Peanut Skin Color: A Biomarker for Total Polyphenolic Content and Antioxidative Capacities of Peanut Cultivars
Abstract
:1. Introduction
2. Results and Discussion
2.1. Peanut Skin Color
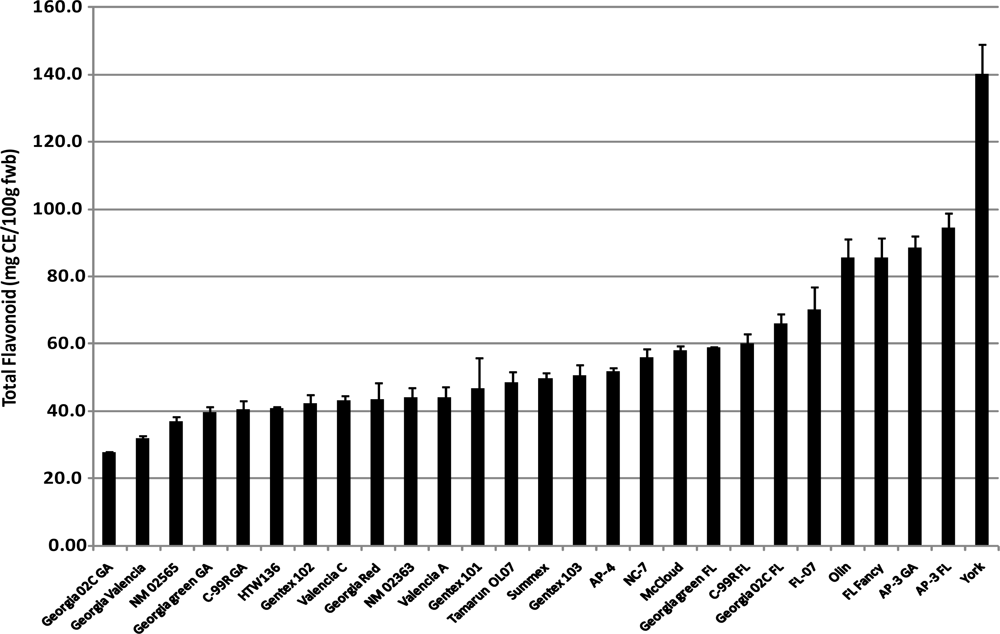
2.2. Total Flavonoids
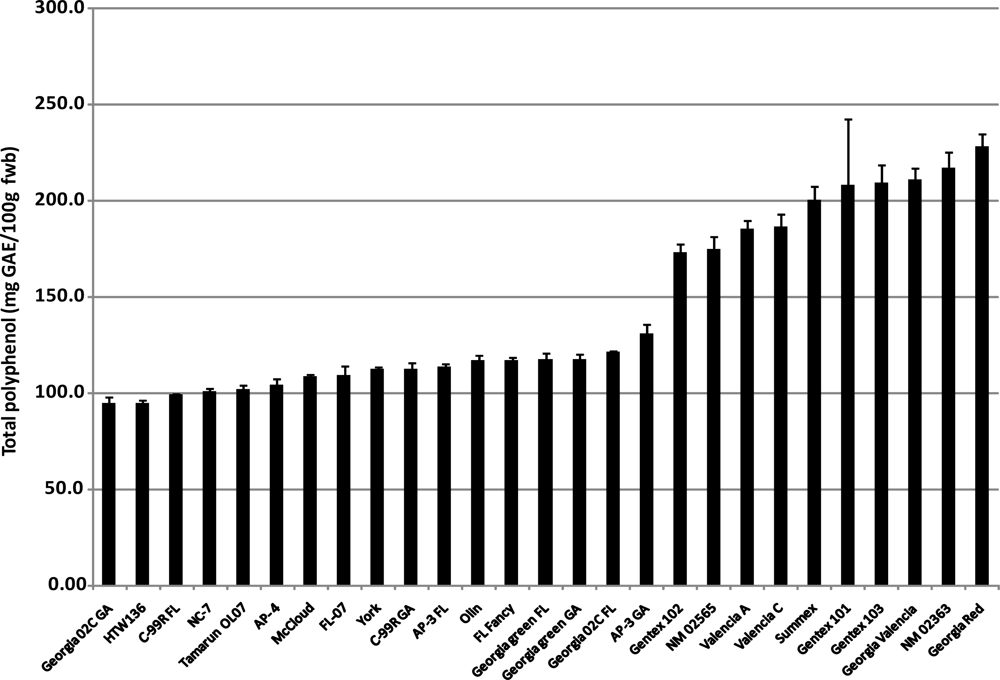
2.3. Total Phenolics
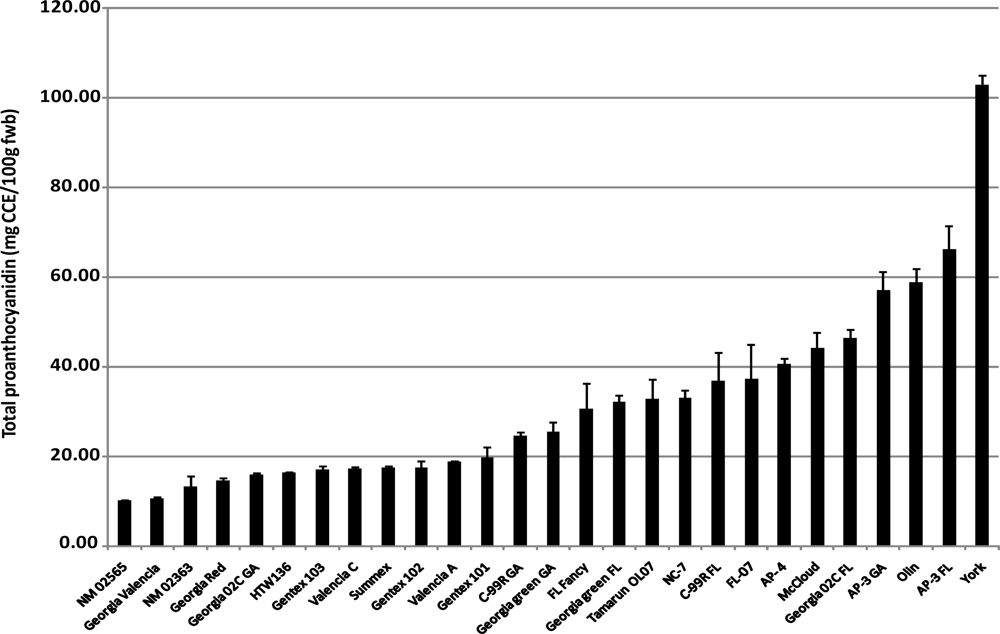
2.4. Total Proanthocyanidins
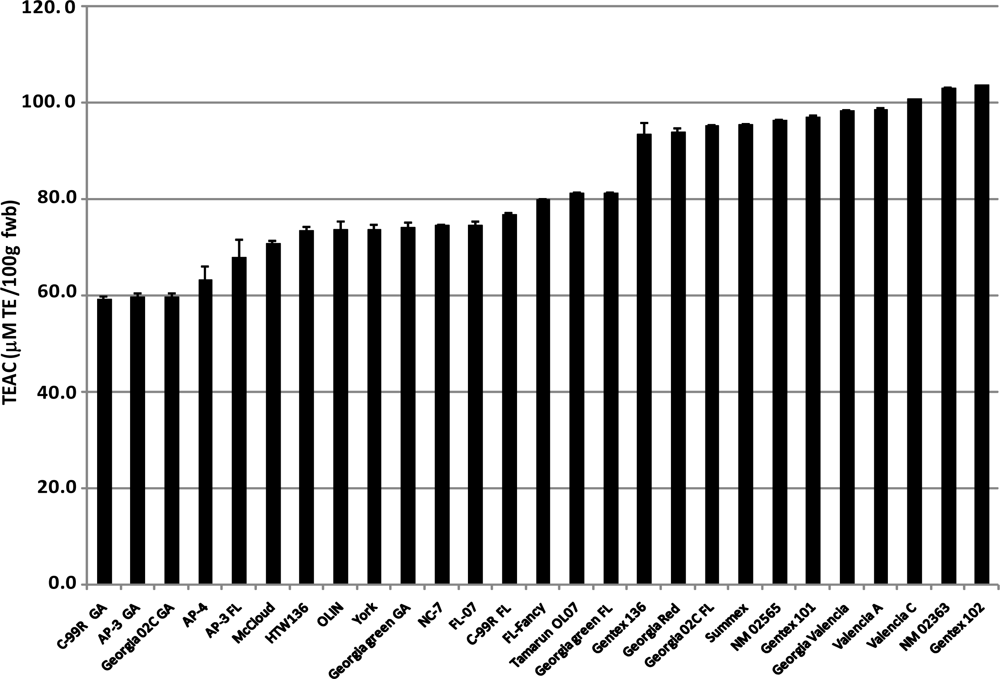
2.5. Trolox® Equivalent Antioxidant Capacity (TEAC)
3. Experimental Section
3.1. Materials
3.2. CEI Color Measurements
3.3. Preparation of Sample Extracts
3.4. Total Flavonoids
3.5. Total Phenolics
3.6. Total Proanthocyanidins
3.7. Trolox Equivalent Antioxidant Capacity (TEAC Assay)
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
References and Notes
- Heron, MP; Hoyert, DL; Xu, J; Scott, C; Tejada-Vera, B. Deaths: Preliminary data for 2006. Natl. Vital Stat. Rep 2008, 56, 1–52. [Google Scholar]
- Willett, WC. Balancing life-style and genomics research for disease prevention. Science 2002, 296, 695–698. [Google Scholar]
- Pellegrini, N; Serafini, M; Colombi, B; Del Rio, D; Salvatore, S; Bianchi, M; Brighenti, F. Total antioxidant capacity of plant foods, beverages and oils consumed in Italy assessed by three different in vitro assays. J. Nutr 2003, 133, 2812–2819. [Google Scholar]
