Effect of Plant Derived Antimicrobials on Salmonella Enteritidis Adhesion to and Invasion of Primary Chicken Oviduct Epithelial Cells in vitro and Virulence Gene Expression
Abstract
:1. Introduction
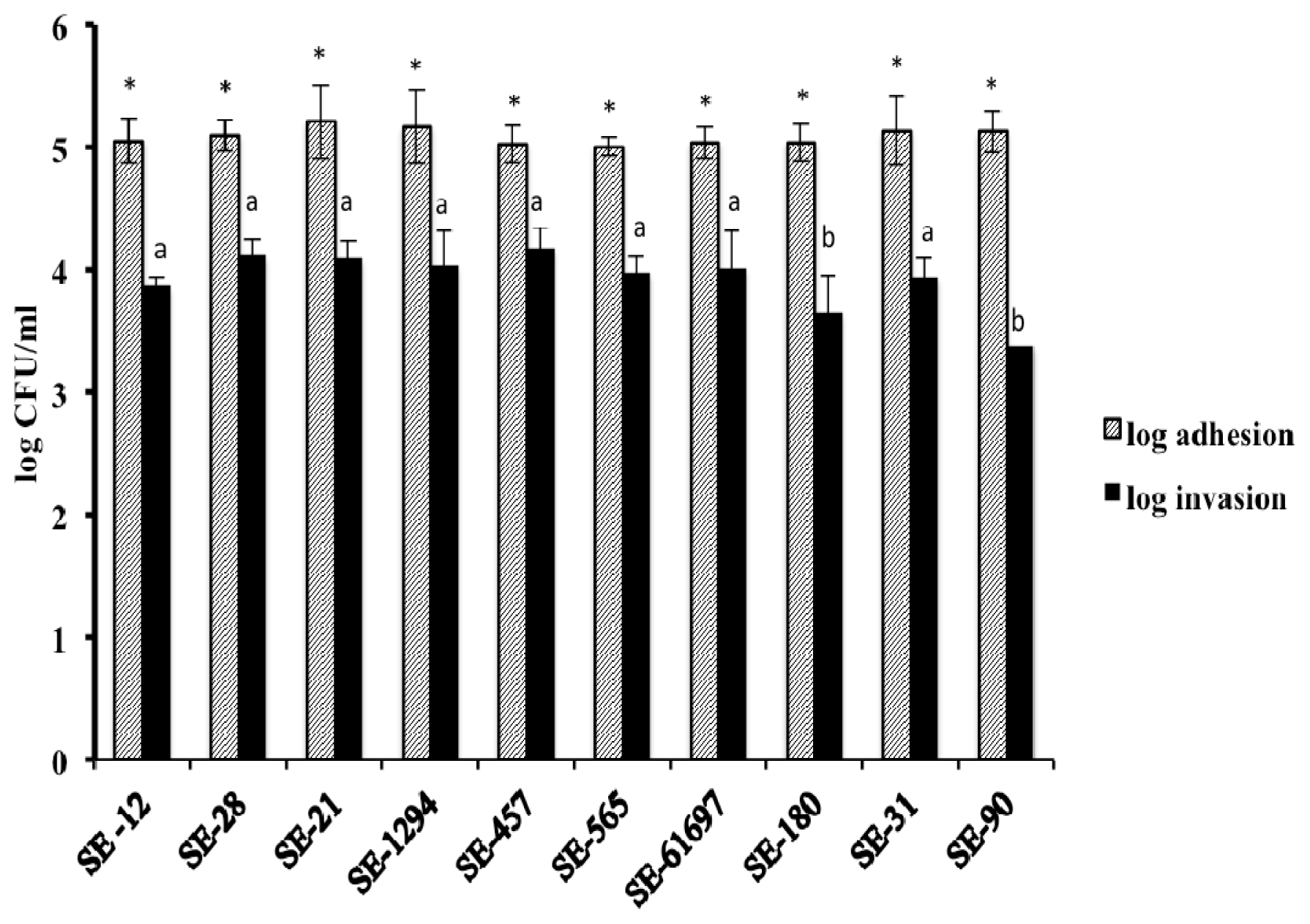
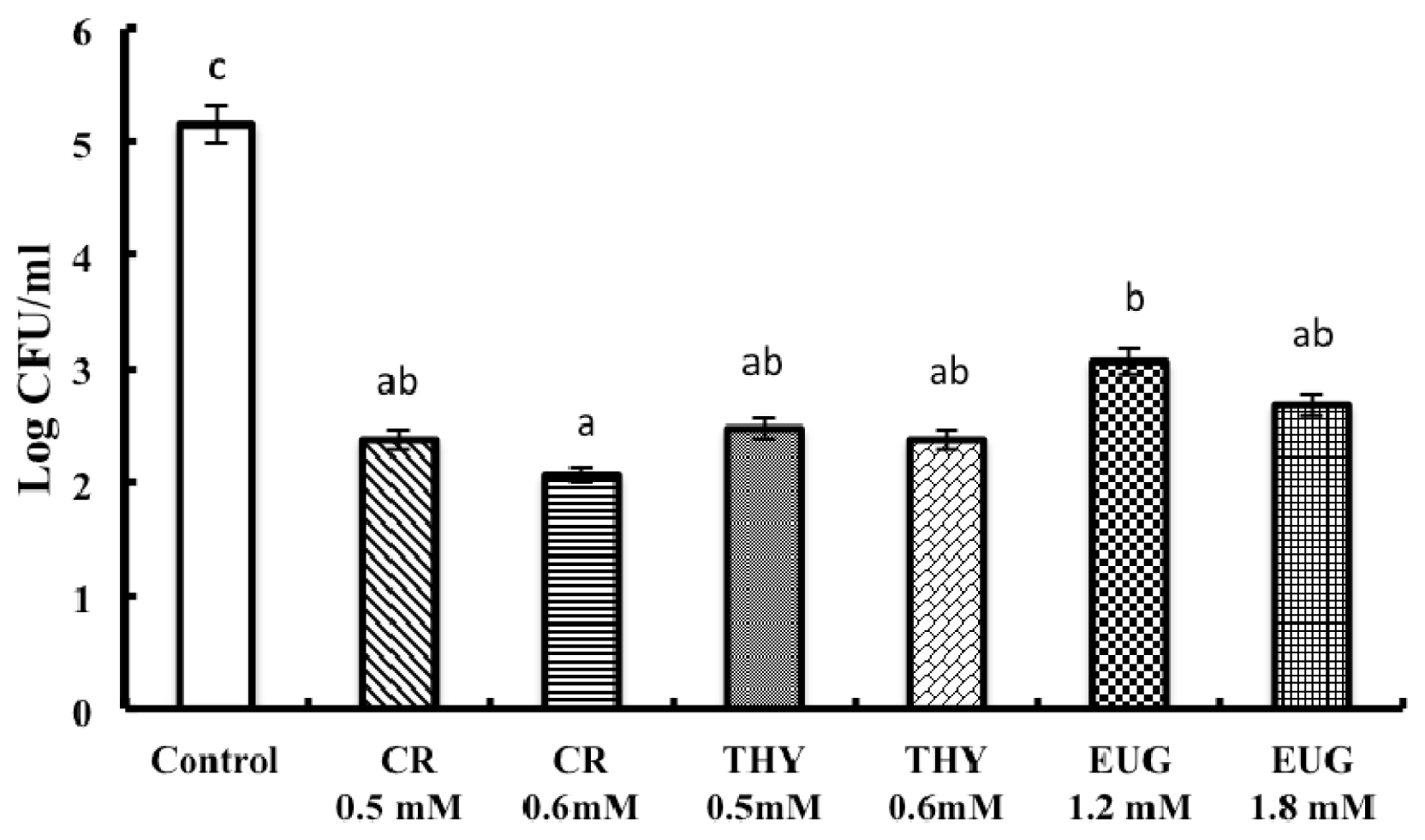
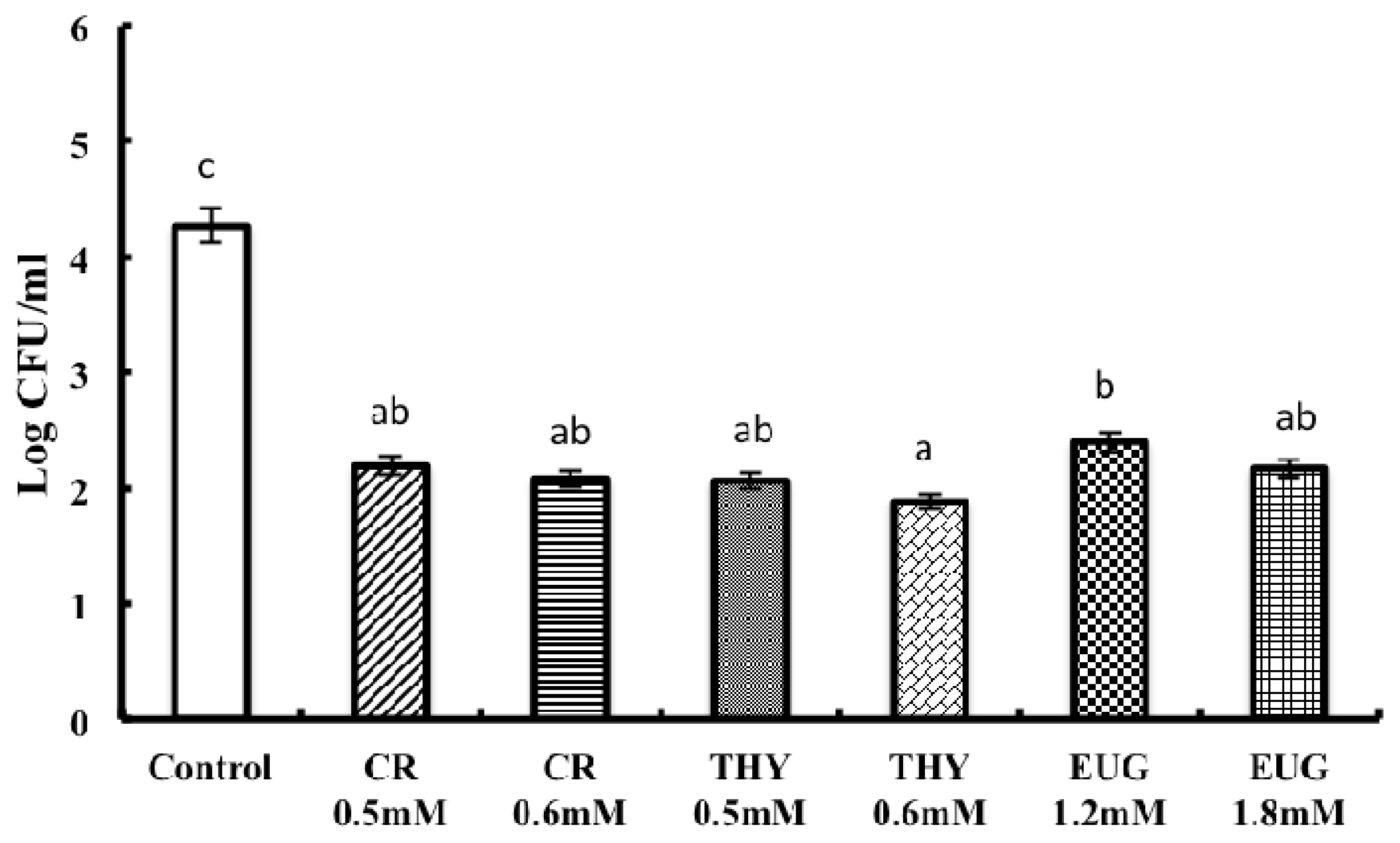
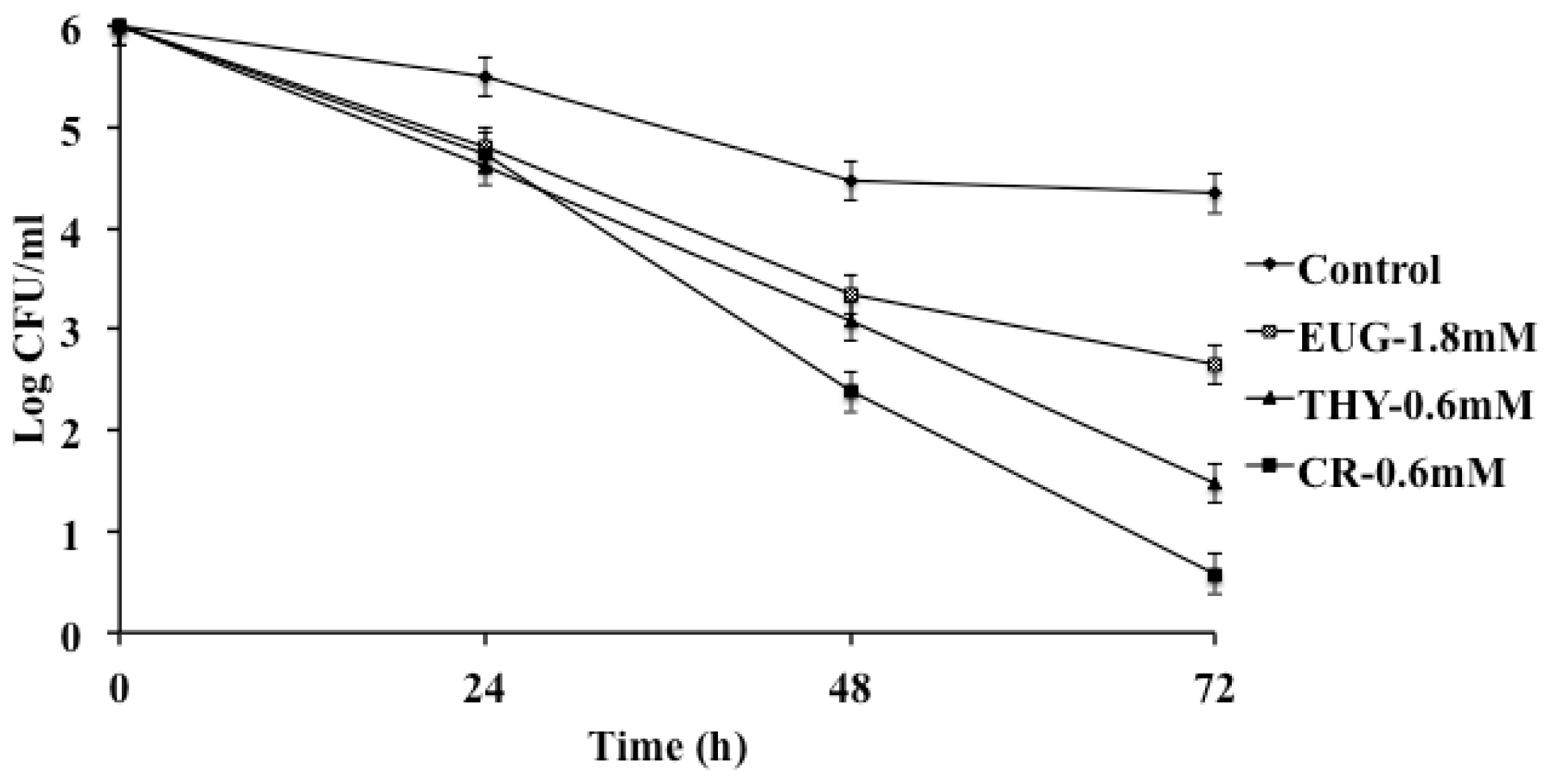
2. Results and Discussion
3. Experimental Section
3.1. Bacterial Strains and Culture Conditions
3.2. PDAs and SIC Determination
3.3. Isolation of Chicken oviduct Epithelial Cells
3.4. SE Adhesion and Invasion Assay
3.5. Macrophage Cultivation and SE Survival Assay
3.6. RNA Isolation and RT-qPCR
