An Efficient, Eco-friendly and Sustainable One-Pot Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones Directly from Alcohols Catalyzed by Heteropolyanion-Based Ionic Liquids
Abstract
:1. Introduction
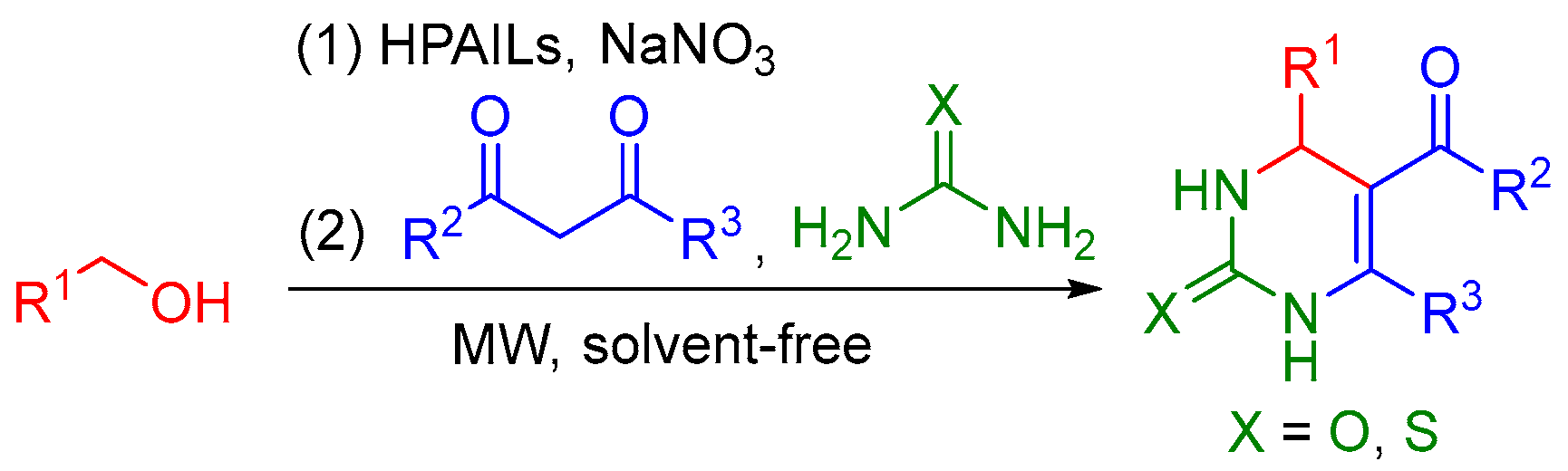
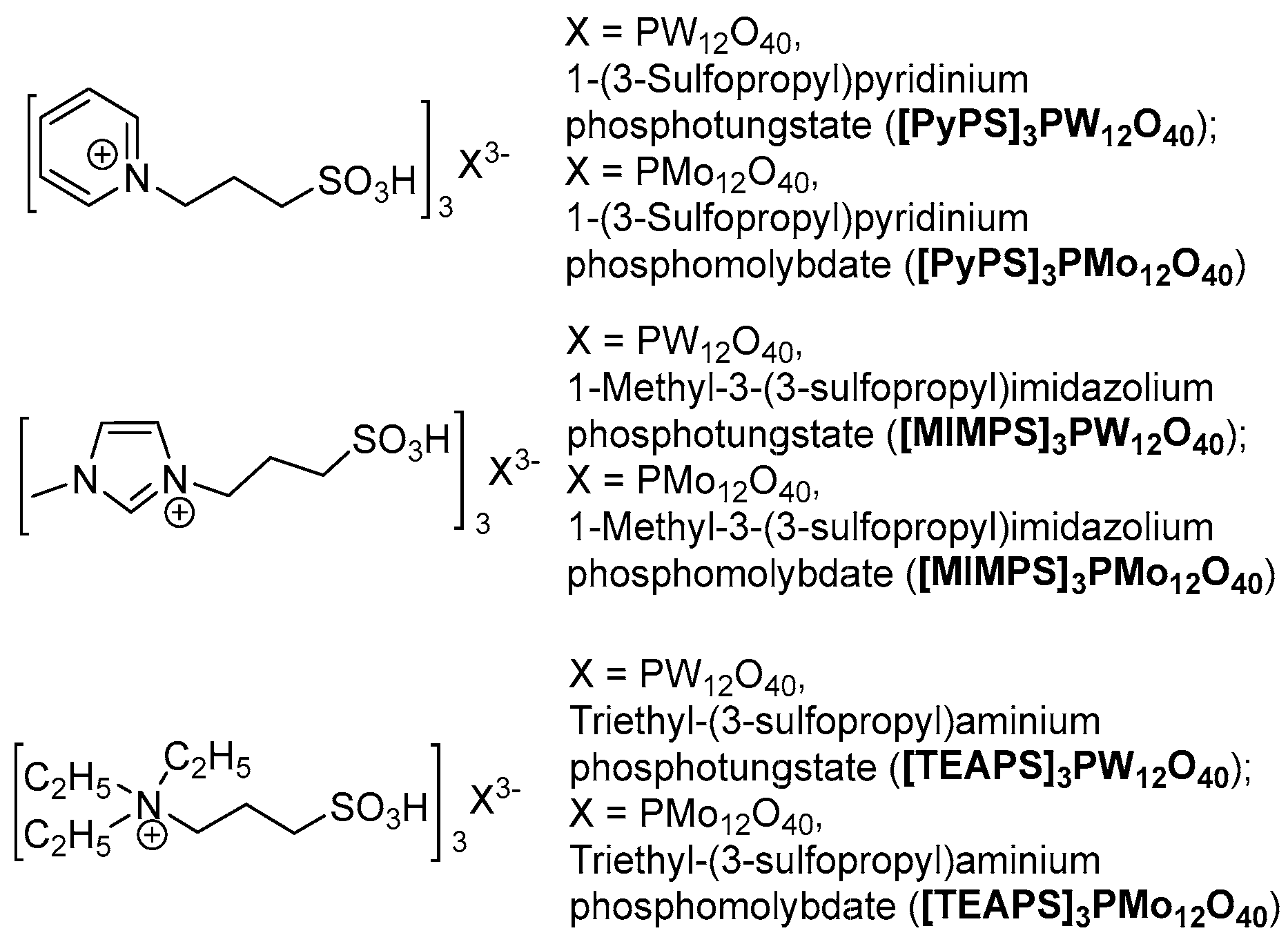
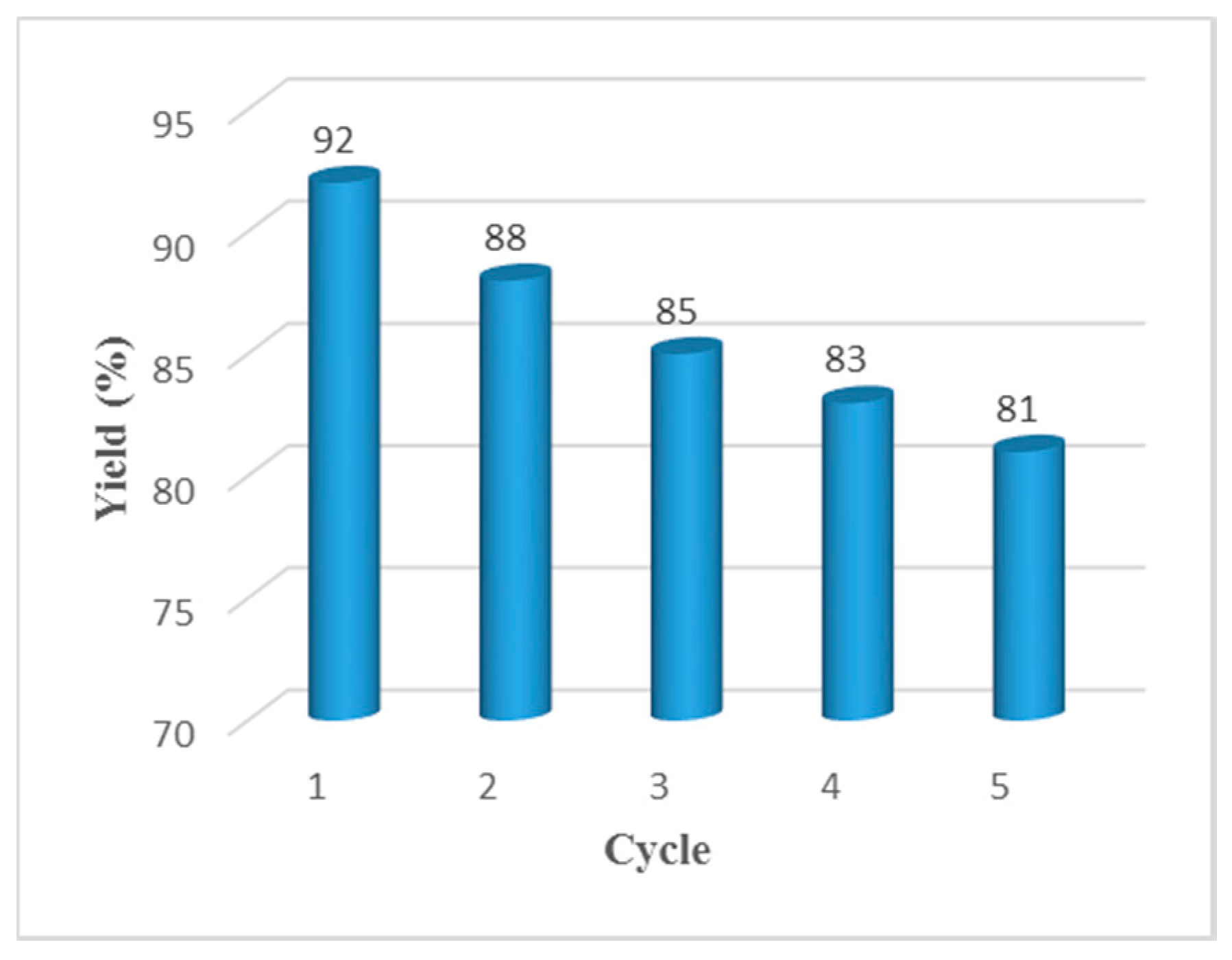
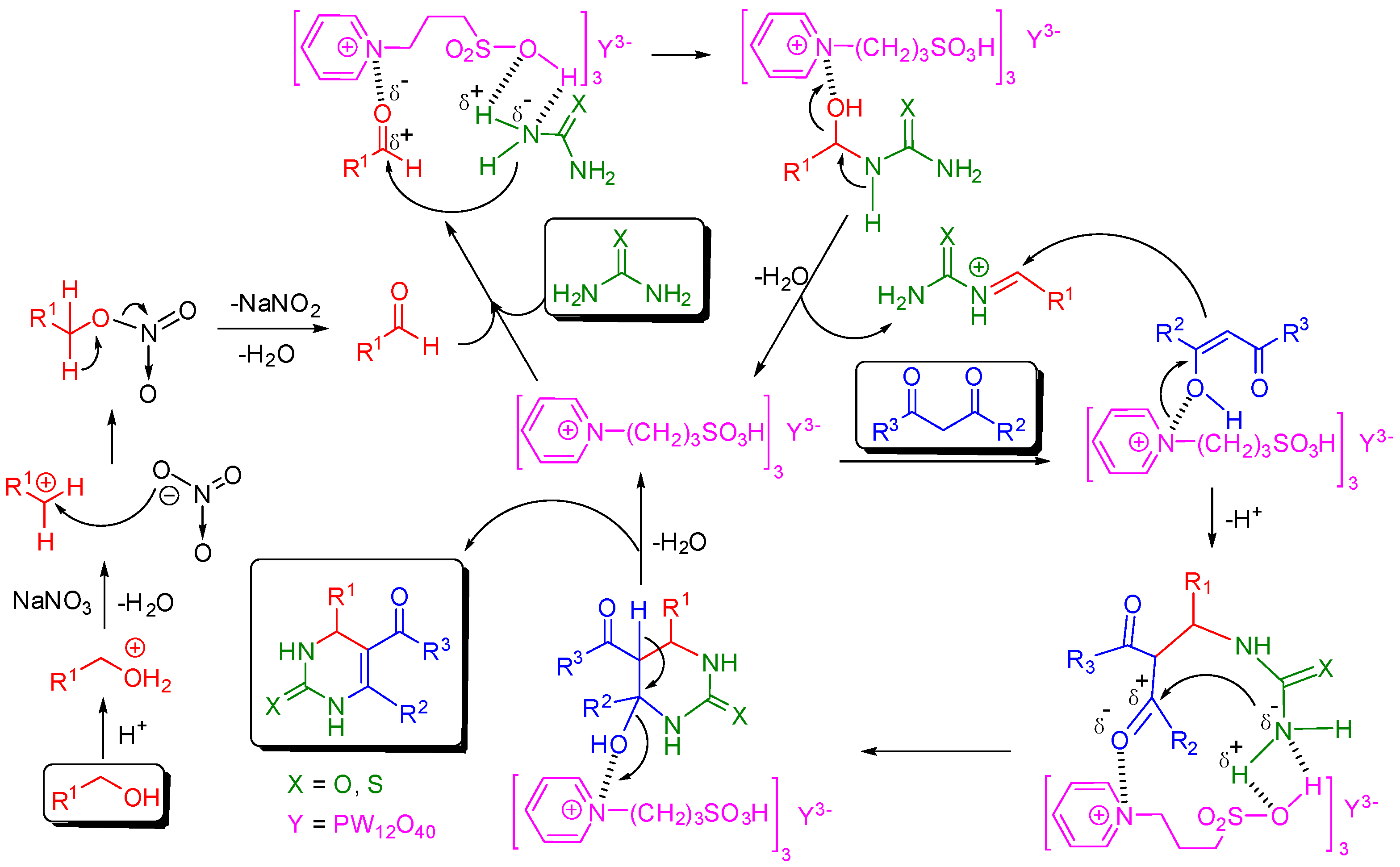
2. Results and Discussion
3. Materials and Methods
3.1. General Methods
3.2. General Procedure for the Synthesis of HPAILs
3.3. General Procedure for the Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tietze, L.F. Domino reactions in organic synthesis. Chem. Rev. 1996, 96, 115–136. [Google Scholar] [CrossRef] [PubMed]
- Climent, M.J.; Corma, A.; Iborra, S. Heterogeneous catalysts for the one-pot synthesis of chemicals and fine chemicals. Chem. Rev. 2011, 111, 1072–1133. [Google Scholar] [CrossRef] [PubMed]
- Pellissier, H. Stereocontrolled domino reactions. Chem. Rev. 2013, 113, 442–524. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, X.; Xue, W.; Chen, G.; Zhang, W.; Zhu, X. Initiator and photocatalyst-free visible light induced one-pot reaction: Concurrent RAFT polymerization and CuAAC Click reaction. Macromol. Rapid Commun. 2016, 37, 799–804. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.; Jiang, L.; Zuo, J.; Hor, T.S.A. Hybrid NS ligands supported Cu(I)/(II) complexes for azide-alkyne cycloaddition reactions. Dalton Trans. 2013, 42, 11319–11326. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.G.; Wang, Q.F.; Song, X.K.; Sun, J. One-step synthesis of pyrido[1,2-a]benzimidazole derivatives by a novel multicomponent reaction of chloroacetonitrile, malononitrile, aromatic aldehyde, and pyridine. J. Org. Chem. 2009, 74, 710–718. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Ermolat’ev, D.S.; Song, G.; Eycken, E.V.V. Synthesis of symmetric 1,4-diamino-2-butynes via a Cu(I)-catalyzed one-pot A3-coupling/decarboxylative coupling of a propiolic acid, an aldehyde, and an amine. J. Org. Chem. 2012, 77, 5149–5154. [Google Scholar] [CrossRef] [PubMed]
- Guchhait, S.K.; Chandgude, A.L.; Priyadarshani, G. CuSO4–glucose for in situ generation of controlled Cu(I)–Cu(II) bicatalysts: Multicomponent reaction of heterocyclic azine and aldehyde with alkyne, and cycloisomerization toward synthesis of N-fused imidazoles. J. Org. Chem. 2012, 77, 4438–4444. [Google Scholar] [CrossRef] [PubMed]
- Reid, M.; Rowe, D.J.; Taylor, R.J.K. Two carbon homologated α,β-unsaturated aldehydes from alcohols using the in situ oxidation–Wittig reaction. Chem. Commun. 2003, 2284–2285. [Google Scholar] [CrossRef]
- Taylor, R.J.K.; Reid, M.; Foot, J.; Raw, S.A. Tandem oxidation processes using manganese dioxide: discovery, applications, and current studies. Acc. Chem. Res. 2005, 38, 851–869. [Google Scholar] [CrossRef] [PubMed]
- Quesada, E.; Taylor, R.J.K. One-pot conversion of activated alcohols into terminal alkynes using manganese dioxide in combination with the Bestmann–Ohira reagent. Tetrahedron Lett. 2005, 46, 6473–6476. [Google Scholar] [CrossRef]
- Phillips, D.J.; Pillinger, K.S.; Wei, L.; Taylor, A.E.; Graham, A.E. Desymmetrization of diols by a tandem oxidation/Wittig olefination reaction. Chem. Commun. 2006, 2280–2282. [Google Scholar] [CrossRef] [PubMed]
- McAllister, G.D.; Oswald, M.F.; Paxton, R.J.; Raw, S.A.; Taylor, R.J.K. The direct preparation of functionalised cyclopropanes from allylic alcohols or α-hydroxyketones using tandem oxidation processes. Tetrahedron 2006, 62, 6681–6694. [Google Scholar] [CrossRef]
- Donald, J.R.; Edwards, M.G.; Taylor, R.J.K. Tandem oxime formation—Epoxide ring opening sequences for the preparation of oxazines related to the trichodermamides. Tetrahedron Lett. 2007, 48, 5201–5204. [Google Scholar] [CrossRef]
- Bromley, W.J.; Gibson, M.; Lang, S.; Raw, S.A.; Whitwood, A.C.; Taylor, R.J.K. Tandem inverse electron demand Diels–Alder, retro-Diels–Alder and intramolecular Diels–Alder sequences: One-pot synthesis of diaza-polycycles. Tetrahedron 2007, 63, 6004–6014. [Google Scholar] [CrossRef]
