Anti-Inflammatory Activity of N-(3-Florophenyl)ethylcaffeamide in Mice
Abstract
:1. Introduction
2. Results
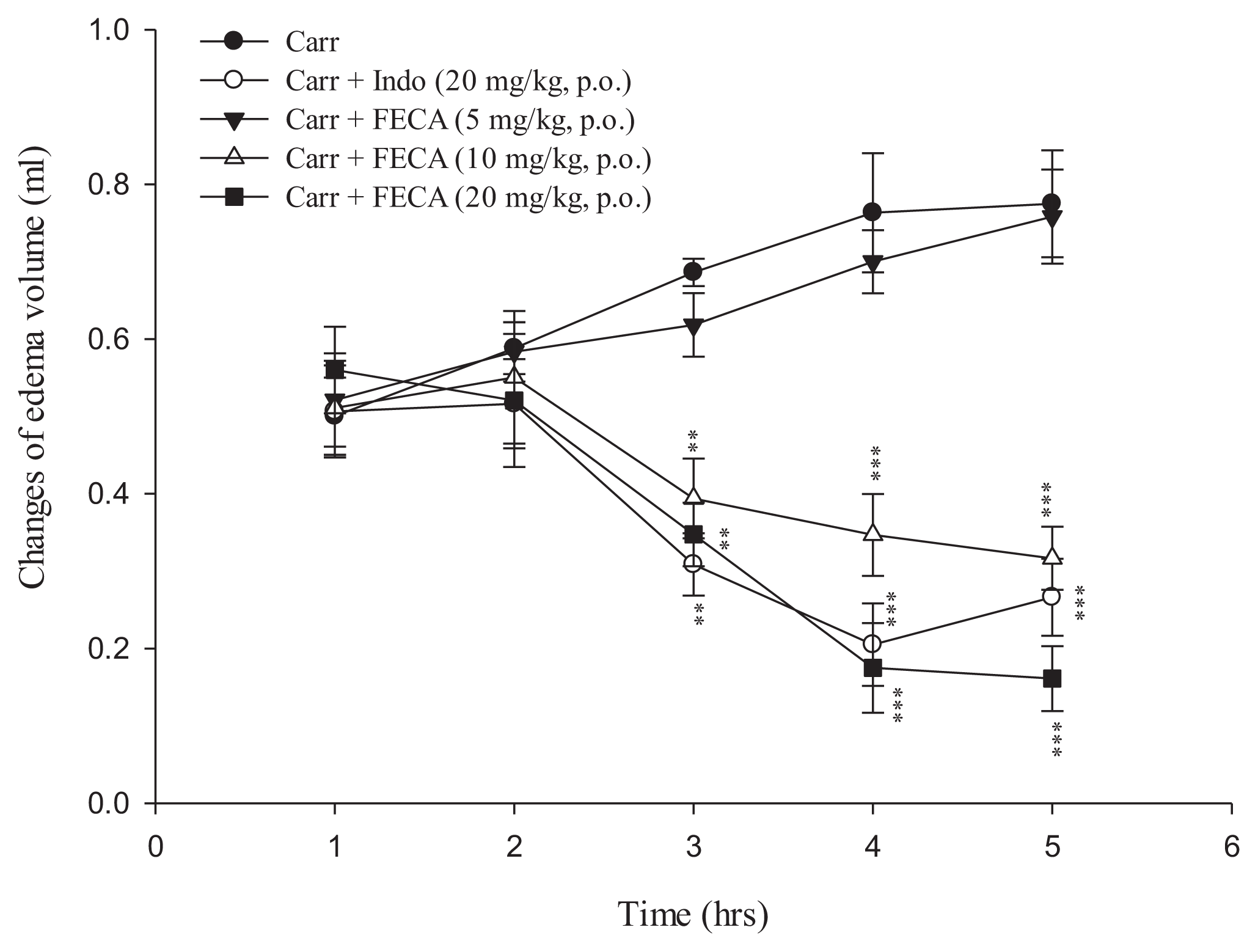
2.1. Effects of FECA on λ-Carrageenan-Induced Mice Paw Edema
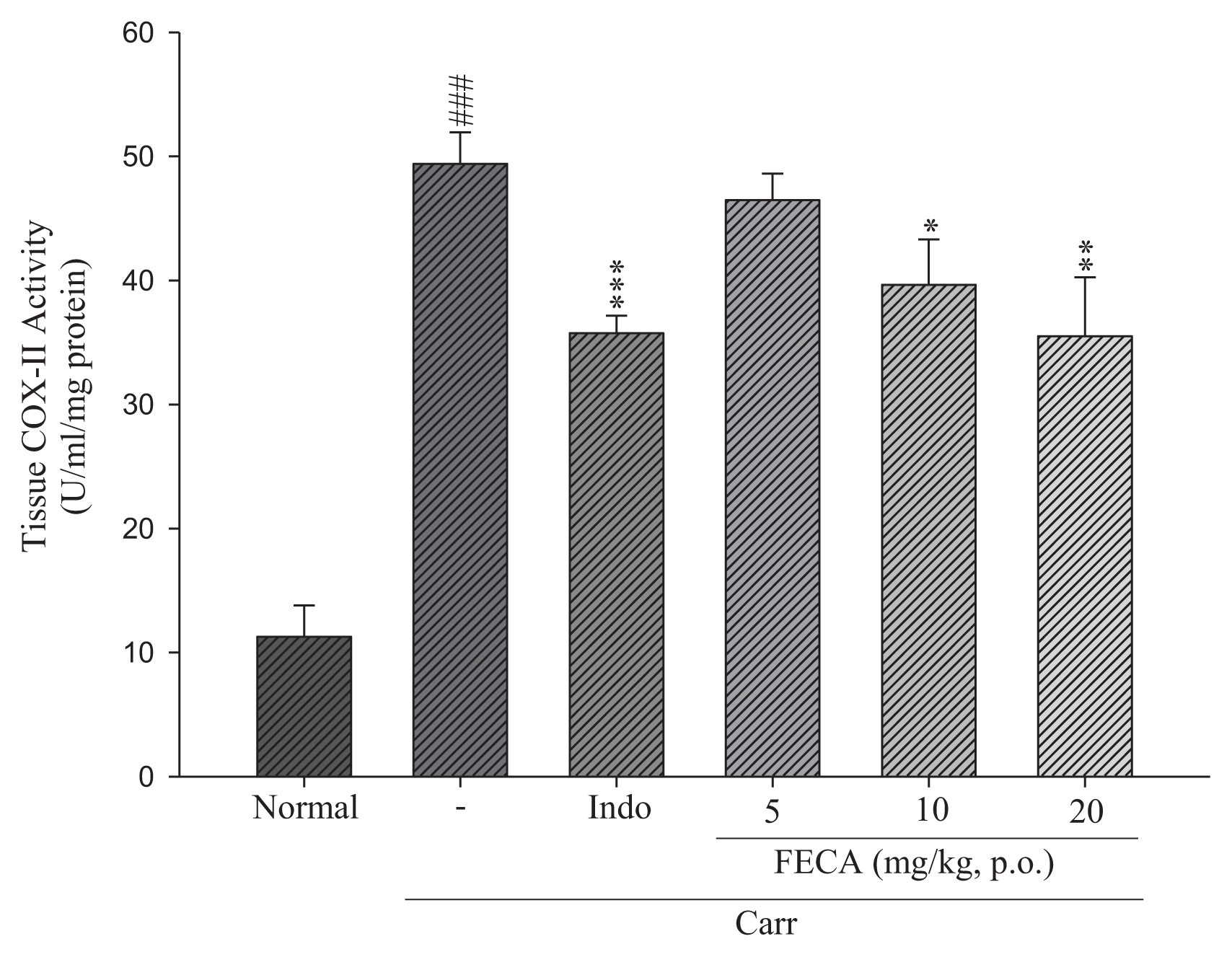
2.2. Effects of FECA on COX-2 Level
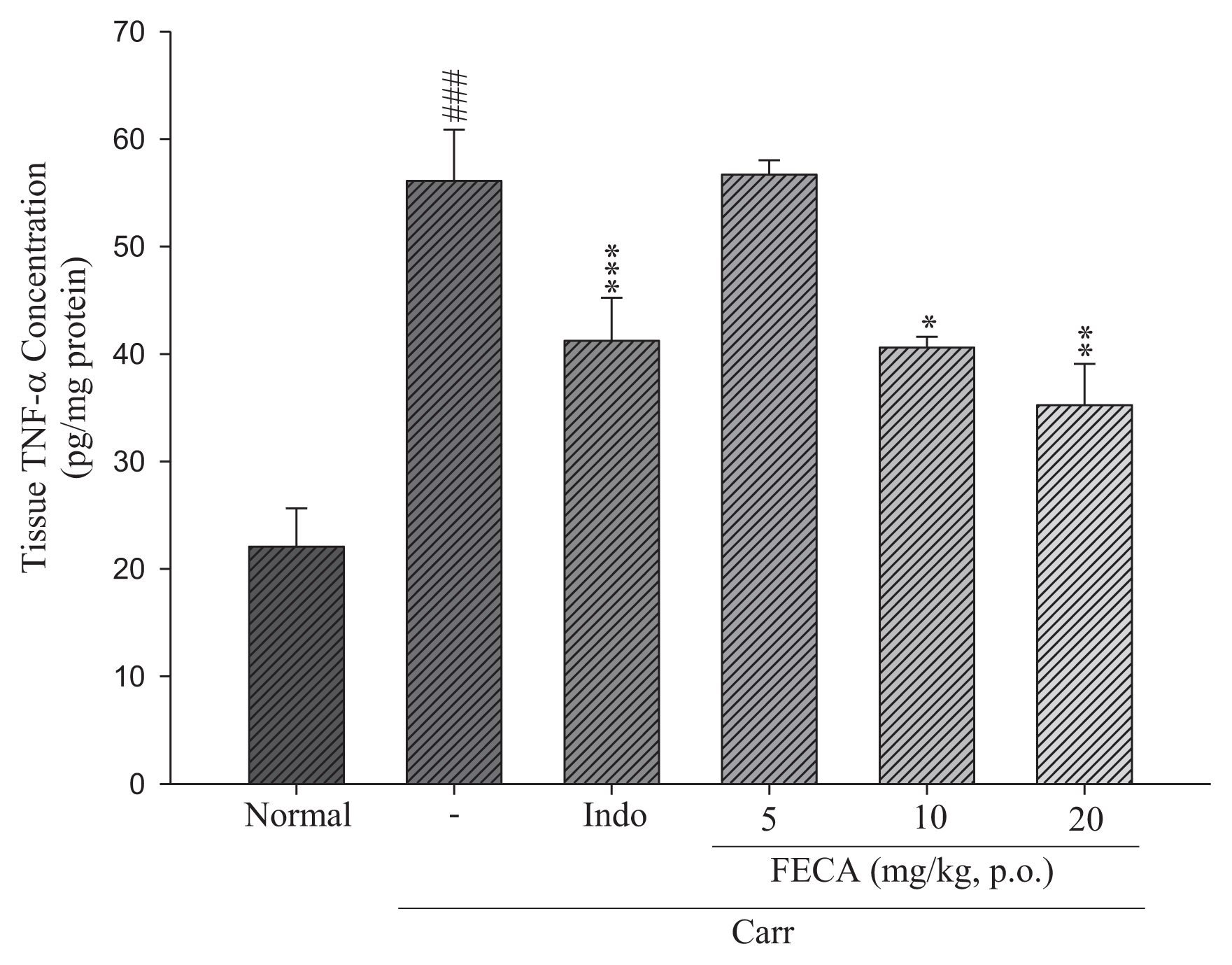
2.3. Effects of FECA on TNF-α and IL-1β Levels
2.4. Effects of FECA on NO Level
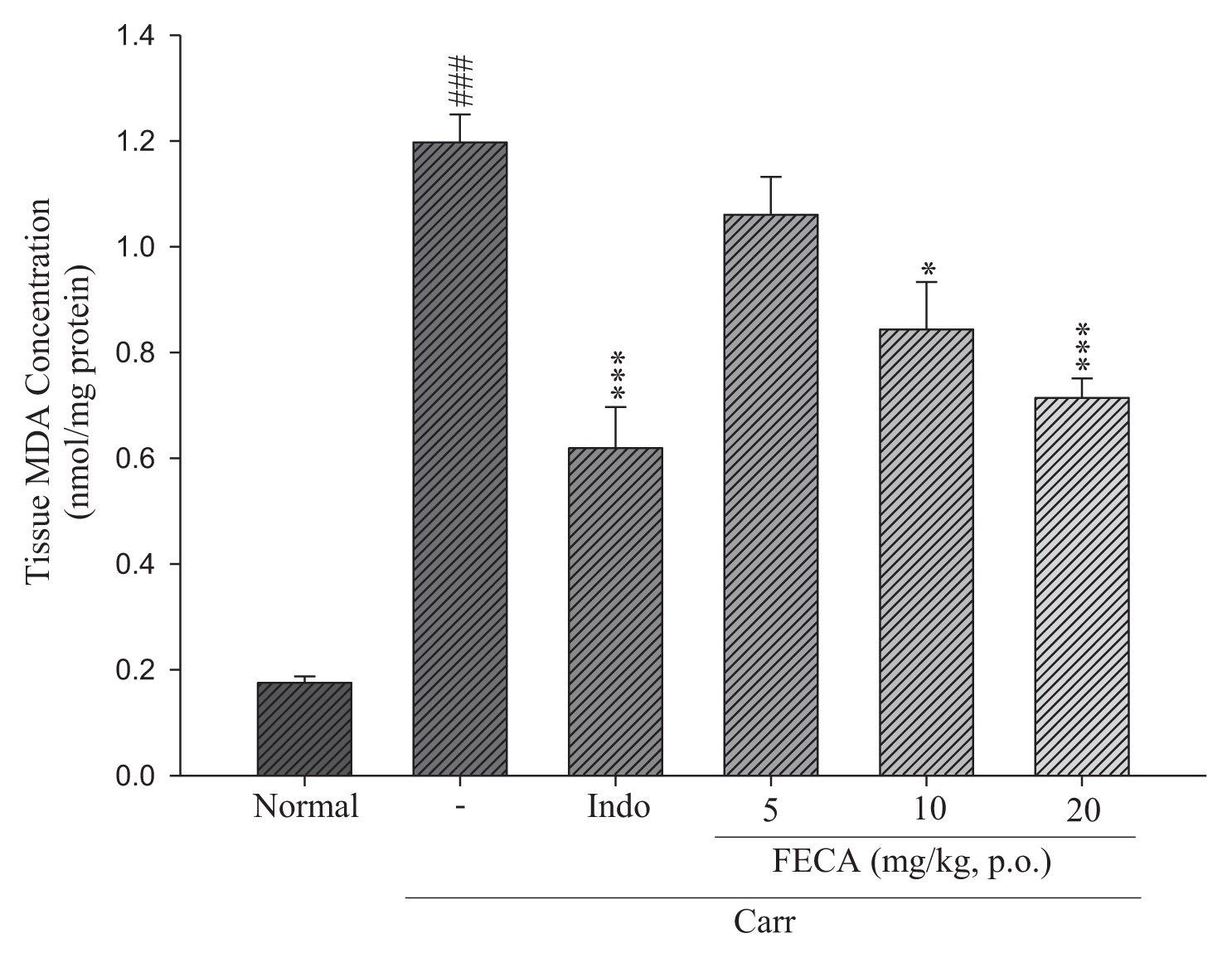
2.5. Effects of FECA on MDA Level Measurements
2.6. Measurements of Antioxidant Enzymatic Activities
3. Discussion
4. Materials and Methods
4.1. Chemicals
FECA
4.2. Experimental Animals
4.3. λ-Carrageenan-Induced Mice Paw Edema
4.4. COX-2 Assay
4.5. NO Assay
4.6. TNF-α and IL-1β Assays
4.7. MDA Assay
4.8. Antioxidant Enzymatic Activity Measurements
4.9. Statistical Analysis
5. Conclusions
Acknowledgements
Conflict of Interest
References
- Janero, D.R. Malondialdehyde and thiobarbituric acid reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic. Biol. Med 1990, 9, 515–540. [Google Scholar]
- Hwang, J.T.; Kwon, D.Y.; Yoon, S.H. AMP-activated protein kinase: A potential target for the diseases prevention by natural occurring polyphenols. N. Biotechnol 2009, 26, 17–22. [Google Scholar]
- Collins, Q.F.; Liu, H.Y.; Pi, J.; Liu, Z.; Quon, M.J.; Cao, W. Epigallocatechin-3-gallate (EGCG), a green tea polyphenol, suppresses hepatic gluconeogenesis through 50-AMP-activated protein kinase. J. Biol. Chem 2007, 282, 30143–30149. [Google Scholar]
