Sequestration of AS-DACA into Acidic Compartments of the Membrane Trafficking System as a Mechanism of Drug Resistance in Rhabdomyosarcoma
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of Endocytic Trafficking Inhibitors on AS-DACA Sensitivity in RMS Cells
2.1.1. RMS Cell line Cytotoxic Response to AS-DACA
2.1.2. Inhibitor Effect on Cell Lines-Determination of Maximum Tolerated Dose (MTD)
2.1.3. Inhibitor Effect on AS-DACA Cytotoxicity
2.2. Effect of Endocytic Trafficking Inhibitors on AS-DACA Distribution in Vesicular Compartments in RMS Cells
2.2.1. Direct Fluorescence of AS-DACA in RMS Cell Lines
2.2.2. AS-DACA Intracellular Distribution with Chlorpromizine Co-Treatment in RMS Cell Lines
2.2.3. AS-DACA Intracellular Distribution with 3-Methyladenine Co-Treatment in RMS Cell Lines
2.3. Alterations to Endosomal Organelle Marker Proteins upon Treatment with AS-DACA or Endosomal Pathway Inhibitors
2.3.1. Effects of Endosomal Protein Expression in RMS Cells following AS-DACA and Chlorpromazine Treatment
2.3.2. Effects of Endosomal Protein Expression in RMS Cells Following AS-DACA and 3-Methyladenine Treatment
2.3.3. Expression of D52 in RMS Cell Lines
3. Experimental Section
3.1. Drugs
3.2. Cell Lines and Culture Conditions
3.3. Cytotoxicity Assay
3.4. Fluorescence Microscopy
3.4.1. AS-DACA Distribution
3.4.2. Acridine Orange
3.5. Western Blotting
3.5.1. Protein Extraction, SDS-PAGE and Transfer
3.5.2. Immunostaining
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Rodeberg, D.; Paidas, C. Childhood rhabdomyosarcoma. Semin. Pediatr. Surg 2006, 15, 57–62. [Google Scholar]
- Wexler, L.H.; Helman, L.J. Rhabdomyosarcoma and the Undifferentiated Sarcomas. In Principles and Practice of Pediatric Oncology; Pizzo, P., Poplack, D.G., Eds.; Lippincott-Raven Publishers: Philadelphia, PA, USA, 1997; pp. 799–829. [Google Scholar]