- Ekoue-Kovi, K.; Wolf, C. One-pot oxidative esterification and amidation of aldehydes. Chem. Eur. J. 2008, 14, 6302–6315. [Google Scholar] [CrossRef] [PubMed]
- Davi, M.; Lebel, H. Copper-Catalyzed Tandem Oxidation-Olefination Process. Org. Lett. 2009, 11, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Maki, B.E.; Scheidt, K.A. Single-flask synthesis of N-acylated indoles by catalytic dehydrogenative coupling with primary alcohols. Org. Lett. 2009, 11, 1651–1654. [Google Scholar] [CrossRef] [PubMed]
- Ngouansavanh, T.; Zhu, J. Alcohols in isonitrile-based multicomponent reaction: passerini reaction of alcohols in the presence of O-iodoxybenzoic acid. Angew. Chem. Int. Ed. 2006, 45, 3495–3497. [Google Scholar] [CrossRef] [PubMed]
- Brioche, J.; Masson, G.; Zhu, J. Passerini three-component reaction of alcohols under catalytic aerobic oxidative conditions. Org. Lett. 2010, 12, 1432–1435. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, M.B.; Salunkhe, S.M.; Patil, D.R.; Anbhule, P.V. A novel and efficient one step synthesis of 2-amino-5-cyano-6-hydroxy-4-aryl pyrimidines and their anti-bacterial activity. Eur. J. Med. Chem. 2009, 44, 2651–2654. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Arora, D.; Poremsk, E.; Lowery, J.; Moreland, R.S. N1-Alkylated 3,4-dihydropyrimidine-2(1H)-ones: Convenient one-pot selective synthesis and evaluation of their calcium channel blocking activity. Eur. J. Med. Chem. 2009, 44, 1997–2001. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.R.P.; Sankar, G.; Baig, R.B.N.; Chandrashekaran, S. Novel Biginelli dihydropyrimidines with potential anticancer activity: A parallel synthesis and CoMSIA study. Eur. J. Med. Chem. 2009, 44, 4192–4198. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, D.L.; Reis, F.S.; Muniz, D.R.; Ruiz, A.L.T.G.; de Carvalho, J.E.; Sabino, A.A.; Modolo, L.V.; de Fatima, A. Free radical scavenging and antiproliferative properties of Biginelli adducts. Bioorg. Med. Chem. 2012, 20, 2645–2650. [Google Scholar] [CrossRef] [PubMed]
- Bagal, S.K.; Brown, A.D.; Cox, P.J.; Omoto, K.; Owen, R.M.; Pryde, D.C.; Sidders, B.; Skerratt, S.E.; Stevens, E.B.; Storer, R.I.; et al. Ion channels as therapeutic targets: A drug discovery perspective. J. Med. Chem. 2013, 56, 593–624. [Google Scholar] [CrossRef] [PubMed]
- Kaur, R.; Chaudhary, S.; Kumar, K.; Gupta, M.K.; Rawal, R.K. Recent synthetic and medicinal perspectives of dihydropyrimidinones: A review. Eur. J. Med. Chem. 2017, 132, 108–134. [Google Scholar] [CrossRef] [PubMed]
- Kappe, C.O. Recent advances in the Biginelli dihydropyrimidine synthesis. New tricks from an old dog. Acc. Chem. Res. 2000, 33, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Dömling, A. Recent developments in isocyanide based multicomponent reactions in applied chemistry. Chem. Rev. 2006, 106, 17–89. [Google Scholar] [CrossRef] [PubMed]
- Dondoni, A.; Massi, A. Design and synthesis of new classes of heterocyclic c-glycoconjugates and carbon-linked sugar and heterocyclic amino acids by asymmetric multicomponent reactions (AMCRs). Acc. Chem. Res. 2006, 39, 451–463. [Google Scholar] [CrossRef] [PubMed]
- Nagarajaiah, H.; Mukhopadhyay, A.; Moorthy, J.N. Biginelli reaction: An overview. Tetrahedron Lett. 2016, 57, 5135–5149. [Google Scholar] [CrossRef]
- Xue, H.; Zhao, Y.; Wu, H.; Wang, Z.; Yang, B.; Wei, Y.; Wang, Z.; Tao, L. Multicomponent combinatorial polymerization via the Biginelli reaction. J. Am. Chem. Soc. 2016, 138, 8690–8693. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Liu, C.; Luo, G. One-pot synthesis of 3,4-dihydropyrimidin-2(1H)-ones using chloroacetic acid as catalyst. Bioorg. Med. Chem. Lett. 2007, 17, 3508–3510. [Google Scholar] [CrossRef] [PubMed]