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- United States Department of Agriculture. Available online: http://www.ers.usda.gov/topics/animal-products/poultry-eggs/background.aspx (accessed on 28 September 2012).
- Latimer, H.K.; Jaykus, L.A.; Morales, R.A.; Cowen, P.; Crawford-Brown, D. Sensitivity analysis of Salmonella Enteritidis levels in contaminated shell eggs using a biphasic growth model. Int. J. Food Microbiol 2002, 75, 71–87. [Google Scholar]
- Bialka, K.L.; Demirci, A.; Knabel, S.J.; Patterson, P.H.; Puri, V.M. Efficacy of electrolyzed oxidizing water for the microbial safety and quality of eggs. Poult. Sci 2004, 83, 2071–2078. [Google Scholar]
- Namata, H.; Méroc, E.; Aerts, M.; Faes, C.; Abrahantes, J.C.; Imberechts, H.; Mintiens, K. Salmonella in Belgian laying hens: An identification of risk factors. Prev. Vet. Med 2008, 83, 323–336. [Google Scholar]
- Thomas, M.E.; Klinkenberg, D.; Ejeta, G.; van Knapen, F.; Bergwerff, A.A.; Stegeman, J.A.; Bouma, A. Quantification of horizontal transmission of Salmonella Enterica serovar Enteritidis bacteria in pair-housed groups of laying hens. Appl. Environ. Microbiol 2009, 75, 6361–6366. [Google Scholar]
- De Vylder, J.; van Hoorebeke, S.; Ducatelle, R.; Pasmans, F.; Haesebrouck, F.; Dewulf, J.; van Immerseel, F. Effect of the housing system on shedding and colonization of gut and internal organs of laying hens with Salmonella Enteritidis. Poult. Sci 2009, 88, 2491–2495. [Google Scholar]
- FDA Improves Egg Safety. 2009. Available online: http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm170640.htm (accessed on 30 September 2012).
