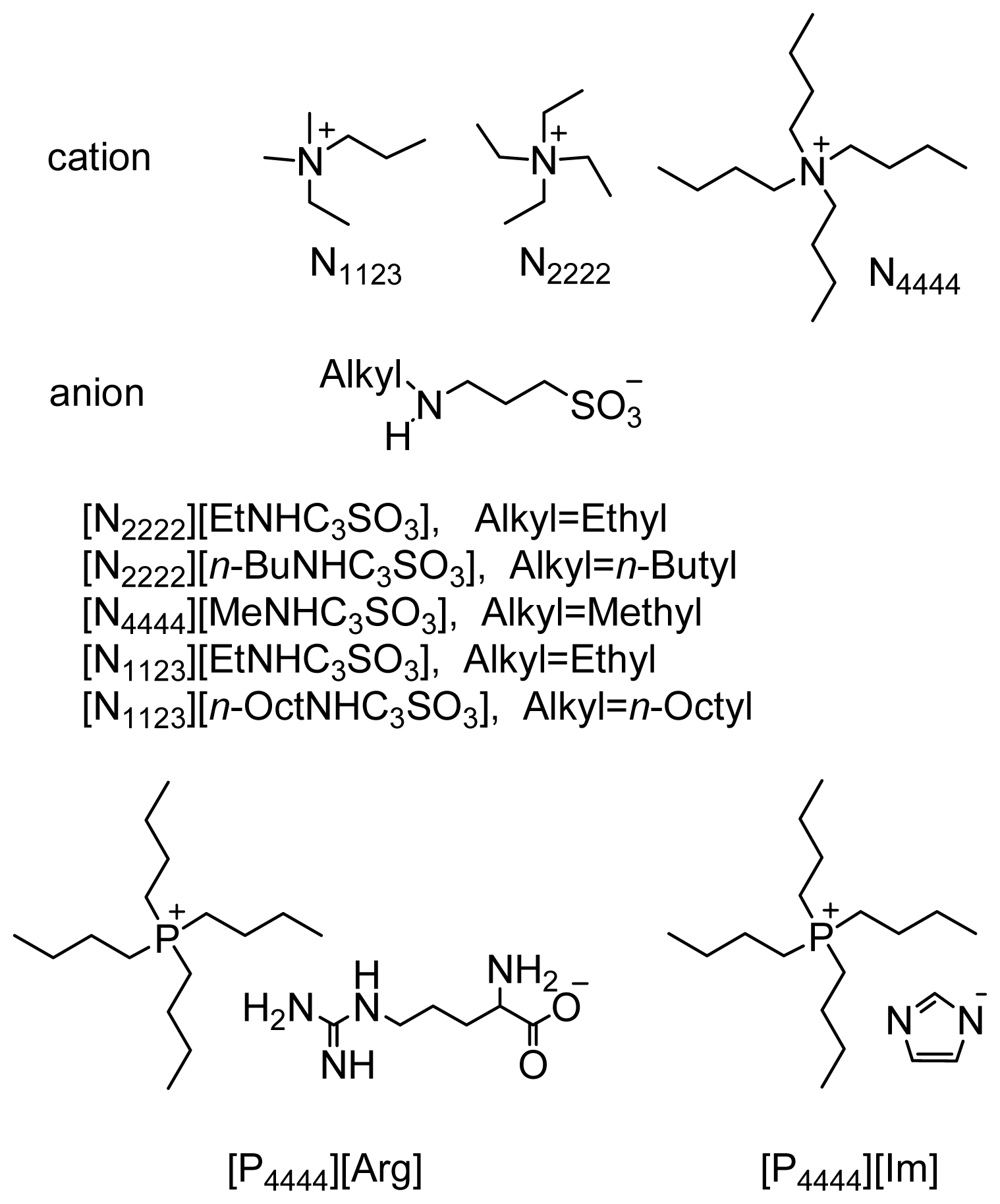
2.3. Comparison of the Aldol Condensation Reactions in Different ILs
2.4. Recycling of the Functionalized ILs
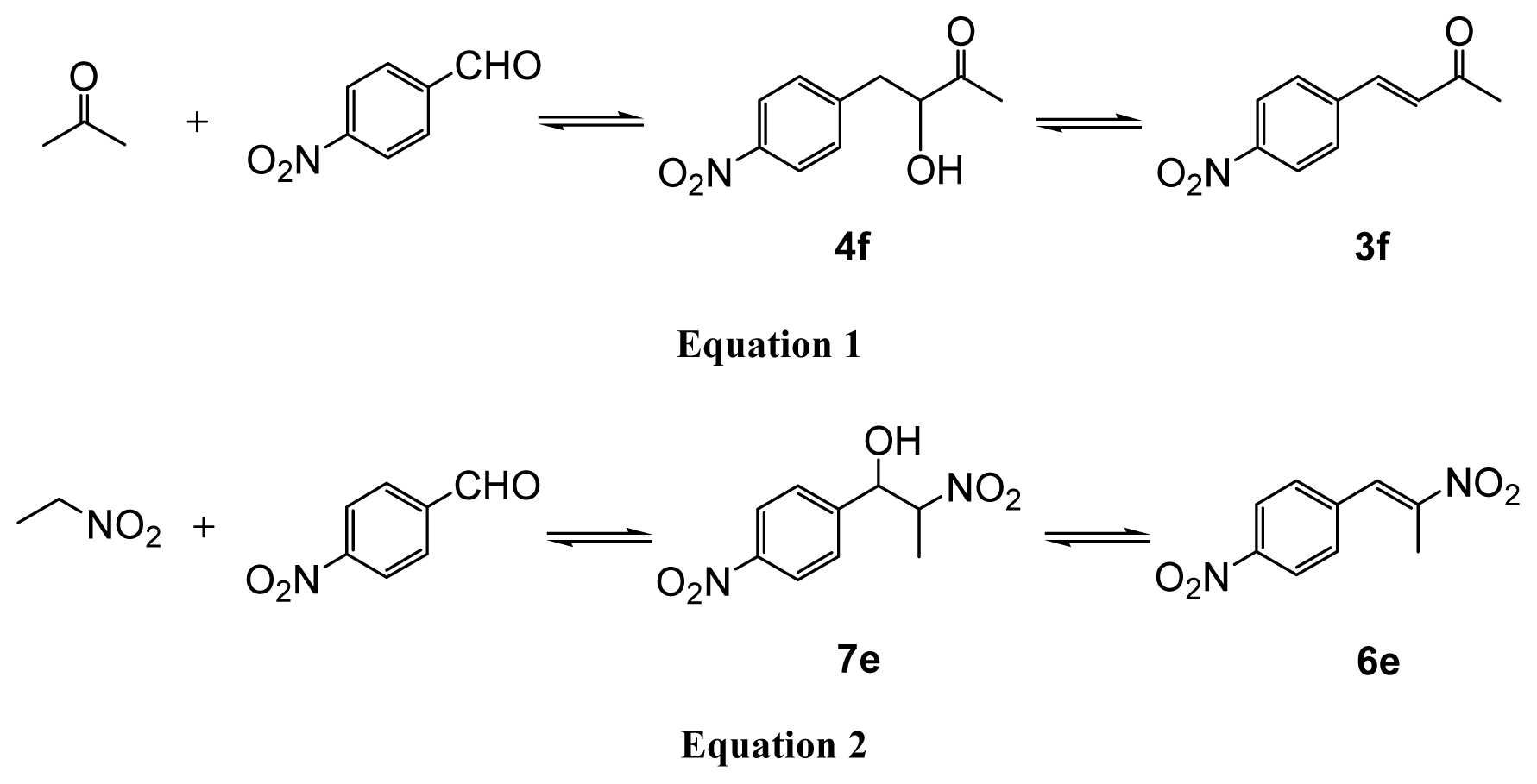
2.5. Influence of Different Substituents on Aromatic Aldehydes
2.6. Extension to the Henry Reactions
3. Experimental Section
3.1. General Information
3.2. Synthesis and Characterization of Functionalized ILs
3.3. General Procedure for the Aldol and Henry Reactions
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Anastas, P.T.; Williamson, T.C. Green Chemistry: An Overview. In Green Chemistry: Designing Chemistry for the Environment; Anastas, P.T., Williamson, T.C., Eds.; American Chemical Society: Washington, DC, USA, 1996; Volume 626, pp. 1–17. [Google Scholar]
- Jessop, P.G.; Heldebrant, D.J.; Li, X.W.; Eckert, C.A.; Liotta, C.L. Green chemistry: Reversible nonpolar-to-polar solvent. Nature 2005, 436, 1102–1102. [Google Scholar]
- Wang, C.; Mahurin, S.M.; Luo, H.; Baker, G.A.; Li, H.; Dai, S. Reversible and robust CO2 capture by equimolar task-specific ionic liquid-superbase mixtures. Green Chem 2010, 12, 870–874. [Google Scholar]
- Davis, J.H. Task-specific ionic liquids. Chem. Lett 2004, 33, 1072–1077. [Google Scholar]
- Welton, T. Room-temperature ionic liquids: Solvents for synthesis and catalysis. Chem. Rev 1999, 99, 2071–2083. [Google Scholar]
- Hallett, J.P.; Welton, T. Room-temperature ionic liquids: Solvents for synthesis and catalysis. 2. Chem. Rev 2011, 111, 3508–3576. [Google Scholar]
- Wasserscheid, P.; Keim, W. Ionic liquids: New “solutions” for transition metal catalysis. Angew. Chem. Int. Ed 2000, 39, 3772–3789. [Google Scholar]
- Cole, A.C.; Jensen, J.L.; Ntai, I.; Tran, K.L.T.; Weaver, K.J.; Forbes, D.C.; Davis, J.H. Novel brønsted acidic ionic liquids and their use as dual solvent-catalysts. J. Am. Chem. Soc 2002, 124, 5962–5963. [Google Scholar]
- Ranu, B.C.; Banerjee, S. Ionic liquid as catalyst and reaction medium. The dramatic influence of a task-specific ionic liquid, [bmim] OH, in Michael addition of active methylene compounds to conjugated ketones, carboxylic esters and nitriles. Org. Lett 2005, 7, 3049–3052. [Google Scholar]
- Saito, S.; Shiozawa, M.; Ito, M.; Yamamoto, H. Conceptually new directed aldol condensation using aluminum tris(2,6-diphenylphenoxide). J. Am. Chem. Soc 1998, 120, 813–814. [Google Scholar]
- Saito, S.; Yamamoto, H. Directed aldol condensation. Chem. Eur. J 1999, 5, 1959–1962. [Google Scholar]
