Role of the Ubiquitin Proteasome System in Regulating Skin Pigmentation
Abstract
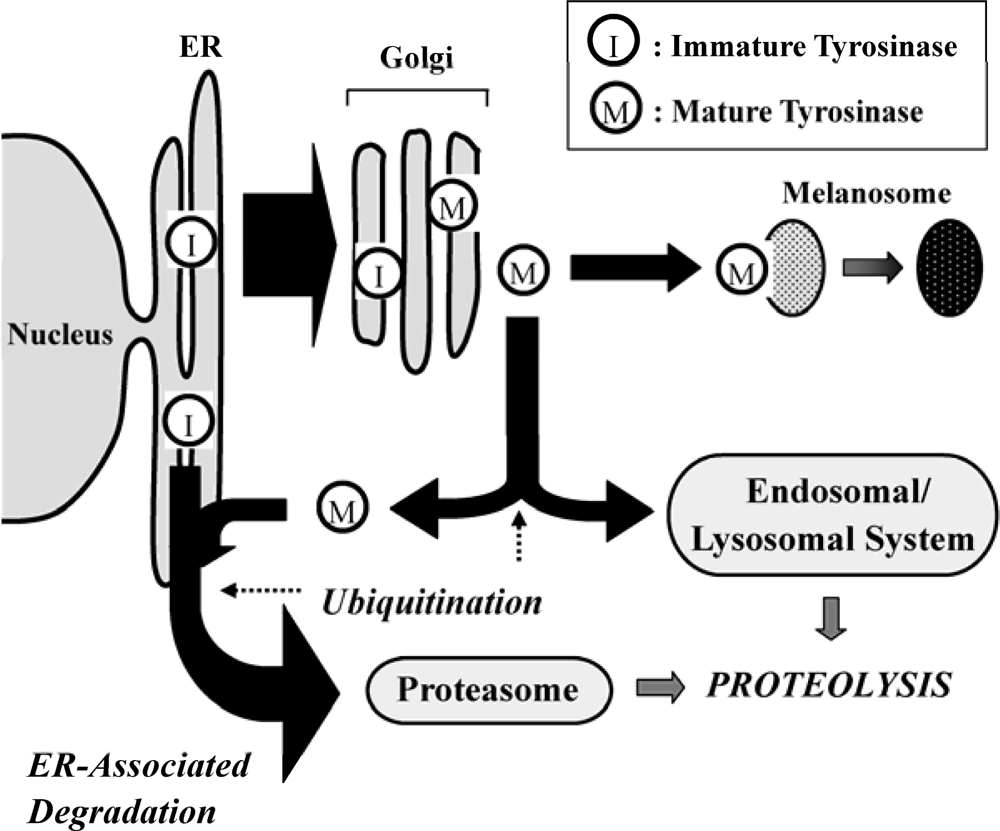
:1. Introduction
2. Role of the UPS in Melanin Synthesis
3. Fatty Acid-Induced Regulation of the UPS on Tyrosinase
4. Conclusions
Acknowledgments
References and Notes
- Ando, H; Kondoh, H; Ichihashi, M; Hearing, VJ. Approaches to identify inhibitors of melanin biosynthesis via the quality control of tyrosinase. J. Invest. Dermatol 2007, 127, 751–761. [Google Scholar]
- Saeki, H; Oikawa, A. Synthesis and degradation of tyrosinase in cultured melanoma cells. J. Cell. Physiol 1980, 104, 171–175. [Google Scholar]
- Martinez-Esparza, M; Jiménez-Cervantes, C; Beermann, F; Aparicio, P; Lozano, JA; García-Borrón, JC. Transforming growth factor-β1 inhibits basal melanogenesis in B16/F10 mouse melanoma cells by increasing the rate of degradation of tyrosinase and tyrosinase-related protein 1. J. Biol. Chem 1997, 272, 3967–3972. [Google Scholar]
- Martinez-Esparza, M; Jiménez-Cervantes, C; Solano, F; Lozano, JA; García-Borrón, JC. Mechanisms of melanogenesis inhibition by tumor necrosis factor-α in B16/F10 mouse melanoma cells. Eur. J. Biochem 1998, 255, 139–146. [Google Scholar]
- Ando, H; Funasaka, Y; Oka, M; Ohashi, A; Furumura, M; Matsunaga, J; Matsunaga, N; Hearing, VJ; Ichihashi, M. Possible involvement of proteolytic degradation of tyrosinase in the regulatory effect of fatty acids on melanogenesis. J. Lipid Res 1999, 40, 1312–1316. [Google Scholar]
- Halaban, R; Cheng, E; Zhang, Y; Moellmann, G; Hanlon, D; Michalak, M; Setaluri, V; Hebert, DN. Aberrant retention of tyrosinase in the endoplasmic reticulum mediates accelerated degradation of the enzyme and contributes to the dedifferentiated phenotype of amelanotic melanoma cells. Proc. Natl. Acad. Sci. USA 1997, 94, 6210–6215. [Google Scholar]
- Wang, Y; Androlewicz, MJ. Oligosaccharide trimming plays a role in the endoplasmic reticulum-associated degradation of tyrosinase. Biochem. Biophys. Res. Commun 2000, 271, 22–27. [Google Scholar]
- Svedine, S; Wang, T; Halaban, R; Hebert, DN. Carbohydrates act as sorting determinants in ER-associated degradation of tyrosinase. J. Cell Sci 2004, 117, 2937–2949. [Google Scholar]
- Bonifacino, JS; Weissman, AM. Ubiquitin and the control of protein fate in the secretory and endocytic pathways. Ann. Rev. Cell Dev. Biol 1998, 14, 19–57. [Google Scholar]
- Plemper, RK; Wolf, DH. Retrograde protein translocation: ERADication of secretory proteins in health and disease. Trends Biochem. Sci 1999, 24, 266–270. [Google Scholar]
- Ellgaard, L; Helenius, A. Quality control in the endoplasmic reticulum. Nature Rev. Mol. Cell Biol 2003, 4, 181–191. [Google Scholar]
- Hall, AM; Krishnamoorthy, L; Orlow, SJ. 25-Hydroxycholesterol acts in the Golgi compartment to induce degradation of tyrosinase. Pigment Cell Res 2004, 17, 396–406. [Google Scholar]
- Hall, AM; Orlow, SJ. Degradation of tyrosinase induced by phenylthiourea occurs following Golgi maturation. Pigment Cell Res 2005, 18, 122–129. [Google Scholar]