- Scalbert, A; Johnson, IT; Saltmarsh, M. Polyphenols: Antioxidants and beyond. Am. J. Clin. Nutr 2005, 81, 215S–217S. [Google Scholar]
- Ferrándiz, ML; Alcaraz, MJ. Anti-inflammatory activity and inhibition of arachidonic acid metabolism by flavonoids. Agents Actions 1991, 32, 283–288. [Google Scholar]
- Middleton, E, Jr; Kandaswami, C. Effects of flavonoids on immune and inflammatory cell functions. Biochem. Pharmacol 1992, 43, 1167–1179. [Google Scholar]
- Gil, B; Sanz, MJ; Terencio, MC; Ferrándiz, ML; Bustos, G; Payá, M; Gunasegaran, R; Alcaraz, MJ. Effects of flavonoids on Naja naja and human recombinant synovial phospholipases A2 and inflammatory responses in mice. Life Sci 1994, 54, 333–338. [Google Scholar]
- Brown, JA. Review of the genetic effects of naturally occurring flavonoids, anthraquinones and related compounds. Mutat. Res 1980, 75, 243–277. [Google Scholar]
- Deschner, EE; Ruperto, J; Wong, G; Newmark, HL. Quercetin and rutin as inhibitors of azoxymethanol-induced colonic neoplasia. Carcinogenesis 1991, 12, 1193–1196. [Google Scholar]