- Allen-Vercoe, E.; Woodward, M.J. Colonization of the chicken caecum by afimbriate and aflagellate derivatives of Salmonella Enterica serotype Enteritidis. Vet. Microbiol 1999, 69, 265–275. [Google Scholar]
- Stern, N.J. Salmonella species and Campylobacter jejuni cecal colonization model in broilers. Poult. Sci 2008, 87, 2399–2403. [Google Scholar]
- Gantois, I.; Ducatelle, R.; Pasmans, F.; Haesebrouck, F.; Gast, R.; Humphrey, T.J.; van Immerseel, F. Mechanisms of egg contamination by Salmonella Enteritidis. FEMS Microbiol. Rev 2009, 33, 718–738. [Google Scholar]
- Miyamoto, T.; Baba, E.; Tanaka, T.; Sasai, K.; Fukata, T.; Arakawa, A. Salmonella Enteritidis contamination of eggs from hens inoculated by vaginal, cloacal, and intravenous routes. Avian Dis 1997, 41, 296–303. [Google Scholar]
- Okamura, M.; Kamijima, Y.; Miyamoto, T.; Tani, H.; Sasai, K.; Baba, E. Differences among six Salmonella serovars in abilities to colonize reproductive organs and to contaminate eggs in laying hens. Avian Dis 2001, 45, 61–69. [Google Scholar]
- Gast, R.K.; Guraya, R.; Guard-Bouldin, J.; Holt, P.S.; Moore, R.W. Colonization of specific regions of the reproductive tract and deposition at different locations inside eggs laid by hens infected with Salmonella Enteritidis or Salmonella Heidelberg. Avian Dis 2007, 51, 40–44. [Google Scholar]
- Gantois, I.; Ducatelle, R.; Pasmans, F.; Haesebrouck, F.; van Immerseel, F. Salmonella Enterica serovar Enteritidis genes induced during oviduct colonization and egg contamination in laying hens. Appl. Environ. Microbiol 2008, 74, 6616–6622. [Google Scholar]
- Thiagarajan, D.; Saeed, A.M.; Asem, E.K. Mechanism of transovarian transmission of Salmonella Enteritidis in laying hens. Poult. Sci 1994, 73, 89–98. [Google Scholar]
- Thiagarajan, D.; Saeed, M.; Turek, J.; Asem, E. In vitro attachment and invasion of chicken ovarian granulosa cells by Salmonella Enteritidis phage type 8. Infect. Immun 1996, 64, 5015–5021. [Google Scholar]
- Altekruse, S.; Koehler, J.; Hickman-Brenner, F.; Tauxe, R.V.; Ferris, K. A comparison of Salmonella Enteritidis phage types from egg-associated outbreaks and implicated laying flocks. Epidemiol. Infect 1993, 110, 17–22. [Google Scholar]
- Keller, L.H.; Benson, C.E.; Krotec, K.; Eckroade, R.J. Salmonella Enteritidis colonization of the reproductive tract and forming and freshly laid eggs of chickens. Infect Immun 1995, 63, 2443–2449. [Google Scholar]
- Clavijo, R.I.; Loui, C.; Andersen, G.L.; Riley, L.W.; Lu, S. Identification of genes associated with survival of Salmonella enterica serovar Enteritidis in chicken egg albumen. Appl. Environ. Microbiol 2006, 72, 1055–1064. [Google Scholar]
- Wollenweber, E. Occurrence of flavonoid aglycones in medicinal plants. Prog. Clin. Biol. Res 1988, 180, 45–55. [Google Scholar]
- Abee, T.; Krockel, L.; Hill, C. Bacteriocins: Modes of action and potentials in food preservation and control of food poisoning. Int. J. Food Microbiol 1995, 28, 169–185. [Google Scholar]
- Salamci, E.; Kordali, S.; Kotan, R.; Cakir, A.; Kaya, Y. Chemical compositions, antimicrobial and herbicidal effects of essential oils isolated from Turkish Tanacetum aucheranum and Tanacetum chiliophyllum var. chiliophyllum. Biochem. Syst. Ecol 2007, 35, 569–581. [Google Scholar]
- Bilgrami, K.S.; Sinha, K.K.; Sinha, A.K. Inhibition of aflatoxin production and growth of Aspergillus flavus by eugenol and onion and garlic extracts. Indian J Med Res 1992, 96, 171–175. [Google Scholar]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol 2004, 94, 223–253. [Google Scholar]
- Holley, R.A.; Patel, D. Improvement of shelf life and safety of perishable foods by plant essential oils and smoke antimicrobials. Food Microbiol 2005, 22, 273–292. [Google Scholar]
- Friedman, M.; Henika, P.R.; Mandrell, R.E. Bactericidal activities of plant essential oils and some of there isolated constituents against Campylobacter jejuni, Escherichia coli, Listeria monocytogenes, and Salmonella enterica. J. Food Prot 2002, 65, 1545–1560. [Google Scholar]
- Chun, O.K.; Kim, D.O.; Smith, N.; Schroeder, D.; Han, J.T.; Lee, C.Y. Daily consumption of phenolics and total antioxi- dant capacity from fruit and vegetables in the American diet. J. Sci. Food Agric 2005, 85, 1715–1724. [Google Scholar]