- Polshettiwar, V.; Varma, R.S. Biginelli reaction in aqueous medium: A greener and sustainable approach to substituted 3,4-dihydropyrimidin-2(1H)-ones. Tetrahedron Lett. 2007, 48, 7343–7346. [Google Scholar] [CrossRef]
- Shobha, D.; Chari, M.A.; Ahn, K.H. An efficient Biginelli one-pot synthesis of new benzoxazole-substituted dihydropyrimidinones and thiones catalysed by trifluoro acetic acid under solvent-free conditions. Chin. Chem. Lett. 2009, 20, 1059–1061. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, J.; Yang, H.; Miao, Z.; Chen, R. Solvent-free Biginelli reaction: A green method for the synthesis of 3,4-dihydropyrimidin-2-ones catalyzed by protic acids in large-scale. Lett. Org. Chem. 2011, 8, 264–267. [Google Scholar] [CrossRef]
- Gore, S.; Baskaran, S.; Koenig, B. Efficient synthesis of 3,4-dihydropyrimidin-2-ones in low melting tartaric acid–urea mixtures. Green Chem. 2011, 13, 1009–1013. [Google Scholar] [CrossRef]
- Ren, Y.; Cai, C.; Yang, R. Molecular iodine-catalyzed multicomponent reactions: An efficient catalyst for organic synthesis. RSC Adv. 2013, 3, 7182–7204. [Google Scholar] [CrossRef]
- Barbero, M.; Cadamuro, S.; Dughera, S. A Brønsted acid catalysed enantioselective Biginelli reaction. Green Chem. 2017, 19, 1529–1535. [Google Scholar] [CrossRef]
- Shen, Z.; Xu, X.; Ji, S. Brønsted base-catalyzed one-pot three-component Biginelli-type reaction: An efficient synthesis of 4,5,6-triaryl-3,4-dihydropyrimidin-2(1H)-one and mechanistic study. J. Org. Chem. 2010, 75, 1162–1167. [Google Scholar] [CrossRef] [PubMed]
- Tamaddon, F.; Razmi, Z.; Jafari, A.A. Synthesis of 3,4-dihydropyrimidin-2(1H)-ones and 1,4-dihydropyridines using ammonium carbonate in water. Tetrahedron Lett. 2010, 51, 1187–1189. [Google Scholar] [CrossRef]
- Lannou, M.; Hélion, F.; Namy, J. Applications of lanthanide trichloride hydrates, prepared from mischmetall, in the Biginelli reaction. Synlett 2008, 2008, 105–107. [Google Scholar]
- Prodius, D.; Macaev, F.; Mereacre, V.; Shova, S.; Lutsenco, Y.; Styngach, E.; Ruiz, P.; Muraviev, D.; Lipkowski, J.; Simonov, Y.A.; et al. Synthesis and characterization of {Fe2CuO} clusters as precursors for nanosized catalytic system for Biginelli reaction. Inorg. Chem. Commun. 2009, 12, 642–645. [Google Scholar] [CrossRef]
- Chitra, S.; Pandiarajan, K. Calcium fluoride: an efficient and reusable catalyst for the synthesis of 3,4-dihydropyrimidin-2(1H)-ones and their corresponding 2(1H)thione: An improved high yielding protocol for the Biginelli reaction. Tetrahedron Lett. 2009, 50, 2222–2224. [Google Scholar] [CrossRef]
- Litvić, M.; Večenaj, I.; Ladišić, Z. M.; Lovrić, M.; Vinković, V.; Litvić, M.F. First application of hexaaquaaluminium(III) tetrafluoroborate as a mild, recyclable, non-hygroscopic acid catalyst in organic synthesis: A simple and efficient protocol for the multigram scale synthesis of 3,4-dihydropyrimidinones by Biginelli reaction. Tetrahedron 2010, 66, 3463–3471. [Google Scholar] [CrossRef]
- Sadek, K.U.; Al-Qalaf, F.; Abdelkhalik, M.M.; Elnagdi, M.H.J. Cerium (IV) ammonium nitrate as an efficient Lewis acid for the one-pot synthesis of 3,4-dihydropyrimidin-2(1H)-ones and their corresponding 2-(1H) thiones. Heterocycl. Chem. 2010, 47, 284–286. [Google Scholar]
- Narsaiah, A.V.; Reddy, A.R.; Yadav, J.S. Samarium triflate–catalyzed Biginelli condensation: An improved method for the synthesis of 3,4-dihydropyrimidin-2(1H)-ones. Synth. Commun. 2011, 41, 2794–2799. [Google Scholar] [CrossRef]
- Lei, M.; Ma, L.; Hu, L. Cu(ClO4)2∙6H2O as an efficient catalyst for the synthesis of 3,4-dihydropyrimidin-2(1H)-ones under solvent-free conditions. Synth. Commun. 2011, 41, 3071–3077. [Google Scholar] [CrossRef]
- Pasunooti, K.K.; Chai, H.; Jensen, C.N.; Gorityala, B.K.; Wang, S.; Liu, X. A microwave-assisted, copper-catalyzed three-component synthesis of dihydropyrimidinones under mild conditions. Tetrahedron Lett. 2011, 52, 80–84. [Google Scholar] [CrossRef]
