The Role of Molecular Biology in the Biomonitoring of Human Exposure to Chemicals
Abstract
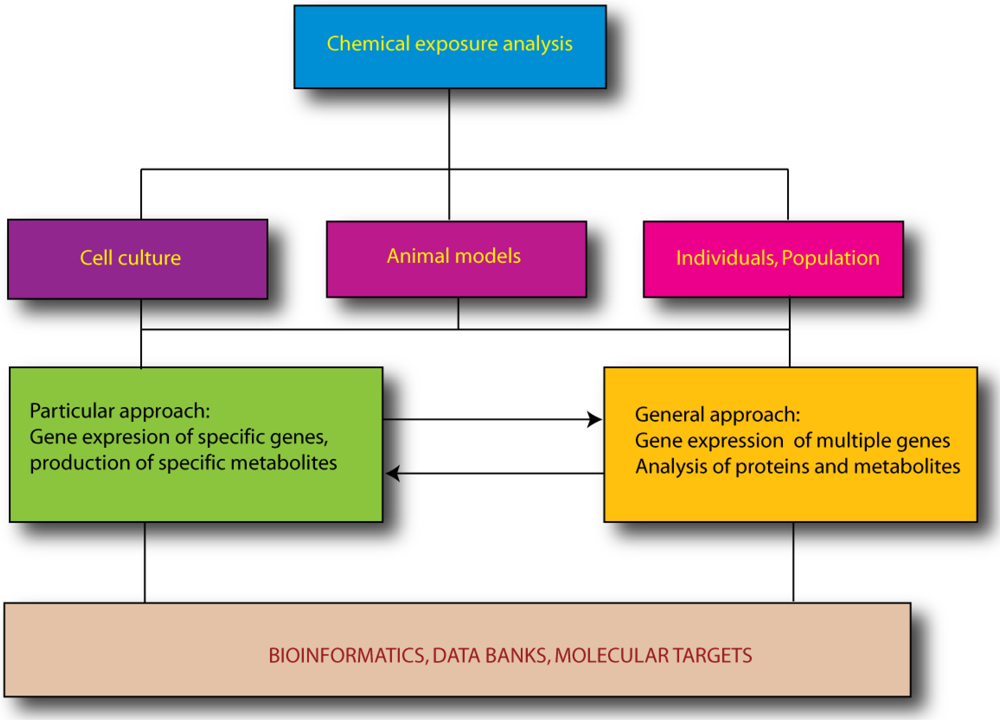
:1. Introduction
2. Cell Cultures
2.1. Primary Cell Culture
2.2. Cell Lines
2.3. Stem Cells
2.4. Three Dimensional-Cultures
3. Evaluation of Gene Expression
3.1. qRT PCR
3.2. ELISA
3.3. Microarrays
4. “Omics” in Analysis of Chemical Exposure
4.1. Genomics and Transcriptomics
4.2. Proteomics
4.3. Metabolomics
5. Bioinformatics
6. Molecular Biology in Evaluation of Risk to Chemical Exposure
6.1. Polycyclic Aromatic Compounds
6.2. Metals and Non-Metals
6.3. Radiactive Materials
7. Conclusions
Acknowledgments
References
- Zucco, F; de Angelis, I; Testai, E; Stammati, A. Toxicology investigations with cell culture systems: 20 years after. Toxicol. in Vitro 2004, 18, 153–163. [Google Scholar]
- Gomez-Lechon, MJ; Donato, MT; Castell, JV; Jover, R. Human hepatocytes in primary culture: the choice to investigate drug metabolism in man. Curr. Drug Metab 2004, 5, 443–462. [Google Scholar]
- Donato, MT; Lahoz, A; Castell, JV; Gomez-Lechon, MJ. Cell lines: a tool for in vitro drug metabolism studies. Curr. Drug Metab 2008, 9, 1–11. [Google Scholar]
- Castell, JV; Donato, MT; Gomez-Lechon, MJ. Metabolism and bioactivation of toxicants in the lung. The in vitro cellular approach. Exp. Toxicol. Pathol 2005, 57, 189–204. [Google Scholar]
- Bergamaschi, E; Bussolati, O; Magrini, A; Bottini, M; Migliore, L; Bellucci, S; Iavicoli, I; Bergamaschi, A. Nanomaterials and lung toxicity: interactions with airways cells and relevance for occupational health risk assessment. Int. J. Immunopathol. Pharmacol 2006, 19, 3–10. [Google Scholar]
- Martinez-Velazquez, M; Maldonado, V; Ortega, A; Melendez-Zajgla, J; Albores, A. Benzene metabolites induce apoptosis in lymphocytes. Exp. Toxicol. Pathol 2006, 58, 65–70. [Google Scholar]
- Windal, I; Denison, MS; Birnbaum, LS; van Wouwe, N; Baeyens, W; Goeyens, L. Chemically activated luciferase gene expression (CALUX) cell bioassay analysis for the estimation of dioxin-like activity: critical parameters of the CALUX procedure that impact assay results. Environ. Sci. Technol 2005, 39, 7357–7364. [Google Scholar]
- Seagrave, J; McDonald, JD; Mauderly, JL. In vitro versus in vivo exposure to combustion emissions. Exp. Toxicol. Pathol 2005, 57, 233–238. [Google Scholar]
- Bae, DS; Gennings, C; Carter, WH, Jr; Yang, RS; Campain, JA. Toxicological interactions among arsenic, cadmium, chromium, and lead in human keratinocytes. Toxicol. Sci 2001, 63, 132–142. [Google Scholar]
- Munoz, B; Huerta, M; Lopez-Bayghen, E. Cilostazol reduces proliferation through c-Myc down-regulation in MDCK cells. Eur. J. Pharmacol 2009, 616, 22–30. [Google Scholar]
