Identification of Compounds in the Essential Oil of Nutmeg Seeds (Myristica fragrans Houtt.) That Inhibit Locomotor Activity in Mice
Abstract
:1. Introduction
2. Results and Discussion
2.1. Composition of Essential Oils
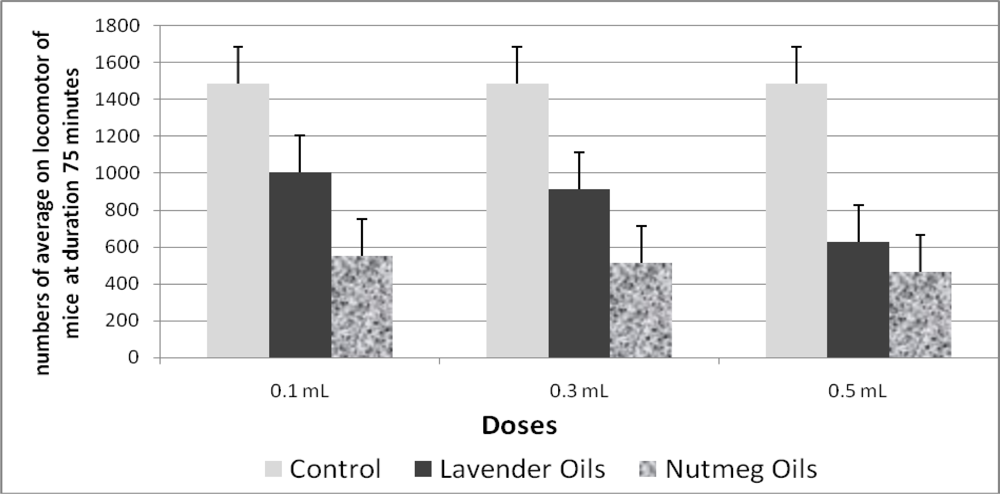
2.2. Locomotor Activity of Mice after Inhalation of Nutmeg Oil
2.3. Active Volatile Compounds
2.4. Locomotor Activity of Single Compounds Identified in Mice
3. Experimental Section
3.1. Materials
3.2. Methods
3.2.1. Isolation of Essential Oil
3.2.2. Mouse Locomotor Activity Tests
3.2.3. GC/MS Analysis
3.2.4. Qualitative Analysis
3.2.5. Quantitative Analysis
| C | concentration (g/g); |
| IS | GC peak area of Internal Standard |
| A | GC peak area of compounds of essential oils |
| %EO | yield of essential oils. |
4. Conclusions
Acknowledgments
References
- van Gils, C; Cox, PA. Ethnobotany of nutmeg in the Spice Islands. J. Ethnopharmacol 1994, 42, 117–124. [Google Scholar]
- Olajide, OA; Ajayi, FF; Ekhelar, AI; Awe, SO; Makinde, JM; Alada, AR. Biological effects of Myristica fragrans (nutmeg) extract. Phytother Res 1999, 13, 344–345. [Google Scholar]
- Sonavane, GS; Sarveiya, VP; Kasture, VS; Kasture, SB. Anxiogenic activity of Myristica fragrans seeds. Pharmacol. Biochem. Behav 2002, 71, 239–244. [Google Scholar]
- Isogai, A; Suzuki, A; Tamura, S. Structure of dimeric phenoxy-propanoids from Myristica fragrans. Agar. Biol. Chem 1973, 37, 193–194. [Google Scholar]
- Stein, U; Greyer, H; Hentschel, H. Nutmeg (myristicin) poisoning—report on a fatal case and a series of cases recorded by a poison information centre. Forensic Sci. Int 2001, 118, 87–90. [Google Scholar]
- Sonavane, A; Vikram, S; Veena, K; Sanjay, BK. Behavioural action of Myristica fragrans Houtt. seeds. Indian J. Pharmacol 2001, 33, 417–424. [Google Scholar]
- Hallstrom, H; Thuvander, A. Toxicological evaluation of myristicin. Nat. Toxins 1997, 5, 186–192. [Google Scholar]
- Oyen, NXD. Plant Resources of South-East Asia, Essential-oil Plants; Proscea: Bogor, Indonesia, 1999; Volume 12. [Google Scholar]
- Buchbauer, G; Jirovetz, L; Jager, W; Dietrich, H; Plank, C. Aromatherapy: Evidence for sedative effects of the essential oil of lavender after inhalation. Z Naturforsch C 1991, 46, 1067–1072. [Google Scholar]
- Buchbauer, G; Jirovetz, L; Jager, W; Plank, C; Dietrich, H. Fragrance compounds and essential oils with sedative effects upon inhalation. J. Pharm. Sci 1993, 82, 660–664. [Google Scholar]
