NAD(P)H:Quinone Oxidoreductase 1 (NQO1) P187S Polymorphism and Prostate Cancer Risk in Caucasians
Abstract
:1. Introduction
2. Results and Discussion
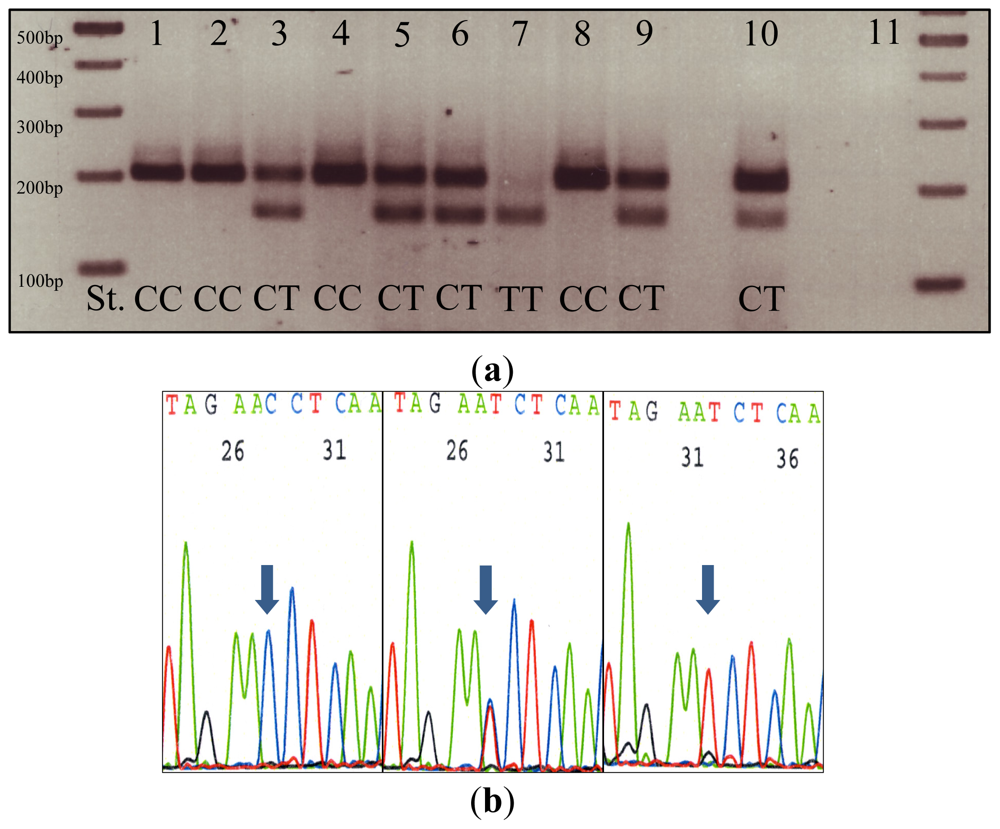
2.1. Results from Method Validation and Cohort Testing
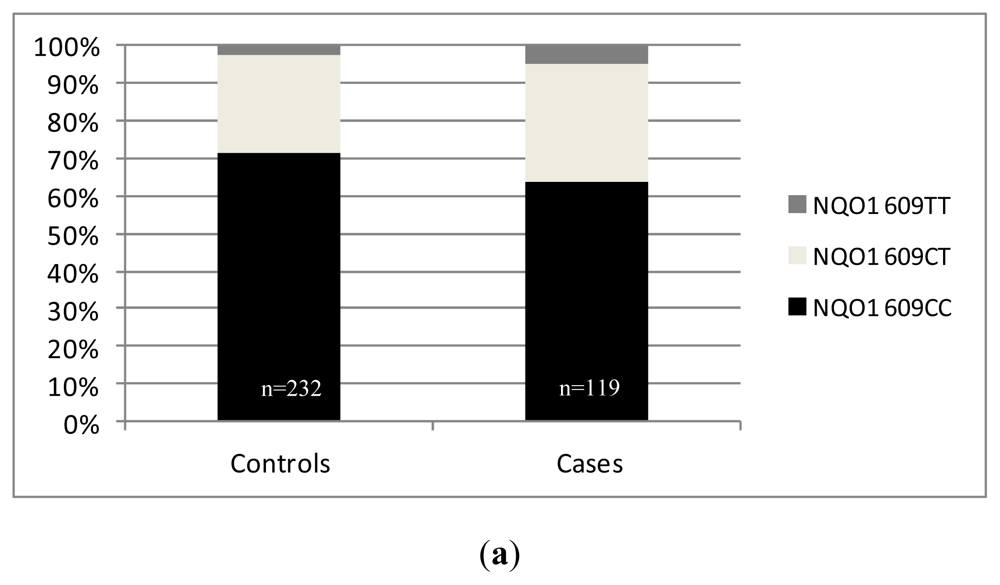