- Starcevich, J.T.; Laughlin, T.J.; Mohan, R.S. Iron(III) tosylate catalyzed synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones via the Biginelli reaction. Tetrahedron Lett. 2013, 54, 983–985. [Google Scholar] [CrossRef]
- Nguyen, N.H.T.; Nguyen, P.P.T.; Nguyen, T.T.; Tran, M.T.; Huynh, T.T.H.; Tran, P.H. Au nanorod: An efficient catalyst for one-pot synthesis of 3,4-dihydropyrimidin-2(1H)-ones via the multicomponent Biginelli reaction. Chemistryselect 2017, 2, 3932–3936. [Google Scholar] [CrossRef]
- Mandhane, P.G.; Joshi, R.S.; Nagargoje, D.R.; Gill, C.H. An efficient synthesis of 3,4-dihydropyrimidin-2(1H)-ones catalyzed by thiamine hydrochloride in water under ultrasound irradiation. Tetrahedron Lett. 2010, 51, 3138–3140. [Google Scholar] [CrossRef]
- Rao, G.B.D.; Acharya, B.N.; Verma, S.K.; Kaushik, M.P. N,N′-Dichlorobis(2,4,6-trichlorophenyl)urea (CC-2) as a new reagent for the synthesis of pyrimidone and pyrimidine derivatives via Biginelli reaction. Tetrahedron Lett. 2011, 52, 809–812. [Google Scholar] [CrossRef]
- Silva, D.L.; Fernandes, S.A.; Sabino, A.A.; Fátima, Â. p-Sulfonic acid calixarenes as efficient and reusable organocatalysts for the synthesis of 3,4-dihydropyrimidin-2(1H)-ones/-thiones. Tetrahedron Lett. 2011, 52, 6328–6330. [Google Scholar] [CrossRef]
- Verma, S.; Jain, S.L. Thiourea dioxide in water as a recyclable catalyst for the synthesis of structurally diverse dihydropyrido[2,3-d]pyrimidine-2,4-diones. Tetrahedron Lett. 2012, 53, 2595–2600. [Google Scholar] [CrossRef]
- Xu, F.; Huang, D.; Lin, X.; Wang, Y. Highly enantioselective Biginelli reaction catalyzed by SPINOL-phosphoric acids. Org. Biomol. Chem. 2012, 10, 4467–4470. [Google Scholar] [CrossRef] [PubMed]
- Nagarapu, L.; Gaikwad, H.K.; Palem, J.D.; Venkatesh, R.; Bantu, R.; Sridhar, B. Convenient approach for the one-pot, three-component synthesis of triheterocyclic 4H-pyrimido[2,1-b]benzothiazole derivatives using TBAHS. Synth. Commun. 2013, 43, 93–104. [Google Scholar] [CrossRef]
- Puripat, M.; Ramozzi, R.; Hatanaka, M.; Parasuk, W.; Parasuk, V. The Biginelli reaction is a urea-catalyzed organocatalytic multicomponent reaction. J. Org. Chem. 2015, 80, 6959–6967. [Google Scholar] [CrossRef] [PubMed]
- Khatri, C.K.; Rekunge, D.S.; Chaturbhuj, G.U. Sulfated polyborate: A new and eco-friendly catalyst for one-pot multicomponent synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones via Biginelli reaction. New J. Chem. 2016, 40, 10412–10417. [Google Scholar] [CrossRef]
- Cheng, Q.; Wang, Q.; Xu, X.; Ruan, M.; Yao, H.; Yang, X. Solvent-free synthesis of monastrol derivatives catalyzed by NaHSO4. J. Heterocycl. Chem. 2010, 47, 624–628. [Google Scholar] [CrossRef]
- Shobha, D.; Chari, M.A.; Mano, A.; Selvan, S.T.; Mukkanti, K.; Vinu, A. Synthesis of 3,4-dihydropyrimidin-2-ones (DHPMs) using mesoporous aluminosilicate (AlKIT-5) catalyst with cage type pore structure. Tetrahedron 2009, 65, 10608–10611. [Google Scholar] [CrossRef]
- Verma, S.; Jain, S.L.; Sain, B. PEG-embedded thiourea dioxide (PEG.TUD) as a novel organocatalyst for the highly efficient synthesis of 3,4-dihydropyrimidinones. Tetrahedron Lett. 2010, 51, 6897–6900. [Google Scholar] [CrossRef]
- Murata, H.; Ishitani, H.; Iwamoto, M. Synthesis of Biginelli dihydropyrimidinone derivatives with various substituents on aluminium-planted mesoporous silica catalyst. Org. Biomol. Chem. 2010, 8, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Regati, S.; Butcher, R.J.; Arman, H.D.; Chen, Z.; Xiang, S.; Chen, B.; Zhao, C. Hydrogen-bonding 2D metal–organic solids as highly robust and efficient heterogeneous green catalysts for Biginelli reaction. Tetrahedron Lett. 2011, 52, 6220–6222. [Google Scholar] [CrossRef] [PubMed]
- Konkala, K.; Sabbavarapu, N.M.; Katla, R.; Durga, N.Y.V.; Reddy, T.V.K.; Bethala, L.A.P.D.; Rachapudi, B.N.P. Revisit to the Biginelli reaction: a novel and recyclable bioglycerol-based sulfonic acid functionalized carbon catalyst for one-pot synthesis of substituted 3,4-dihydropyrimidin-2-(1H)-ones. Tetrahedron Lett. 2012, 53, 1968–1973. [Google Scholar] [CrossRef]