- Ali, S.M.; Khan, A.A.; Ahmed, I.; Musaddiq, M.; Ahmed, K.S.; Polasa, H.; Venkateswar Rao, L.; Habibullah, C.M.; Sechi, L.A.; Ahmed, N. Antimicrobial activities of Eugenol and Cinnamaldehyde against the human gastric pathogen Helicobacter pylori. Ann. Clin. Microbiol. Antimicrob 2005, 4, 20–27. [Google Scholar]
- Arrebola, M.L.; Navarro, M.C.; Jimenez, J.; Ocana, F.A. Yield and composition of the essential oil of Thymus serpylloides subsp. serpylloides. Phytochemistry 1994, 36, 67–72. [Google Scholar]
- Adams, T.B.; Cohen, S.M.; Doull, J.; Feron, V.J.; Goodman, J.I.; Marnett, L.J.; Munro, I.C.; Portoghese, P.S.; Smith, R.L.; Waddell, W.J.; et al. The FEMA GRAS assessment of cinnamyl derivatives used as flavor ingredients. Food Chem. Toxicol 2004, 42, 157–185. [Google Scholar]
- Adams, T.B.; Cohen, S.M.; Doull, J.; Feron, V.J.; Goodman, J.I.; Marnett, L.J.; Munro, I.C.; Portoghese, P.S.; Smith, R.L.; Waddell, W.J.; et al. The FEMA GRAS assessment of hydroxyl- and alkoxy-substituted benzyl derivatives used as flavor ingredients. Food Chem. Toxicol 2005, 43, 1241–1271. [Google Scholar]
- Baskaran, Y.; Periyasamy, V.; Venkatraman, A.C. Investigation of antioxidant, anti- inflammatory and DNA-protective properties of eugenol in thioacetamide-induced liver injury in rats. Toxicology 2010, 268, 204–212. [Google Scholar]
- Kollanoor-Johny, A.; Darre, M.J.; Hoagland, T.A.; Schreiber, D.T.; Donoghue, A.M.; Donoghue, D.J.; Venkitanarayanan, K. Antibacterial effect of trans- cinnamaldehyde on Salmonella Enteritidis and Campylobacter jejuni in chicken drinking water. J. Appl. Poult. Res 2008, 17, 490–497. [Google Scholar]
- Kollanoor-Johny, A.; Darre, M.J.; Donoghue, A.M.; Donoghue, D.J.; Venkitanarayanan, K. Antibacterial effect of trans-cinnamaldehyde, eugenol, thymol and carvacrol against Salmonella Enteritidis and Campylobacter jejuni in vitro. J. Appl. Poult. Res 2010, 19, 237–244. [Google Scholar]
- Kollanoor-Johny, A.; Mattson, T.; Baskaran, S.A.; Amalaradjou, M.A.; Babapoor, S.; March, B.; Valipe, S.; Darre, M.; Hoagland, T.; Schreiber, D.; et al. Reduction of Salmonella enterica serovar Enteritidis colonization in 20-day-old broiler chickens by the plant-derived compounds trans-cinnamaldehyde and eugenol. Appl. Environ. Microbiol 2012, 8, 2981–2987. [Google Scholar]
- Amalaradjou, M.A.; Narayanan, A.; Venkitanarayanan, K. Trans-cinnamaldehyde decreases attachment and invasion of uropathogenic Escherichia coli in urinary tract epithelial cells by modulating virulence gene expression. J. Urol 2011, 185, 1526–1531. [Google Scholar]
- De Buck, J.; van Immerseel, F.; Meulemans, G.; Haesebrouck, F.; Ducatelle, R. Adhesion of Salmonella enterica serotype Enteritidis isolates to chicken isthmal glandular secretions. Vet. Microbiol 2003, 93, 223–233. [Google Scholar]
- Hoop, R.K.; Pospischil, A. Bacteriological, serological, histological and immunohistochemical findings in laying hens with naturally acquired Salmonella Enteritidis phage type 4 infection. Vet. Rec 1993, 133, 391–393. [Google Scholar]
- De Buck, J.; Pasmans, F.; van Immerseel, F.; Haesebrouck, F.; Ducatelle, R. Tubular glands of the isthmus are the predominant colonization sites of Salmonella Enteritidis in the upper oviduct of laying hens. Poult. Sci 2004, 83, 352–358. [Google Scholar]
- Cox, N.A.; Berrang, M.E.; Cason, J.A. Salmonella penetration of eggshells and proliferation in broiler hatching eggs-a review. Poult. Sci 2000, 79, 1571–1574. [Google Scholar]
- De Buck, J.; van Immerseel, F.; Haesebrouck, F.; Ducatelle, R. Colonization of the chicken reproductive tract and egg contamination by Salmonella. J. Appl. Microbiol 2004, 97, 233–245. [Google Scholar]
- Ebers, K.L.; Zhang, C.Y.; Zhang, M.Z.; Bailey, R.H.; Zhang, S. Transcriptional profiling avian beta-defensins in chicken oviduct epithelial cells before and after infection with Salmonella entericaserovar Enteritidis. BMC Microbiol 2009, 9. [Google Scholar] [CrossRef]
- Jung, J.G.; Park, T.S.; Kim, J.N.; Han, B.K.; Lee, S.D.; Song, G.; Han, J.Y. Characterization and application of oviductal epithelial cells in vitro in Gallus domesticus. Biol. Reprod 2011, 85, 798–807. [Google Scholar]