- Liu, Y; Glatt, H. Human cytochrome P450 2E1 and sulfotransferase 1A1 coexpressed in Chinese hamster V79 cells enhance spontaneous mutagenesis. Environ. Mol. Mutagen 2010, 51, 23–30. [Google Scholar]
- Chen, J; Yang, XX; Huang, M; Hu, ZP; He, M; Duan, W; Chan, E; Sheu, FS; Chen, X; Zhou, SF. Small interfering RNA-mediated silencing of cytochrome P450 3A4 gene. Drug Metab. Dispos 2006, 34, 1650–1657. [Google Scholar]
- Rohwedel, J; Guan, K; Hegert, C; Wobus, AM. Embryonic stem cells as an in vitro model for mutagenicity, cytotoxicity and embryotoxicity studies: present state and future prospects. Toxicol. in Vitro 2001, 15, 741–753. [Google Scholar]
- Heng, BC; Richards, M; Shu, Y; Gribbon, P. Induced pluripotent stem cells: A new tool for toxicology screening? Arch. Toxicol 2009, 83, 641–644. [Google Scholar]
- Yu, X; Hong, S; Moreira, EG; Faustman, EM. Improving in vitro Sertoli cell/gonocyte co-culture model for assessing male reproductive toxicity: Lessons learned from comparisons of cytotoxicity versus genomic responses to phthalates. Toxicol. Appl. Pharmacol 2009, 239, 325–336. [Google Scholar]
- Pamies, D; Reig, JA; Vilanova, E; Sogorb, MA. Expression of Neuropathy Target Esterase in mouse embryonic stem cells during differentiation. Arch. Toxicol 2010, 84, 481–491. [Google Scholar]
- Davila, JC; Cezar, GG; Thiede, M; Strom, S; Miki, T; Trosko, J. Use and application of stem cells in toxicology. Toxicol. Sci 2004, 79, 214–223. [Google Scholar]
- Lee, MY; Kumar, RA; Sukumaran, SM; Hogg, MG; Clark, DS; Dordick, JS. Three-dimensional cellular microarray for high-throughput toxicology assays. Proc. Natl. Acad. Sci. USA 2008, 105, 59–63. [Google Scholar]
- Mazzoleni, G; Di Lorenzo, D; Steimberg, N. Modelling tissues in 3D: The next future of pharmaco-toxicology and food research? Genes Nutr 2009, 4, 13–22. [Google Scholar]
- Nakamura, K; Mizutani, R; Sanbe, A; Enosawa, S; Kasahara, M; Nakagawa, A; Ejiri, Y; Murayama, N; Miyamoto, Y; Torii, T; Kusakawa, S; Yamauchi, J; Fukuda, M; Yamazaki, H; Tanoue, A. Evaluation of drug toxicity with hepatocytes cultured in a micro-space cell culture system. J Biosci Bioeng 2010, (in press). [Google Scholar]
- Lin, YF; Nagasawa, H; Peng, Y; Chuang, EY; Bedford, JS. Comparison of several radiation effects in human MCF10A mammary epithelial cells cultured as 2D monolayers or 3D acinar stuctures in matrigel. Radiat. Res 2009, 171, 708–715. [Google Scholar]
- Pampaloni, F; Stelzer, EH; Masotti, A. Three-dimensional tissue models for drug discovery and toxicology. Recent Pat. Biotechnol 2009, 3, 103–117. [Google Scholar]
- Lee, J; Lilly, GD; Doty, RC; Podsiadlo, P; Kotov, NA. In vitro toxicity testing of nanoparticles in 3D cell culture. Small 2009, 5, 1213–1221. [Google Scholar]