- Breen, D.M.; Sanli, T.; Giacca, A.; Tsiani, E. Stimulation of muscle cell glucose uptake by resveratrol through sirtuins and AMPK. Biochem. Biophys. Res. Commun 2008, 374, 117–122. [Google Scholar]
- Fang, X.K.; Gao, J.; Zhu, D.N. Kaempferol and quercetin isolated from Euonymus alatus improve glucose uptake of 3T3-L1 cells without adipogenesis activity. Life Sci 2008, 82, 615–622. [Google Scholar]
- Hwang, J.T.; Kim, S.H.; Lee, M.S.; Kim, S.H.; Yang, H.J.; Kim, M.J.; Kim, H.S.; Ha, J.; Kim, M.S.; Kwon, D.Y. Anti-obesity effects of ginsenoside Rh2 are associated with the activation of AMPK signaling pathway in 3T3-L1 adipocyte. Biochem. Biophys. Res. Commun 2007, 364, 1002–1008. [Google Scholar]
- Lee, E.S.; Uhm, K.O.; Lee, Y.M.; Han, M.; Lee, M.; Park, J.M.; Suh, P.G.; Park, S.H.; Kim, H.S. CAPE (caffeic acid phenethyl ester) stimulates glucose uptake through AMPK (AMP-activated protein kinase) activation in skeletal muscle cells. Biochem. Biophys. Res. Commun 2007, 361, 854–858. [Google Scholar]
- Cheng, Z.; Pang, T.; Gu, M.; Gao, A.H.; Xie, C.M.; Li, J.Y.; Nan, F.J.; Li, J. Berberine-stimulated glucose uptake in L6 myotubes involves both AMPK and p38 MAPK. Biochem. Biophys. Res. Commun 2006, 1760, 1682–1689. [Google Scholar]
- Lee, Y.S.; Kim, W.S.; Kim, K.H.; Yoon, M.J.; Cho, H.J.; Shen, Y.; Ye, J.M.; Lee, C.H.; Oh, W.K.; Kim, C.T.; et al. Berberine, a natural plant product, activates AMP-activated protein kinase with beneficial metabolic effects in diabetic and insulin-resistant states. Diabetes 2006, 55, 2256–2264. [Google Scholar]