- Narahari, S.R.; Reguri, B.R.; Gudaparthi, O.; Mukkanti, K. Synthesis of dihydropyrimidinones via Biginelli multicomponent reaction. Tetrahedron Lett. 2012, 53, 1543–1545. [Google Scholar] [CrossRef]
- Sahu, P.K.; Sahu, P.K.; Agarwal, D.D. Efficient and facile synthesis of heterocycles and their mechanistic consideration using kaolin. RSC Adv. 2013, 3, 9854–9864. [Google Scholar] [CrossRef]
- Shi, X.; Yang, H.; Tao, M.; Zhang, W. Sulfonic acid-functionalized polypropylene fiber: highly efficient and recyclable heterogeneous Brønsted acid catalyst. RSC Adv. 2013, 3, 3939–3945. [Google Scholar] [CrossRef]
- Shen, P.; Xu, M.; Yin, D.; Xie, S.; Zhou, C.; Li, F. Halogenated macroporous sulfonic resins as efficient catalysts for the Biginelli reaction. Catal. Commun. 2016, 77, 18–21. [Google Scholar] [CrossRef]
- Sheykhan, M.; Yahyazadeh, A.; Ramezani, L. A novel cooperative Lewis acid/Brønsted base catalyst Fe3O4@SiO2-APTMS-Fe(OH)2: An efficient catalyst for the Biginelli reaction. Mol. Catal. 2017, 435, 166–173. [Google Scholar] [CrossRef]
- Zolfagharinia, S.; Kolvari, E.; Koukabi, N. A new type of magnetically-recoverable heteropolyacid nanocatalyst supported on zirconia-encapsulated Fe3O4 nanoparticles as a stable and strong solid acid for multicomponent reactions. Catal. Lett. 2017, 147, 1551–1566. [Google Scholar] [CrossRef]
- Ghosh, B. K.; Moitra, D.; Chandel, M.; Patra, M.K.; Vadera, S.R.; Ghosh, N. N. CuO nanoparticle immobilised mesoporous TiO2–cobalt ferrite nanocatalyst: A versatile, magnetically separable and reusable catalyst. Catal. Lett. 2017, 147, 1061–1076. [Google Scholar] [CrossRef]
- Khosropour, A.R.; Khodaei, M.M.; Beygzadeh, M.; Jokar, M. A one-pot synthesis of 3,4-dihydropyrimidin-2-(1H)-ones from primary alcohols promoted by Bi(NO3)3·5H2O in two different media: organic solvent and ionic liquid. Heterocycles 2005, 65, 767–773. [Google Scholar] [CrossRef]
- Garima; Srivastava, V.P.; Yadav, L.D.S. Biginelli reaction starting directly from alcohols. Tetrahedron Lett. 2010, 51, 6436–6438. [Google Scholar] [CrossRef]
- Kolvari, E.; Zolfigol, M.A.; Mirzaeean, M. Aluminium nitrate nonahydrate (Al(NO3)3∙9H2O): An efficient oxidant catalyst for the one-pot synthesis of Biginelli compounds from benzyl alcohols. Helv. Chim. Acta 2012, 95, 115–119. [Google Scholar] [CrossRef]
- Sahoo, P.K.; Bose, A.; Mal, P. Solvent-free ball-milling Biginelli reaction by subcomponent synthesis. Eur. J. Org. Chem. 2015, 2015, 6994–6998. [Google Scholar] [CrossRef]
- Simon, M.; Li, C. Green chemistry oriented organic synthesis in water. Chem. Soc. Rev. 2012, 41, 1415–1427. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Poliakoff, M. Continuous reactions in supercritical carbon dioxide: Problems, solutions and possible ways forward. Chem. Soc. Rev. 2012, 41, 1428–1436. [Google Scholar] [CrossRef] [PubMed]
- Baig, R.B.N.; Varma, R. S. Alternative energy input: Mechanochemical, microwave and ultrasound-assisted organic synthesis. Chem. Soc. Rev. 2012, 41, 1559–1584. [Google Scholar] [CrossRef] [PubMed]
- Xin, B.W.; Hao, J.C. Imidazolium-based ionic liquids grafted on solid surfaces. Chem. Soc. Rev. 2014, 43, 7171–7187. [Google Scholar] [CrossRef] [PubMed]
- Wagh, K.V.; Bhanage, B.M. Synthesis of 2-phenylnaphthalenes from styrene oxides using a recyclable Brønsted acidic [HNMP]+HSO4− ionic liquid. Green Chem. 2015, 17, 4446–4451. [Google Scholar] [CrossRef]
- Bourlinos, A.B.; Raman, K.; Herrera, R.; Zhang, Q.; Archer, L.A.; Giannelis, E.P. A liquid derivative of 12-tungstophosphoric acid with unusually high conductivity. J. Am. Chem. Soc. 2004, 126, 15358–15359. [Google Scholar] [CrossRef] [PubMed]
- Rickert, P.G.; Antonio, M.R.; Firestone, M.A.; Kubatko, K.A.; Szreder, T.; Wishart, J.F.; Dietz, M.L. Tetraalkylphosphonium polyoxometalate ionic liquids: Novel, organic-inorganic hybrid materials. J. Phys. Chem. B 2007, 111, 4685–4692. [Google Scholar] [CrossRef] [PubMed]
