The Role of Sox Genes in Lung Morphogenesis and Cancer
Abstract
:1. Introduction
2. Sox Transcriptional Factors
3. Sox Gene and Lung Morphogenesis
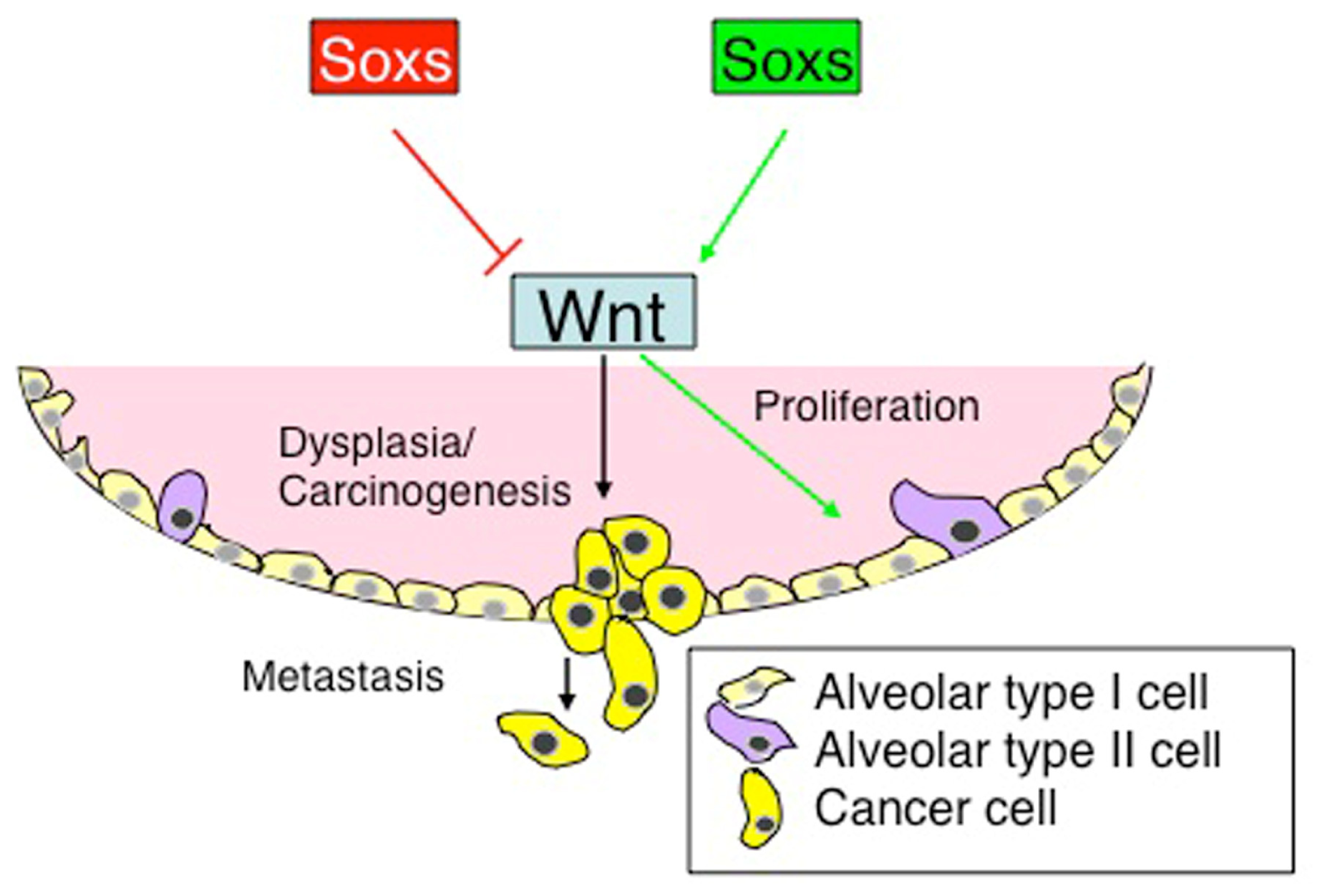
4. Sox Genes and Lung Cancer
4.1. Sox Genes and Cancers
4.2. Oncogenic Potential of Sox Genes in Lung Cancer
5. Sox Gene and Lung Cancer Stem Cells
6. Sox genes for Diagnosis and Therapeutic Targets in Lung Cancer
7. Concluding Remarks
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
Reference
- Jemal, A.; Siegel, R.; Xu, J.; Ward, E. Cancer statistics, 2010. CA Cancer J. Clin 2010, 60, 277–300. [Google Scholar]
- Broers, J.L.; Rot, M.K.; Oostendorp, T.; Huysmans, A.; Wagenaar, S.S.; Wiersma-van Tilburg, A.J.; Vooijs, G.P.; Ramaekers, F.C. Immunocytochemical detection of human lung cancer heterogeneity using antibodies to epithelial, neuronal, and neuroendocrine antigens. Cancer Res 1987, 47, 3225–3234. [Google Scholar]
- Mukherjee, T.K.; Paul, K.; Mukhopadhyay, S. Cell signaling molecules as drug targets in lung cancer: An overview. Curr. Opin. Pulm. Med 2011, 17, 286–291. [Google Scholar]
- Larsen, J.E.; Cascone, T.; Gerber, D.E.; Heymach, J.V.; Minna, J.D. Targeted therapies for lung cancer: Clinical experience and novel agents. Cancer J 2011, 17, 512–527. [Google Scholar]
- Warburton, D.; El-Hashash, A.; Carraro, G.; Tiozzo, C.; Sala, F.; Rogers, O.; De Langhe, S.; Kemp, P.J.; Riccardi, D.; Torday, J.; et al. Lung organogenesis. Curr. Top. Dev. Biol 2010, 90, 73–158. [Google Scholar]
- Dang, X.; Ma, A.; Yang, L.; Hu, H.; Zhu, B.; Shang, D.; Chen, T.; Luo, Y. Microrna-26a regulates tumorigenic properties of ezh2 in human lung carcinoma cells. Cancer Genet 2012, 205, 113–123. [Google Scholar]
- Lee, S.; Kim, Y.; Sun, J.M.; Choi, Y.L.; Kim, J.G.; Shim, Y.M.; Park, Y.H.; Ahn, J.S.; Park, K.; Han, J.H.; et al. Molecular profiles of egfr, k-ras, c-met, and fgfr in pulmonary pleomorphic carcinoma, a rare lung malignancy. J. Cancer Res. Clin. Oncol 2011, 137, 1203–1211. [Google Scholar]
- Chang, Y.L.; Wu, C.T.; Shih, J.Y.; Lee, Y.C. Egfr and p53 status of pulmonary pleomorphic carcinoma: Implications for egfr tyrosine kinase inhibitors therapy of an aggressive lung malignancy. Ann. Surg. Oncol 2011, 18, 2952–2960. [Google Scholar]
- Wen, J.; Fu, J.; Zhang, W.; Guo, M. Genetic and epigenetic changes in lung carcinoma and their clinical implications. Mod. Pathol 2011, 24, 932–943. [Google Scholar]
- Schepers, G.E.; Teasdale, R.D.; Koopman, P. Twenty pairs of Sox: Extent, homology, and nomenclature of the mouse and human Sox transcription factor gene families. Dev. Cell 2002, 3, 167–170. [Google Scholar]
- Kormish, J.D.; Sinner, D.; Zorn, A.M. Interactions between Sox factors and wnt/beta-catenin signaling in development and disease. Dev. Dyn 2010, 239, 56–68. [Google Scholar]
- Bowles, J.; Schepers, G.; Koopman, P. Phylogeny of the Sox family of developmental transcription factors based on sequence and structural indicators. Dev. Biol 2000, 227, 239–255. [Google Scholar]
- Mertin, S.; McDowall, S.G.; Harley, V.R. The DNA-binding specificity of Sox9 and other Sox proteins. Nucleic Acids Res 1999, 27, 1359–1364. [Google Scholar]
- Van Beest, M.; Dooijes, D.; van De Wetering, M.; Kjaerulff, S.; Bonvin, A.; Nielsen, O.; Clevers, H. Sequence-specific high mobility group box factors recognize 10–12-base pair minor groove motifs. J. Biol. Chem 2000, 275, 27266–27273. [Google Scholar]
- Harley, V.R.; Lovell-Badge, R.; Goodfellow, P.N. Definition of a consensus DNA binding site for sry. Nucleic Acids Res 1994, 22, 1500–1501. [Google Scholar]