- Ando, H; Wen, ZM; Kim, HY; Valencia, JC; Costin, GE; Watabe, H; Yasumoto, K; Niki, Y; Kondoh, H; Ichihashi, M; Hearing, VJ. Intracellular composition of fatty acid affects the processing and function of tyrosinase through the ubiquitin-proteasome pathway. Biochem. J 2006, 394, 43–50. [Google Scholar]
- Oetting, WS; King, RA. Molecular basis of albinism: Mutations and polymorphisms of pigmentation genes associated with albinism. Hum. Mutat 1999, 13, 99–115. [Google Scholar]
- Toyofuku, K; Wada, I; Valencia, JC; Kushimoto, T; Ferrans, VJ; Hearing, VJ. Oculocutaneous albinism types 1 and 3 are ER retention diseases: Mutation of tyrosinase or Tyrp1 can affect the processing of both mutant and wild-type proteins. FASEB J 2001, 15, 2149–2161. [Google Scholar]
- Berson, JF; Frank, DW; Calvo, PA; Bieler, BM; Marks, MS. A common temperature-sensitive allelic form of human tyrosinase is retained in the endoplasmic reticulum at the nonpermissive temperature. J. Biol. Chem 2000, 275, 12281–12289. [Google Scholar]
- Hammond, C; Helenius, A. Quality control in the secretory pathway. Curr. Opin. Cell Biol 1995, 7, 523–529. [Google Scholar]
- Toyofuku, K; Wada, I; Spritz, RA; Hearing, VJ. The molecular basis of oculocutaneous albinism type 1 (OCA1): Sorting failure and degradation of mutant tyrosinase results in a lack of pigmentation. Biochem. J 2001, 355, 259–269. [Google Scholar]
- Mosse, CA; Hsu, W; Engelhard, VH. Tyrosinase degradation via two pathways during reverse translocation to the cytosol. Biochem. Biophys. Res. Commun 2001, 285, 313–319. [Google Scholar]
- Petrescu, SM; Petrescu, AJ; Titu, HN; Dwek, RA; Platt, FM. Inhibition of N-glycan processing in B16 melanoma cells results in inactivation of tyrosinase but does not prevent its transport to the melanosome. J. Biol. Chem 1997, 272, 15796–15803. [Google Scholar]
- Branza-Nichita, N; Petrescu, AJ; Dwek, RA; Wormald, MR; Platt, FM; Petrescu, SM. Tyrosinase folding and copper loading in vivo: A crucial role for calnexin and α-glucosidase II. Biochem. Biophys. Res. Commun 1999, 261, 720–725. [Google Scholar]
- de Virgilio, M; Weninger, H; Ivessa, NE. Ubiquitination is required for the retro-translocation of a short-lived luminal endoplasmic reticulum glycoprotein to the cytosol for degradation by the proteasome. J. Biol. Chem 1998, 273, 9734–9743. [Google Scholar]
- Arvan, P; Zhao, X; Ramos-Castaneda, J; Chang, A. Secretory pathway quality control operating in Golgi, plasmalemmal and endosomal systems. Traffic 2002, 3, 771–780. [Google Scholar]
- Tanaka, K; Ii, K; Ichihara, A; Waxman, L; Goldberg, AL. A high molecular weight protease in the cytosol of rat liver. I. Purification, enzymological properties, and tissue distribution. J. Biol. Chem 1986, 261, 15197–15203. [Google Scholar]
- McGuire, MJ; McCullough, ML; Croall, DE; DeMartino, GN. The high molecular weight multicatalytic proteinase, macropain, exists in a latent form in human erythrocytes. Biochim. Biophys. Acta 1989, 995, 181–186. [Google Scholar]
- Dahlmann, B; Becher, B; Sobek, A; Ehlers, C; Kopp, F; Kuehn, L. In vitro activation of the 20S proteasome. Enzyme Protein 1993, 47, 274–284. [Google Scholar]
- Rivett, AJ. Proteasomes: Multicatalytic proteinase complexes. Biochem. J 1993, 291, 1–10. [Google Scholar]