- Beyer, J; Ehlers, D; Maurer, HH. Abuse of nutmeg (Myristica fragrans Houtt.): Studies on the metabolism and the toxicologic detection of its ingredients elemicin, myristicin, and safrole in rat and human urine using gas chromatography/mass spectrometry. Ther. Drug Monit 2006, 28, 568–575. [Google Scholar]
- Foye, W. Principles of Medicinal Chemistry; LEA & FEBRIGER: Philadelphia, PA, USA, 1981; Volume 2, p. 931. [Google Scholar]
- Hossain, SJ; Hamamoto, K; Aoshima, H; Hara, Y. Effects of tea components on the response of GABA(A) receptors expressed in Xenopus Oocytes. J. Agric. Food Chem 2002, 50, 3954–3960. [Google Scholar]
- Aoshima, H; Hossain, SJ; Hamamoto, K; Yokoyama, T; Yamada, M; Shingai, R. Kinetic analyses of alcohol-induced potentiation of the response of GABA(A) receptors composed of alpha(1) and beta(1) subunits. J. Biochem 2001, 130, 703–709. [Google Scholar]
- Adams, RP. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy; Allured Publishing Corporation: Carol Stream, IL, USA, 1995. [Google Scholar]
- Jirovetz, L; Jager, W; Buchbauer, G; Nikiforov, A; Raverdino, V. Investigations of animal blood samples after fragrance drug inhalation by gas chromatography/mass spectrometry with chemical ionization and selected ion monitoring. Biol. Mass Spectrom 1991, 20, 801–803. [Google Scholar]
- Jirovetz, L; Buchbauer, G; Jager, W; Woidich, A; Nikiforov, A. Analysis of fragrance compounds in blood samples of mice by gas chromatography, mass spectrometry, GC/FTIR and GC/AES after inhalation of sandalwood oil. Biomed. Chromatogr 1992, 6, 133–134. [Google Scholar]
- Kovar, KA; Gropper, B; Friess, D; Ammon, HP. Blood levels of 1,8-cineole and locomotor activity of mice after inhalation and oral administration of rosemary oil. Planta Med 1987, 53, 315–318. [Google Scholar]


| No. | Retention Time | LRI expa | LRI Refb | Compounds | Percentage % |
|---|---|---|---|---|---|
| 1. | 5.967 | 920 | 931 | α-thujene | 0.78 |
| 2. | 6.246 | 931 | 939 | α-pinene | 10.23 |
| 3. | 6.583 | 943 | 953 | Camphene | 0.16 |
| 4. | 7.508 | 978 | 976 | Sabinene | 21.38 |
| 5. | 7.792 | 989 | 991 | α-myrcena | 2.38 |
| 6. | 8.493 | 1017 | 1018 | α-terpinene | 2.72 |
| 7. | 8.843 | 1032 | 1031 | Limonene | 5.57 |
| 8. | 9.142 | 1045 | 1040 | β-ocimene | 0.03 |
| 9. | 9.525 | 1061 | 1062 | γ-terpinene | 3.98 |
| 10. | 9.728 | 1070 | 1097 | trans-sabinene hydrate | 0.03 |
| 11. | 10.097 | 1085 | 1088 | Terpinolene | 1.62 |
| 12. | 10.393 | 1098 | 1098 | Linalool | 0.75 |
| 13. | 10.821 | 1119 | - | Fenchyl alcohol | 0.05 |
| 14. | 11.603 | 1158 | 1219 | cis-sabinene hydrate | 0.06 |