- Gurney, J.G.; Young, J.L.; Roffers, S.D.; Smith, M.A.; Bunin, G.R. Soft Tissue Sarcomas. In Cancer Incidence and Survival among Children and Adolescents: United States SEER Program 1975–1995; Ries, L.A.G., Smith, M.A., Gurney, J.G., Linet, M., Tamra, T., Young, J.L., Bunin, G.R., Eds.; National Cancer Institute, SEER Program: Bethesda, MD, USA, 1999; pp. 111–124. [Google Scholar]
- Qualman, S.J.; Coffin, C.M.; Newton, W.A.; Hojo, H.; Triche, T.J.; Parham, D.M.; Crist, W.M. Intergroup rhabdomyosarcoma study: Update for pathologists. Pediatr. Dev. Pathol 1998, 1, 550–561. [Google Scholar]
- Breitfeld, P.P.; Meyer, W.H. Rhabdomyosarcoma: New windows of opportunity. Oncologist 2005, 10, 518–527. [Google Scholar]
- Sandler, E.; Lyden, E.; Ruymann, F.; Maurer, H.; Wharam, M.; Parham, D.; Link, M.; Crist, W. Efficacy of ifosfamide and doxorubicin given as a phase II “window” in children with newly diagnosed metastatic rhabdomyosarcoma: A Report from the Intergroup Rhabdomyosarcoma Study Group. Med. Pediatr. Oncol 2001, 37, 442–448. [Google Scholar]
- Arndt, C.A.S.; Crist, W.M. Common musculoskeletal tumors of childhood and adolescence. N. Engl. J. Med 1999, 341, 42–52. [Google Scholar]
- Wolf, S.; Wakelin, L.P.G.; He, Z.; Stewart, B.W.; Catchpoole, D.R. In vitro assessment of novel transcription inhibitors and topoisomerase poisons in rhabdomyosarcoma cell lines. Cancer Chemother. Pharmacol 2009, 64, 1059–1069. [Google Scholar]
- Gottesman, M.M. Mechanisms of cancer drug resistance. Ann. Rev. Med 2002, 53, 615–627. [Google Scholar]
- Simon, S.; Rou, D.; Schindler, M. Intracellular pH and the control of multidrug resistance. Cell Biol 1994, 91, 1128–1132. [Google Scholar]
- Wolf, S.; Huyh, T.; Bryce, N.; Hambley, T.; Walkelin, L.; Stewart, B.; Catchpoole, D. Intracellular trafficking as a determinant of AS-DACA cytotoxixity in rhabdomyosarcoma cells. BMC Cell Biol 2011, 12, 36–47. [Google Scholar]
