Solid-Phase Synthesis of Methyl N-(pyrimidin-2-yl)glycinate
Abstract
:Introduction
Results and Discussion
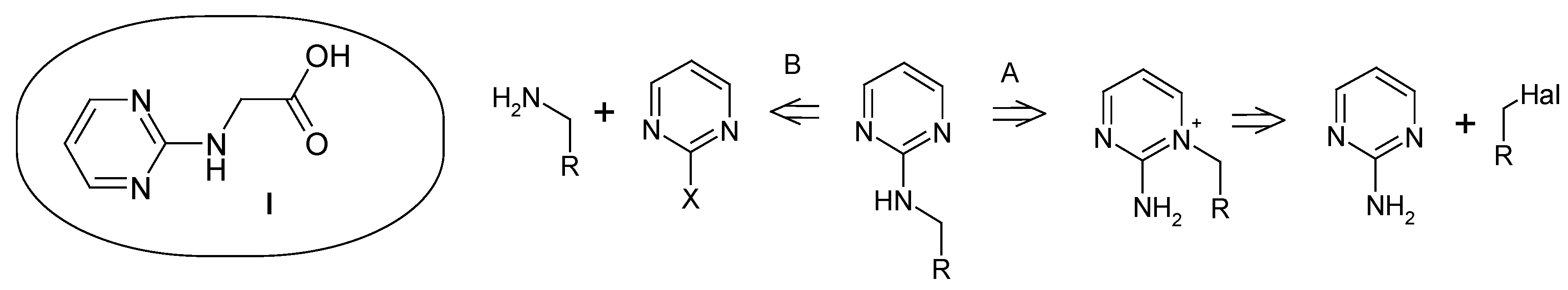
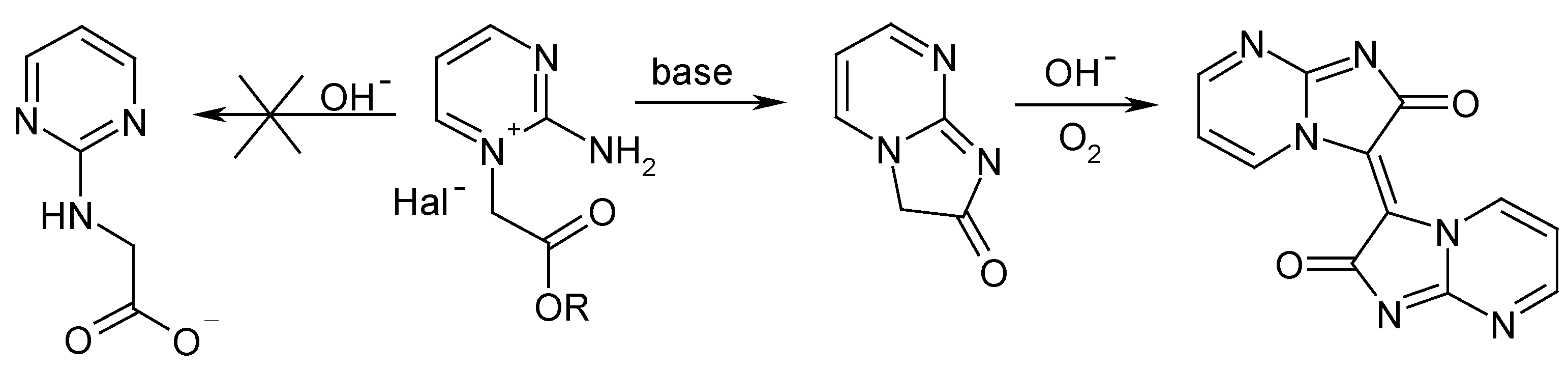
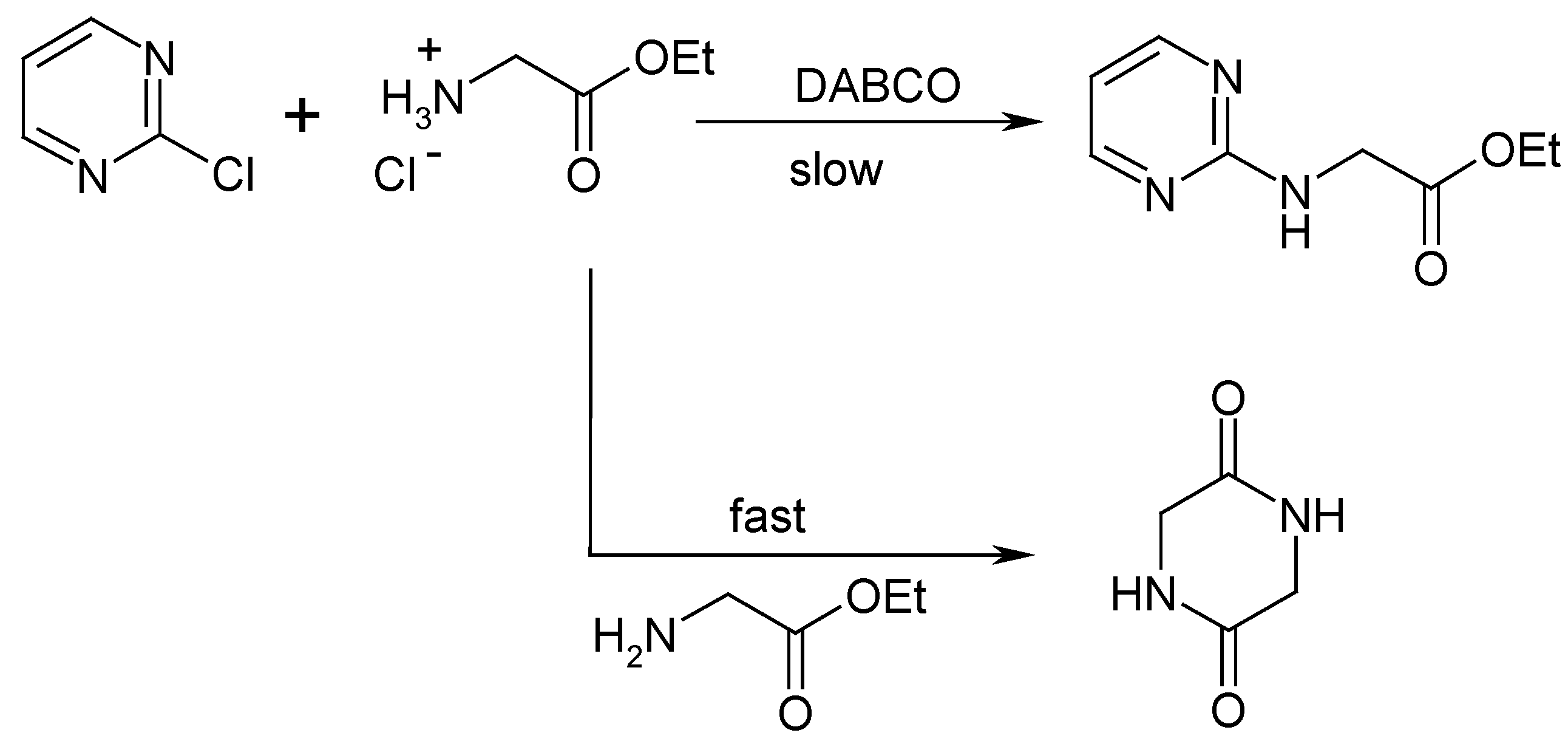
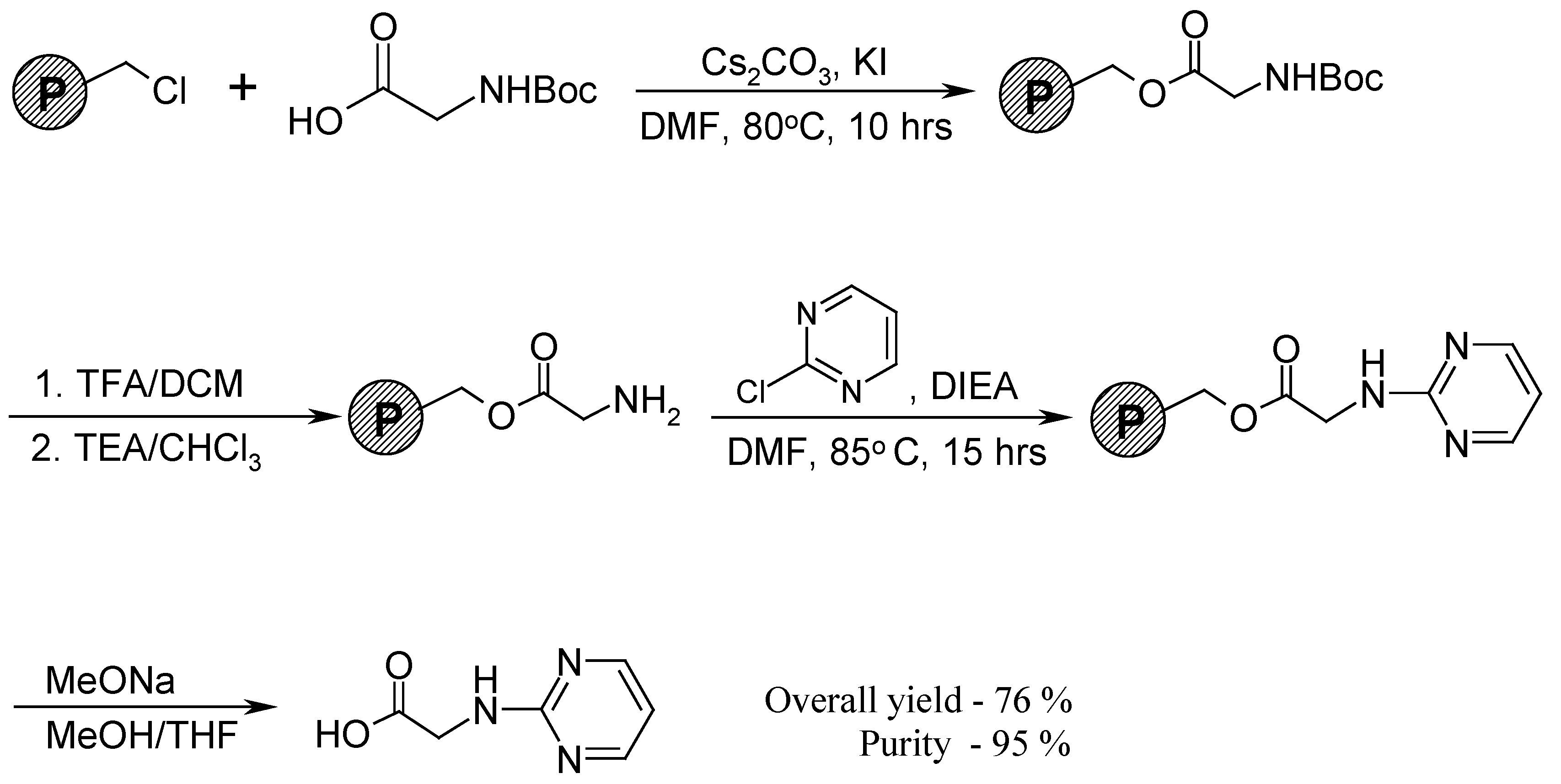
Conclusions
Experimental
General
Synthetic procedure
Acknowledgments
References
- Illuminati, G. Nucleophilic heteroaromatic substitution. Adv. Heterocycl. Chem. 1964, Vol. 3, 275–283. [Google Scholar]