- Tanaka, S.; Kamachi, Y.; Tanouchi, A.; Hamada, H.; Jing, N.; Kondoh, H. Interplay of Sox and pou factors in regulation of the nestin gene in neural primordial cells. Mol. Cell. Biol 2004, 24, 8834–8846. [Google Scholar]
- Wissmuller, S.; Kosian, T.; Wolf, M.; Finzsch, M.; Wegner, M. The high-mobility-group domain of Sox proteins interacts with DNA-binding domains of many transcription factors. Nucleic Acids Res 2006, 34, 1735–1744. [Google Scholar]
- Chen, Q.L.; Zheng, W.L.; Yao, W.J.; Nie, L.W.; Cheng, S.H.; Ma, W.L. Analysis of Sox4 gene mutation in non-small cell lung cancer tissues. Zhonghua Yi Xue Yi Chuan Xue Za Zhi 2007, 24, 505–509. [Google Scholar]
- Castillo, S.D.; Sanchez-Cespedes, M. The Sox family of genes in cancer development: Biological relevance and opportunities for therapy. Expert Opin. Ther. Targets 2012, 16, 903–919. [Google Scholar]
- Dong, C.; Wilhelm, D.; Koopman, P. Sox genes and cancer. Cytogen. Genome Res 2004, 105, 442–447. [Google Scholar]
- Melichar, H.J.; Narayan, K.; Der, S.D.; Hiraoka, Y.; Gardiol, N.; Jeannet, G.; Held, W.; Chambers, C.A.; Kang, J. Regulation of gammadelta versus alphabeta T lymphocyte differentiation by the transcription factor Sox13. Science 2007, 315, 230–233. [Google Scholar]
- Fernandez-Lloris, R.; Osses, N.; Jaffray, E.; Shen, L.N.; Vaughan, O.A.; Girwood, D.; Bartrons, R.; Rosa, J.L.; Hay, R.T.; Ventura, F. Repression of Sox6 transcriptional activity by sumo modification. FEBS Lett 2006, 580, 1215–1221. [Google Scholar]
- Pan, X.; Li, H.; Zhang, P.; Jin, B.; Man, J.; Tian, L.; Su, G.; Zhao, J.; Li, W.; Liu, H.; et al. Ubc9 interacts with Sox4 and represses its transcriptional activity. Biochem. Biophys. Res. Commun 2006, 344, 727–734. [Google Scholar]
- Kaplan, F. Molecular determinants of fetal lung organogenesis. Mol. Genet. Metab 2000, 71, 321–341. [Google Scholar]
- Lange, A.W.; Keiser, A.R.; Wells, J.M.; Zorn, A.M.; Whitsett, J.A. Sox17 promotes cell cycle progression and inhibits tgf-beta/smad3 signaling to initiate progenitor cell behavior in the respiratory epithelium. PLoS One 2009, 4, e5711. [Google Scholar]
- Park, J.; Zhang, J.J.; Moro, A.; Kushida, M.; Wegner, M.; Kim, P.C. Regulation of Sox9 by sonic hedgehog (shh) is essential for patterning and formation of tracheal cartilage. Dev. Dyn 2010, 239, 514–526. [Google Scholar]
- Park, K.S.; Wells, J.M.; Zorn, A.M.; Wert, S.E.; Whitsett, J.A. Sox17 influences the differentiation of respiratory epithelial cells. Dev. Biol 2006, 294, 192–202. [Google Scholar]
- Tompkins, D.H.; Besnard, V.; Lange, A.W.; Keiser, A.R.; Wert, S.E.; Bruno, M.D.; Whitsett, J.A. Sox2 activates cell proliferation and differentiation in the respiratory epithelium. Am. J. Respir. Cell Mol. Biol 2011, 45, 101–110. [Google Scholar]
- Sock, E.; Rettig, S.D.; Enderich, J.; Bosl, M.R.; Tamm, E.R.; Wegner, M. Gene targeting reveals a widespread role for the high-mobility-group transcription factor Sox11 in tissue remodeling. Mol. Cell. Biol 2004, 24, 6635–6644. [Google Scholar]
- Gontan, C.; de Munck, A.; Vermeij, M.; Grosveld, F.; Tibboel, D.; Rottier, R. Sox2 is important for two crucial processes in lung development: Branching morphogenesis and epithelial cell differentiation. Dev. Biol 2008, 317, 296–309. [Google Scholar]
- Tompkins, D.H.; Besnard, V.; Lange, A.W.; Wert, S.E.; Keiser, A.R.; Smith, A.N.; Lang, R.; Whitsett, J.A. Sox2 is required for maintenance and differentiation of bronchiolar clara, ciliated, and goblet cells. PLoS One 2009, 4, e8248. [Google Scholar]
