Synthesis and Smo Activity of Some Novel Benzamide Derivatives
Abstract
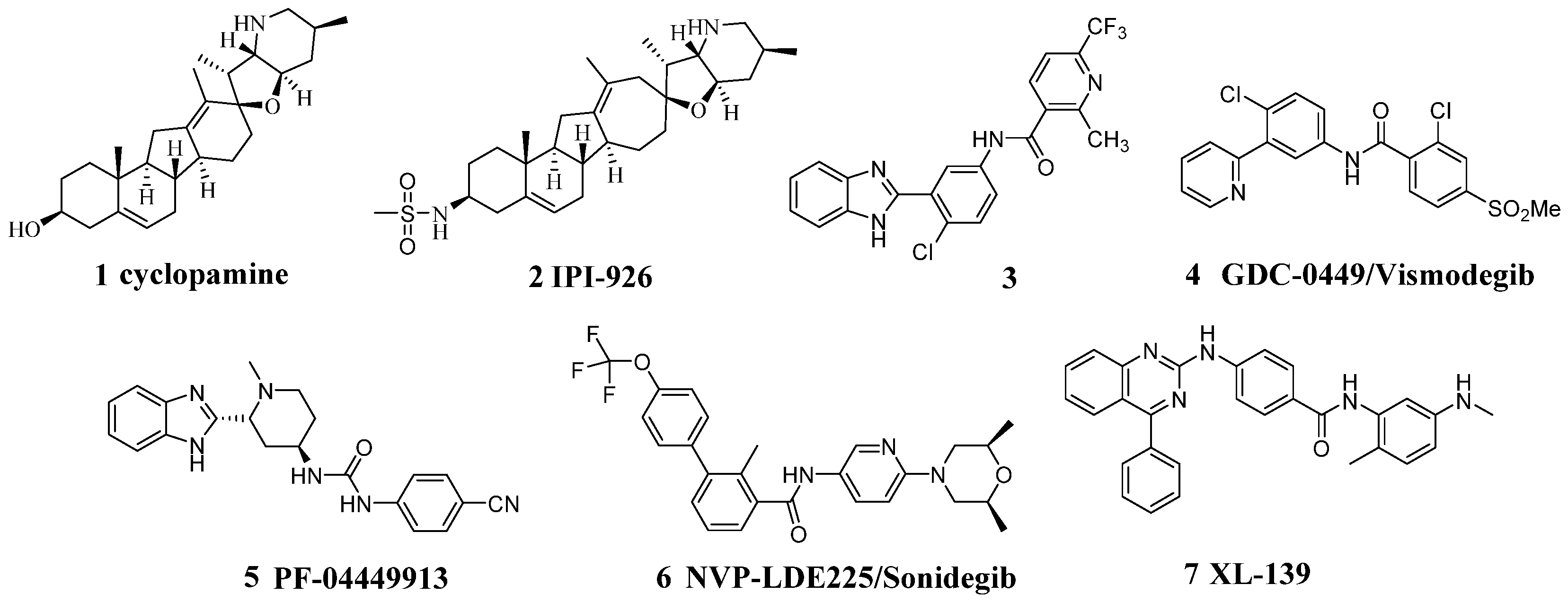
:1. Introduction
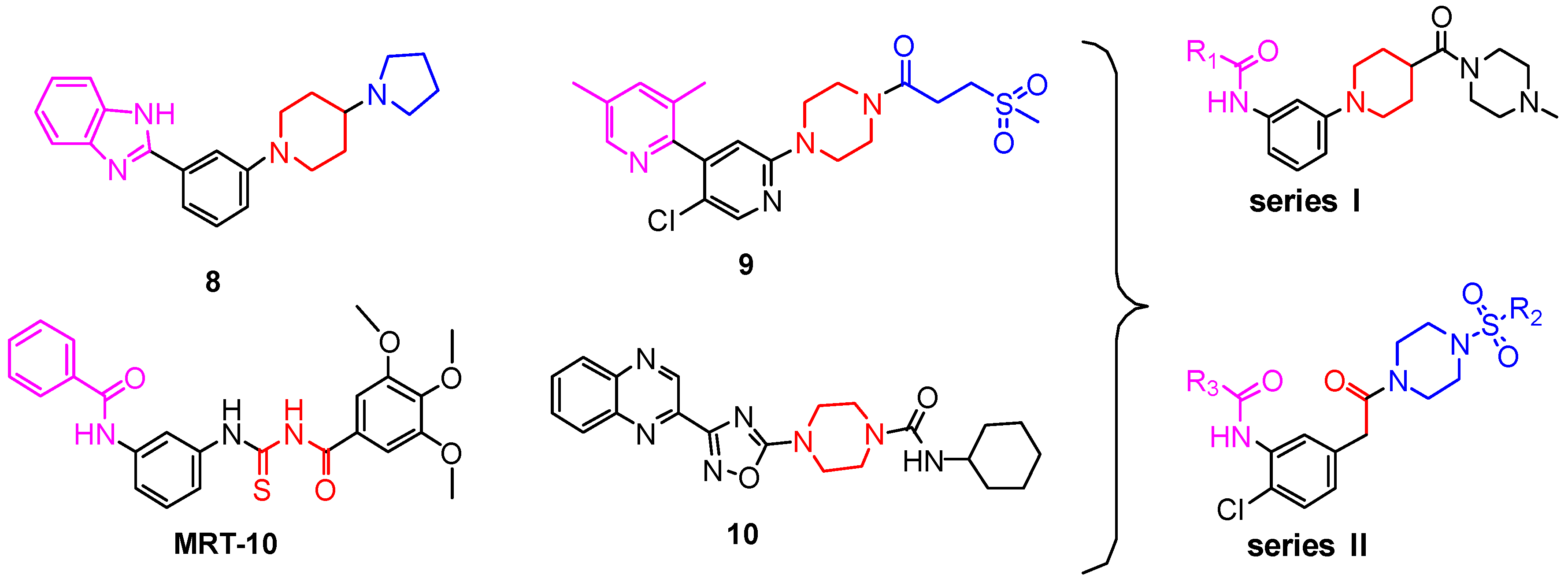
2. Results and Discussion
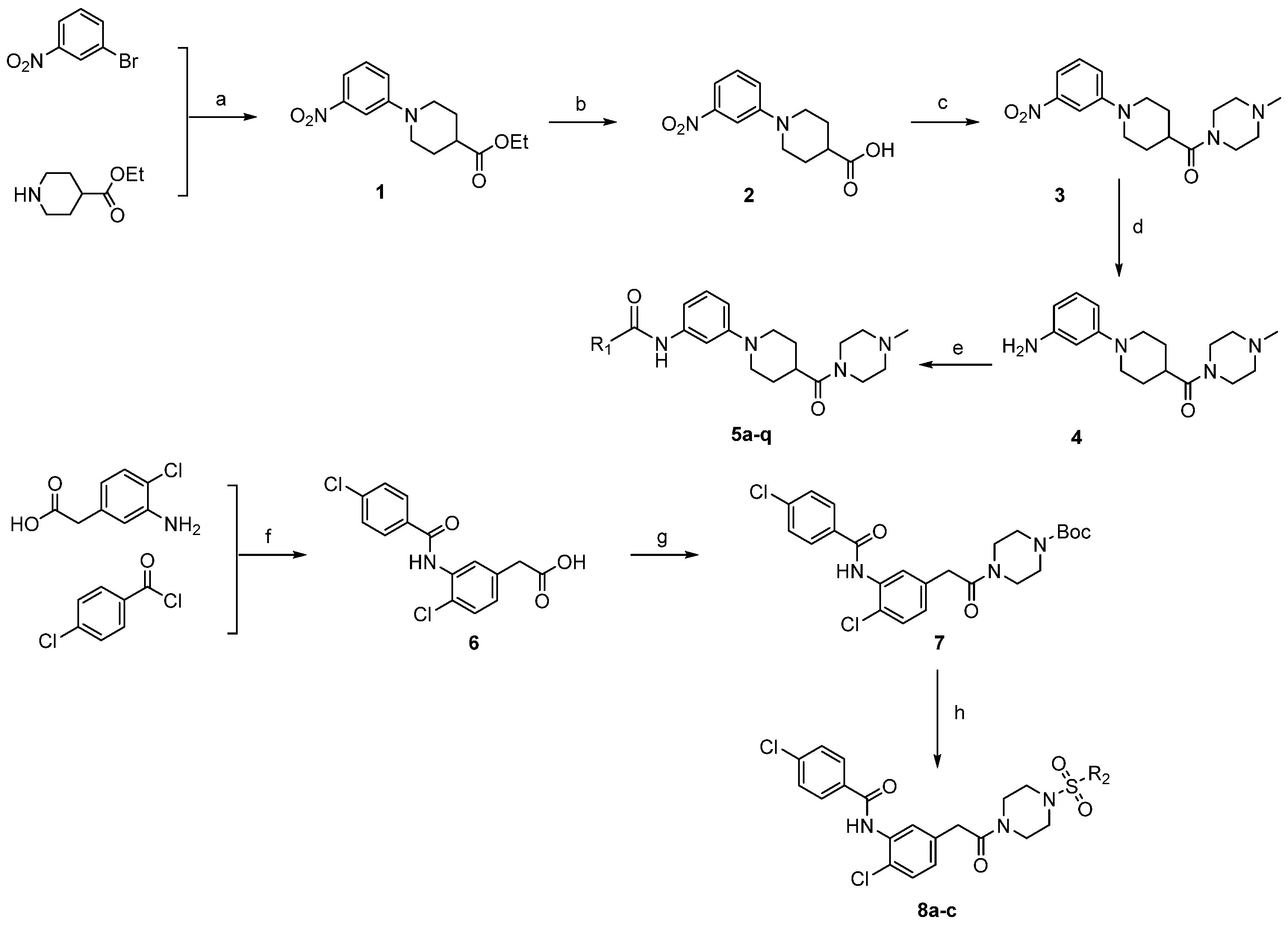
2.1. Chemistry
2.2. In Vitro Bioactivity
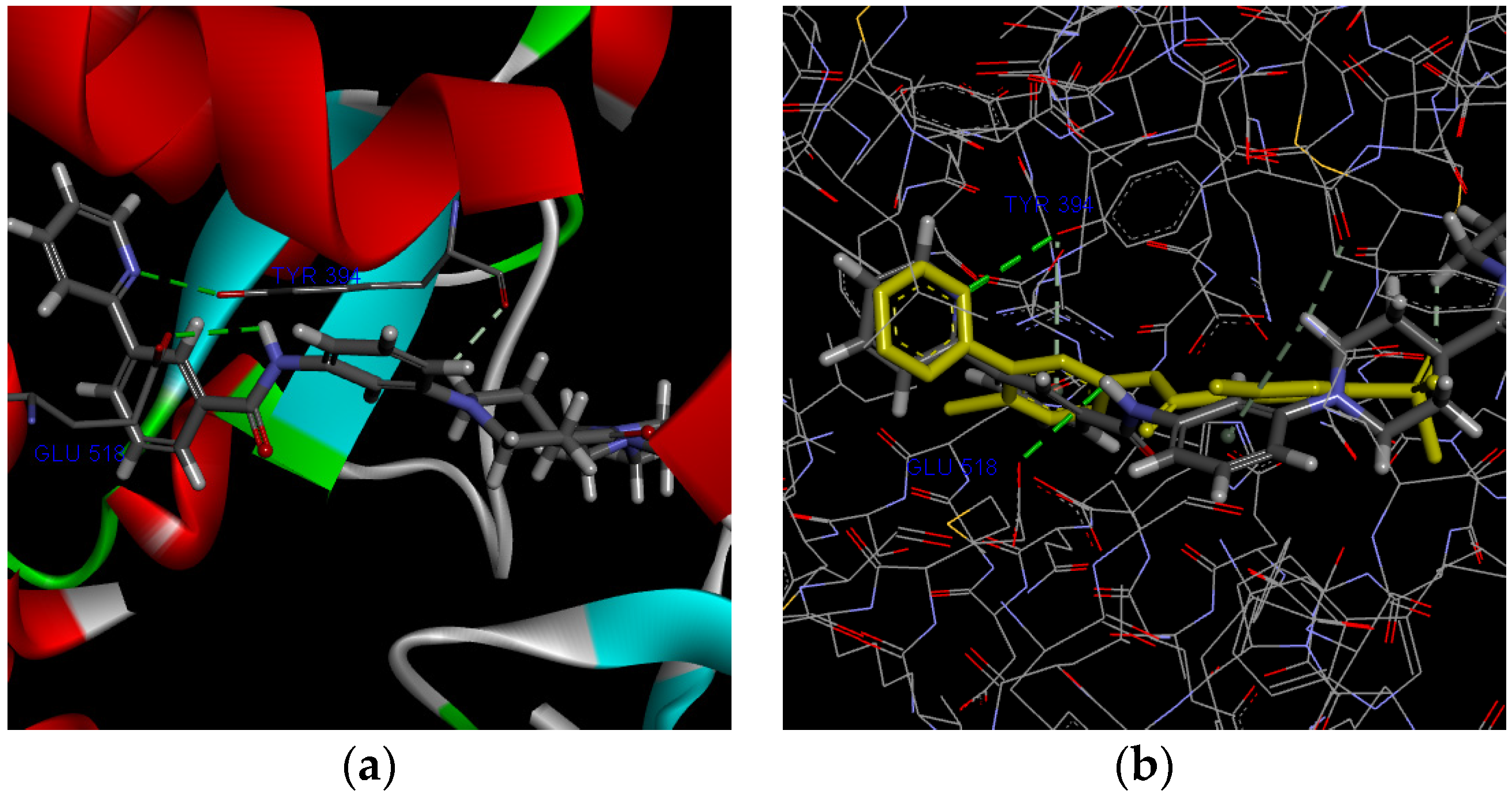
2.3. Molecular Docking and Simulation
3. Experimental
3.1. Materials and Reagents
3.2. Chemical Synthesis
3.2.1. Procedure for Preparation Intermediates of 1–4
3.2.2. General Procedure for Preparation of Target Compounds of 5a–q
3.2.3. Procedure for Preparation Intermediates of 6–7
3.2.4. General Procedure for Preparation of Target Compounds of 8a–c
3.3. Biological Methods
3.3.1. NIH3T3-Gli-Luciferase Reporter Assay
Cell Culture
Plating Cells
Transfection
Compound Treatment
Dual-Luciferase Assay
Data Analysis
3.3.2. Hsmo-BC Binding Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ma, X.L.; Sun, H.J.; Wang, Y.S.; Huang, S.H.; Xie, J.W.; Zhang, H.W. Study of Sonic hedgehog signaling pathway related molecules in gastric carcinoma. World J. Gastroenterol. 2006, 12, 3965–3969. [Google Scholar] [CrossRef] [PubMed]