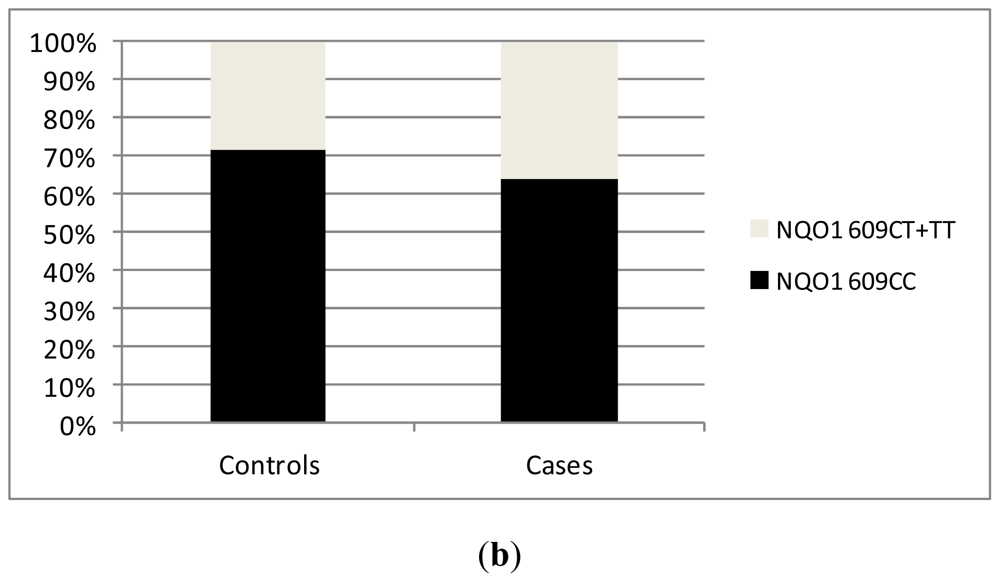
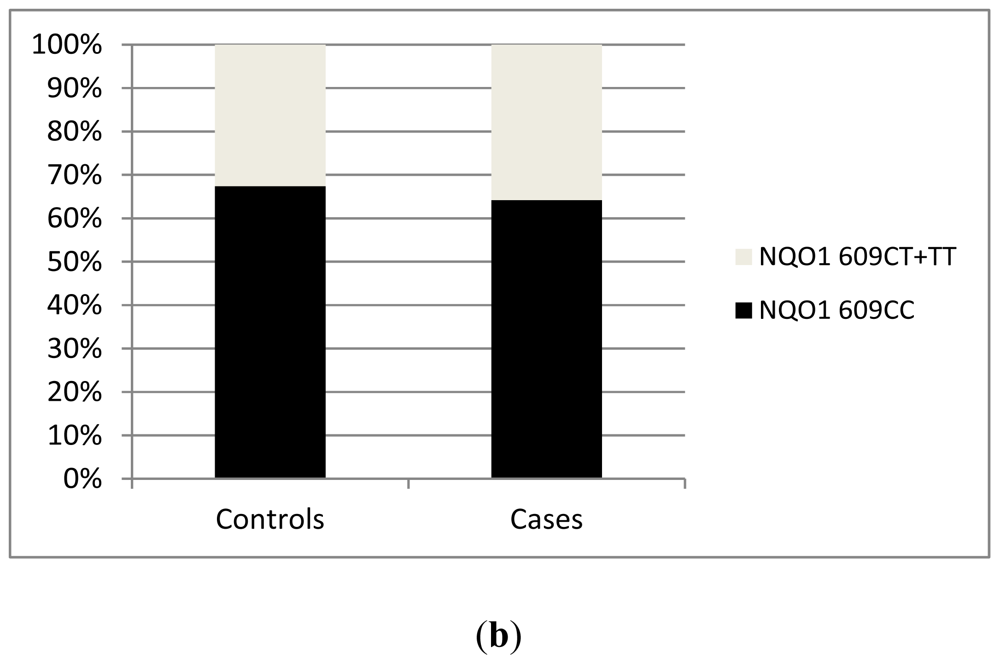
2.2. Results from Our Single Center Analysis
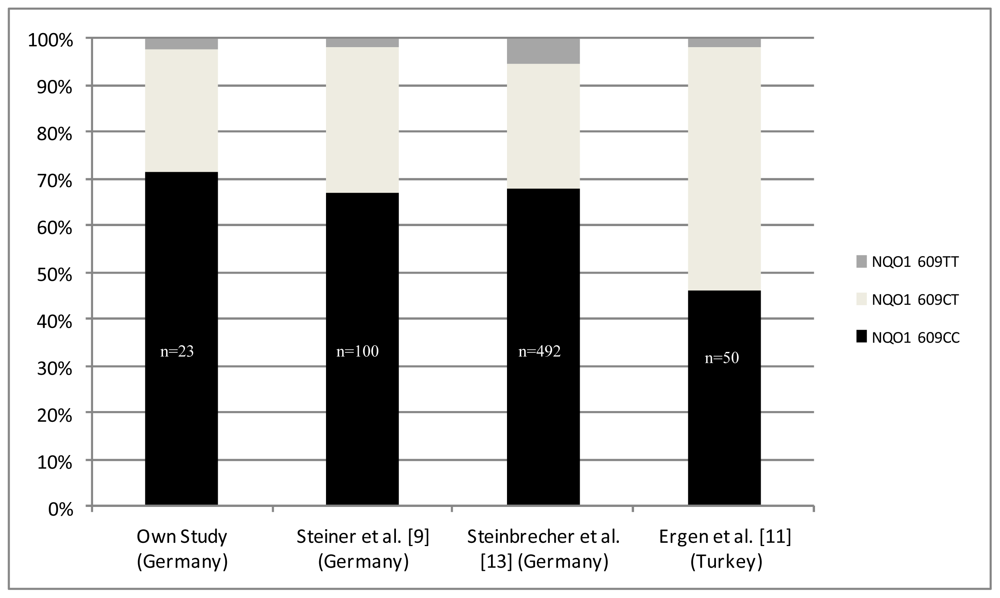
2.3. Results from Combined Data of Published European Studies
3. Discussion
4. Experimental Section
4.1. Samples
4.2. Tissue Microdissection and DNA Isolation
4.3. NQO1 P187S SNP Analysis
4.4. Amplification of NQO1 Variants and RFLP Analysis
4.5. Sequencing Analysis
4.6. Statistical Analysis
5. Conclusions
Acknowledgments
- Conflicts of InterestThe authors declare no conflict of interest.