- Wang, E.; Lee, M.D.; Dunn, K.W. Lysosomal accumulation of drugs in drug-sensitive MES-SA but not multidrug-resistant MES-SA/Dx5 uterine sarcoma cells. J. Cell. Physiol 2000, 184, 263–274. [Google Scholar]
- Wang, L.; Rothberg, K.; Anderson, R. Mis-assembly of clathrin lattices on endosomes reveals a regulatory switch for coated pit formation. J. Cell Biol 1993, 123, 1107–1117. [Google Scholar]
- Drose, S.; Altendorf, K. Bafilomycins and concanamycins as inhibitors of V-ATPases and P-ATPases. J. Exp. Biol 1997, 3, 39–47. [Google Scholar]
- Ouar, Z.; Bens, M.; Vignes, C.; Paulais, M.; Pringel, C.; Fleury, J.; Cluzeaud, F.; Lacave, R.; Vandewalle, A. Inhibitors of vacuolar H+-ATPase impair the preferential accumulation of daunomycin in lysosomes and reverse the resistance to anthracyclines in drug-resistant renal epithelial cells. Biochem. J. 2003, 370, 185–193. [Google Scholar]
- Yoshimori, T.; Yamamoto, A.; Moriyama, Y.; Futa, M.; Tashiro, Y. Bafilomycin A1, a specific inhibitor of vacuolar-type H+-ATPase, inhibits acidification and protein degradation in lysosomes of cultured cells. J. Biol. Chem 1991, 266, 17707–17712. [Google Scholar]
- Yumoto, R.; Nishikawa, H.; Okamoto, M.; Katayama, H.; Nagai, J.; Takano, M. Clathrin-mediated endocytosis of FITC-albumin in alveolar type II epithelial cell line RLE-6TN. Am. J. Physiol. Lung Physiol 2006, 290, L946–L955. [Google Scholar]
- Punnonen, E.L.; Marjomäki, V.S.; Reunanen, H. 3-Methyladenine inhibits transport from late endosomes to lysosomes in cultured rat and mouse fibroblasts. Eur. J. Cell Biol 1994, 65, 14–25. [Google Scholar]
- Seglen, P.O.; Gordon, P.B. 3-Methyladenine: Specific inhibitor of autophagic/lysosomal protein degradation in isolated rat hepatocytes. Proc. Natl. Acad. Sci. USA 1982, 79, 1889–1892. [Google Scholar]