- Pasca di Magliano, M.; Hebrok, M. Hedgehog signalling in cancer formation and maintenance. Nat. Rev. Cancer 2003, 3, 903–911. [Google Scholar] [CrossRef] [PubMed]
- Park, K.S.; Martelotto, L.G.; Peifer, M.; Sos, M.L.; Karnezis, A.N.; Mahjoub, M.R.; Bernard, K.; Conklin, J.F.; Szczepny, A.; Yuan, J.; et al. A crucial requirement for Hedgehog signaling in small cell lung cancer. Nat. Med. 2011, 17, 1504–1508. [Google Scholar] [CrossRef] [PubMed]
- Peacock, C.D.; Wang, Q.; Gesell, G.S.; Corcoran-Schwartz, I.M.; Jones, E.; Kim, J.; Devereux, W.L.; Rhodes, J.T.; Huff, C.A.; Beachy, P.A.; et al. Hedgehog Signaling Maintains a Tumor Stem Cell Compartment in Multiple Myeloma. Proc. Natl. Acad. Sci. USA 2007, 104, 4048–4053. [Google Scholar] [CrossRef] [PubMed]
- Winkler, J.D.; Isaacs, A.K.; Xiang, C.; Baubet, V.; Dahmane, N. Design, synthesis, and biological evaluation of estrone-derived hedgehog signaling inhibitors. Tetrahedron 2011, 67, 10261–10266. [Google Scholar] [CrossRef] [PubMed]
- Rudin, C.M. Vismodegib. Clin. Cancer Res. 2012, 18, 3218–3222. [Google Scholar] [CrossRef] [PubMed]
- Dessole, G.; Jones, P.; Bufi, L.L.; Muraglia, E.; Ontoria, J.M.O.; Torrisi, C. 1,2,4-OXADIAZOLE Substituted Piperidine and Piperazine Derivatives as Smo Antagonists. WO Patent WO/2010/013037 A1, 4 February 2010. [Google Scholar]