- Rickert, P.G.; Antonio, M.R.; Firestone, M.; Kubatko, A.K.A.; Szreder, T.; Wishart, J.F.; Dietz, M.L. Tetraalkylphosphonium polyoxometalates: Electroactive, “task-specific” ionic liquids. Dalton Trans. 2007, 529–531. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.; Kai, D.; Ke, K.L.; Lin, M.; Jiang, L.; Jiang, Y.; Young, D.J.; Loh, X.J.; Li, X.; Hor, T.S.A. A triazolyl-pyridine-supported CuI dimer: Tunable luminescence and fabrication of composite fibers. ChemPlusChem 2015, 80, 1235–1240. [Google Scholar] [CrossRef]
- Teeuwen, R.L.M.; van Berkel, S.S.; van Dulmen, T.H.H.; Schoffelen, S.; Meeuwissen, S.A.; Zuilhof, H.; de Wolf, F.A.; van Hest, J.C.M. “Clickable” elastins: Elastin-like polypeptides functionalized with azide or alkyne groups. Chem. Commun. 2009, 0, 4022–4024. [Google Scholar] [CrossRef] [PubMed]
- Leng, Y.; Wang, J.; Zhu, D.; Ren, X.; Ge, H.; Shen, L. Heteropolyanion-based ionic liquids: Reaction-induced self-separation catalysts for esterification. Angew. Chem. Int. Ed. 2009, 48, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Leng, Y.; Zhu, D.; Wu, Y.; Wang, J. Phosphotungstic acid salt of triphenyl(3-sulfopropyl)phosphonium: An efficient and reusable solid catalyst for esterification. Catal. Commun. 2009, 11, 151–154. [Google Scholar] [CrossRef]
- Li, H.; Qiao, Y.; Hua, L.; Hou, Z.; Feng, B.; Pan, Z.; Hu, Y.; Wang, X.; Zhao, X.; Yu, Y. Imidazolium polyoxometalate: An ionic liquid catalyst for esterification and oxidative esterification. ChemCatChem 2010, 2, 1165–1170. [Google Scholar] [CrossRef]
- Zhang, W.; Leng, Y.; Zhao, P.; Wang, J.; Zhu, D.; Huang, J. Heteropolyacid salts of N-methyl-2-pyrrolidonium as highly efficient and reusable catalysts for Prins reactions of styrenes with formalin. Green Chem. 2011, 13, 832–834. [Google Scholar] [CrossRef]
- Fang, D.; Wang, F.; Wang, L.; Wu, Y.; Yang, J.; Qian, C. Regioselective mononitration of aromatic compounds catalyzed by heteropolyanions-based acidic ionic liquids. Curr. Catal. 2012, 1, 197–201. [Google Scholar] [CrossRef]
- Zhang, X.; Mao, D.; Leng, Y.; Zhou, Y.; Wang, J. Heterogeneous beckmann rearrangements catalyzed by a sulfonated imidazolium salt of phosphotungstate. Catal. Lett. 2013, 143, 193–199. [Google Scholar] [CrossRef]
- Qiao, Y.; Hou, Z.; Li, H.; Hu, Y.; Feng, B.; Wang, X.; Hua, L.; Huang, Q. Polyoxometalate-based protic alkylimidazolium salts as reaction-induced phase-separation catalysts for olefin epoxidation. Green Chem. 2009, 11, 1955–1960. [Google Scholar] [CrossRef]
- Leng, Y.; Wang, J.; Zhu, D.; Zhang, M.; Zhao, P.; Long, Z.; Huang, J. Polyoxometalate-based amino-functionalized ionic solid catalysts lead to highly efficient heterogeneous epoxidation of alkenes with H2O2. Green Chem. 2011, 13, 1636–1639. [Google Scholar] [CrossRef]
- Leng, Y.; Wang, J.; Zhu, D.; Shen, L.; Zhao, P.; Zhang, M. Heteropolyanion-based ionic hybrid solid: A green bulk-type catalyst for hydroxylation of benzene with hydrogen peroxide. Chem. Eng. J. 2011, 173, 620–626. [Google Scholar] [CrossRef]
- Leng, Y.; Zhang, W.; Wang, J.; Jiang, P. A novel heteropolyanion-based amino-containing cross-linked ionic copolymer catalyst for epoxidation of alkenes with H2O2. Appl. Catal. A-Gen. 2012, 445–446, 306–311. [Google Scholar] [CrossRef]
- Leng, Y.; Liu, J.; Zhang, C.; Jiang, P. A polyhedral oligomeric silsesquioxane (POSS)-bridged oxo-molybdenum Schiff base complex with enhanced heterogeneous catalytic activity in epoxidation. Catal. Sci. Technol. 2014, 4, 997–1004. [Google Scholar] [CrossRef]
- Long, Z. Y.; Zhou, Y.; Chen, G. J.; Ge, W.L.; Wang, J. C3N4-H5PMo10V2O40: A dual-catalysis system for reductant-free aerobic oxidation of benzene to phenol. Sci. Rep. 2014, 4, 3651. [Google Scholar] [CrossRef] [PubMed]