- Mukaiyama, T. Explorations into new reaction chemistry. Angew. Chem. Int. Ed 2004, 43, 5590–5614. [Google Scholar]
- Palomo, C.; Oiarbide, M.; Garcia, J.M. Current progress in the asymmetric aldol addition reaction. Chem. Soc. Rev 2004, 33, 65–75. [Google Scholar]
- Alonso, D.M.; Bond, J.Q.; Dumesic, J.A. Catalytic conversion of biomass to biofuels. Green Chem 2010, 12, 1493–1513. [Google Scholar]
- Hathaway, B.A. An aldol condensation experiment using a number of aldehydes and ketones. J. Chem. Educ 1987, 64, 367–368. [Google Scholar]
- Huitric, A.C.; Kumler, W.D. The dipole moments, spectra and structure of some new 2-phenyl-cyclohexanones, 2-benzyl-cyclohexanones, 2-(para-halobenzylidene)-cyclohexanones and 2,6-bis(para-halobenzylidene)-cyclohexanones. J. Am. Chem. Soc 1956, 78, 614–622. [Google Scholar]
- Bigdeli, M.A.; Mahdavinia, G.H.; Jafari, S.; Hazarkhani, H. Wet 2,4,6-trichloro-1,3,5-triazine (TCT) an efficient catalyst for synthesis of α,α′-bis(substituted-benzylidene) cycloalkanones under solvent-free conditions. Catal. Commun 2007, 8, 2229–2231. [Google Scholar]
- Bhagat, S.; Sharma, R.; Chakraborti, A.K. Dual-activation protocol for tandem cross-aldol condensation: An easy and highly efficient synthesis of α,α′-bis(aryl/alkylmethylidene)ketones. J. Mol. Catal. A Chem 2006, 260, 235–240. [Google Scholar]
- Miura, K.; Nakagawa, T.; Hosomi, A. Lewis base-promoted aldol reaction of dimethylsilyl enolates in aqueous dimethylformamide: Use of calcium chloride as a Lewis base catalyst. J. Am. Chem. Soc 2002, 124, 536–537. [Google Scholar]
- Shrikhande, J.J.; Gawande, M.B.; Jayaram, R.V. Cross-aldol and Knoevenagel condensation reactions in aqueous micellar media. Catal. Commun 2008, 9, 1010–1016. [Google Scholar]
- Fischer, A.; Makowski, P.; Mueller, J.O.; Antonietti, M.; Thomas, A.; Goettmann, F. High-surface-area TiO2 and TiN as catalysts for the C–C coupling of alcohols and ketones. ChemSusChem 2008, 1, 444–449. [Google Scholar]
- Mukaiyam, T.; Narasaka, K.; Hokonok, H. Reactions of bivalent sulfur compounds-copper(II) complexes. J. Am. Chem. Soc 1969, 91, 4315–4317. [Google Scholar]
- Nakano, T.; Irifune, S.; Umano, S.; Inada, A.; Ishii, Y.; Ogawa, M. Cross-condensation reactions of cycloalkanones with aldehydes and primary alcohols under the influence of zirconocene complexes. J. Org. Chem 1987, 52, 2239–2244. [Google Scholar]