- Lin, C.L.; Huang, H.C.; Lin, J.K. Theaflavins attenuate hepatic lipid accumulation through activating AMPK in human HepG2 cells. J. Lipid Res 2007, 48, 2334–2243. [Google Scholar]
- Sud’ina, G.F.; Mirzoeva, O.K.; Pushkareva, M.A.; Korshunova, G.A.; Sumbatyan, N.V.; Varfolomeev, S.D. Caffeic acid phenethyl ester as a lipoxygenase inhibitor with antioxidant properties. FEBS Lett 1993, 329, 21–24. [Google Scholar]
- Michaluart, P.; Masferrer, J.L.; Carothers, A.M.; Subbaramaiah, K.; Zweifel, B.S.; Koboldt, C.; Mestre, J.R.; Grunberger, D.; Sacks, P.G.; Tanabe, T.; et al. Inhibitory effects of caffeic acid phenethyl ester on the activity and expression of cyclooxygenase-2 in human oral epithelial cells and in a rat model of inflammation. Cancer Res 1999, 59, 2347–2352. [Google Scholar]
- Hung, M.W.; Shiao, M.S.; Tsai, L.C.; Chang, G.G.; Chang, T.C. Apoptotic effect of caffeic acid phenethyl ester and its ester and amide analogues in human cervical cancer ME180 cells. Anticancer Res 2003, 23, 4773–4780. [Google Scholar]
- Fesen, M.R.; Pommier, Y.; Leteurtre, F.; Hiroguchi, S.; Yung, J.; Kohn, K.W. Inhibition of HIV-1 integrase by flavones, caffeic acid phenethyl ester (CAPE) and related compounds. Biochem. Pharmacol 1994, 48, 595–608. [Google Scholar]
- Park, E.H.; Kahng, J.H. Suppressive effects of propolis in rat adjuvant arthritis. Arch. Pharm. Res 1999, 22, 554–558. [Google Scholar]