- Que, J.; Luo, X.; Schwartz, R.J.; Hogan, B.L. Multiple roles for Sox2 in the developing and adult mouse trachea. Development 2009, 136, 1899–1907. [Google Scholar]
- Elluru, R.G.; Thompson, F.; Reece, A. Fibroblast growth factor 18 gives growth and directional cues to airway cartilage. Laryngoscope 2009, 119, 1153–1165. [Google Scholar]
- Perl, A.K.; Kist, R.; Shan, Z.; Scherer, G.; Whitsett, J.A. Normal lung development and function after Sox9 inactivation in the respiratory epithelium. Genesis 2005, 41, 23–32. [Google Scholar]
- Park, K.S.; Wells, J.M.; Zorn, A.M.; Wert, S.E.; Laubach, V.E.; Fernandez, L.G.; Whitsett, J.A. Transdifferentiation of ciliated cells during repair of the respiratory epithelium. Am. J. Respir. Cell Mol. Biol 2006, 34, 151–157. [Google Scholar]
- Yin, D.; Jia, Y.; Yu, Y.; Brock, M.V.; Herman, J.G.; Han, C.; Su, X.; Liu, Y.; Guo, M. Sox17 methylation inhibits its antagonism of wnt signaling pathway in lung cancer. Discovery Med 2012, 14, 33–40. [Google Scholar]
- Seguin, C.A.; Draper, J.S.; Nagy, A.; Rossant, J. Establishment of endoderm progenitors by Sox transcription factor expression in human embryonic stem cells. Cell Stem Cell 2008, 3, 182–195. [Google Scholar]
- Kim, I.; Saunders, T.L.; Morrison, S.J. Sox17 dependence distinguishes the transcriptional regulation of fetal from adult hematopoietic stem cells. Cell 2007, 130, 470–483. [Google Scholar]
- Matsui, T.; Kanai-Azuma, M.; Hara, K.; Matoba, S.; Hiramatsu, R.; Kawakami, H.; Kurohmaru, M.; Koopman, P.; Kanai, Y. Redundant roles of Sox17 and Sox18 in postnatal angiogenesis in mice. J. Cell Sci 2006, 119, 3513–3526. [Google Scholar]
- Sakamoto, Y.; Hara, K.; Kanai-Azuma, M.; Matsui, T.; Miura, Y.; Tsunekawa, N.; Kurohmaru, M.; Saijoh, Y.; Koopman, P.; Kanai, Y. Redundant roles of Sox17 and Sox18 in early cardiovascular development of mouse embryos. Biochem. Biophys. Res. Commun 2007, 360, 539–544. [Google Scholar]
- Otsubo, T.; Akiyama, Y.; Yanagihara, K.; Yuasa, Y. Sox2 is frequently downregulated in gastric cancers and inhibits cell growth through cell-cycle arrest and apoptosis. Br. J. Cancer 2008, 98, 824–831. [Google Scholar]
- Sun, M.; Uozaki, H.; Hino, R.; Kunita, A.; Shinozaki, A.; Ushiku, T.; Hibiya, T.; Takeshita, K.; Isogai, M.; Takada, K.; et al. Sox9 expression and its methylation status in gastric cancer. Virchows Arch 2012, 460, 271–279. [Google Scholar]
- Cox, J.L.; Wilder, P.J.; Desler, M.; Rizzino, A. Elevating Sox2 levels deleteriously affects the growth of medulloblastoma and glioblastoma cells. PLoS One 2012, 7, e44087. [Google Scholar]
- Swartling, F.J.; Savov, V.; Persson, A.I.; Chen, J.; Hackett, C.S.; Northcott, P.A.; Grimmer, M.R.; Lau, J.; Chesler, L.; Perry, A.; et al. Distinct neural stem cell populations give rise to disparate brain tumors in response to n-myc. Cancer Cell 2012, 21, 601–613. [Google Scholar]
- Burgos-Ojeda, D.; Rueda, B.R.; Buckanovich, R.J. Ovarian cancer stem cell markers: Prognostic and therapeutic implications. Cancer Lett 2012, 322, 1–7. [Google Scholar]
- Jiang, S.S.; Fang, W.T.; Hou, Y.H.; Huang, S.F.; Yen, B.L.; Chang, J.L.; Li, S.M.; Liu, H.P.; Liu, Y.L.; Huang, C.T.; et al. Upregulation of Sox9 in lung adenocarcinoma and its involvement in the regulation of cell growth and tumorigenicity. Clin. Cancer Res 2010, 16, 4363–4373. [Google Scholar]