- Iranpoor, N.; Kazemi, F. RuCl3 catalyses aldol condensations of aldehydes and ketones. Tetrahedron 1998, 54, 9475–9480. [Google Scholar]
- Zhang, L.J.; Wang, S.W.; Sheng, E.H.; Zhou, S.L. A solvent-free synthesis of α,α′-bis(substituted benzylidene) cycloalkanones catalyzed by lanthanide amides [(Me3Si)2N]3Ln(μ-Cl)Li(THF)3 under microwave irradiation. Green Chem 2005, 7, 683–686. [Google Scholar]
- Mestres, R. A green look at the aldol reaction. Green Chem 2004, 6, 583–603. [Google Scholar]
- Lee, S.G. Functionalized imidazolium salts for task-specific ionic liquids and their applications. Chem. Commun 2006, 1049–1063. [Google Scholar]
- Mehnert, C.P.; Dispenziere, N.C.; Cook, R.A. Preparation of C9-aldehyde via aldol condensation reactions in ionic liquid media. Chem. Commun 2002, 1610–1611. [Google Scholar]
- Rosamilia, A.E.; Giarrusso, M.A.; Scott, J.L.; Strauss, C.R. A direct, efficient synthesis of unsymmetrically substituted bis(arylidene)alkanones. Green Chem 2006, 8, 1042–1050. [Google Scholar]
- Kreher, U.P.; Rosamilia, A.E.; Raston, C.L.; Scott, J.L.; Strauss, C.R. Direct preparation of monoarylidene derivatives of aldehydes and enolizable ketones with DIMCARB. Org. Lett 2003, 5, 3107–3110. [Google Scholar]
- Zhu, A.L.; Jiang, T.; Wang, D.; Han, B.X.; Liu, L.; Huang, J.; Zhang, J.C.; Sun, D.H. Direct aldol reactions catalyzed by 1,1,3,3-tetramethylguanidine lactate without solvent. Green Chem 2005, 7, 514–517. [Google Scholar]
- Zhu, A.L.; Jiang, T.; Han, B.X.; Huang, J.; Zhang, J.C.; Ma, X.M. Study on guanidine-based task-specific ionic liquids as catalysts for direct aldol reactions without solvent. New J. Chem 2006, 30, 736–740. [Google Scholar]
- Hu, S.; Jiang, T.; Zhang, Z.; Zhu, A.; Han, B.; Song, J.; Xie, Y.; Li, W. Functional ionic liquid from biorenewable materials: Synthesis and application as a catalyst in direct aldol reactions. Tetrahedron Lett 2007, 48, 5613–5617. [Google Scholar]
- Yang, S.D.; Wu, L.Y.; Yan, Z.Y.; Pan, Z.L.; Liang, Y.M. A novel ionic liquid supported organocatalyst of pyrrolidine amide: Synthesis and catalyzed Claisen-Schmidt reaction. J. Mol. Catal. A Chem 2007, 268, 107–111. [Google Scholar]
- Formentin, P.; Garcia, H.; Leyva, A. Assessment of the suitability of imidazolium ionic liquids as reaction medium for base-catalysed reactions: Case of Knoevenagel and Claisen-Schmidt reactions. J. Mol. Catal. A Chem 2004, 214, 137–142. [Google Scholar]