- Harwig, S.S.; Swiderek, K.; Kokryakov, V.N.; Tan, L.; Lee, T.D.; Panyutich, E.A.; Aleshina, G.M.; Shamova, O.V.; Lehrer, R.I. Gallinacins: Cysteine- rich antimicrobial peptides of chicken leukocytes. FEBS Lett 1994, 342, 281–285. [Google Scholar]
- Lynn, D.J.; Higgs, R.; Gaines, S.; Tierney, J.; James, T.; Lloyd, A.T.; Fares, M.A.; Mulcahy, G.; O’Farrelly, C. Bioinformatic discovery and initial characterisation of nine novel antimicrobial peptide genes in the chicken. Immunogenetics 2004, 56, 170–177. [Google Scholar]
- Xiao, Y.; Hughes, A.L.; Ando, J.; Matsuda, Y.; Cheng, J.F.; Skinner-Noble, D.; Zhang, G. A genome-wide screen identifies a single beta-defensin gene cluster in the chicken: Implications for the origin and evolution of mammalian defensins. BMC Genomics 2004, 5, 56–67. [Google Scholar]
- Michailidis, G.; Avdi, M.; Argiriou, A. Transcriptional profiling of antimicrobial peptides avian β-defensins in the chicken ovary during sexual maturation and in response to Salmonella Enteritidis infection. Res. Vet. Sci 2012, 92, 60–65. [Google Scholar]
- Mageed, A.M.; Isobe, N.; Yoshimura, Y. Expression of avian beta defensins in the oviduct and effects of lipopolysaccharide on their expression in the vagina of hens. Poult. Sci 2008, 87, 979–984. [Google Scholar]
- Li, S.; Zhang, Z.; Pace, L.; Lillehoj, H.; Zhang, S. Functions exerted by the virulence-associated type-three secretion systems during Salmonella enterica serovar Enteritidis invasion into and survival within chicken oviduct epithelial cells and macrophages. Avian Pathol 2009, 38, 97–106. [Google Scholar]
- Goh, E.B.; Yim, G.; Tsui, W.; McClure, J.; Surette, M.G.; Davies, J. Transcriptional modulation of bacterial gene expression by subinhibitory concentrations of antibiotics. Proc. Natl. Acad. Sci. USA 2002, 99, 17025–17030. [Google Scholar]
- Fonseca, A.P.; Extremina, C.; Fonseca, A.F.; Sousa, J.C. Effect of subinhibitory concentration of piperacillin/tazobactam on Pseudomonas aeruginosa. J. Med. Microbiol 2004, 53, 903–910. [Google Scholar]
- De Buck, J.; van Immerseel, F.; Haesebrouck, F.; Ducatelle, R. Effect of type 1 fimbriae of Salmonella enterica serotype Enteritidis on bacteremia and reproductive tract infection in laying hens. Avian Pathol 2004, 33, 314–320. [Google Scholar]
- Porter, S.B.; Curtiss, R., III. Effect of Inv mutations on Salmonella virulence and colonization in 1-day-old White Leghorn chicks. Avian Dis 1997, 41, 45–57. [Google Scholar]
- Lu, S.; Killoran, P.B.; Riley, L.W. Association of Salmonella enterica serovar Enteritidis yafD with resistance to chicken egg albumen. Infect. Immun 2003, 71, 6734–6741. [Google Scholar]
- Gantois, I.; Ducatelle, R.; Pasmans, F.; Haesebrouck, F.; van Immerseel, F. The Salmonella Enteritidis lipopolysaccharide biosynthesis gene rfbH is required for survival in egg albumen. Zoonoses Public Health 2009, 56, 145–149. [Google Scholar]
- Shah, D.H.; Casavant, C.; Hawley, Q.; Addwebi, T.; Call, D.R.; Guard-Petter, J. Salmonella Enteritidis strains from poultry exhibit differential responses to acid stress, oxidative stress, and survival in the egg albumen. Foodborne Pathog. Dis 2012, 9, 258–264. [Google Scholar]
- Bohez, L.; Gantois, I.; Ducatelle, R.; Pasmans, F.; Dewulf, J.; Haesebrouck, F.; van Immerseel, F. The Salmonella Pathogenicity Island 2 regulator ssrA promotes reproductive tract but not intestinal colonization in chickens. Vet. Microbiol 2008, 126, 216–224. [Google Scholar]
- De Groote, M.A.; Ochsner, U.A.; Shiloh, M.U.; Nathan, C.; McCord, J.M.; Dinauer, M.C.; Libby, S.J.; Vazquez-Torres, A.; Xu, Y.; Fang, F.C. Periplasmic superoxide dismutase protects Salmonella from products of phagocyte NADPH-oxidase and nitric oxide synthase. Proc. Natl. Acad. Sci. USA 1997, 94, 3997–14001. [Google Scholar]
- Lesnick, M.L.; Reiner, N.E.; Fierer, J.; Guiney, D.G. The Salmonella spvB virulence gene encodes an enzyme that ADP-ribosylates actin and destabilizes the cytoskeleton of eukaryotic cells. Mol. Microbiol 2001, 39, 1464–1470. [Google Scholar]