- Petrova, E.; Rios-Esteves, J.; Ouerfelli, O.; Glickman, J.F.; Resh, M.D. Inhibitors of Hedgehog acyltransferase block Sonic Hedgehog signaling. Nat. Chem. Biol. 2013, 9, 247–249. [Google Scholar] [CrossRef] [PubMed]
- Lauth, M.; Bergström, A.; Shimokawa, T.; Toftgård, R. Inhibition of GLI-mediated transcription and tumor cell growth by small-molecule antagonists. Proc. Natl. Acad. Sci. USA 2007, 104, 8455–8460. [Google Scholar] [CrossRef] [PubMed]
- Dahmane, N.; Winkler, J.D. Steroid-Derived Cyclopamine Analogs and Methods for Using the Same in the Prevention or Treatment of Cancer. U.S. Patent US8669243 B2, 11 March 2014. [Google Scholar]
- Winkler, J.D.; Isaacs, A.; Holderbaum, L.; Tatard, V.; Dahmane, N. Design and synthesis of inhibitors of Hedgehog signaling based on the alkaloid cyclopamine. Org. Lett. 2009, 11, 2824–2827. [Google Scholar] [CrossRef] [PubMed]
- Taipale, J.; Chen, J.K.; Cooper, M.K.; Wang, B.; Mann, R.K.; Milenkovic, L.; Scott, M.P.; Beachy, P.A. Effects of oncogenic mutations in Smoothened and Patchedcan be reversed by cyclopamine. Nature 2000, 406, 1005–1009. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, M.; Lescarbeau, A.; Grogan, M.; Tan, E.; Lin, G.; Austad, B.; Yu, L.; Behnke, M.; Nair, S.; Hagel, M.; et al. Discovery of a potent and orally active hedgehog pathway antagonist (IPI-926). J. Med. Chem. 2009, 52, 4400–4418. [Google Scholar] [CrossRef] [PubMed]
- Robarge, K.D.; Brunton, S.A.; Castanedo, G.M.; Cui, Y.; Dina, M.S.; Goldsmith, R.; Gould, S.E.; Guichert, O.; Gunzner, J.L.; Halladay, J.; et al. GDC-0449-a potent inhibitor of the hedgehog pathway. Bioorg. Med. Chem. Lett. 2009, 19, 5576–5581. [Google Scholar] [CrossRef] [PubMed]
- Munchhof, M.J.; Li, Q.; Shavnya, A.; Borzillo, G.V.; Boyden, T.L.; Jones, C.S.; Lagreca, S.D.; Martinez-Alsina, L.; Patel, N.; Pelletier, K.; et al. Discovery of PF-04449913, a Potent and Orally Bioavailable Inhibitor of Smoothened. ACS Med. Chem. Lett. 2011, 3, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.; Wu, X.; Jiang, J.; Gao, W.; Wan, Y.; Cheng, D.; Han, D.; Liu, J.; Englund, N.P.; Wang, Y.; et al. Discovery of NVP-LDE225, a Potent and Selective Smoothened Antagonist. ACS Med. Chem. Lett. 2010, 1, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, M.R.; Nesler, M.; Weatherhead, R.; Castro, A.C. Recent patents for Hedgehog pathway inhibitors for the treatment of malignancy. Expert Opin. Ther. Pat. 2009, 19, 1039–1056. [Google Scholar] [CrossRef] [PubMed]