- Wu, Y.T.; Tan, H.L.; Shui, G.; Bauvy, C.; Huang, Q.; Wenk, M.R.; Ong, C.N.; Codogno, P.; Shen, H.M. Dual role of 3-Methyladenine in modulation of autophagy via different temporal patterns of inhibition on class I and III phosphoinositide 3-kinase. J. Biol. Chem 2010, 285, 10850–10861. [Google Scholar]
- Vercauteren, D.; Vandenbroucke, R.E.; Jones, A.T.; Rejman, J.; Demeester, J.; de Smedt, S.C.; Sanders, N.N.; Braeckmans, K. The use of inhibitors to study endocytic pathways of gene carriers: Optimization and pitfalls. Mol. Ther 2010, 18, 561–569. [Google Scholar]
- Weisz, O.A. Organelle acidification and disease. Traffic 2003, 4, 57–64. [Google Scholar]
- Belhoussine, R.; Morjani, H.; Sharonov, S.; Ploton, D.; Manfait, M. Characterisation of intracellular pH gradients in human multidrug-resistant tumour cells by means of scanning microspectrofluorometry and dual-emission-ratio probes. Int. J. Cancer 1999, 81, 81–89. [Google Scholar]
- Hurwitz, S.J.; Terasim, M.; Mizunuma, M.; Slapak, C.K. Vesicular anthracycline accumulation in doxorubcin-selected U-937 cells: Participation of lysosomes. Blood 1997, 89, 345–3754. [Google Scholar]
- Lee, C.M.; Tannock, I.F. Inhibition of endosomal sequestration of basic anticancer drugs: Influence on cytoxicity and tissue penetration. Br. J. Cancer 2006, 94, 863–869. [Google Scholar]
- Mahoney, B.P.; Raghunand, N.; Baggett, B.; Gillies, R.J. Tumour acidity, ion trapping and chemotherapeutics I. Acid pH affects the distribution of chemotherapeutic agents in vitro. Biochem. Pharmacol 2003, 66, 1207–1218. [Google Scholar]
- Schindler, M.; Grabski, S.; Hoff, E.; Simon, S.M. Defective pH regulation of acidic compartments in human breast cancer cells (MCF-7) is normalized in adriamycin-resistant cells. Biochemistry 1996, 35, 2811–2817. [Google Scholar]
- Larsen, A.K.; Escargueil, A.E.; Skladanowski, A. Resistance mechanisms associated with altered intracellular distribution of anticancer agents. Pharmacol. Ther 2000, 85, 271–229. [Google Scholar]