- Otto, H.; Tezcan-Merdol, D.; Girisch, R.; Haag, F.; Rhen, M.; Koch-Nolte, F. The spvB gene-product of the Salmonella enterica virulence plasmid is a mono (ADP-ribosyl) transferase. Mol. Microbiol 2000, 37, 1106–1115. [Google Scholar]
- Retamal, P.; Castillo-Ruiz, M.; Mora, G.C. Characterization of MgtC, a virulence factor of Salmonella enterica serovar Typhi. PloS One 2009, 4, e5551. [Google Scholar]
- Townsend, S.M.; Hurrell, E.; Gonzalez-Gomez, I.; Lowe, J.; Frye, J.G.; Forsythe, S.; Badger, J.L. Enterobacter sakazakii invades brain capillary endothelial cells, persists in human macrophages influencing cytokine secretion and induces severe brain pathology in the neonatal rat. Microbiology 2007, 153, 3538–3547. [Google Scholar]
- Erturk, H.N. Responses of Superoxide Dismutases to Oxidative Stress in Arabidopsis thaliana. Ph.D. Thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA, USA, January 1999. [Google Scholar]
- Kollanoor-Johny, A.; Baskaran, S.A.; Charles, A.S.; Amalaradjou, M.A.; Darre, M.J.; Khan, M.I.; Hoagland, T.A.; Schreiber, D.T.; Donoghue, A.M.; Donoghue, D.J.; et al. Prophylactic supplementation of caprylic acid in feed reduces Salmonella enteritidis colonization in commercial broiler chicks. J. Food Prot 2009, 72, 722–727. [Google Scholar]
- Moroni, O.; Kheadr, E.; Boutin, Y.; Lacroix, C.; Fliss, I. Inactivation of adhesion and invasion of food-borne Listeria monocytogenes by bacteriocin-producing Bifidobacterium strains of human origin. Appl. Environ. Microbiol 2006, 72, 6894–6901. [Google Scholar]
- Kannan, L.; Rath, N.C.; Liyanage, R.; Lay, J.O., Jr. Identification and characterization of thymosin beta-4 in chicken macrophages using whole cell MALDI-TOF. Ann. N. Y. Acad. Sci 2007, 1112, 425–434. [Google Scholar]

| Gene | CR 0.50 mM | CR 0.60 mM | THY 0.50 mM | THY 0.60 mM | EUG 1.2 mM | EUG 1.8 mM |
|---|---|---|---|---|---|---|
| fimD | −1.88 | −1.95 | −3.04 | −3.67 | −6.11 | −6.87 |
| flgG | −1.81 | −2.19 | −1.13 | −1.24 | −3.55 | −3.83 |
| hflK | −1.40 | −2.44 | −0.30 | −1.90 | −3.18 | −3.32 |
| invH | −16.39 | −17.78 | −28.55 | −29.76 | −18.89 | −19.81 |
| lrpF | −10.05 | −13.15 | −41.66 | −47.61 | −1.08 | −1.14 |
| mrr1 | −4.98 | −6.22 | −8.20 | −10.88 | −23.95 | −24.13 |
| ompR | −3.10 | −3.66 | −5.75 | −6.13 | −0.15 | −0.18 |
| orf245 | −21.39 | −25.37 | −60.32 | −64.81 | −73.29 | −76.92 |
| pipB | −15.34 | −19.16 | −65.48 | −66.90 | −54.38 | −54.59 |
| prot6E | −1.99 | −3.25 | −5.99 | −8.65 | −7.11 | −7.92 |
| rfbH | −10.41 | −11.11 | −12.09 | −16.66 | −1.17 | −1.20 |
| rpoS | −4.20 | −6.89 | −3.33 | −5.23 | −1.33 | −1.48 |
| sipA | −29.66 | −35.24 | −78.59 | −88.86 | −76.30 | −77.14 |
| sipB | −7.12 | −8.33 | −14.50 | −17.48 | −50.29 | −53.42 |
| sodC | −4.14 | −4.20 | −8.67 | −9.25 | −0.22 | −0.27 |
| spvB | −0.28 | −0.44 | −0.14 | −0.21 | −0.12 | −0.19 |
| mgtC | −4.09 | −6.99 | −4.52 | −7.69 | −0.12 | −0.23 |
| sopB | −22.49 | −24.10 | −43.22 | −46.60 | −48.58 | −49.25 |
| ssaV | −5.05 | −6.93 | −0.10 | −0.10 | −1.07 | −1.27 |
| ssrA | −5.97 | −7.04 | −3.29 | −4.11 | 0.09 | −0.11 |
| tatA | −0.61 | −1.74 | −0.78 | −2.08 | −0.94 | −1.15 |
| xthA | −5.02 | −5.74 | −8.01 | −8.26 | −0.19 | −0.27 |
| Strains of SE | Source |
|---|---|
| SE-21 | Chicken intestine isolate (Connecticut Veterinary Diagnostic Medical Laboratory) |
| SE-28 | Chicken oviduct isolate (Connecticut Veterinary Diagnostic Medical Laboratory) |
| SE-12 | Chicken liver isolate (Connecticut Veterinary Diagnostic Medical Laboratory) |
| SE-31 | Chicken gut isolate (Connecticut Veterinary Diagnostic Medical Laboratory) |
| SE-457 | Chicken egg yolk isolate (University of Pennsylvania) |
| SE-1294 | Human egg outbreak (New York Department of Health) |
| SE-565 | Food outbreak (Lunch–II) |
| SE-61697 | Human isolate (University of Pennsylvania) |
| SE-180 | Human isolate (New York Department of Health) |
| SE-90 | Human isolate (Connecticut Veterinary Diagnostic Medical Laboratory) |
| Gene | Sequence |
|---|---|
| β-Actin-F | 5′-TGCGTGACATCAAGGAGAAG-3′ |
| β-Actin-R | 5′-GACCATCAGGGAGTTCATAGC-3′ |
| AvBD-4-F | 5′-CATCTCAGTGTCGTTTCTCTGC-3′ |
| AvBD-4-R | 5′-CGCGATATCCACATTGCATG-3′ |
| AvBD-5-F | 5′-CTGCCAGCAAGAAAGGAACCTG-3′ |
| AvBD-5-R | 5′-GTAATCCTCGAGCAAGGGACA-3′ |
| AvBD-9-F | 5′-GCAAAGGCTATTCCACAGCAG-3′ |