- Mendoza-Cantu, A; Castorena-Torres, F; Bermudez de Leon, M; Cisneros, B; Lopez-Carrillo, L; Rojas-Garcia, AE; Aguilar-Salinas, A; Manno, M; Albores, A. Occupational toluene exposure induces cytochrome P450 2E1 mRNA expression in peripheral lymphocytes. Environ. Health Perspect 2006, 114, 494–499. [Google Scholar]
- Gwinn, MR; Weston, A. Application of oligonucleotide microarray technology to toxic occupational exposures. 2008, 71, 315–324. [Google Scholar]
- Walker, NJ. Real-time and quantitative PCR: applications to mechanism-based toxicology. J. Biochem. Mol. Toxicol 2001, 15, 121–127. [Google Scholar]
- Watson, DE; Li, B. TaqMan applications in genetic and molecular toxicology. Int. J. Toxicol 2005, 24, 139–145. [Google Scholar]
- Fan, CY; Cowden, J; Simmons, SO; Padilla, S; Ramabhadran, R. Gene expression changes in developing zebrafish as potential markers for rapid developmental neurotoxicity screening. Neurotoxicol. Teratol 2010, 32, 91–98. [Google Scholar]
- Prat, O; Berenguer, F; Steinmetz, G; Ruat, S; Sage, N; Quemeneur, E. Alterations in gene expression in cultured human cells after acute exposure to uranium salt: Involvement of a mineralization regulator. Toxicol. in Vitro 2010, 24, 160–168. [Google Scholar]
- Bunderson, M; Brooks, DM; Walker, DL; Rosenfeld, ME; Coffin, JD; Beall, HD. Arsenic exposure exacerbates atherosclerotic plaque formation and increases nitrotyrosine and leukotriene biosynthesis. Toxicol. Appl. Pharmacol 2004, 201, 32–39. [Google Scholar]
- Heinrich, A; Balszuweit, F; Thiermann, H; Kehe, K. Rapid simultaneous determination of apoptosis, necrosis, and viability in sulfur mustard exposed HaCaT cell cultures. Toxicol. Lett 2009, 191, 260–267. [Google Scholar]
- Liebers, V; Stubel, H; Duser, M; Bruning, T; Raulf-Heimsoth, M. Standardization of whole blood assay for determination of pyrogenic activity in organic dust samples. Int. J. Hyg. Environ. Health 2009, 212, 547–556. [Google Scholar]
- Mishra, PK; Panwar, H; Bhargava, A; Gorantla, VR; Jain, SK; Banerjee, S; Maudar, KK. Isocyanates induces DNA damage, apoptosis, oxidative stress, and inflammation in cultured human lymphocytes. J. Biochem. Mol. Toxicol 2008, 22, 429–440. [Google Scholar]
- Niot-Mansart, V; Muhamedi, A; Arnould, JP. A competitive ELISA detecting 7-methylguanosine adduct induced by N-nitrosodimethylamine exposure. Hum. Exp. Toxicol 2005, 24, 89–94. [Google Scholar]
- Rothman, N; Poirier, MC; Haas, RA; Correa-Villasenor, A; Ford, P; Hansen, JA; O’Toole, T; Strickland, PT. Association of PAH-DNA adducts in peripheral white blood cells with dietary exposure to polyaromatic hydrocarbons. Environ. Health Perspect 1993, 99, 265–267. [Google Scholar]
- Lettieri, T. Recent applications of DNA microarray technology to toxicology and ecotoxicology. Environ. Health Perspect 2006, 114, 4–9. [Google Scholar]