- Raghunand, N.; Martinez-Zaguilan, R.; Wright, S.H.; Gillies, R. pH and drug resistance. II. Turnover of acidic vesicles and resistance to weakly basic and chemotherapeutic drugs. Biochem. Pharmacol 1999, 57, 1047–1058. [Google Scholar]
- Raghunand, N.; Gillies, R.J. pH and drug resistance in tumours. Drug Resist. Updates 2000, 3, 39–47. [Google Scholar]
- Kang, H.C.; Samsonova, O.; Bae, Y.H. Trafficking microenvironmental pHs of polycationic gene vectors in drug-sensitive and multidrug-resistant MCF7 breast cancer cells. Biomaterials 2010, 31, 3071–3078. [Google Scholar]
- Altan, N.; Chen, Y.; Schindler, M.; Simon, S.M. Defective acidification in human breast tumor cells and implications for chemotherapy. J. Exp. Med 1998, 187, 1589–1598. [Google Scholar]
- Chapuy, B.; Koch, R.; Radunski, U.; Corsham, S.; Cheong, N.; Inagaki, N.; Ban, N.; Wenzel, D.; Reinhardt, D.; Zapf, A.; et al. Intracellular ABC transporter A3 confers multidrug resistance in leukemia cells by lysosomal drug sequestration. Leukemia 2008, 22, 1576–1586. [Google Scholar]
- Kones, A.T.; Clague, M.C. Phosphatidylinositol 3-kinase activity is required for early endosomal fusion. Res. Commun. Biochem. J 1995, 311, 31–34. [Google Scholar]
- Safaei, R.; Katano, K.; Larson, B.J.; Samimi, G.; Holzer, A.K.; Naerdemann, W.; Timioka, M.; Goodman, M.; Howell, S.B. Intracellular localization and trafficking of fluorescein-labeled cisplatin in human ovarian carcinoma cells. Clin. Cancer Res 2005, 11, 756–757. [Google Scholar]
- Jin, J.; Pastrello, D.; Penning, N.A.; Jones, A.T. Clustering of endocytic organelles in parental and drug-resistant myeloid leukaemia cell lines lacking centrosomally organised microtubule arrays. Int. J. Biochem. Cell Biol 2008, 40, 2240–2252. [Google Scholar]