| AvBD-9-R | 5′-GGAGCACGGCATGCAACAA-3′ |
| AvBD-10-F | 5′-TGGGGCACGCAGTCCACAAC-3′ |
| AvBD-10-R | 5′-CATGCCCCAGCACGGCAGAA-3′ |
| AvBD-11-F | 5′-ACTGCATCCGTTCCAAAGTCTG-3′ |
| AvBD-11-R | 5′-GTCCCAGCTGTTCTTCCAG-3′ |
| AvBD-12-F | 5′-CCCAGCAGGACCAAAGCAATG-3′ |
| AvBD-12-R | 5′-AGTACTTAGCCAGGTATTCC-3′ |
| Accession Number | Gene | Gene Function | Sequence (5′-3′) |
|---|---|---|---|
| NC_011294.1 | fimDF fimDR | Outer membrane usher protein FimD | 5′CGCGGCGAAAGTTATTTCAA 3′ 5′CCACGGACGCGGTATCC 3′ |
| NC_011294.1 | flgGF flgGR | Flagellar basal body rod protein | 5′GCGCCGGACGATTGC 3′ 5′CCGGGCTGGAAAGCATT 3′ |
| NC_011294.1 | hflKF hflKR | FtsH protease regulator | 5′AGCGCGGCGTTGTGA 3′ 5′TCAGACCTGGCTCTACCAGATG 3′ |
| NC_011294.1 | invHF invHR | Cell adherence/invasion protein | 5′ CCCTTCCTCCGTGAGCAAA 3′ 5′TGGCCAGTTGCTCTTTCTGA 3′ |
| NC_011294.1 | lrpF lrpR | Leucine-responsive transcriptional regulator | 5′TTAATGCCGCCGTGCAA 3′ 5′GCCGGAAACCAAATGACACT 3′ |
| NC_011294.1 | mrr1F mrr1R | Pseudo/restriction endonuclease gene | 5′CCATCGCTTCCAGCAACTG 3′ 5′TCTCTACCATGAACCCGTACAAATT 3′ |
| NC_011294.1 | ompRF ompRR | Osmolarity response regulator | 5′TGTGCCGGATCTTCTTCCA 3′ 5′CTCCATCGACGTCCAGATCTC 3′ |
| NC_011294.1 | orf245F orf245R | Pathogenicity island protein | 5′CAGGGTAATATCGATGTGGACTACA 3′ 5′GCGGTATGTGGAAAACGAGTTT 3′ |
| NC_011294.1 | pipBF pipBR | Pathogenicity island protein | 5′GCTCCTGTTAATGATTTCGCTAAAG3′ 5′GCTCAGACTTAACTGACACCAAACTAA 3′ |
| NC_011294.1 | prot6EF prot6ER | Fimbrial biosynthesis | 5′GAACGTTTGGCTGCCTATGG 3′ 5′CGCAGTGACTGGCATCAAGA 3′ |
| NC_011294.1 | rfbHF rfbHR | DehydrataseRfbH | 5′ACGGTCGGTATTTGTCAACTCA 3′ 5′TCGCCAACCGTATTTTGCTAA 3′ |
| NC_011294.1 | rpoSF rpoSR | RNA polymerase sigma factor RpoS | 5′TTTTTCATCGGCCAGGATGT 3′ 5′CGCTGGGCGGTGATTC 3′ |
| NC_011294.1 | sipAF sipAR | Pathogenicity island 1 effector protein | 5′CAGGGAACGGTGTGGAGGTA 3′ 5′AGACGTTTTTGGGTGTGATACGT 3′ |
| NC_011294.1 | sipBF sipBR | Pathogenicity island 1 effector protein | 5′GCCACTGCTGAATCTGATCCA 3′ 5′CGAGGCGCTTGCTGATTT 3′ |
| NC_011294.1 | sodCF sodCR | Superoxide dismutase | 5′CACATGGATCATGAGCGCTTT 3′ 5′CTGCGCCGCGTCTGA3′ |
| NC_011294.1 | sopBF sopBR | Cell invasion protein | 5′GCGTCAATTTCATGGGCTAAC 3′ 5′GGCGGCGAACCCTATAAACT 3′ |
| NC_011294.1 | ssaVF ssaVR | Secretion system apparatus protein SsaV | 5′GCGCGATACGGACATATTCTG 3′ 5′TGGGCGCCACGTGAA3′ |
| NC_011294.1 | ssrAF ssrAR | Sensor Kinase | 5′CGAGTATGGCTGGATCAAAACA 3′ 5′TGTACGTATTTTTTGCGGGATGT 3′ |
| NC_011294.1 | tatAF tatAR | Twin arginine translocase protein A | 5′AGTATTTGGCAGTTGTTGATTGTTG 3′ 5′ACCGATGGAACCGAGTTTTTT 3′ |
| NC_011294.1 | xthAF xthAR | Exonuclease III | 5′CGCCCGTCCCCATCA 3′ 5′CACATCGGGCTGGTGTTTT 3′ |
| NC_011294.1 | 16S f 16S r | SENr010, 16S ribosomal RNA | 5′CCAGGGCTACACACGTGCTA 3′ 5′TCTCGCGAGGTCGCTTCT 3′ |
| NC_011294.1 | mgtCF mgtCR | Mg (2+) transport ATPase protein C | 5′CGAACCTCGCTTTCATCTTCTT 3′ 5′CCGCCGAGGGAGAAAAAC 3′ |
| NC_019120.1 | spvBF spvBR | Actin ADP ribosyltransferase 2C toxin SpvB | 5′TGGGTGGGCAACAGCAA 3′ 5′GCAGGATGCCGTTACTGTCA 3′ |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Upadhyaya, I.; Upadhyay, A.; Kollanoor-Johny, A.; Darre, M.J.; Venkitanarayanan, K. Effect of Plant Derived Antimicrobials on Salmonella Enteritidis Adhesion to and Invasion of Primary Chicken Oviduct Epithelial Cells in vitro and Virulence Gene Expression. Int. J. Mol. Sci. 2013, 14, 10608-10625. https://doi.org/10.3390/ijms140510608
Upadhyaya I, Upadhyay A, Kollanoor-Johny A, Darre MJ, Venkitanarayanan K. Effect of Plant Derived Antimicrobials on Salmonella Enteritidis Adhesion to and Invasion of Primary Chicken Oviduct Epithelial Cells in vitro and Virulence Gene Expression. International Journal of Molecular Sciences. 2013; 14(5):10608-10625. https://doi.org/10.3390/ijms140510608
Chicago/Turabian StyleUpadhyaya, Indu, Abhinav Upadhyay, Anup Kollanoor-Johny, Michael J. Darre, and Kumar Venkitanarayanan. 2013. "Effect of Plant Derived Antimicrobials on Salmonella Enteritidis Adhesion to and Invasion of Primary Chicken Oviduct Epithelial Cells in vitro and Virulence Gene Expression" International Journal of Molecular Sciences 14, no. 5: 10608-10625. https://doi.org/10.3390/ijms140510608