- Di Rosa, M. Biological properties of carrageenan. J. Pharm. Pharmacol 1972, 24, 89–102. [Google Scholar]
- Seibert, K.; Zhang, Y.; Leahy, K.; Hauser, S.; Masferrer, J.; Perkins, W.; Lee, L.; Isakson, P. Pharmacological and biochemical demonstration of the role of cyclooxygenase 2 in inflammation and pain. Proc. Natl. Acad. Sci. USA 1994, 91, 12013–12017. [Google Scholar]
- Spector, W.G.; Willoughby, D.A. The inflammatory response. Bacteriol. Rev 1963, 27, 117–154. [Google Scholar]
- Sawatzky, D.A.; Megson, I.L.; Rossi, A.G. Sildenafil offers protection against NSAID-induced gastric injury. Br. J. Pharmacol 2005, 146, 477–478. [Google Scholar]
- Dinarello, C.A. Proinflammatory and anti-inflammatory cytokines as mediators in the pathogenesis of septic shock. Chest 1997, 112, 321–329. [Google Scholar]
- Beutler, B.; Cerami, A. The biology of cachectin/TNF—A primary mediator of the host response. Annu. Rev. Immunol 1989, 7, 625–655. [Google Scholar]
- Park, W.H.; Park, S.Y.; Kim, H.M.; Kim, C.H. Effect of a Korean traditional formulation, Hwaotang, on superoxide generation in human neutrophils, platelet aggregation in human blood, and nitric oxide, prostaglandin E2 production and paw oedema induced by carrageenan in mice. Immunopharmacol. Immunotoxicol 2004, 26, 53–73. [Google Scholar]
- Strieter, R.M.; Belperio, J.A.; Keane, M.P. Cytokines in innate host defense in the lung. Clin. Invest 2002, 109, 699–705. [Google Scholar]