- Li, B.; Ge, Z.; Song, S.; Zhang, S.; Yan, H.; Huang, B.; Zhang, Y. Decreased expression of Sox7 is correlated with poor prognosis in lung adenocarcinoma patients. Pathol. Oncol. Res 2012, 18, 1039–1045. [Google Scholar]
- Sholl, L.M.; Barletta, J.A.; Yeap, B.Y.; Chirieac, L.R.; Hornick, J.L. Sox2 protein expression is an independent poor prognostic indicator in stage i lung adenocarcinoma. Am. J. Surg. Pathol 2010, 34, 1193–1198. [Google Scholar]
- Yousem, S.A. Role of molecular studies in the diagnosis of lung adenocarcinoma. Mod. Pathol 2012, 25, S11–S17. [Google Scholar]
- Guo, X.; Xiong, L.; Sun, T.; Peng, R.; Zou, L.; Zhu, H.; Zhang, J.; Li, H.; Zhao, J. Expression features of Sox9 associate with tumor progression and poor prognosis of hepatocellular carcinoma. Diagn. Pathol 2012, 7, 44. [Google Scholar]
- Chen, Y.; Shi, L.; Zhang, L.; Li, R.; Liang, J.; Yu, W.; Sun, L.; Yang, X.; Wang, Y.; Zhang, Y.; et al. The molecular mechanism governing the oncogenic potential of Sox2 in breast cancer. J Biol. Chem 2008, 283, 17969–17978. [Google Scholar]
- Fu, D.Y.; Wang, Z.M.; Li, C.; Wang, B.L.; Shen, Z.Z.; Huang, W.; Shao, Z.M. Sox17, the canonical wnt antagonist, is epigenetically inactivated by promoter methylation in human breast cancer. Breast Cancer Res. Treat 2009, 119, 601–612. [Google Scholar]
- Acloque, H.; Ocana, O.H.; Matheu, A.; Rizzoti, K.; Wise, C.; Lovell-Badge, R.; Nieto, M.A. Reciprocal repression between Sox3 and snail transcription factors defines embryonic territories at gastrulation. Dev. Cell 2011, 21, 546–558. [Google Scholar]
- Passeron, T.; Valencia, J.C.; Namiki, T.; Vieira, W.D.; Passeron, H.; Miyamura, Y.; Hearing, V.J. Upregulation of Sox9 inhibits the growth of human and mouse melanomas and restores their sensitivity to retinoic acid. J. Clin. Invest 2009, 119, 954–963. [Google Scholar]
- Liu, P.; Ramachandran, S.; Ali Seyed, M.; Scharer, C.D.; Laycock, N.; Dalton, W.B.; Williams, H.; Karanam, S.; Datta, M.W.; Jaye, D.L.; et al. Sex-determining region y box 4 is a transforming oncogene in human prostate cancer cells. Cancer Res 2006, 66, 4011–4019. [Google Scholar]
- Scharer, C.D.; McCabe, C.D.; Ali-Seyed, M.; Berger, M.F.; Bulyk, M.L.; Moreno, C.S. Genome-wide promoter analysis of the Sox4 transcriptional network in prostate cancer cells. Cancer Res 2009, 69, 709–717. [Google Scholar]
- Wang, H.; McKnight, N.C.; Zhang, T.; Lu, M.L.; Balk, S.P.; Yuan, X. Sox9 is expressed in normal prostate basal cells and regulates androgen receptor expression in prostate cancer cells. Cancer Res 2007, 67, 528–536. [Google Scholar]
- Zhong, W.D.; Qin, G.Q.; Dai, Q.S.; Han, Z.D.; Ling, X.H.; Fu, X.; Cai, C.; Chen, J.H.; Chen, X.B.; Lin, Z.Y.; et al. Soxs in human prostate cancer: Implication as progression and prognosis factors. BMC Cancer 2012, 12, 248. [Google Scholar]
- Sinner, D.; Kordich, J.J.; Spence, J.R.; Opoka, R.; Rankin, S.; Lin, S.C.; Jonatan, D.; Zorn, A.M.; Wells, J.M. Sox17 and Sox4 differentially regulate beta-catenin/T-cell factor activity and proliferation of colon carcinoma cells. Mol. Cell. Biol 2007, 27, 7802–7815. [Google Scholar]
- Azhikina, T.; Kozlova, A.; Skvortsov, T.; Sverdlov, E. Heterogeneity and degree of timp4, gata4, Sox18, and egfl7 gene promoter methylation in non-small cell lung cancer and surrounding tissues. Cancer Genet 2011, 204, 492–500. [Google Scholar]
- Castillo, S.D.; Matheu, A.; Mariani, N.; Carretero, J.; Lopez-Rios, F.; Lovell-Badge, R.; Sanchez-Cespedes, M. Novel transcriptional targets of the sry-HMG box transcription factor Sox4 link its expression to the development of small cell lung cancer. Cancer Res 2012, 72, 176–186. [Google Scholar]