- Leng, Y.; Liu, J.; Jiang, P.; Wang, J. POSS-derived mesostructured amphiphilic polyoxometalate-based ionic hybrids as highly efficient epoxidation catalysts. ACS Sustain. Chem. Eng. 2015, 3, 170–176. [Google Scholar] [CrossRef]
- Leng, Y.; Zhao, J.; Jiang, P.; Wang, J. Amphiphilic porous polyhedral oligomeric silsesquioxanes (POSS) incorporated polyoxometalate-paired polymeric hybrids: Interfacial catalysts for epoxidation reactions. RSC Adv. 2015, 5, 17709–17715. [Google Scholar] [CrossRef]
- Dallinger, D.; Kappe, C.O. Microwave-assisted synthesis in water as solvent. Chem. Rev. 2007, 107, 2563–2591. [Google Scholar] [CrossRef] [PubMed]
- Kappe, C.O. Microwave dielectric heating in synthetic organic chemistry. Chem. Soc. Rev. 2008, 37, 1127–1139. [Google Scholar] [CrossRef] [PubMed]
- Polshettiwar, V.; Varma, R.S. Microwave-Assisted Organic Synthesis and Transformations using Benign Reaction Media. Acc. Chem. Res. 2008, 41, 629–639. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Fu, R.; Chai, W.; Zheng, H.; Sun, L.; Lu, Q.; Yuan, R. An eco-benign and highly efficient procedure for N-acylation catalyzed by heteropolyanion-based ionic liquids using carboxylic acid under solvent-free conditions. Tetrahedron 2014, 70, 2237–2245. [Google Scholar] [CrossRef]
- Fu, R.; Yang, Y.; Chen, Z.; Lai, W.; Ma, Y.; Wang, Q.; Yuan, R. Microwave-assisted heteropolyanion-based ionic liquids catalyzed transamidation of non-activated carboxamides with amines under solvent-free conditions. Tetrahedron 2014, 70, 9492–9499. [Google Scholar] [CrossRef]
- Wang, Q.; Feng, J.; Chai, W.; Geng, H.; Xu, M.; Wang, K.; Xu, C.; Fu, R.; Yuan, R. Bu4NI-catalyzed construction of C–O bonds by oxidative coupling of alcohols with ethers. Tetrahedron Lett. 2014, 55, 4785–4789. [Google Scholar] [CrossRef]
- Wang, Q.; Zheng, H.; Chai, W.; Chen, D.; Zeng, X.; Fu, R.; Yuan, R. Copper catalyzed C–O bond formation via oxidative cross-coupling reaction of aldehydes and ethers. Org. Biomol. Chem. 2014, 12, 6549–6553. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Geng, H.; Chai, W.; Zeng, X.; Xu, M.; Zhu, C.; Fu, R.; Yuan, R. Copper-catalyzed formation of C–O bonds by oxidative coupling of benzylic alcohols with ethers. Eur. J. Org. Chem. 2014, 6850–6853. [Google Scholar] [CrossRef]
- Fu, R.; Yang, Y.; Ma, Y.; Yang, F.; Li, J.; Chai, W.; Wang, Q.; Yuan, R. Microwave-promoted direct amidation of unactivated esters catalyzed by heteropolyanion-based ionic liquids under solvent-free conditions. Tetrahedron Lett. 2015, 56, 4527–4531. [Google Scholar] [CrossRef]
- Fu, R.; Yang, Y.; Lai, W.; Ma, Y.; Chen, Z.; Zhou, J.; Chai, W.; Wang, Q.; Yuan, R. Efficient and green microwave-assisted multicomponent Biginelli reaction for the synthesis of dihydropyrimidinones catalyzed by heteropolyanion-based ionic liquids under solvent-free conditions. Synth. Commun. 2015, 45, 477–487. [Google Scholar] [CrossRef]
- Fu, R.; Yang, Y.; Zhang, J.; Shao, J.; Xia, X.; Ma, Y.; Yuan, R. Direct oxidative amidation of aldehydes with amines catalyzed by heteropolyanion-based ionic liquids under solvent-free conditions via a dual-catalysis process. Org. Biomol. Chem. 2016, 14, 1784–1793. [Google Scholar] [CrossRef] [PubMed]
- Fu, R.; Yang, Y.; Jin, W.; Gu, H.; Zeng, X.; Chai, W.; Ma, Y.; Wang, Q.; Yi, J.; Yuan, R. Microwave-assisted heteropolyanion-based ionic liquid promoted sustainable protocol to N-heteroaryl amides via N-directing dual catalyzed oxidative amidation of aldehydes. RSC Adv. 2016, 6, 107699–107707. [Google Scholar] [CrossRef]
- Fu, R.; Yang, Y.; Feng, W.; Ge, Q.; Feng, Y.; Zeng, X.; Chai, W.; Yi, J.; Yuan, R. An efficient, eco-friendly and sustainable tandem oxidative amidation of alcohols with amines catalyzed by heteropolyanion-based ionic liquids via a bifunctional catalysis process. Tetrahedron 2016, 72, 8319–8326. [Google Scholar] [CrossRef]
- Folkers, K.; Harwood, H.J.; Johnson, T.B. Researches on pyrimidines. Cxxx. Synthesis of 2-keto-1,2,3,4-tetrahydropyrimidines. J. Am. Chem. Soc. 1932, 54, 3751–3758. [Google Scholar] [CrossRef]