References
- Ziech, D.; Franco, R.; Pappa, A.; Panayiotidis, M.I. Reactive oxygen species (ROS)-induced genetic and epigenetic alterations in human carcinogenesis. Mutat. Res 2011, 711, 167–173. [Google Scholar]
- Khandrika, L.; Kumar, B.; Koul, S.; Maroni, P.; Koul, H.K. Oxidative stress in prostate cancer. Cancer Lett 2009, 282, 125–136. [Google Scholar]
- Dinkova-Kostova, A.T.; Talaly, P. NAD(P)H: Quinone acceptor oxidoreductase 1 (NQO1), a multifunctional antioxidant enzyme and exceptionally versatile cytoprotector. Arch. Biochem. Biophys 2010, 501, 116–123. [Google Scholar]
- Siegel, D.; Anwar, A.; Winski, S.L.; Kepa, J.K.; Zolman, K.L.; Ross, D. Rapid polyubiquitination and proteasomal degradation of a mutant form of NAD(P)H: Quinone oxidoreductase 1. Mol. Pharmacol 2001, 59, 263–268. [Google Scholar]
- Ross, D.; Kepa, J.K.; Winski, S.L.; Beal, H.D.; Anwar, A.; Siegel, D. NAD(P)H: Quinone oxidoreductase 1 (NQO1): Chemoprotection, bioactivation, gene regulation and genetic polymorphisms. Chem. Biol. Interact 2000, 129, 77–97. [Google Scholar]
- Chen, J.; Lin, Y.; Zhang, R.; Huang, Z.J.; Pan, X.G. Contribution of the NAD(P)H quinone oxidoreductase 1 (NQO1) Pro187Ser polymorphism and the risk of colorectal adenoma and colorectal cancer in Caucasians: A meta-analysis. Arch. Med. Res 2012, 43, 58–66. [Google Scholar]
- Yuan, W.; Xu, L.; Chen, W.; Wang, L.; Fu, Z.; Pang, D.; Li, D. Evidence on the association between NQO1 Pro187Ser polymorphism and breast cancer risk in the current studies: A meta-analysis. Breast Cancer Res. Treat 2011, 125, 467–472. [Google Scholar]
- Chao, C.; Zhang, Z.F.; Berthiller, J.; Boffetta, P.; Hashibe, M. NAD(P)H: Quinone oxidoreductase 1 Pro187Ser polymorphism and the risk of lung, bladder, and colorectal cancer: A meta-analysis. Cancer Epidemiol. Biomark. Prev 2006, 15, 979–987. [Google Scholar]
- Steiner, M.; Hillenbrand, M.; Borkowski, M.; Seiter, H.; Schuff-Werner, P. 609 C→T polymorphism in NAD(P)H: Quinone oxidoreductase gene in patients with prostatic adenocarcinoma or benign prostatic hyperplasia. Cancer Lett 1999, 135, 67–71. [Google Scholar]
- Cunningham, J.M.; Hebbring, S.J.; McDonnell, S.K.; Cicek, M.S.; Christensen, G.B.; Wang, L.; Jacobsen, S.J.; Cerhan, J.R.; Blute, M.L.; Schaid, D.J.; et al. Evaluation of genetic variants in the androgen and estrogen metabolic pathways as risk factors for sporadic and familial prostate cancer. Cancer Epidemiol. Biomark. Prev 2007, 16, 969–978. [Google Scholar]
- Ergen, H.A.; Gormus, U.; Narter, F.; Zeybek, U.; Bulgurcuoglu, S.; Isbir, T. Investigation of NAD(P)H: Quinone oxidoreductase 1 (NQO1) C609T polymorphism in prostate cancer. Anticancer Res 2007, 27, 4107–4110. [Google Scholar]
- Hamajima, N.; Matsuo, K.; Iwata, H.; Shinoda, M.; Yamamura, Y.; Kato, T.; Hatooka, S.; Mitsudomi, T.; Suyama, M.; Kagami, Y.; et al. NAD(P)H: Quinone oxidoreductase 1 (NQO1) C609T polymorphism and the risk of eight cancers for Japanese. Int. J. Clin. Oncol 2002, 7, 103–108. [Google Scholar]
- Steinbrecher, A.; Rohrmann, S.; Timofeeva, M.; Risch, A.; Jansen, E.; Linseisen, J. Dietary glucosinolate intake, polymorphisms in selected biotransformation enzymes, and risk of prostate cancer. Cancer Epidemiol. Biomark. Prev 2010, 19, 135–143. [Google Scholar]
- Crawford, A.; Fassett, R.G.; Geraghty, D.P.; Kunde, D.A.; Ball, M.B.; Robertson, I.K.; Coombes, J.S. Relationships between single nucleotide polymorphisms of antioxidant enzymes and disease. Gene 2012, 501, 89–103. [Google Scholar]
- Martin, F.L.; Patell, I.I.; Sozeri, O.; Singh, P.B.; Ragavan, N.; Nicholson, C.M.; Frei, E.; Meinl, W.; Glatt, H; Phillips, D.H.; et al. Prostate 2010, 70, 1586–1599.