- Manetti, F.; Faure, H.; Roudaut, H.; Gorojankina, T.; Traiffort, E.; Schoenfelder, A.; Mann, A.; Solinas, A.; Taddei, M.; Ruat, M. Virtual screening-based discovery and mechanistic characterization of the acylthiourea MRT-10 family as smoothened antagonists. Mol. Pharmacol. 2010, 78, 658–665. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Li, Y.; Shi, A.; Zhang, J.; Li, Y.; Zhao, M.; Zhang, L.; Zheng, H.; Meng, Y.; Ding, H. Synthesis and evaluation of novel N-3-benzimidazolephenylbisamide derivatives for antiproliferative and Hedgehog pathway inhibitory activity. Med. Chem. Commun. 2015, 6, 1137–1142. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds 5a–5q and 8a–8c are available from the authors. |

| Compd. | R | Gli-luc Reporter IC50 a (μmol/L) | Compd. | R | Gli-luc Reporter IC50 a (μmol/L) |
|---|---|---|---|---|---|
| 5a |  | 28.52 ± 0.02 | 5l |  | 1.83 ± 0.03 |
| 5b |  | 12.47 ± 0.11 | 5m |  | 10.49 ± 0.05 |
| 5c |  | 14.63 ± 0.01 | 5n |  | 5.32 ± 0.13 |
| 5d |  | 15.51 ± 0.03 | 5o |  | 4.28 ± 0.01 |
| 5e |  | 17.59 ± 0.15 | 5p |  | 3.43 ± 0.08 |