- Bour-Dill, C.; Gramain, M.; Merlin, J.; Marchal, S.; Guillemin, F. Determination of intracellular organelles implicated in daunorubicin cytoplasmic sequestration in multidrug-resistant MCF-7 cells using fluorescence microscopy image analysis. Cytometry 2000, 39, 16–25. [Google Scholar]
- Lou, P.; Lai, P.; Sheh, M.; MacRobert, A.; Berg, K.; Bown, S. Reversal of doxorubicin in cancer cells by photochemical internalization. Int. J. Cancer 2006, 119, 2692–2698. [Google Scholar]
- Krolenko, S.A.; Adamyan, S.Y.; Belyaeva, T.N.; Mozhenok, T.P. Acridine orange accumulation in acid organelles of normal and vacuolated frog skeletal muscle fibres. Cell Biol. Int 2006, 30, 933–939. [Google Scholar]
- Niederau, C.; Grendell, J.H. Intracellular acuoles in experimental acute pancreatitis in rats and mice are an acidified compartment. J. Clin. Investig 1988, 81, 229–236. [Google Scholar]
- Blommaart, E.F.C.; Krause, U.; Schellens, J.P.M.; Vreeling-Sindelarova, H.; Meijer, A. The phosphatidylinositol 3-kinase inhibitors wortmannin and LY294002 inhibit autophagy in isolated rat heoticytes. Eur. J. Biochem. 1997, 243, 240–246. [Google Scholar]
- Ndolo, R.A.; Forrest, L.; Krise, J. The role of lysosomes in limiting drug toxicity in mice. J. Pharmacol. Exp. Ther 2010, 333, 120–128. [Google Scholar]
- Ouar, Z.; Lacave, R.; Bens, M.; Vandewalle, A. Mechanisms of altered sequestration and efflux of chemotherapeutic drugs by multidrug-resistant cells. Cell Biol. Toxicol 1999, 15, 91–100. [Google Scholar]
- Prasad, D.V.R.; Parekh, V.V.; Joshi, B.N.; Banderjee, P.P.; Parab, P.B.; Chattopadhyay, S.; Kumar, A.; Mishra, G.C. The Th1-specific costimulatory molecule M150 is a posttranslational isoform of lysosome-associated membrane protein-1. J. Immunol. 2002, 269, 1801–1809. [Google Scholar]
- Safaei, R.; Larson, B.J.; Cheng, T.C.; Gibson, M.A.; Otani, S.; Naerdemann, W.; Howell, S.B. Abnormal lysosomal trafficking and enhanced exosomal export of cisplatin in drug-resistant human ovarian carcinoma cells. Mol. Cancer Ther 2005, 4, 1595–1604. [Google Scholar]