- Elangovan, V; Sekar, N; Govindasamy, S. Chemopreventive potential of dietary bioflavonoids against 20-methylcholanthrene induced tumorigenesis. Cancer Lett 1994, 87, 107–113. [Google Scholar]
- Rump, AF; Schussler, M; Acar, D; Cordes, A; Ratke, R; Theisohn, M; Rosen, R; Klaus, W; Fricke, U. Effects of different inotropes with antioxidant properties on acute regional myocardial ischemia in isolated rabbit hearts. Gen. Pharmacol 1995, 26, 603–611. [Google Scholar]
- Sobolev, VS; Cole, RJ. trans-Resveratrol content in commercial peanuts and peanut products. J. Agric. Food Chem 1999, 47, 1435–1439. [Google Scholar]
- Gu, L; Kelm, MA; Hammerstone, JF; Beecher, G; Holden, J; Haytowitz, D; Gebhardt, S; Prior, RL. Concentrations of proanthocyanidins in common foods and estimation of normal consumption. J. Nutr 2004, 134, 613–617. [Google Scholar]
- Wang, ML; Gillaspie, AG; Morris, JB; Pittman, RN; Davis, J; Pederson, GA. Flavonoid content in different legume germplasm seeds quantified by HPLC. Plant Gen. Res 2008, 6, 62–69. [Google Scholar]
- Todd, JJ; Vodkin, LO. Pigmented soybean (Glycine max) seed coats accumulate proanthocyanidins during development. Plant Physiol 1993, 102, 663–670. [Google Scholar]
- American Peanut Council. About the peanut industry.