- Chen, S.; Xu, Y.; Chen, Y.; Li, X.; Mou, W.; Wang, L.; Liu, Y.; Reisfeld, R.A.; Xiang, R.; Lv, D.; et al. Sox2 gene regulates the transcriptional network of oncogenes and affects tumorigenesis of human lung cancer cells. PLoS One 2012, 7, e36326. [Google Scholar]
- Li, X.; Wang, J.; Xu, Z.; Ahmad, A.; Li, E.; Wang, Y.; Qin, S.; Wang, Q. Expression of Sox2 and Oct4 and their clinical significance in human non-small-cell lung cancer. Int. J. Mol. Sci 2012, 13, 7663–7675. [Google Scholar]
- Maddison, P.; Thorpe, A.; Silcocks, P.; Robertson, J.F.; Chapman, C.J. Autoimmunity to Sox2, clinical phenotype and survival in patients with small-cell lung cancer. Lung Cancer 2010, 70, 335–339. [Google Scholar]
- Medina, P.P.; Castillo, S.D.; Blanco, S.; Sanz-Garcia, M.; Largo, C.; Alvarez, S.; Yokota, J.; Gonzalez-Neira, A.; Benitez, J.; Clevers, H.C.; et al. The sry-HMG box gene, Sox4, is a target of gene amplification at chromosome 6p in lung cancer. Hum. Mol. Genet 2009, 18, 1343–1352. [Google Scholar]
- Rudin, C.M.; Durinck, S.; Stawiski, E.W.; Poirier, J.T.; Modrusan, Z.; Shames, D.S.; Bergbower, E.A.; Guan, Y.; Shin, J.; Guillory, J.; et al. Comprehensive genomic analysis identifies Sox2 as a frequently amplified gene in small-cell lung cancer. Nat. Genet 2012, 44, 1111–1116. [Google Scholar]
- Titulaer, M.J.; Klooster, R.; Potman, M.; Sabater, L.; Graus, F.; Hegeman, I.M.; Thijssen, P.E.; Wirtz, P.W.; Twijnstra, A.; Smitt, P.A.; et al. Sox antibodies in small-cell lung cancer and lambert-eaton myasthenic syndrome: Frequency and relation with survival. J. Clin. Oncol 2009, 27, 4260–4267. [Google Scholar]
- Wilbertz, T.; Wagner, P.; Petersen, K.; Stiedl, A.C.; Scheble, V.J.; Maier, S.; Reischl, M.; Mikut, R.; Altorki, N.K.; Moch, H.; et al. Sox2 gene amplification and protein overexpression are associated with better outcome in squamous cell lung cancer. Mod. Pathol 2011, 24, 944–953. [Google Scholar]
- Xiang, R.; Liao, D.; Cheng, T.; Zhou, H.; Shi, Q.; Chuang, T.S.; Markowitz, D.; Reisfeld, R.A.; Luo, Y. Downregulation of transcription factor Sox2 in cancer stem cells suppresses growth and metastasis of lung cancer. Br. J. Cancer 2011, 104, 1410–1417. [Google Scholar]
- Wang, L.; He, S.; Yuan, J.; Mao, X.; Cao, Y.; Zong, J.; Tu, Y.; Zhang, Y. Oncogenic role of Sox9 expression in human malignant glioma. Med. Oncol. 2012. [Google Scholar] [CrossRef]
- Yan, H.T.; Shinka, T.; Sato, Y.; Yang, X.J.; Chen, G.; Sakamoto, K.; Kinoshita, K.; Aburatani, H.; Nakahori, Y. Overexpression of Sox15 inhibits proliferation of nt2/d1 cells derived from a testicular embryonal cell carcinoma. Mol. Cells 2007, 24, 323–328. [Google Scholar]
- Aleman, A.; Adrien, L.; Lopez-Serra, L.; Cordon-Cardo, C.; Esteller, M.; Belbin, T.J.; Sanchez-Carbayo, M. Identification of DNA hypermethylation of Sox9 in association with bladder cancer progression using cpg microarrays. Br. J. Cancer 2008, 98, 466–473. [Google Scholar]
- Matheu, A.; Collado, M.; Wise, C.; Manterola, L.; Cekaite, L.; Tye, A.J.; Canamero, M.; Bujanda, L.; Schedl, A.; Cheah, K.S.; et al. Oncogenicity of the developmental transcription factor Sox9. Cancer Res 2012, 72, 1301–1315. [Google Scholar]
- Alonso, M.M.; Diez-Valle, R.; Manterola, L.; Rubio, A.; Liu, D.; Cortes-Santiago, N.; Urquiza, L.; Jauregi, P.; Lopez de Munain, A.; Sampron, N.; et al. Genetic and epigenetic modifications of Sox2 contribute to the invasive phenotype of malignant gliomas. PLoS One 2011, 6, e26740. [Google Scholar]
