A Convenient Method to Prepare Labile FMN Derivatives
Abstract
:Introduction
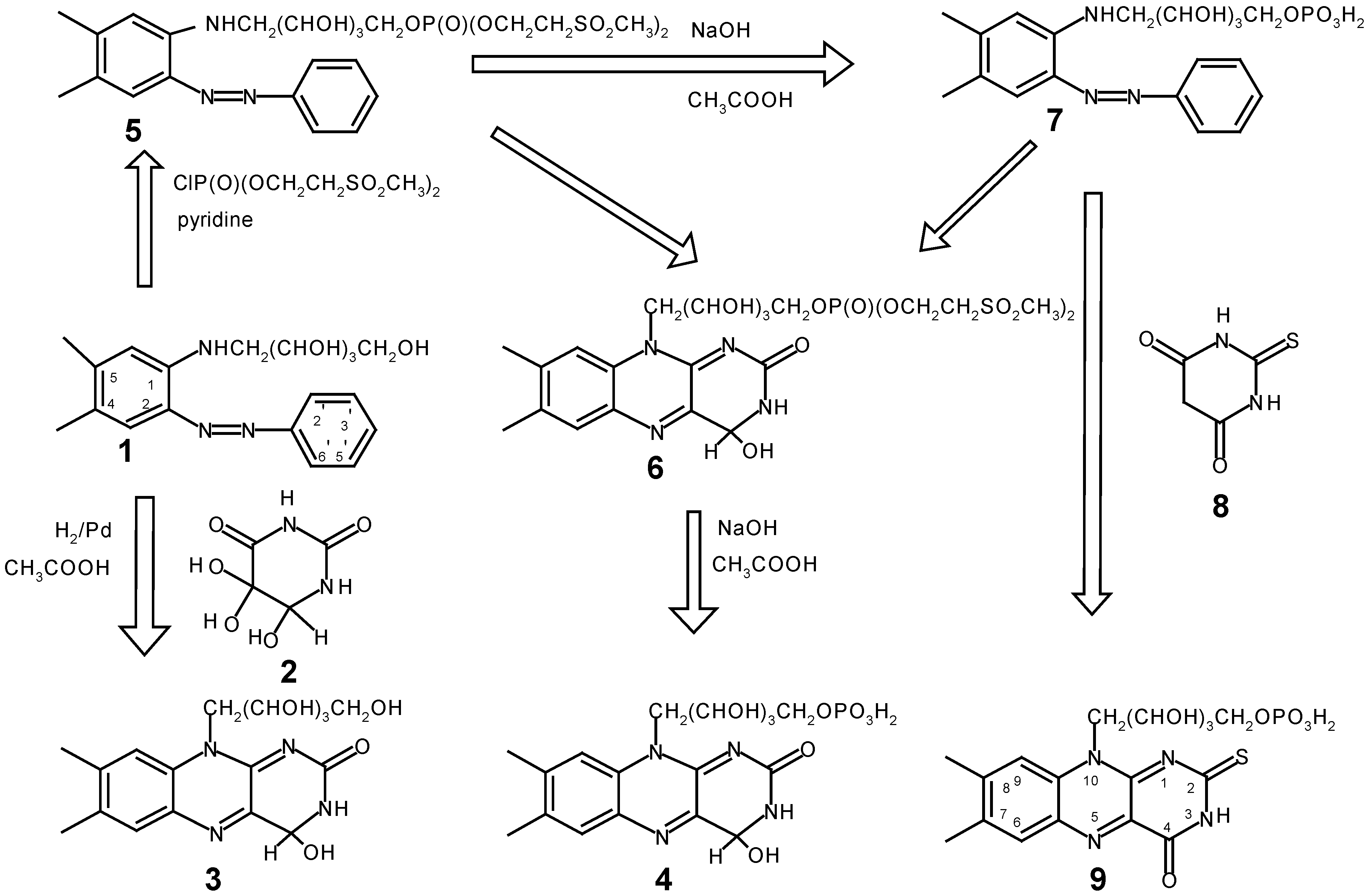
Results and Discussion
Conclusions
Experimental
General
4,5-dimethyl-2-phenylazo-N-ribityl 5'phosphate-bis[2-(methylsulfonyl)ethyl]-aniline (5)
7,8-dimethyl-10-ribityl-5'phosphate-bis(2-(methylsulfonyl)ethyl)-3,4-dihydro-isoalloxazine (6)
3,4-Dihydro-riboflavin (3):
4,5-Dimethyl-2-phenylazo-N-ribityl 5'-phosphate-aniline (7)
3,4-Dihydro-FMN (4)
2-Thio-FMN (9)
Acknowledgements
References
- Flexner, L. A.; Farkas, W. G. Preparation of Riboflavin Phosphate Intermediates. U.S. Patent 1952, 2,610,176, [Chem. Abstr.1953, 47,8781a]. [Google Scholar]
- Scola-Nagelschneider, G; Hemmerich, P. Synthesis, Separation, Identification and Interconversion of Riboflavin Phosphates and Their Acetyl Derivatives: A Reinvestigation. Eur. J. Biochem. 1976, 66, 567–577. [Google Scholar]
- Nielsen, P.; Rauschenbach, P.; Bacher, A. Phosphates of Riboflavin and Riboflavin Analogs: A Reinvestigation by High-Performance Liquid Chromatography. Anal. Biochem. 1983, 130, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Föry, W.; Hemmerich, P. Synthese Funktionell Abgewandelter Flavokoenzyme: 3-Alkyl und 2-Alkyliminoflavin-mononucleotide and Flavin-adenin-dinucleotide. Helv. Chim. Acta 1967, 50, 1766–1774. [Google Scholar] [CrossRef] [PubMed]
- Beld, A.; Claesen, C. A. A.; Roersma, E. S.; Schippers, W. J. M.; Keizer, L. M.; Tesser, G. I. Bis[2-(methylsulfonyl)methyl] phosphochloridate, a New Phosphorylating Agent. Recl. Trav. Chim. Pays-Bas 1984, 103, 196–202. [Google Scholar] [CrossRef]
- Müller, F. Chemistry and Biochemistry of Flavoenzymes; Müller, F., Ed.; CRC Press: Boca Raton, FL, 1991; Vol. 1, Chapter 1; p. 4. [Google Scholar]
- Behrend, R.; Rosen, O. Synthese der Harnsäure. Justus Liebigs Ann. Chem. 1889, 251, 235–256. [Google Scholar] [CrossRef]
- Müller, F.; Massey, V.; Heizmann, C.; Hemmerich, P.; Lhoste, J. M.; Gould, D. C. The Reduction of Flavins by Borohydride: 3,4-Dihydroflavins. Structure, Absorption and Luminescence. Eur. J. Biochem. 1969, 9, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples are available from MDPI.
© 2001 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Müller, F.; Lee, J. A Convenient Method to Prepare Labile FMN Derivatives. Molecules 2001, 6, 825-830. https://doi.org/10.3390/61000825
Müller F, Lee J. A Convenient Method to Prepare Labile FMN Derivatives. Molecules. 2001; 6(10):825-830. https://doi.org/10.3390/61000825
Chicago/Turabian StyleMüller, Franz, and John Lee. 2001. "A Convenient Method to Prepare Labile FMN Derivatives" Molecules 6, no. 10: 825-830. https://doi.org/10.3390/61000825



