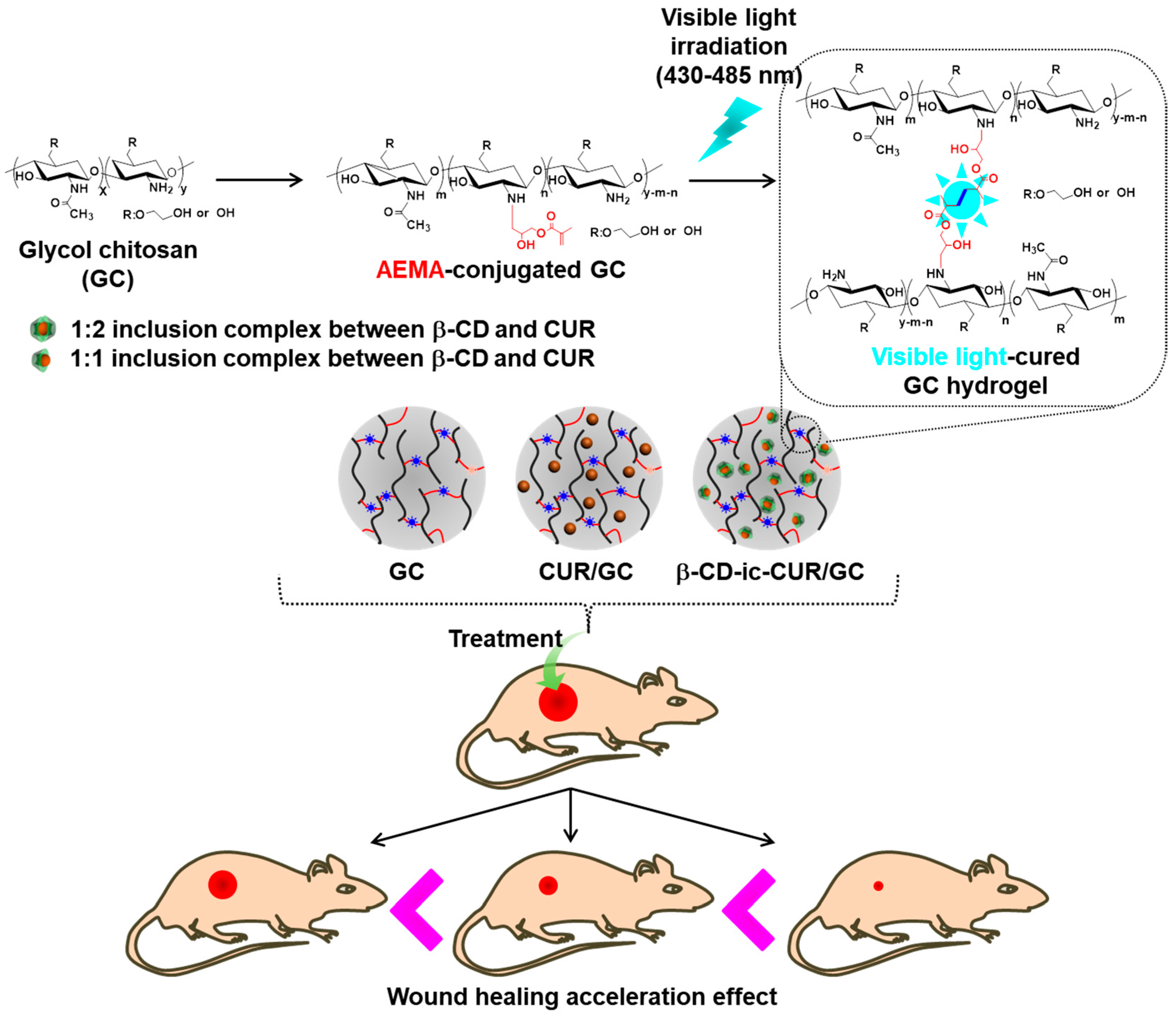
Visible Light-Cured Glycol Chitosan Hydrogel Containing a Beta-Cyclodextrin-Curcumin Inclusion Complex Improves Wound Healing In Vivo
Abstract