- Cai, Y.R.; Zhang, H.Q.; Qu, Y.; Mu, J.; Zhao, D.; Zhou, L.J.; Yan, H.; Ye, J.W.; Liu, Y. Expression of met and Sox2 genes in non-small cell lung carcinoma with egfr mutation. Oncol. Rep 2011, 26, 877–885. [Google Scholar]
- Yuan, P.; Kadara, H.; Behrens, C.; Tang, X.; Woods, D.; Solis, L.M.; Huang, J.; Spinola, M.; Dong, W.; Yin, G.; et al. Sex determining region y-box 2 (Sox2) is a potential cell-lineage gene highly expressed in the pathogenesis of squamous cell carcinomas of the lung. PLoS One 2010, 5, e9112. [Google Scholar]
- Sabater, L.; Titulaer, M.; Saiz, A.; Verschuuren, J.; Gure, A.O.; Graus, F. Sox1 antibodies are markers of paraneoplastic lambert-eaton myasthenic syndrome. Neurology 2008, 70, 924–928. [Google Scholar]
- Gure, A.O.; Stockert, E.; Scanlan, M.J.; Keresztes, R.S.; Jager, D.; Altorki, N.K.; Old, L.J.; Chen, Y.T. Serological identification of embryonic neural proteins as highly immunogenic tumor antigens in small cell lung cancer. Proc. Natl. Acad. Sci. USA 2000, 97, 4198–4203. [Google Scholar]
- Vural, B.; Chen, L.C.; Saip, P.; Chen, Y.T.; Ustuner, Z.; Gonen, M.; Simpson, A.J.; Old, L.J.; Ozbek, U.; Gure, A.O. Frequency of Sox group b (Sox1, 2, 3) and zic2 antibodies in turkish patients with small cell lung carcinoma and their correlation with clinical parameters. Cancer 2005, 103, 2575–2583. [Google Scholar]
- Lu, Y.; Futtner, C.; Rock, J.R.; Xu, X.; Whitworth, W.; Hogan, B.L.; Onaitis, M.W. Evidence that Sox2 overexpression is oncogenic in the lung. PLoS One 2010, 5, e11022. [Google Scholar]
- Friedman, R.S.; Bangur, C.S.; Zasloff, E.J.; Fan, L.; Wang, T.; Watanabe, Y.; Kalos, M. Molecular and immunological evaluation of the transcription factor Sox4 as a lung tumor vaccine antigen. J. Immunol 2004, 172, 3319–3327. [Google Scholar]
- D’Angelo, S.P.; Pietanza, M.C. The molecular pathogenesis of small cell lung cancer. Cancer Biol. Ther 2010, 10, 1–10. [Google Scholar]
- Sholl, L.M.; Long, K.B.; Hornick, J.L. Sox2 expression in pulmonary non-small cell and neuroendocrine carcinomas. Appl. Immunohistochem. Mol. Morphol 2010, 18, 55–61. [Google Scholar]
- Bass, A.J.; Watanabe, H.; Mermel, C.H.; Yu, S.; Perner, S.; Verhaak, R.G.; Kim, S.Y.; Wardwell, L.; Tamayo, P.; Gat-Viks, I.; et al. Sox2 is an amplified lineage-survival oncogene in lung and esophageal squamous cell carcinomas. Nat. Genet 2009, 41, 1238–1242. [Google Scholar]
- Aaboe, M.; Birkenkamp-Demtroder, K.; Wiuf, C.; Sorensen, F.B.; Thykjaer, T.; Sauter, G.; Jensen, K.M.; Dyrskjot, L.; Orntoft, T. Sox4 expression in bladder carcinoma: Clinical aspects and in vitro functional characterization. Cancer Res 2006, 66, 3434–3442. [Google Scholar]
- Beer, D.G.; Kardia, S.L.; Huang, C.C.; Giordano, T.J.; Levin, A.M.; Misek, D.E.; Lin, L.; Chen, G.; Gharib, T.G.; Thomas, D.G.; et al. Gene-expression profiles predict survival of patients with lung adenocarcinoma. Nat. Med 2002, 8, 816–824. [Google Scholar]
- Tavazoie, S.F.; Alarcon, C.; Oskarsson, T.; Padua, D.; Wang, Q.; Bos, P.D.; Gerald, W.L.; Massague, J. Endogenous human micrornas that suppress breast cancer metastasis. Nature 2008, 451, 147–152. [Google Scholar]
- Pramoonjago, P.; Baras, A.S.; Moskaluk, C.A. Knockdown of Sox4 expression by rnai induces apoptosis in acc3 cells. Oncogene 2006, 25, 5626–5639. [Google Scholar]
- Dammann, R.; Strunnikova, M.; Schagdarsurengin, U.; Rastetter, M.; Papritz, M.; Hattenhorst, U.E.; Hofmann, H.S.; Silber, R.E.; Burdach, S.; Hansen, G. Cpg island methylation and expression of tumour-associated genes in lung carcinoma. Eur. J. Cancer 2005, 41, 1223–1236. [Google Scholar]