- Economic Research Service ERS/USDA. Food consumption per capita data 2007.
- Yu, J; Ahmedna, M; Goktepe, I; Dai, J. Peanut skin procyanidins: Composition and antioxidant activities as affected by processing. J. Food Comp. Anal 2006, 19, 364–371. [Google Scholar]
- Cao, G; Sofic, E; Prior, RL. Antioxidant and prooxidant behavior of flavonoids: Structure-activity relationships. Free Radic. Biol. Med 1997, 22, 749–760. [Google Scholar]
- Jia, Z; Tang, M; Wu, J. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 1999, 64, 555–559. [Google Scholar]
- Singleton, VL; Rossi, JA, Jr. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Viticul 1965, 16, 144–153. [Google Scholar]
- Porter, LJ; Hristch, LN; Chan, BC. The conversion of procyanidins and prodelphinidins to cyanidins and delphinidins. Phytochemistry 1986, 25, 225–230. [Google Scholar]
- Shahidi, F; Alasalvar, C; Liyana-Pathirana, CM. Phytochemicals in hazelnut kernel (Corylus avellana L.) and its byproducts. J. Agric. Food Chem 2007, 55, 1212–1220. [Google Scholar]

| Peanut Cultivar | StateW | Market typex | CIE Colory | |||
|---|---|---|---|---|---|---|
| L* | a* | b* | Hue angle | |||
| AP-3 | FL | R | 49.34 ± 0.14h | 14.24 ± 0.20j | 23.84 ± 0.20bc | 0.54 ± 0.01k |
| AP-4 | FL | R | 47.74 ± 0.08i | 15.97 ± 0.09fghi | 22.21 ± 0.25de | 0.62 ± 0.01hij |
| C-99R | FL | R | 50.32 ± 0.04g | 12.97 ± 0.03k | 21.54 ± 0.06ef | 0.54 ± 0.00k |
| Georgia Green | FL | R | 43.53 ± 0.09k | 19.29 ± 0.17d | 18.42 ± 0.20g | 0.81 ± 0.01f |
| Georgia 02C | FL | R | 50.46 ± 0.06fg | 15.21 ± 0.06hij | 22.92 ± 0.05cd | 0.59 ± 0.00ijk |
| FL-07 | FL | R | 45.92 ± 0.20j | 19.43 ± 0.09d | 22.03 ± 0.17de | 0.72 ± 0.01g |
| FL Fancy | FL | R | 51.01 ± 0.19ef | 14.37 ± 0.09j | 21.72 ± 0.14ef | 0.58 ± 0.01ijk |
| McCloud | FL | R | 53.21 ± 0.12c | 15.45 ± 0.21hi | 24.04 ± 0.28b | 0.57 ± 0.01k |
| York | FL | R | 51.08 ± 0.08e | 14.99 ± 0.13ij | 23.82 ± 0.15bc | 0.56 ± 0.01k |
| Olin | TX | S | 51.81 ± 0.16d | 17.37 ± 0.07e | 23.72 ± 0.17bc | 0.63 ± 0.01hi |
| NC-7 | TX | Vir | 54.63 ± 0.42a | 13.19 ± 0.64k | 21.34 ± 0.62ef | 0.55 ± 0.03k |
| HTW136 | TX | Val | 37.97 ± 0.31n | 21.69 ± 0.62bc | 14.06 ± 0.60jk | 1.00 ± 0.03cd |
| Tamarun OL07 | TX | R | 53.87 ± 0.14b | 16.07 ± 0.85fgh | 25.67 ± 0.06a | 0.56 ± 0.00k |
| AP-3 | GA | R | 48.75 ± 0.12h | 15.88 ± 0.16ghi | 24.56 ± 0.15b | 0.57 ± 0.01jk |
| C-99R | GA | R | 43.65 ± 0.16k | 16.58 ± 0.19efg | 22.01 ± 0.22de | 0.65 ± 0.01h |
| Georgia Green | GA | R | 47.05 ± 0.03i | 16.92 ± 0.19ef | 21.74 ± 0.17e | 0.66 ± 0.01h |
| Georgia 02C | GA | R | 40.10 ± 0.30l | 16.54 ± 0.45efg | 20.70 ± 0.53f | 0.67 ± 0.03gh |