:1. Introduction
2. Results
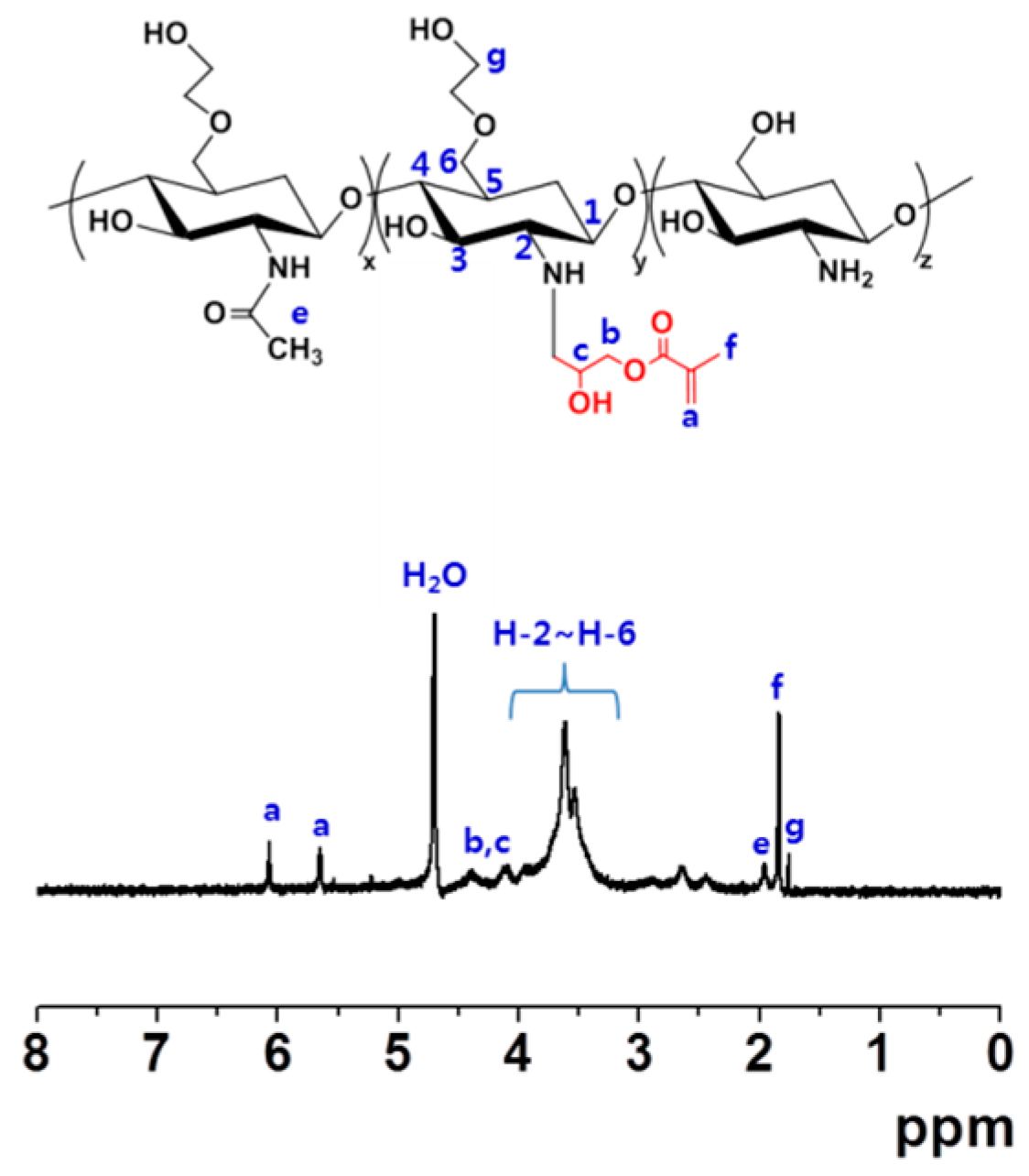
2.1. Proton Nuclear Magnetic Resonance (1H-NMR) Analysis
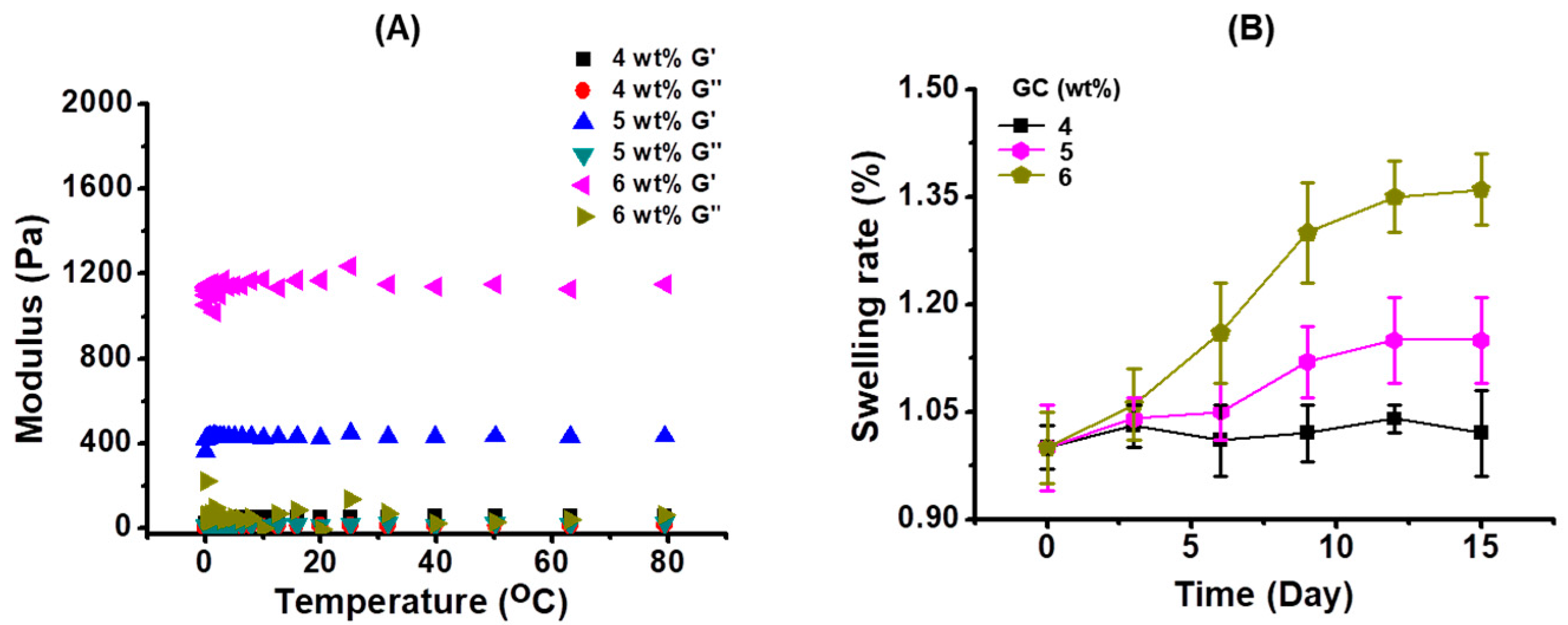