- Ling, R.; Yoshida, M.; Mariano, P.S. Exploratory investigations probing a preparatively versatile, pyridinium salt photoelectrocyclization-solvolytic aziridine ring opening sequence. J. Org. Chem. 1996, 61, 4439–4449. [Google Scholar] [CrossRef] [PubMed]
- Kappe, C.O. A reexamination of the mechanism of the Biginelli dihydropyrimidine synthesis. Support for an N-acyliminium ion intermediate. J. Org. Chem. 1997, 62, 7201–7204. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: All samples are available from the authors. |
| Entry | Catalyst (mol %) | T1 b/T2 c (°C) | t1 d/t2 e (min) | Yield f (%) |
|---|---|---|---|---|
| 1 g | [PyPS]3PW12O40 (3) | 80 | 120 | 13 |
| 2 | [PyPS]3PW12O40 (3) | 80/80 | 5/30 | 51 |
| 3 | [PyPS]3PW12O40 (3) | 100/80 | 5/30 | 35 |
| 4 | [PyPS]3PW12O40 (3) | 80/100 | 5/10 | 78 |
| 5 | [PyPS]3PW12O40 (3) | 80/120 | 5/5 | 90 |
| 6 | [PyPS]3PW12O40 (4) | 80/120 | 5/5 | 92 |
| 7 | [PyPS]3PW12O40 (5) | 80/120 | 5/5 | 91 |
| 8 | [PyPS]3PMo12O40 (4) | 80/120 | 5/5 | 84 |
| 9 | [MIMPS]3PW12O40 (4) | 80/120 | 5/5 | 90 |
| 10 | [MIMPS]3PMo12O40 (4) | 80/120 | 5/5 | 80 |
| 11 | [TEAPS]3PW12O40 (4) | 80/120 | 5/5 | 78 |
| 12 | [TEAPS]3PMo12O40 (4) | 80/120 | 5/5 | 71 |
| 13 | H3PW12O40 (4) | 80/120 | 5/5 | 63 |

| Entry | R1 | R2 | X | t1 b/t2 c (min) | Yield d (%) |
|---|---|---|---|---|---|
| 1 | C6H5 | OC2H5 | O | 5/5 | 92 |
| 2 | 4-CH3O-C6H4 | OC2H5 | O | 5/5 | 94 |
| 3 | 4-CH3-C6H4 | OC2H5 | O | 5/5 | 93 |
| 4 | 4-Cl-C6H4 | OC2H5 | O | 5/5 | 90 |
| 5 | 4-NO2-C6H4 | OC2H5 | O | 5/8 | 85 |
| 6 | 4-OH-C6H4 | OC2H5 | O | 5/5 | 90 |
| 7 | 2-OH-C6H4 | OC2H5 | O | 5/8 | 83 |
| 8 | 2-Furyl | OC2H5 | O | 5/8 | 84 |
| 9 | n-C6H13 | OC2H5 | O | 5/8 | 73 |
| 10 | i-Pr | OC2H5 | O | 5/8 | 71 |
| 11 | C6H5 | OC2H5 | S | 5/10 | 90 |
| 12 | 4-CH3-C6H4 | OC2H5 | S | 5/10 | 91 |
| 13 | 4-Cl-C6H4 | OC2H5 | S | 5/10 | 90 |
| 14 | 4-NO2-C6H4 | OC2H5 | S | 8/10 | 83 |
| 15 | C6H5 | OCH3 | O | 5/5 | 92 |
| 16 | 4-CH3O-C6H4 | OCH3 | O | 5/5 | 95 |
| 17 | 4-CH3-C6H4 | OCH3 | O | 5/5 | 91 |
| 18 | 4-Cl-C6H4 | OCH3 | O | 5/5 | 89 |
| 19 | 4-NO2-C6H4 | OCH3 | O | 8/8 | 80 |
| 20 | C6H5 | OCH3 | S | 5/10 | 88 |
| 21 | 4-CH3-C6H4 | OCH3 | S | 5/10 | 90 |
| 22 | 4-NO2-C6H4 | OCH3 | S | 5/10 | 78 |
| 23 | C6H5 | CH3 | O | 5/5 | 91 |
| 24 | 4-CH3-C6H4 | CH3 | O | 5/5 | 92 |
| 25 | 4-NO2-C6H4 | CH3 | O | 8/5 | 80 |
| 26 | C6H5 | CH3 | S | 5/5 | 88 |
| 27 | 4-CH3-C6H4 | CH3 | S | 5/5 | 90 |
| 28 | 4-NO2-C6H4 | CH3 | S | 8/5 | 77 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, R.; Yang, Y.; Ma, X.; Sun, Y.; Li, J.; Gao, H.; Hu, H.; Zeng, X.; Yi, J. An Efficient, Eco-friendly and Sustainable One-Pot Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones Directly from Alcohols Catalyzed by Heteropolyanion-Based Ionic Liquids. Molecules 2017, 22, 1531. https://doi.org/10.3390/molecules22091531
Fu R, Yang Y, Ma X, Sun Y, Li J, Gao H, Hu H, Zeng X, Yi J. An Efficient, Eco-friendly and Sustainable One-Pot Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones Directly from Alcohols Catalyzed by Heteropolyanion-Based Ionic Liquids. Molecules. 2017; 22(9):1531. https://doi.org/10.3390/molecules22091531
Chicago/Turabian StyleFu, Renzhong, Yang Yang, Xudong Ma, Yu Sun, Jin Li, Hang Gao, Huaxing Hu, Xiaojun Zeng, and Jun Yi. 2017. "An Efficient, Eco-friendly and Sustainable One-Pot Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones Directly from Alcohols Catalyzed by Heteropolyanion-Based Ionic Liquids" Molecules 22, no. 9: 1531. https://doi.org/10.3390/molecules22091531
APA StyleFu, R., Yang, Y., Ma, X., Sun, Y., Li, J., Gao, H., Hu, H., Zeng, X., & Yi, J. (2017). An Efficient, Eco-friendly and Sustainable One-Pot Synthesis of 3,4-Dihydropyrimidin-2(1H)-ones Directly from Alcohols Catalyzed by Heteropolyanion-Based Ionic Liquids. Molecules, 22(9), 1531. https://doi.org/10.3390/molecules22091531