- Sen, B; Mahadevan, B; DeMarini, DM. Transcriptional responses to complex mixtures: a review. Mutat. Res 2007, 636, 144–177. [Google Scholar]
- Omura, S; Koike, E; Kobayashi, T. Microarray analysis of gene expression in rat alveolar epithelial cells exposed to fractionated organic extracts of diesel exhaust particles. Toxicology 2009, 262, 65–72. [Google Scholar]
- Umbright, C; Sellamuthu, R; Li, S; Kashon, M; Luster, M; Joseph, P. Blood gene expression markers to detect and distinguish target organ toxicity. Mol. Cell. Biochem 2010, 335, 223–234. [Google Scholar]
- Fachin, AL; Mello, SS; Sandrin-Garcia, P; Junta, CM; Ghilardi-Netto, T; Donadi, EA; Passos, GA; Sakamoto-Hojo, ET. Gene expression profiles in radiation workers occupationally exposed to ionizing radiation. J. Radiat. Res 2009, 50, 61–71. [Google Scholar]
- Castorena-Torres, F; Bermudez de Leon, M; Cisneros, B; Zapata-Perez, O; Salinas, JE; Albores, A. Changes in gene expression induced by polycyclic aromatic hydrocarbons in the human cell lines HepG2 and A549. Toxicol. in Vitro 2008, 22, 411–421. [Google Scholar]
- Harrill, AH; Rusyn, I. Systems biology and functional genomics approaches for the identification of cellular responses to drug toxicity. Expert Opin. Drug Metab. Toxicol 2008, 4, 1379–1389. [Google Scholar]
- Smith, MT; Vermeulen, R; Li, G; Zhang, L; Lan, Q; Hubbard, AE; Forrest, MS; McHale, C; Zhao, X; Gunn, L; Shen, M; Rappaport, SM; Yin, S; Chanock, S; Rothman, N. Use of ‘Omic’ technologies to study humans exposed to benzene. Chem Biol Interact 2005. [Google Scholar]
- Malard, V; Gaillard, JC; Berenguer, F; Sage, N; Quemeneur, E. Urine proteomic profiling of uranium nephrotoxicity. Biochim. Biophys. Acta 2009, 1794, 882–891. [Google Scholar]
- Chen, X; Ma, XM; Ma, SW; Coenraads, PJ; Zhang, CM; Liu, J; Zhao, LJ; Sun, M; Tang, NJ. Proteomic analysis of the rat ovary following chronic low-dose exposure to 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD). J. Toxicol. Environ. Health 2009, 72, 717–726. [Google Scholar]
- Lin, PP; Yang, MH; Liao, PC; Wu, HY; Chang, LW; Tsai, HT; Tyan, YC. Proteomic analysis of proteins associated with tt-DDE induced toxicity in BEAS-2B cells. Biochem. Biophys. Res. Commun 2008, 376, 519–524. [Google Scholar]
- Sumner, S; Snyder, R; Burgess, J; Myers, C; Tyl, R; Sloan, C; Fennell, T. Metabolomics in the assessment of chemical-induced reproductive and developmental outcomes using non-invasive biological fluids: application to the study of butylbenzyl phthalate. J. Appl. Toxicol 2009, 29, 703–714. [Google Scholar]
- Heijne, WH; Kienhuis, AS; van Ommen, B; Stierum, RH; Groten, JP. Systems toxicology: applications of toxicogenomics, transcriptomics, proteomics and metabolomics in toxicology. Expert Rev. Proteomics 2005, 2, 767–780. [Google Scholar]