| Valencia A | NM | Val | 35.43 ± 0.18q | 24.62 ± 0.43a | 14.79 ± 0.29hij | 1.03 ± 0.02abc |
| Valencia C | NM | Val | 36.12 ± 0.18op | 24.96 ± 0.38a | 14.37 ± 0.33ijk | 1.05 ± 0.02ab |
| Gentex 101 | NM | Val | 33.79 ± 0.07r | 24.15 ± 0.06a | 13.83 ± 0.14jk | 1.05 ± 0.01a |
| Gentex 102 | NM | Val | 35.72 ± 0.25pq | 24.14 ± 0.06a | 15.58 ± 0.16h | 1.00 ± 0.01bcd |
| Gentex 136 | NM | Val | 37.52 ± 0.32n | 24.67 ± 0.71a | 15.38 ± 0.61hi | 1.01 ± 0.03abc |
| Sunmex | NM | Val | 36.45 ± 0.22o | 22.60 ± 0.52b | 14.77 ± 0.51hij | 0.99 ± 0.03cd |
| Georgia Red | NM | Val | 32.71 ± 0.08s | 22.28 ± 0.19b | 13.58 ± 0.02k | 1.02 ± 0.01abc |
| Georgia Val | NM | Val | 38.91 ± 0.04m | 20.69 ± 0.14c | 14.74 ± 0.27hij | 0.95 ± 0.01de |
| NM 02363 | NM | Val | 35.81 ± 0.29pq | 20.77 ± 0.58c | 13.89 ± 0.60jk | 0.98 ± 0.03cd |
| NM 02565 | NM | Val | 39.49 ± 0.02m | 18.75 ± 0.03d | 14.84 ± 0.07hij | 0.90 ± 0.00e |
| Mean ± SEM | 43.81 ± 0.78 | 18.66 ± 0.43 | 19.26 ± 0.47 | 0.77 ± 0.02 | ||
| Pearson’s Correlation coefficient (r2) | ||||
|---|---|---|---|---|
| L* | a* | b* | Hue | |
| Total Polyphenols | −0.78 | 0.77 | −0.80 | 0.82 |
| Total Flavonoids | 0.57 | −0.46 | 0.57 | −0.54 |
| Total Proanthocyanidins | 0.70 | −0.60 | 0.74 | −0.70 |
| TEAC | −0.62 | 0.70 | −0.75 | 0.75 |
| TP | TF | TPC | TEAC | |
| Total Polyphenols | 1.00 | −0.35 | −0.52 | 0.80 |
| Total Flavonoids | 1.00 | 0.90 | −0.33Z | |
| Total Proanthocyanidins | 1.00 | −0.51 | ||
| TEAC | 1.00 | |||
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chukwumah, Y.; Walker, L.T.; Verghese, M. Peanut Skin Color: A Biomarker for Total Polyphenolic Content and Antioxidative Capacities of Peanut Cultivars. Int. J. Mol. Sci. 2009, 10, 4941-4952. https://doi.org/10.3390/ijms10114941
Chukwumah Y, Walker LT, Verghese M. Peanut Skin Color: A Biomarker for Total Polyphenolic Content and Antioxidative Capacities of Peanut Cultivars. International Journal of Molecular Sciences. 2009; 10(11):4941-4952. https://doi.org/10.3390/ijms10114941
Chicago/Turabian StyleChukwumah, Yvonne, Lloyd T. Walker, and Martha Verghese. 2009. "Peanut Skin Color: A Biomarker for Total Polyphenolic Content and Antioxidative Capacities of Peanut Cultivars" International Journal of Molecular Sciences 10, no. 11: 4941-4952. https://doi.org/10.3390/ijms10114941
APA StyleChukwumah, Y., Walker, L. T., & Verghese, M. (2009). Peanut Skin Color: A Biomarker for Total Polyphenolic Content and Antioxidative Capacities of Peanut Cultivars. International Journal of Molecular Sciences, 10(11), 4941-4952. https://doi.org/10.3390/ijms10114941