- Guo, H.M.; Cun, L.F.; Gong, L.Z.; Mi, A.Q.; Jiang, Y.Z. Asymmetric direct aldol reaction catalyzed by an l-prolinamide derivative: Considerable improvement of the catalytic efficiency in the ionic liquid. Chem. Commun 2005, 1450–1452. [Google Scholar]
- Cordova, A. Direct catalytic asymmetric cross-aldol reactions in ionic liquid media. Tetrahedron Lett 2004, 45, 3949–3952. [Google Scholar]
- Kotrusz, P.; Alemayehu, S.; Toma, T.; Schmalz, H.G.; Adler, A. Enantioselective organocatalysis in ionic liquids: Addition of aliphatic aldehydes and ketones to diethyl azodicarboxylate. Eur. J. Org. Chem 2005, 2005, 4904–4911. [Google Scholar]
- Lombardo, M.; Easwar, S.; Pasi, F.; Trombini, C.; Dhavale, D.D. Protonated arginine and lysine as catalysts for the direct asymmetric aldol reaction in ionic liquids. Tetrahedron 2008, 64, 9203–9207. [Google Scholar]
- Davey, P.N.; Forsyth, S.A.; Gunaratne, H.Q.N.; Hardacre, C.; McKeown, A.; McMath, S.E.J.; Rooney, D.W.; Seddon, K.R. Synthesis of 3-(4-tert-butylphenyl)-2-propen-1-one, a precursor to Lilial®, via an aldol condensation in an ionic liquid. Green Chem 2005, 7, 224–229. [Google Scholar]
- Gruttadauria, M.; Riela, S.; Lo Meo, P.; D’Anna, F.; Noto, R. Supported ionic liquid asymmetric catalysis. A new method for chiral catalysts recycling. The case of proline-catalyzed aldol reaction. Tetrahedron Lett 2004, 45, 6113–6116. [Google Scholar]
- Zhang, X.Y.; Fan, X.S.; Niu, H.Y.; Wang, J.J. An ionic liquid as a recyclable medium for the green preparation of α,α′-bis(substituted benzylidene)cycloalkanones catalyzed by FeCl3·6H2O. Green Chem 2003, 5, 267–269. [Google Scholar]
- Curnow, O.J.; Holmes, M.T.; Ratten, L.C.; Walst, K.J.; Yunis, R. A facile route to functionalised, protic and chiral ionic liquids based on the triaminocyclopropenium cation. RSC Adv 2012, 2, 10794–10797. [Google Scholar]
- Kong, Y.; Tan, R.; Zhao, L.; Yin, D. l-Proline supported on ionic liquid-modified magnetic nanoparticles as a highly efficient and reusable organocatalyst for direct asymmetric aldol reaction in water. Green Chem 2013, 15, 2422–2433. [Google Scholar]
- Aprile, C.; Giacalone, F.; Gruttadauria, M.; Marculescu, A.M.; Noto, R.; Revell, J.D.; Wennemers, H. New ionic liquid-modified silica gels as recyclable materials for l-prolineor H-Pro-Pro-Asp-NH2-catalyzed aldol reaction. Green Chem 2007, 9, 1328–1334. [Google Scholar]