- Fujibuchi, W; Kim, H; Okada, Y; Taniguchi, T; Sone, H. High-performance gene expression module analysis tool and its application to chemical toxicity data. Methods Mol. Biol 2009, 577, 55–65. [Google Scholar]
- Wetmore, BA; Merrick, BA. Toxicoproteomics: proteomics applied to toxicology and pathology. Toxicol. Pathol 2004, 32, 619–642. [Google Scholar]
- Chen, M; Martin, J; Fang, H; Isukapalli, S; Georgopoulos, PG; Welsh, WJ; Tong, W. ebTrack: an environmental bioinformatics system built upon ArrayTrack. BMC Proc 2009, 3, 5. [Google Scholar]
- Davis, AP; Murphy, CG; Rosenstein, MC; Wiegers, TC; Mattingly, CJ. The Comparative Toxicogenomics Database facilitates identification and understanding of chemical-gene-disease associations: arsenic as a case study. BMC Med. Genet 2008, 1, 48. [Google Scholar]
- Mattingly, CJ; Rosenstein, MC; Colby, GT; Forrest, JN, Jr; Boyer, JL. The Comparative Toxicogenomics Database (CTD): a resource for comparative toxicological studies. J. Exp. Zool 2006, 305, 689–692. [Google Scholar]
- Sone, H; Okura, M; Zaha, H; Fujibuchi, W; Taniguchi, T; Akanuma, H; Nagano, R; Ohsako, S; Yonemoto, J. Profiles of Chemical Effects on Cells (pCEC): a toxicogenomics database with a toxicoinformatics system for risk evaluation and toxicity prediction of environmental chemicals. J. Toxicol. Sci 2010, 35, 115–123. [Google Scholar]
- Angerer, J; Ewers, U; Wilhelm, M. Human biomonitoring: state of the art. Int. J. Hyg. Environ.Health 2007, 210, 201–228. [Google Scholar]
- Brandt, HC; Watson, WP. Monitoring human occupational and environmental exposures to polycyclic aromatic compounds. Ann. Occup. Hyg 2003, 47, 349–378. [Google Scholar]
- Fowler, BA. Monitoring of human populations for early markers of cadmium toxicity: a review. Toxicol. Appl. Pharmacol 2009, 238, 294–300. [Google Scholar]
- Koller, LD. In vitro assessment of humoral immunity following exposure to heavy metals. Environ. Health Perspect 1982, 43, 37–39. [Google Scholar]
- Blakely, WF; Prasanna, PG; Grace, MB; Miller, AC. Radiation exposure assessment using cytological and molecular biomarkers. Radiat. Prot. Dosim 2001, 97, 17–23. [Google Scholar]

© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Muñoz, B.; Albores, A. The Role of Molecular Biology in the Biomonitoring of Human Exposure to Chemicals. Int. J. Mol. Sci. 2010, 11, 4511-4525. https://doi.org/10.3390/ijms11114511
Muñoz B, Albores A. The Role of Molecular Biology in the Biomonitoring of Human Exposure to Chemicals. International Journal of Molecular Sciences. 2010; 11(11):4511-4525. https://doi.org/10.3390/ijms11114511
Chicago/Turabian StyleMuñoz, Balam, and Arnulfo Albores. 2010. "The Role of Molecular Biology in the Biomonitoring of Human Exposure to Chemicals" International Journal of Molecular Sciences 11, no. 11: 4511-4525. https://doi.org/10.3390/ijms11114511