- Fan, R.; Zhang, L.Y.; Wang, H.; Yang, B.; Han, T.; Zhao, X.L.; Wang, W.; Wang, X.Q.; Lin, G.W. Methylation of the cpg island near Sox7 gene promoter is correlated with the poor prognosis of patients with myelodysplastic syndrome. Tohoku J. Exp. Med 2012, 227, 119–128. [Google Scholar]
- Peacock, C.D.; Watkins, D.N. Cancer stem cells and the ontogeny of lung cancer. J. Clin. Oncol 2008, 26, 2883–2889. [Google Scholar]
- Xu, Y.; Hu, Y.D. Lung cancer stem cells research. Clues from ontogeny. Saudi Med. J 2009, 30, 1381–1389. [Google Scholar]
- Asselin-Labat, M.L.; Filby, C.E. Adult lung stem cells and their contribution to lung tumourigenesis. Open Biol 2012, 2, 120094. [Google Scholar]
- Kim, C.F.; Jackson, E.L.; Woolfenden, A.E.; Lawrence, S.; Babar, I.; Vogel, S.; Crowley, D.; Bronson, R.T.; Jacks, T. Identification of bronchioalveolar stem cells in normal lung and lung cancer. Cell 2005, 121, 823–835. [Google Scholar]
- Park, I.H.; Zhao, R.; West, J.A.; Yabuuchi, A.; Huo, H.; Ince, T.A.; Lerou, P.H.; Lensch, M.W.; Daley, G.Q. Reprogramming of human somatic cells to pluripotency with defined factors. Nature 2008, 451, 141–146. [Google Scholar]
- Miyoshi, N.; Ishii, H.; Nagai, K.; Hoshino, H.; Mimori, K.; Tanaka, F.; Nagano, H.; Sekimoto, M.; Doki, Y.; Mori, M. Defined factors induce reprogramming of gastrointestinal cancer cells. Proc. Natl. Acad. Sci. USA 2010, 107, 40–45. [Google Scholar]
- Ikushima, H.; Todo, T.; Ino, Y.; Takahashi, M.; Miyazawa, K.; Miyazono, K. Autocrine TGF-β signaling maintains tumorigenicity of glioma-initiating cells through sry-related hmg-box factors. Cell Stem Cell 2009, 5, 504–514. [Google Scholar]
- Neumann, J.; Bahr, F.; Horst, D.; Kriegl, L.; Engel, J.; Luque, R.M.; Gerhard, M.; Kirchner, T.; Jung, A. Sox2 expression correlates with lymph-node metastases and distant spread in right-sided colon cancer. BMC Cancer 2011, 11, 518. [Google Scholar]
- Giangreco, A.; Lu, L.; Vickers, C.; Teixeira, V.H.; Groot, K.R.; Butler, C.R.; Ilieva, E.V.; George, P.J.; Nicholson, A.G.; Sage, E.K.; et al. Beta-catenin determines upper airway progenitor cell fate and preinvasive squamous lung cancer progression by modulating epithelial-mesenchymal transition. J. Pathol 2012, 226, 575–587. [Google Scholar]
- Clevers, H. Wnt/β-catenin signaling in development and disease. Cell 2006, 127, 469–480. [Google Scholar]
- Logan, C.Y.; Nusse, R. The wnt signaling pathway in development and disease. Annu. Rev. Cell Dev. Biol 2004, 20, 781–810. [Google Scholar]
- Pacheco-Pinedo, E.C.; Morrisey, E.E. Wnt and Kras signaling-dark siblings in lung cancer. Oncotarget 2011, 2, 569–574. [Google Scholar]
- Pacheco-Pinedo, E.C.; Durham, A.C.; Stewart, K.M.; Goss, A.M.; Lu, M.M.; Demayo, F.J.; Morrisey, E.E. Wnt/β-catenin signaling accelerates mouse lung tumorigenesis by imposing an embryonic distal progenitor phenotype on lung epithelium. J. Clin. Invest 2011, 121, 1935–1945. [Google Scholar]
- Tsuta, K.; Tanabe, Y.; Yoshida, A.; Takahashi, F.; Maeshima, A.M.; Asamura, H.; Tsuda, H. Utility of 10 immunohistochemical markers including novel markers (desmocollin-3, glypican 3, s100a2, s100a7, and Sox2) for differential diagnosis of squamous cell carcinoma from adenocarcinoma of the lung. J. Thorac. Oncol 2011, 6, 1190–1199. [Google Scholar]
| Sox genes | Evidence or role in lung cancer |
|---|---|