| 15. | 12.306 | 1193 | 1177 | 4-terpineol | 13.92 |
| 16. | 12.492 | 1203 | 1189 | α-terpineol | 3.11 |
| 17. | 12.67 | 1212 | 1228 | Citronellol | 0.77 |
| 18. | 13.297 | 1247 | 1257 | Linalyl acetate | 0.06 |
| 19. | 13.949 | 1282 | 1285 | Bornyl acetate | 0.24 |
| 20. | 14.158 | 1293 | 1285 | Safrole | 4.28 |
| 21. | 15.76 | 1392 | 1401 | Methyl eugeunol | 0.77 |
| 22. | 16.568 | 1447 | 1447 | Isoeugeunol | 1.74 |
| 23. | 17.773 | 1530 | 1520 | Myristicin | 13.57 |
| 24. | 18 | 1551 | 1554 | Elimicin | 1.42 |
| 25. | 18.573 | 1586 | - | Metoxyeugeunol | 0.10 |
| 26. | 18.742 | 1598 | 1622 | β-asaron | 0.03 |
| 27. | 20.625 | 1767 | - | Myristic acid | 0.11 |
| 28. | 21.01 | 1789 | 1793 | Ethyl miristate | 0.04 |
| 29. | 21.352 | 1716 | - | Palmitic acid | 0.03 |
| 30. | 22.946 | 1954 | 1993 | Ethyl palmitate | 0.07 |
| 31. | 25.103 | 2181 | - | Stearic acid | 0.01 |
| 32. | 25.183 | 2193 | 2194 | Ethyl oleate | 0.01 |
| No | Mean instead of Number of average | Inhibitory Effectc (%) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Treatment | Doses (mL) | Minute | |||||||
| 0–15 ±SD | 15–30 ±SD | 30–45 ±SD | 45–60 ±SD | 60–75 ±SD | Number of average | ||||
| 1 | Controlb | 0 | 280.4 ± 20.6 | 294.4 ± 4.3 | 311.4 ± 17.4 | 303.4 ± 14.7 | 297.4 ± 9.8 | 1,487.0 | 0 |
| 2 | Lavender oil | 0.1 | 217.6 ± 18.2 | 200.4 ± 12.4 | 195.6 ± 9.7 | 197.4 ± 7.9 | 193.2 ± 11.9 | 1,004.2 | 32.46 |
| 0.3 | 187.8 ± 19.0 | 181.4 ± 20.5 | 186.2 ± 15.0 | 180.4 ± 16.5 | 178.4 ± 19.4 | 914.2 | 38.52 | ||
| 0.5 | 139.6 ± 11.2 | 128.8 ± 10.9 | 116.8 ± 10.3 | 123.2 ± 8.9 | 118.2 ± 8.7 | 626.6 | 57.86 | ||
| 3 | Nutmeg oil | 0.1 | 120.0 ± 35.5 | 113.6 ± 32.9 | 114.2 ± 22.8 | 105.0 ± 22.3 | 100.2 ± 22.1 | 553.0 | 62.81 |
| 0.3 | 121.4 ± 41.8 | 119.2 ± 37.1 | 110.8 ± 36.5 | 81.8 ± 23.2 | 82.2 ± 18.6 | 515.4 | 65.33 | ||
| 0.5 | 107.8 ± 27.3 | 104.0 ± 26.1 | 84.4 ± 27.4 | 85.8 ± 23.1 | 84.6 ± 23.2 | 466.6 | 68.62 | ||
| Compound | Duration of Inhalation | LRI Refa | |||||
|---|---|---|---|---|---|---|---|
| ½ hour (Rd = 84%) | 1 hour (Rd = 90%) | 2 hours (Rd = 86%) | |||||
| LRI Expb | Conc. μg/mL | LRI Expb | Conc. μg/mL | LRI Expb | Conc. μg/mL | ||
| 4-Terpineol | 1181 | 1.5 | 1181 | 2.9 | 1183 | 6.3 | 1177 |
| Safrole | nd | nd | nd | nd | 1292 | 1.3 | 1285 |
| Myristicin | 1521 | 3.8 | 1521 | 5.2 | 1523 | 7.1 | 1520 |
| Methyl myristate | 1718 | 1.6 | 1719 | 1.4 | 1720 | 1.2 | 1726 |
| Methyl palmitate | 1919 | 67.8 | 1920 | 72.2 | 1922 | 58.7 | 1927 |
| Palmitic acid | 1952 | 2.8 | nd | nd | nd | nd | 1961c |