- Thapa, D.; Ghosh, R. Antioxidants for prostate cancer chemoprevention: Challenges and opportunities. Biochem. Pharmacol 2012, 83, 1319–1330. [Google Scholar]
- Dong, Y.; Bey, E.A.; Li, L.S.; Kabbani, W.; Yan, J.; Xie, X.J.; Hsieh, J.T.; Gao, J.; Boothman, D.A. Prostate cancer radiosensitization through Poly(ADP-ribose) polymerase-1 hyperactivation. Cancer Res 2010, 70, 8088–8096. [Google Scholar]
- Saramäki, O.R.; Porkka, K.P.; Vesella, R.L.; Visakorpi, T. Genetic aberrations in prostate cancer by microarray analysis. Int. J. Cancer 2006, 119, 1322–1329. [Google Scholar]
- Planchon, S.M.; Pink, J.J.; Tagliarino, C.; Bornmann, W.G.; Varnes, M.E.; Boothman, D.A. β-lapachone-induced apoptosis in human prostate cancer cells: Involvement of NQO1/xip3. Exp. Cell Res 2001, 267, 95–106. [Google Scholar]
- Kim, M.Y.; Zhang, T.; Kraus, W.L. Poly(ADP-ribosyl)ation by PARP-1: “PAR-laying” NAD+ into a nuclear signal. Genes Dev 2005, 19, 1951–1967. [Google Scholar]
- Thapa, D.; Kumar, A.P.; Gosh, R. Implication of NQO1 knockdown on prostate carcinogenesis. Cancer Res 2012, 72. [Google Scholar] [CrossRef]
- Epstein, J.I.; Algaba, F.; Allsbrook, W.C.; Bastacky, S.; Boccon-Gibod, L.; de Marzo, A.M.; Egevad, L.; Furusato, M.; Hamper, U.M.; Helpap, B.; et al. Tumours of the Prostate. In World Health Organization, Classification of Tumours. Pathology and Genetics. Tumours of the Urinary System and Male Genital Organs, 1st ed; Eble, J.N., Sauter, G., Epstein, J.I., Sesterhenn, I.A., Eds.; IARC: Lyon, France, 2004; pp. 159–216. [Google Scholar]
- Sobin, L.H.; Wittekind, C. TNM Classification of Malignant Tumors; Wiley & Sons: Hoboken, NJ, USA; p. 2002.
- Hardy-Weinberg equilibrium. Available online: http://ihg.gsf.de/cgi-bin/hw/hwa1.pl accessed on April 13, 2012.

| Cases | Controls | |||
|---|---|---|---|---|
| Number | n = 119 | n = 232 | ||
| Age (years) | Median: 66 | Range: 46–74 | Median: 69.5 | Range: 25–94 |
| Mean: 64.3 (± 6.4) | Mean: 67.9 (± 10.2) | |||
| Stage | Organ-confined | n = 59 | ||
| Non-Organ-confined | n = 55 | |||
| No data available | n = 5 | |||
| Gleason Score | Median: 7 | Range: 3–10 | ||
| Gleason Sum | 3–4 | n = 3 | ||
| 5–7 | n = 80 | |||
| 8–10 | n = 28 | |||
| No data available | n = 8 |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Stoehr, C.G.; Nolte, E.; Wach, S.; Wieland, W.F.; Hofstaedter, F.; Hartmann, A.; Stoehr, R. NAD(P)H:Quinone Oxidoreductase 1 (NQO1) P187S Polymorphism and Prostate Cancer Risk in Caucasians. Int. J. Mol. Sci. 2012, 13, 10959-10969. https://doi.org/10.3390/ijms130910959
Stoehr CG, Nolte E, Wach S, Wieland WF, Hofstaedter F, Hartmann A, Stoehr R. NAD(P)H:Quinone Oxidoreductase 1 (NQO1) P187S Polymorphism and Prostate Cancer Risk in Caucasians. International Journal of Molecular Sciences. 2012; 13(9):10959-10969. https://doi.org/10.3390/ijms130910959
Chicago/Turabian StyleStoehr, Christine G., Elke Nolte, Sven Wach, Wolf F. Wieland, Ferdinand Hofstaedter, Arndt Hartmann, and Robert Stoehr. 2012. "NAD(P)H:Quinone Oxidoreductase 1 (NQO1) P187S Polymorphism and Prostate Cancer Risk in Caucasians" International Journal of Molecular Sciences 13, no. 9: 10959-10969. https://doi.org/10.3390/ijms130910959