- Herlevsen, M.; Oxford, G.; Owens, C.R.; Conaway, M.; Theodorescu, D. Depletion of major vault protein increases doxorubicin sensitivity and nuclear accumulation and disrupts its sequestration in lysosomes. Mol. Cancer Ther 2007, 6, 1804–1813. [Google Scholar]
- Thomas, D.D.; Martin, C.L.; Weng, N.; Byrne, J.A.; Groblewski, G.E. Tumour protein D52 expression and Ca2+-dependent phosphorylation modulates lysosomal membrane protein trafficking to the plasma membrane. Am. J. Cell Physiol 2010, 298, 725–739. [Google Scholar]
- Weylandt, K.H.; Nebrig, M.; Jansen-Rosseck, N.; Amey, J.S.; Carmena, D.; Wiedenmann, B.; Higgins, C.F.; Sardini, A. ClC-3 expression enhances etoposide resistance by increasing acidification of the late endocytic compartment. Mol. Cancer Ther 2007, 6, 979–986. [Google Scholar]
- Almquist, K.C.; Loe, D.W.; Hipfner, D.R.; Mackie, J.E.; Cole, S.P.C.; Deeley, R.G. Characterisation of the Mr 190,000 Multi Drug Resistance Protein (MRP) in drug-selected and transfected human tumour cells. Cancer Res 1995, 55, 102–110. [Google Scholar]
- Gong, Y.; Duvvuri, M.; Duncan, M.B.; Liu, J.; Krise, J.P. Niemann-Pick C1 protein facilitates the efflux of the anticancer drug daunorubicin from cells according to a novel vesicle-mediated pathway. J. Pharmacol. Exp. Ther 2006, 316, 242–247. [Google Scholar]
- Ko, D.C.; Gordon, M.D.; Jin, J.Y.; Scott, M.P. Dynamic movements of organelles containing Niemann-Pick C1 protein: NPC1 involvement in late endocytic events. Mol. Biol. Cell 2001, 12, 601–614. [Google Scholar]
- Ifergan, I.; Scheffer, G.L.; Assaraf, Y.G. Novel extracellular vesicles mediate an ABCG2-dependent anticancer drug sequestration and resistance. Cancer Res 2005, 65, 10952–10958. [Google Scholar]
- Gong, Y.; Duvvuri, M.; Krise, J. Separate roles for the golgi apparatus and lysosomes in the sequestration of drugs in the multidrug-resistant human leukemic cell line HL-60. J. Biol. Chem 2003, 278, 50234–50239. [Google Scholar]