- Cui, X.; Zhang, S.; Shi, F.; Zhang, Q.; Ma, X.; Lu, L.; Deng, Y. The influence of the acidity of ionic liquids on catalysis. ChemSusChem 2010, 3, 1043–1047. [Google Scholar]
- Karmee, S.K.; Hanefeld, U. Ionic liquid catalysed synthesis of β-hydroxy ketones. ChemSusChem 2011, 4, 1118–1123. [Google Scholar]
- Luo, S.; Xu, H.; Li, J.; Zhang, L.; Mi, X.; Zheng, X.; Cheng, J.P. Facile evolution of asymmetric organocatalysts in water assisted by surfactant Brønsted acids. Tetrahedron 2007, 63, 11307–11314. [Google Scholar]
- Luo, S.; Mi, X.; Zhang, L.; Liu, S.; Xu, H.; Cheng, J.P. Functionalized ionic liquids catalyzed direct aldol reactions. Tetrahedron 2007, 63, 1923–1930. [Google Scholar]
- Wang, C.; Liu, X.; Yang, M.; Ma, H.; Yan, P.; Slattery, J.M.; Gao, Y.A. A green and efficient amine-functionalized ionic liquid/H2O catalytic system for the synthesis of α,α′-bis(substituted benzylidene)cyclopentanones. RSC Adv 2013, 3, 8796–8804. [Google Scholar]
- Kappe, C.O.; Stadler, A. Microwaves Theory. In Microwaves in Organic and Medicinal Chemistry; Raimund, M., Hugo, K., Gerd, F., Eds.; Wiely-VCH: Weinheim, Germany, 2005; Volume Chapter 2, pp. 9–28. [Google Scholar]
- Gedye, R.; Smith, F.; Westaway, K.; Ali, H.; Baldisera, L.; Laberge, L.; Rousell, J. The use of microwave-ovens for rapid organic-synthesis. Tetrahedron Lett 1986, 27, 279–282. [Google Scholar]
- Giguere, R.J.; Bray, T.L.; Duncan, S.M.; Majetich, G. Application of commercial microwave-ovens to organic synthesis. Tetrahedron Lett 1986, 27, 4945–4948. [Google Scholar]
- Loupy, A.; Perreux, L.; Liagre, M.; Burle, K.; Moneuse, M. Reactivity and selectivity under microwaves in organic chemistry. Relation with medium effects and reaction mechanisms. Pure Appl. Chem 2001, 73, 161–166. [Google Scholar]
- Chen, Y.; Markina, N.A.; Larock, R.C. An efficient, microwave-assisted, one-pot synthesis of indoles under Sonogashira conditions. Tetrahedron 2009, 65, 8908–8915. [Google Scholar]
- Sajith, A.M.; Muralidharan, A. Exploration of copper and amine-free Sonogashira cross coupling reactions of 2-halo-3-alkyl imidazo [4,5-b]pyridines using tetrabutyl ammonium acetate as an activator under microwave enhanced conditions. Tetrahedron Lett 2012, 53, 5206–5210. [Google Scholar]
- Huang, H.; Liu, H.; Jiang, H.; Chen, K. Rapid and efficient Pd-catalyzed sonogashira coupling of aryl chlorides. J. Org. Chem 2008, 73, 6037–6040. [Google Scholar]