- Salvemini, D.Z.; Wang, D.M.; Bourdon, M.K.; Stern, M.K.; Curne, M.G.; Manning, P.T. Evidence of peroxynitrite involvement in the carrageenan induced rat paw edema. Eur. J. Pharmacol 1996, 303, 217–220. [Google Scholar]
- Daniela, S.; Wang, Z.Q.; Wyatt, P.S.; Bourdon, D.M.; Marino, M.H.; Manning, P.T.; Currie, M.G. Nitric oxide: A key mediator in the early and late phase of carrageenan-induced rat paw inflammation. Br. J. Pharmacol 1996, 118, 829–838. [Google Scholar]
- Dawson, J.; Sedgwick, A.D.; Edwards, J.C.; Lees, P. A comparative study of the cellular, exudative and histological responses to carrageenan, dextran and zymosan in the mouse. Int. J. Tissue React 1991, 13, 171–185. [Google Scholar]
- Lu, T.C.; Ko, Y.Z.; Huang, H.W.; Hung, Y.C.; Lin, Y.C.; Peng, W.H. Analgesic and anti-inflamatory activities of aqueous extract from Glycine tomentella root in mice. J. Ethnopharmacol 2007, 113, 142–148. [Google Scholar]
- Flemming, N.; Mikkelsen, B.B.; Nielsen, J.B.; Andersen, H.R.; Grandjean, P. Plasma malondialdehyde as biomarker for oxidative stress: Reference interval and effects of life-style factors. Clin. Chem 1997, 43, 1209–1214. [Google Scholar]
- Boudreau, L.H.; Maillet, J.; LeBlanc, L.M.; Jean-François, J.; Touaibia, M.; Flamand, N.; Surette, M.E. Caffeic acid phenethyl ester and its amide analogue are potent inhibitors of leukotriene biosynthesis in human polymorphonuclear leukocytes. PLoS One 2012, 7, e31833. [Google Scholar]