| Methyl octadeca-10-oate | 2094 | 23.3 | 2096 | 24.7 | 2097 | 18.9 | - |
| Methyl oleate | 2100 | 12.1 | 2102 | 13.8 | 2105 | 10.7 | - |
| Methyl stearate | 2129 | 13.1 | 2132 | 13.2 | 2134 | 10.8 | 2128 |
| Treatment | Dose (mL) | Mean Instead of Number of Average | Inhibitory Effect (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Minutes | |||||||||
| 0–15 | 15–30 | 30–45 | 45–60 | 60–75 | 75–90 | Total Number | |||
| Control | Control | 321.5 ±30.1 | 305.64 ±13.3 | 299.89 ±10.2 | 287.96 ±18.2 | 314.6 ±35.6 | 255.46 ±25.34 | 1529.59 | 0 |
| 0.1 mL | 129.5 ±9.88 | 139.9 ±12.56 | 126.7 ±9.89 | 128.2 ±10.23 | 129.7 ±8.77 | 115.43 ±10.22 | 654 | 57.24 | |
| Myristicin | 0.3 mL | 105.8 ±19.10 | 120.4 ±20.5 | 129.5 ±15.08 | 117.6 ±16.5 | 109.2 ±19.4 | 76.45 ±3.20 | 582.5 | 61.92 |
| 0.5 mL | 55.23 ±11.2 | 60.8 ±10.9 | 45.4 ±10.3 | 39.2 ±8.9 | 8.33 ±8.7 | 0.81 ±0.01 | 208.96 | 86.34 | |
| 0.1 mL | 120 ±35.5 | 113.6 ±32.9 | 114.2 ±22.8 | 105 ±22.3 | 100.2 ±22.1 | 98.43 ±15.67 | 553 | 63.85 | |
| Safrole | 0.3 mL | 95.4 ±11.12 | 91.34 ±22.23 | 83.26 ±16.34 | 90.45 ±22.22 | 88.2 ±13.16 | 45.29 ±10.23 | 448.65 | 70.67 |
| 0.5 mL | 40.2 ±7.2 | 49.5 ±6.14 | 30.55 ±7.35 | 18.6 ±6.78 | 14.7 ±8.2 | 0.00 ±0.00 | 153.55 | 89.96 | |
| 0.1 mL | 132.22 ±33.55 | 133.21 ±23.4 | 124.98 ±22.8 | 125.65 ±32.3 | 105.2 ±22.1 | 111.56 ±32.32 | 621.26 | 59.38 | |
| 4-Terpineol | 0.3 mL | 110.1 ±31.18 | 109.32 ±27.5 | 80.81 ±26.22 | 85.48 ±12.2 | 82.2 ±11.8 | 78.65 ±16.54 | 467.91 | 69.41 |
| 0.5 mL | 78.85 ±7.60 | 60.25 ±5.37 | 64.4 ±7.25 | 55.78 ±5.54 | 54.16 ±9.25 | 40.20 ±8.50 | 313.44 | 79.51 | |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Muchtaridi; Subarnas, A.; Apriyantono, A.; Mustarichie, R. Identification of Compounds in the Essential Oil of Nutmeg Seeds (Myristica fragrans Houtt.) That Inhibit Locomotor Activity in Mice. Int. J. Mol. Sci. 2010, 11, 4771-4781. https://doi.org/10.3390/ijms11114771
Muchtaridi, Subarnas A, Apriyantono A, Mustarichie R. Identification of Compounds in the Essential Oil of Nutmeg Seeds (Myristica fragrans Houtt.) That Inhibit Locomotor Activity in Mice. International Journal of Molecular Sciences. 2010; 11(11):4771-4781. https://doi.org/10.3390/ijms11114771
Chicago/Turabian StyleMuchtaridi, Anas Subarnas, Anton Apriyantono, and Resmi Mustarichie. 2010. "Identification of Compounds in the Essential Oil of Nutmeg Seeds (Myristica fragrans Houtt.) That Inhibit Locomotor Activity in Mice" International Journal of Molecular Sciences 11, no. 11: 4771-4781. https://doi.org/10.3390/ijms11114771