- Limnios, D.; Kokotos, C.G. Microwave-assisted organocatalytic cross-aldol condensation of aldehydes. RSC Adv 2013, 3, 4496–4499. [Google Scholar]
- Hu, H.Y.; Yang, H.; Huang, P.; Cui, D.X.; Peng, Y.Q.; Zhang, J.C.; Lu, F.Y.; Lian, J.; Shi, D.L. Unique role of ionic liquid in microwave-assisted synthesis of monodisperse magnetite nanoparticles. Chem. Commun 2010, 46, 3866–3868. [Google Scholar]
- Leadbeater, N.E.; Torenius, H.M. A study of the ionic liquid mediated microwave heating of organic solvents. J. Org. Chem 2002, 67, 3145–3148. [Google Scholar]
- Kagimoto, J.; Fukumoto, K.; Ohno, H. Effect of tetrabutylphosphonium cation on the physico-chemical properties of amino-acid ionic liquids. Chem. Commun 2006, 2254–2256. [Google Scholar]
- Soutullo, M.D.; Odom, C.I.; Wicker, B.F.; Henderson, C.N.; Stenson, A.C.; Davis, J.H., Jr. Reversible CO2 capture by unexpected plastic-, resin-, and gel-like ionic soft materials discovered during the combi-click generation of a TSIL library. Chem. Mater 2007, 19, 3581–3583. [Google Scholar]
- Wang, C.; Luo, X.; Luo, H.; Jiang, D.E.; Li, H.; Dai, S. Tuning the basicity of ionic liquids for equimolar CO2 capture. Angew. Chem. Int. Ed 2011, 50, 4918–4922. [Google Scholar]
- Lindenschmidt, A.; Will, D.W.; Jaehne, G.; Wollmann, T.A.; Frick, W.; Junker, B.; Rigal, D.; Billen, G.; Jendralla, H. Method for producing 1,4-diphenyl azetidinone derivatives. WO Patent 2005113496, 1 Dec 2005. [Google Scholar]
- Zhang, Y.; Wang, M.G.; Liang, J.; Shang, Z.C. An environment-friendly and efficient method for aldol condensation catalyzed by l-lysine in pure water. Lett. Org. Chem 2010, 7, 27–31. [Google Scholar]
- Fernandez-Lopez, R.; Kofoed, J.; Machuqueiro, M.; Darbre, T. A selective direct aldol reaction in aqueous media catalyzed by zinc-proline. Eur. J. Org. Chem 2005, 2005, 5268–5276. [Google Scholar]
- Wang, B.; Liu, X.W.; Liu, L.Y.; Chang, W.X.; Li, J. Highly efficient direct asymmetric aldol reactions catalyzed by a prolinethioamide derivative in aqueous media. Eur. J. Org. Chem 2010, 2010, 5951–5954. [Google Scholar]
- Yan, S.; Gao, Y.; Xing, R.; Shen, Y.; Liu, Y.; Wu, P.; Wu, H. An efficient synthesis of (E)-nitroalkenes catalyzed by recoverable diamino-functionalized mesostructured polymers. Tetrahedron 2008, 64, 6294–6299. [Google Scholar]
- Allini, R.; Noe, M.; Perosa, A.; Selva, M. Selective nitroaldol condensations over heterogeneous catalysts in the presence of supercritical carbon dioxide. J. Org. Chem 2008, 73, 8520–8528. [Google Scholar]