| Sox1 | Autoantibody to Sox1 detected in the serum of patients with SCLC [67,77–79]. |
| Sox2 | Autoimmunity found in patients with SCLC [64,67,78,79]; strongly expressed in both of NSCLC and SCLC tumor cells determined by IHC and/or FISH assays [62,63,66,76]; overexpression leads to lung cancer with poor prognostic outcome [48,80]; gene mutation(s) observed in SCLC [66]; inhibition of expression in cancer stem cells suppress the growth and metastasis of lung cancer [69]. |
| Sox3 | Autoantibody detected in patients with SCLC [67,78,79]; gene mutation(s) observed in SCLC [66]. |
| Sox4 | Autoantibody detected in the serum of patients with SCLC [81]; strong expression detected in SCLC tumor cells by IHC [61]; gene mutations found in SCLC and might be correlated with the lung cancerogenesis and tumor metastasis [18,65,66,82]. |
| Sox5 | Gene mutation (s) determined in SCLC [66] |
| Sox6 | Gene mutation (s) observed in SCLC [66] |
| Sox7 | Downregulation was correlated with a poor prognosis in patients with lung AC [47]. |
| Sox9 | Overexpression promoted lung adenocarcinoma cell proliferation [46]; gene mutation(s) detected in SCLC [66]. |
| Sox11 | Gene mutation (s) found in SCLC [66] |
| Sox14 | Gene mutation (s) observed in SCLC [66] |
| Sox17 | Gene mutation (s) detected in SCLC [66] |
| Sox18 | Heterogeneous methylation was found in the promoter of gene [60]. |
| Sox21 | Autoantibody detected in the serum of patients with SCLC [67,78,79] |
| Sox gene | Abnormality or mutation | Potential clinical implication | Reference |
|---|---|---|---|
| Sox2 | Gene amplification | Associated with the prognosis of SCLC and lung SCC | [61,66,68,74,84] |
| Gene methylation | Associated with the progression of malignant gliobastoma and gastric cancer | [41,74] | |
| Sox3 | N161K mutation | Associated with the progression of SCLC | [66] |
| Sox4 | Point mutations | Correlated with the stages of NSCLC | [18] |
| Gene amplification | Associated with the prognosis of SCLC and NSCLC | [61,65] | |
| S395X mutation | Increase the ability of transform in cell line | [65] | |
| D398V mutation | Found in SCLC samples | [66] | |
| Sox5 | V316F, R547L mutations | Found in SCLC samples | [66] |
| Sox6 | D494Y, G728V mutations | Found in SCLC samples | [66] |
| Sox7 | Methylation of CpG islands near the gene promoter | Correlated with the poor prognosis of patients with myelodysplastic syndrome | [90] |
| Sox9 | S64R mutation | Found in SCLC samples | [66] |
| CpG methylation of promoter gene | Associated with progression of bladder cancer and gastric cancer | [42,72] | |
| Sox11 | Gene amplification | Associated with the prognosis of SCLC | [61] |
| E101 * nonsense change | Found in SCLC samples | [66] | |
| Sox14 | M240I mutation | Found in SCLC samples | [66] |
| Sox18 | Promoter gene CpG methylation | Found in NSCLC tissues | [60] |
© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhu, Y.; Li, Y.; Jun Wei, J.W.; Liu, X. The Role of Sox Genes in Lung Morphogenesis and Cancer. Int. J. Mol. Sci. 2012, 13, 15767-15783. https://doi.org/10.3390/ijms131215767
Zhu Y, Li Y, Jun Wei JW, Liu X. The Role of Sox Genes in Lung Morphogenesis and Cancer. International Journal of Molecular Sciences. 2012; 13(12):15767-15783. https://doi.org/10.3390/ijms131215767
Chicago/Turabian StyleZhu, Yongzhao, Yong Li, Jun Wei Jun Wei, and Xiaoming Liu. 2012. "The Role of Sox Genes in Lung Morphogenesis and Cancer" International Journal of Molecular Sciences 13, no. 12: 15767-15783. https://doi.org/10.3390/ijms131215767