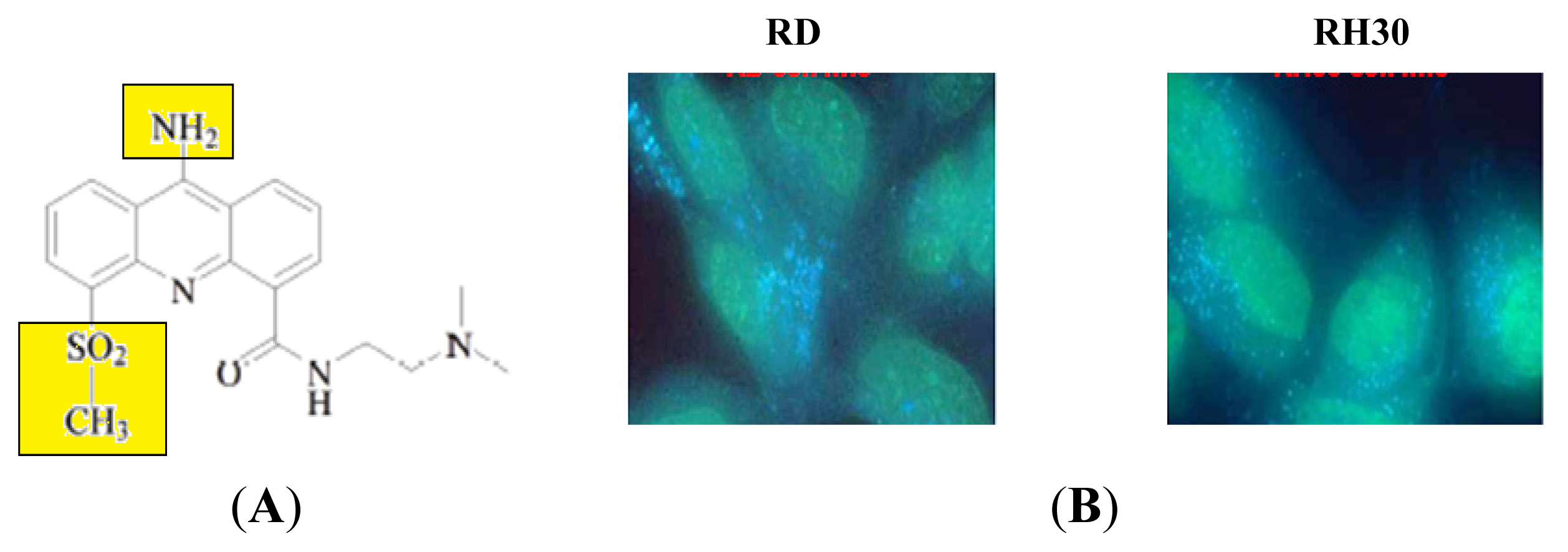
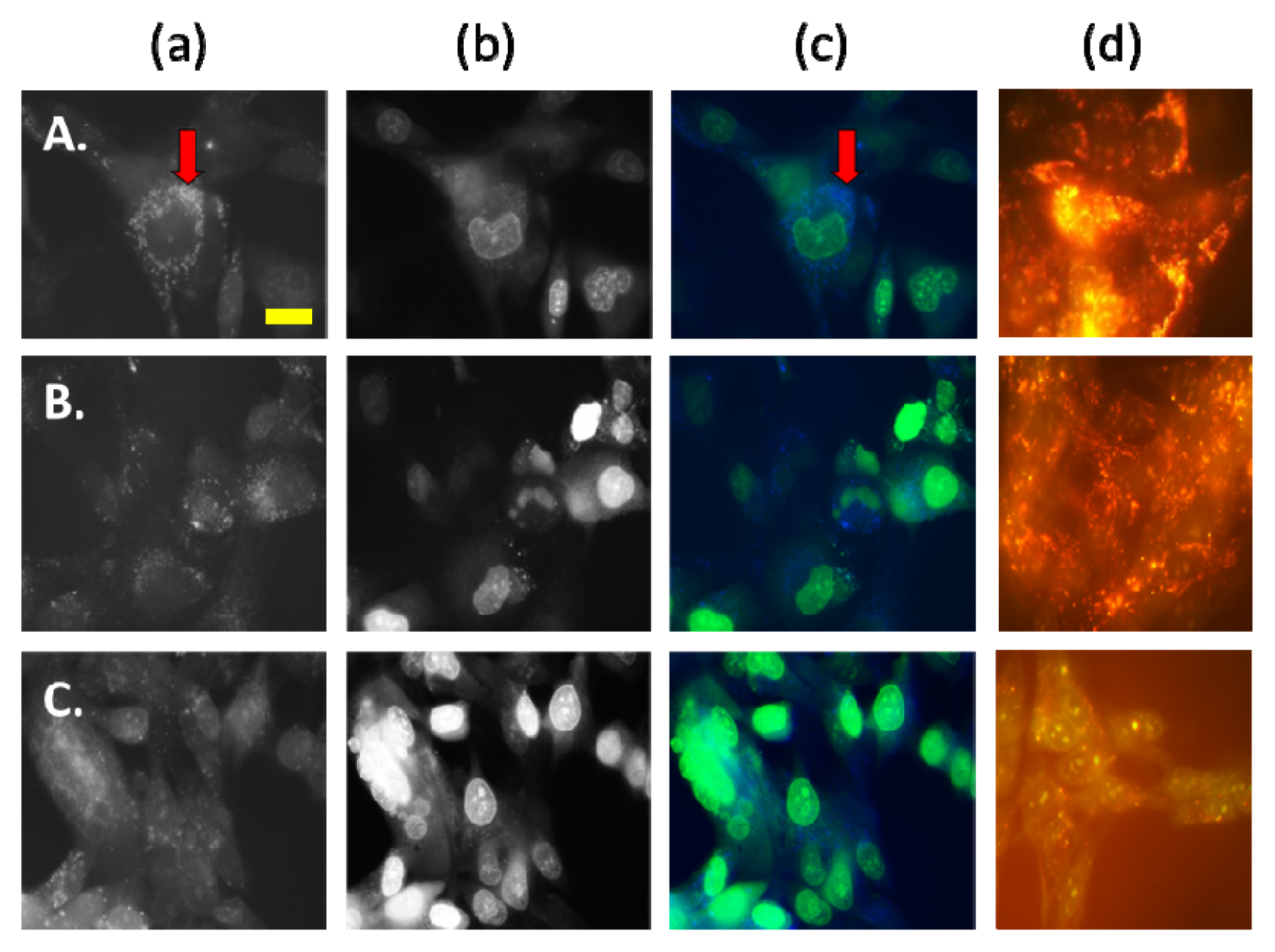

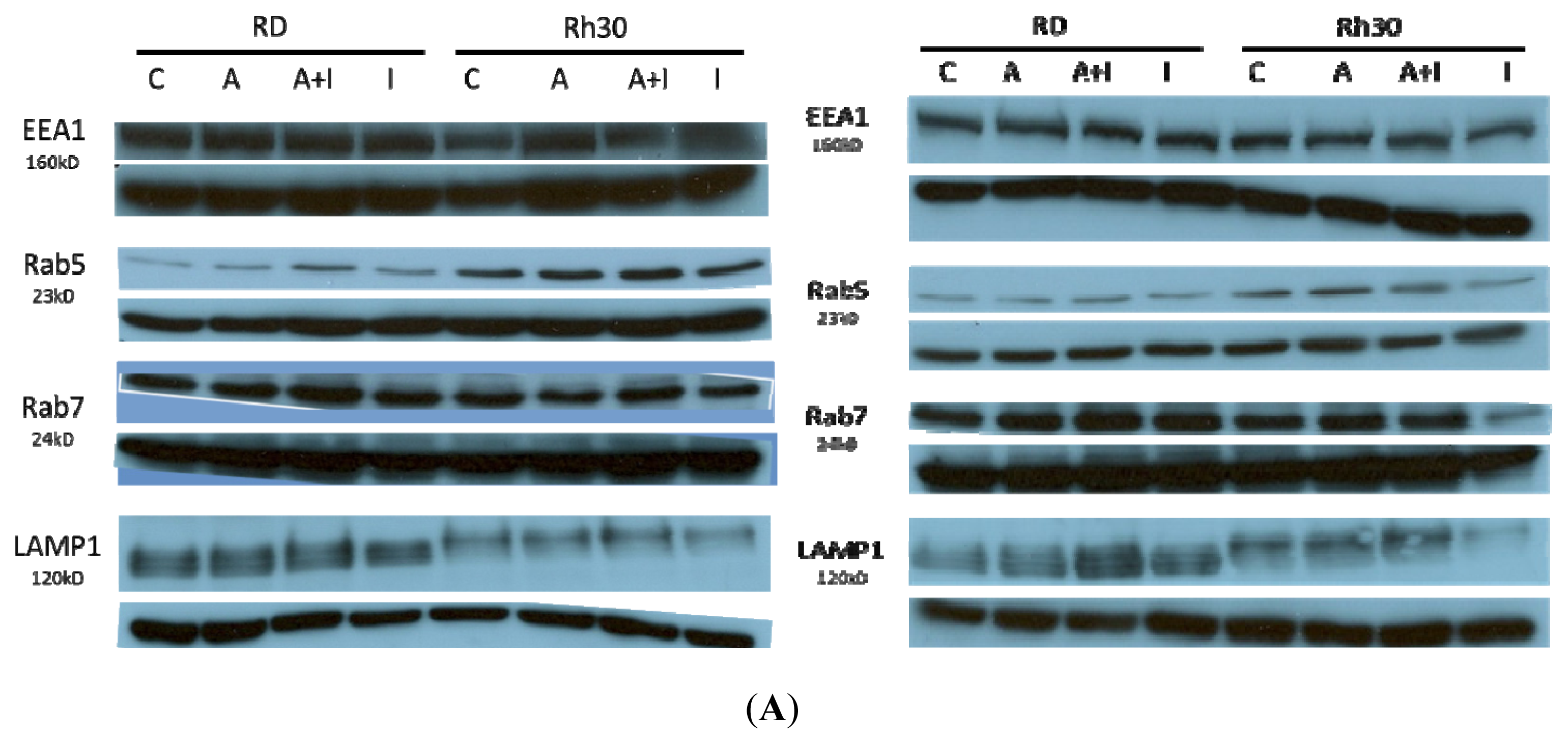
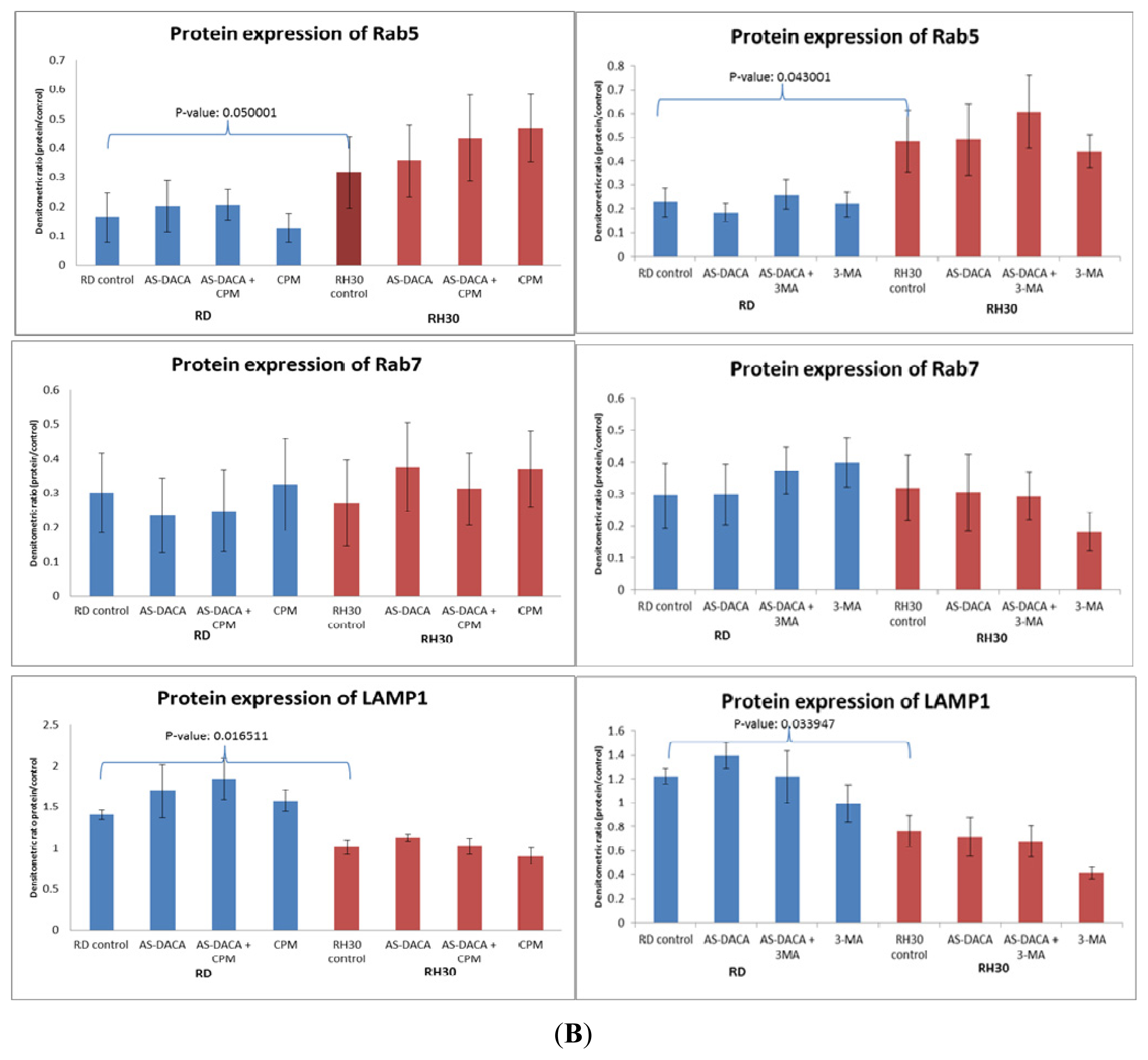

| Treatment | RD | RH30 | Fold change | T-test |
|---|---|---|---|---|
| AS-DACA | 0.92 ± 0.047 | 0.039 ± 0.007 | 23.5 | <0.05 * |
| Chlorpromazine | 39.3 ± 1.11 | 50.6 ± 9.1 | 0.78 | - |
| Bafilomycin A1 | 0.0015 ± 0.0003 | 0.0013 ± 0.0002 | 1.18 | - |
| Chloroquine | 9.61 ± 0.24 | 8.94 ± 3.0 | 1.07 | - |
| 3-Methyladenine | 823 ±178 | 692 ± 262 | 1.19 | - |
| AS-DACA plus | ||||
| Chlorpromazine | 0.51 ± 0.053 | 1.81 | 0.008 * | |
| 0.038 ± 0.0095 | 1.03 | 0.771 | ||
| Bafilomycin A1 | 1.45 ± 0.34 | 0.63 | 0.315 | |
| 0.054 ± 0.0084 | 0.72 | 0.132 | ||
| Chloroquine | 0.96 ± 0.55 | 0.96 | 0.562 | |
| 0.053 ± 0.0084 | 0.74 | 0.425 | ||
| 3-Methyladenine | 0.31 ± 0.20 | 2.97 | 0.033 * | |
| 0.041 ± 0.0073 | 0.95 | 0.803 | ||
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Williams, M.; Catchpoole, D. Sequestration of AS-DACA into Acidic Compartments of the Membrane Trafficking System as a Mechanism of Drug Resistance in Rhabdomyosarcoma. Int. J. Mol. Sci. 2013, 14, 13042-13062. https://doi.org/10.3390/ijms140713042
Williams M, Catchpoole D. Sequestration of AS-DACA into Acidic Compartments of the Membrane Trafficking System as a Mechanism of Drug Resistance in Rhabdomyosarcoma. International Journal of Molecular Sciences. 2013; 14(7):13042-13062. https://doi.org/10.3390/ijms140713042
Chicago/Turabian StyleWilliams, Marissa, and Daniel Catchpoole. 2013. "Sequestration of AS-DACA into Acidic Compartments of the Membrane Trafficking System as a Mechanism of Drug Resistance in Rhabdomyosarcoma" International Journal of Molecular Sciences 14, no. 7: 13042-13062. https://doi.org/10.3390/ijms140713042