| 5f |  | 53.34 ± 0.07 | 5q |  | 1.26 ± 0.03 |
| 5g |  | 60.56 ± 0.04 | 8a |  | 6.88 ± 0.13 |
| 5h |  | 7.85 ± 0.01 | 8b |  | 12.7 ± 0.16 |
| 5i |  | 16.32 ± 0.05 | 8c |  | 8.62 ± 0.09 |
| 5j |  | 4.74 ± 0.02 | MRT10 b | 2.15 | |
| 5k |  | 16.61 ± 0.12 | Vismodegib b | 0.025 |
| Compound | Smo-BCB IC50 a (μM) |
|---|---|
| 5l | 2.41 ± 0.01 |
| 5n | 6.35 ± 0.07 |
| 5o | 6.23 ± 0.12 |
| 5q | 1.61 ± 0.02 |
| Vismodegib b | 0.0051 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ding, H.; Chen, K.; Song, B.; Deng, C.; Li, W.; Niu, L.; Bai, M.; Song, H.; Zhang, L. Synthesis and Smo Activity of Some Novel Benzamide Derivatives. Molecules 2018, 23, 85. https://doi.org/10.3390/molecules23010085
Ding H, Chen K, Song B, Deng C, Li W, Niu L, Bai M, Song H, Zhang L. Synthesis and Smo Activity of Some Novel Benzamide Derivatives. Molecules. 2018; 23(1):85. https://doi.org/10.3390/molecules23010085
Chicago/Turabian StyleDing, Huaiwei, Kai Chen, Bingke Song, Chenglong Deng, Wei Li, Li Niu, Mengxuan Bai, Hongrui Song, and Lijuan Zhang. 2018. "Synthesis and Smo Activity of Some Novel Benzamide Derivatives" Molecules 23, no. 1: 85. https://doi.org/10.3390/molecules23010085