- Merrifield, R.B. Solid phase peptide synthesis. I. The synthesis of a tetrapeptide. J. Am. Chem. Soc. 1963, 85, 2149–2154. [Google Scholar]
- Kobayashi, S.; Akiyama, R.; Kitagawa, H. Polymer-Supported α-Imino Acetates. Versatile Reagents for the Synthesis of alpha-Amino Acid Libraries. J. Comb. Chem. 2000, 2, 438–440. [Google Scholar]
- Mukaiyama, T. Oxidation-Reduction Condensation a New Method for Peptide Synthesis. Synth. Comm. 1972, 2, 243–265. [Google Scholar]
© 2003 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Ermolat'ev, D.S.; Babaev, E.V. Solid-Phase Synthesis of Methyl N-(pyrimidin-2-yl)glycinate. Molecules 2003, 8, 467-471. https://doi.org/10.3390/80600467
Ermolat'ev DS, Babaev EV. Solid-Phase Synthesis of Methyl N-(pyrimidin-2-yl)glycinate. Molecules. 2003; 8(6):467-471. https://doi.org/10.3390/80600467
Chicago/Turabian StyleErmolat'ev, Denis S., and Eugene V. Babaev. 2003. "Solid-Phase Synthesis of Methyl N-(pyrimidin-2-yl)glycinate" Molecules 8, no. 6: 467-471. https://doi.org/10.3390/80600467