- Bulbule, V.J.; Deshpande, V.H.; Velu, S.; Sudalai, A.; Sivasankar, S.; Sathe, V.T. Heterogeneous Henry reaction of aldehydes: Diastereoselective synthesis of nitroalcohol derivatives over Mg–Al hydrotalcites. Tetrahedron 1999, 55, 9325–9332. [Google Scholar]


| Solvent | Conversion (%) | Yield (%) |
|---|---|---|
| None | 28 | 13 |
| Ethanol | 5 | 44 |
| Acetone | 74 | 46 |
| Water | >99 | 98 |
| Entry | IL | Conversion (%) | Yield (%) |
|---|---|---|---|
| 1 | [N2222][EtNHC3SO3] | >99 | 98 |
| 2 | [N2222][n-BuNHC3SO3] | 95 | 85 |
| 3 | [N4444][MeNHC3SO3] | >99 | 98 |
| 4 | [N1123][EtNHC3SO3] | 90 | 80 |
| 5 | [N1123][n-OctNHC3SO3] | 68 | 64 |
| 6 | [P4444][Im] | 96 | 88 |
| 7 | [P4444][Arg] | 97 | 87 |
| Entry | R | Product | Conversion (%) | Yield (%) |
|---|---|---|---|---|
| 1 | H | 3a | >99 | 98 |
| 2 | 4-OMe | 3b | 88 | 87 |
| 3 | 4-Me | 3c | 92 | 89 |
| 4 | 2-Cl | 3d | >99 | 96 |
| 5 | 3-Br | 3e | >99 | 91 |
| 6 | 4-NO2 | 3f | <5 | 0 |
| 7 | 4-NO2a | 3f | 98 | 8 |
| Entry | R | Product | Conversion (%) | Yield (%) |
|---|---|---|---|---|
| 1 | H | 6a | >99 | 98 |
| 2 | 4-OMe | 6b | >99 | 87 |
| 3 | 4-Me | 6c | >99 | 89 |
| 4 | 2-Cl | 6d | >99 | 96 |
| 5 | 4-NO2 | 6e | <5 | 0 |
| 6 | 4-NO2a | 6e | 88 | 12 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, C.; Liu, J.; Leng, W.; Gao, Y. Rapid and Efficient Functionalized Ionic Liquid-Catalyzed Aldol Condensation Reactions Associated with Microwave Irradiation. Int. J. Mol. Sci. 2014, 15, 1284-1299. https://doi.org/10.3390/ijms15011284
Wang C, Liu J, Leng W, Gao Y. Rapid and Efficient Functionalized Ionic Liquid-Catalyzed Aldol Condensation Reactions Associated with Microwave Irradiation. International Journal of Molecular Sciences. 2014; 15(1):1284-1299. https://doi.org/10.3390/ijms15011284
Chicago/Turabian StyleWang, Chang, Jing Liu, Wenguang Leng, and Yanan Gao. 2014. "Rapid and Efficient Functionalized Ionic Liquid-Catalyzed Aldol Condensation Reactions Associated with Microwave Irradiation" International Journal of Molecular Sciences 15, no. 1: 1284-1299. https://doi.org/10.3390/ijms15011284
APA StyleWang, C., Liu, J., Leng, W., & Gao, Y. (2014). Rapid and Efficient Functionalized Ionic Liquid-Catalyzed Aldol Condensation Reactions Associated with Microwave Irradiation. International Journal of Molecular Sciences, 15(1), 1284-1299. https://doi.org/10.3390/ijms15011284