- Zimmermann, M. Ethical guidelines for investigations of experimental pain in conscious animals. Pain 1983, 16, 109–110. [Google Scholar]
- Vinegar, R.; Schreiber, W.; Hugo, R. Biphasic development of carrageenan oedema in rats. J. Pharmacol. Exp. Ther 1969, 166, 96–103. [Google Scholar]
- Hung, S.H.; Shen, K.H.; Wu, C.H.; Liu, C.L.; Shih, Y.W. α-Mangostin suppresses PC-3 human prostate carcinoma cell metastasis by inhibiting matrix metalloproteinase-2/9 and urokinase-plasminogen expression through the JNK signaling pathway. J. Agric. Food Chem 2009, 57, 1291–1298. [Google Scholar]
- Moshage, H.; Kok, B.; Huizenga, J.R.; Jansen, P.L. Nitrite and nitrate determinations in plasma: A critical evaluation. Clin. Chem 1995, 41, 892–896. [Google Scholar]
- Nims, R.W.; Darbyshire, J.F.; Saavedra, J.E.; Christodoulou, D.; Hanbauer, I.; Cox, G.W.; Grisham, M.B.; Laval, F.; Cook, J.A.; Krishna, M.C.; et al. Colorimetric methods for the determination of nitric oxide concentration in neutral aqueous solutions. Methods 1995, 7, 48–54. [Google Scholar]
- Tatum, V.L.; Changchit, C.C.; Chow, K. Measurement of malondialdehyde by high performance liquid chromatography with fluorescence detection. Lipids 1990, 25, 226–229. [Google Scholar]
- Misra, H.P.; Fridovich, I. The univalent reduction of oxygen by reduced flavins and quinones. J. Biol. Chem 1972, 247, 188–192. [Google Scholar]
- Flohe, L.; Günzler, W.A. Assays of glutathione peroxidase. Methods Enzymol 1984, 105, 114–121. [Google Scholar]