- Hamel, FG; Upward, JL; Siford, GL; Duckworth, WC. Inhibition of proteasome activity by selected amino acids. Metabolism 2003, 52, 810–814. [Google Scholar]
- Dahlmann, B; Rutschmann, M; Kuehn, L; Reinauer, H. Activation of the multicatalytic proteinase from rat skeletal muscle by fatty acids or sodium dodecyl sulphate. Biochem. J 1985, 228, 171–177. [Google Scholar]
- Watanabe, N; Yamada, S. Activation of 20S proteasomes from spinach leaves by fatty acids. Plant Cell Physiol 1996, 37, 147–151. [Google Scholar]
- Whitehouse, AS; Khal, J; Tisdale, MJ. Induction of protein catabolism in myotubes by 15(S)-hydroxyeicosatetraenoic acid through increased expression of the ubiquitin-proteasome pathway. Br. J. Cancer 2003, 89, 737–745. [Google Scholar]
- Smith, J; Su, X; El-Maghrabi, R; Stahl, PD; Abumrad, NA. Opposite regulation of CD36 ubiquitination by fatty acids and insulin–effects on fatty acid uptake. J. Biol. Chem 2008, 283, 13578–13585. [Google Scholar]
- Tsujishita, Y; Asaoka, Y; Nishizuka, Y. Regulation of phospholipase A2 in human leukemia cell lines: Its implication for intracellular signaling. Proc. Natl. Acad. Sci. USA 1994, 91, 6274–6278. [Google Scholar]
- Chawla, A; Repa, JJ; Evans, RM; Mangelsdorf, DJ. Nuclear receptors and lipid physiology: Opening the X-files. Science 2001, 294, 1866–1870. [Google Scholar]
- Clarke, SD. The multi-dimensional regulation of gene expression by fatty acids: Polyunsaturated fats as nutrient sensors. Curr. Opin. Lipidol 2004, 15, 13–18. [Google Scholar]
- Ando, H; Watabe, H; Valencia, JC; Yasumoto, K; Furumura, M; Funasaka, Y; Oka, M; Ichihashi, M; Hearing, VJ. Fatty acids regulate pigmentation via proteasomal degradation of tyrosinase—A new aspect of ubiquitin-proteasome function. J. Biol. Chem 2004, 279, 15427–15433. [Google Scholar]
- Ando, H; Ryu, A; Hashimoto, A; Oka, M; Ichihashi, M. Linoleic acid and α-linolenic acid lightens ultraviolet-induced hyperpigmentation of the skin. Arch. Dermatol. Res 1998, 290, 375–381. [Google Scholar]
- Shigeta, Y; Imanaka, H; Ando, H; Ryu, A; Oku, N; Baba, N; Makino, T. Skin whitening effect of linoleic acid is enhanced by liposomal formulations. Biol. Pharm. Bull 2004, 27, 591–594. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ando, H.; Ichihashi, M.; Hearing, V.J. Role of the Ubiquitin Proteasome System in Regulating Skin Pigmentation. Int. J. Mol. Sci. 2009, 10, 4428-4434. https://doi.org/10.3390/ijms10104428
Ando H, Ichihashi M, Hearing VJ. Role of the Ubiquitin Proteasome System in Regulating Skin Pigmentation. International Journal of Molecular Sciences. 2009; 10(10):4428-4434. https://doi.org/10.3390/ijms10104428
Chicago/Turabian StyleAndo, Hideya, Masamitsu Ichihashi, and Vincent J. Hearing. 2009. "Role of the Ubiquitin Proteasome System in Regulating Skin Pigmentation" International Journal of Molecular Sciences 10, no. 10: 4428-4434. https://doi.org/10.3390/ijms10104428
APA StyleAndo, H., Ichihashi, M., & Hearing, V. J. (2009). Role of the Ubiquitin Proteasome System in Regulating Skin Pigmentation. International Journal of Molecular Sciences, 10(10), 4428-4434. https://doi.org/10.3390/ijms10104428