- Carlberg, I.; Mannervik, B. Glutathione reductase. Methods Enzymol 1985, 113, 484–490. [Google Scholar]



| Groups | SOD (U/mg protein) | GPx (U/mg protein) | GRd (U/mg protein) |
|---|---|---|---|
| Normal | 61.70 ± 5.37 | 1.66 ± 0.12 | 0.114 ± 0.007 |
| Carr. | 44.15 ± 3.34 ## | 1.17 ± 0.13 # | 0.069 ± 0.004 ### |
| Carr. + Indo | 58.48 ± 4.61 ** | 1.52 ± 0.03 * | 0.099 ± 0.005 ** |
| Carr. + FECA 5 | 47.61 ± 2.70 | 1.22 ± 0.06 | 0.073 ± 0.002 |
| Carr. + FECA 10 | 54.46 ± 4.11 * | 1.36 ± 0.11 | 0.089 ± 0.006 * |
| Carr. + FECA 20 | 57.62 ± 2.50 ** | 1.51 ± 0.07 * | 0.097 ± 0.008 ** |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liao, J.-C.; Tsai, J.-C.; Peng, W.-H.; Chiu, Y.-J.; Sung, P.-J.; Tsuzoki, M.; Kuo, Y.-H. Anti-Inflammatory Activity of N-(3-Florophenyl)ethylcaffeamide in Mice. Int. J. Mol. Sci. 2013, 14, 15199-15211. https://doi.org/10.3390/ijms140815199
Liao J-C, Tsai J-C, Peng W-H, Chiu Y-J, Sung P-J, Tsuzoki M, Kuo Y-H. Anti-Inflammatory Activity of N-(3-Florophenyl)ethylcaffeamide in Mice. International Journal of Molecular Sciences. 2013; 14(8):15199-15211. https://doi.org/10.3390/ijms140815199
Chicago/Turabian StyleLiao, Jung-Chun, Jen-Chieh Tsai, Wen-Huang Peng, Yung-Jia Chiu, Ping-Jyun Sung, Minoru Tsuzoki, and Yueh-Hsiung Kuo. 2013. "Anti-Inflammatory Activity of N-(3-Florophenyl)ethylcaffeamide in Mice" International Journal of Molecular Sciences 14, no. 8: 15199-15211. https://doi.org/10.3390/ijms140815199