2.2. Characterization of GC Hydrogels Formed by Different Concentrations of GC
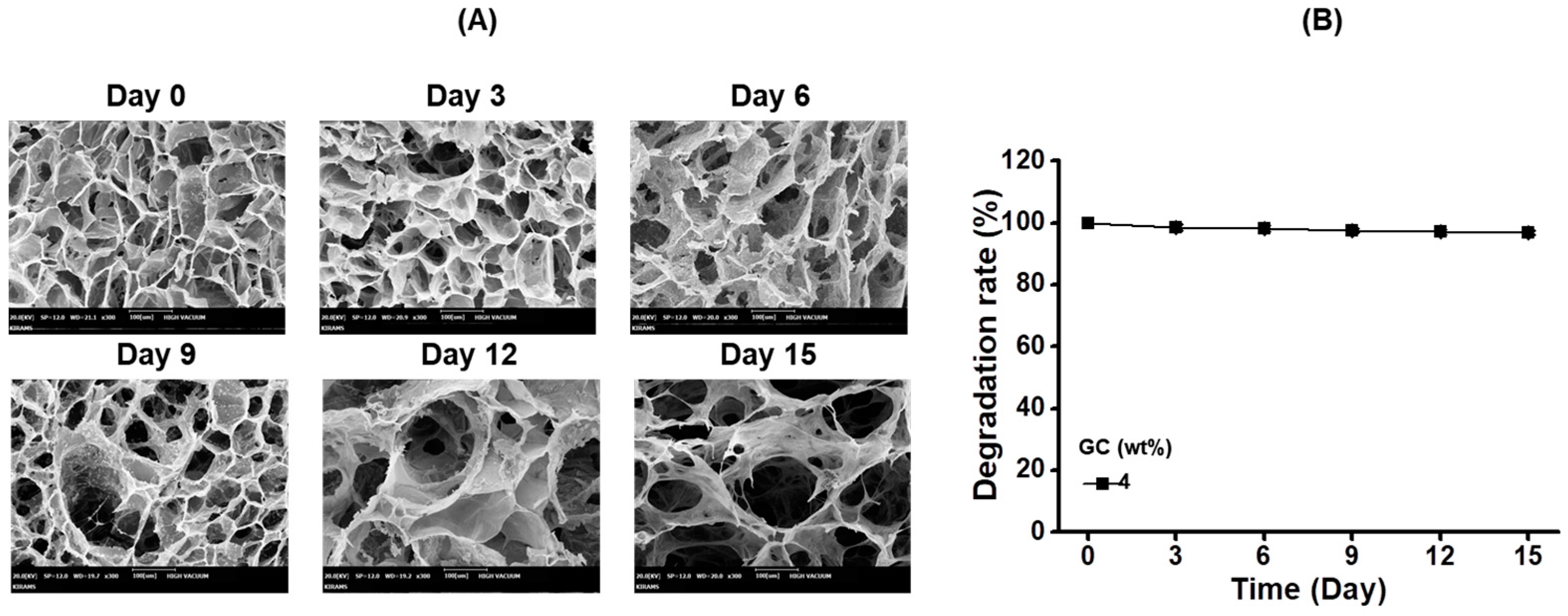
2.3. SEM Images and Degradation Ratio of Hydrogel Prepared by 4 wt % GC as a Function of Time
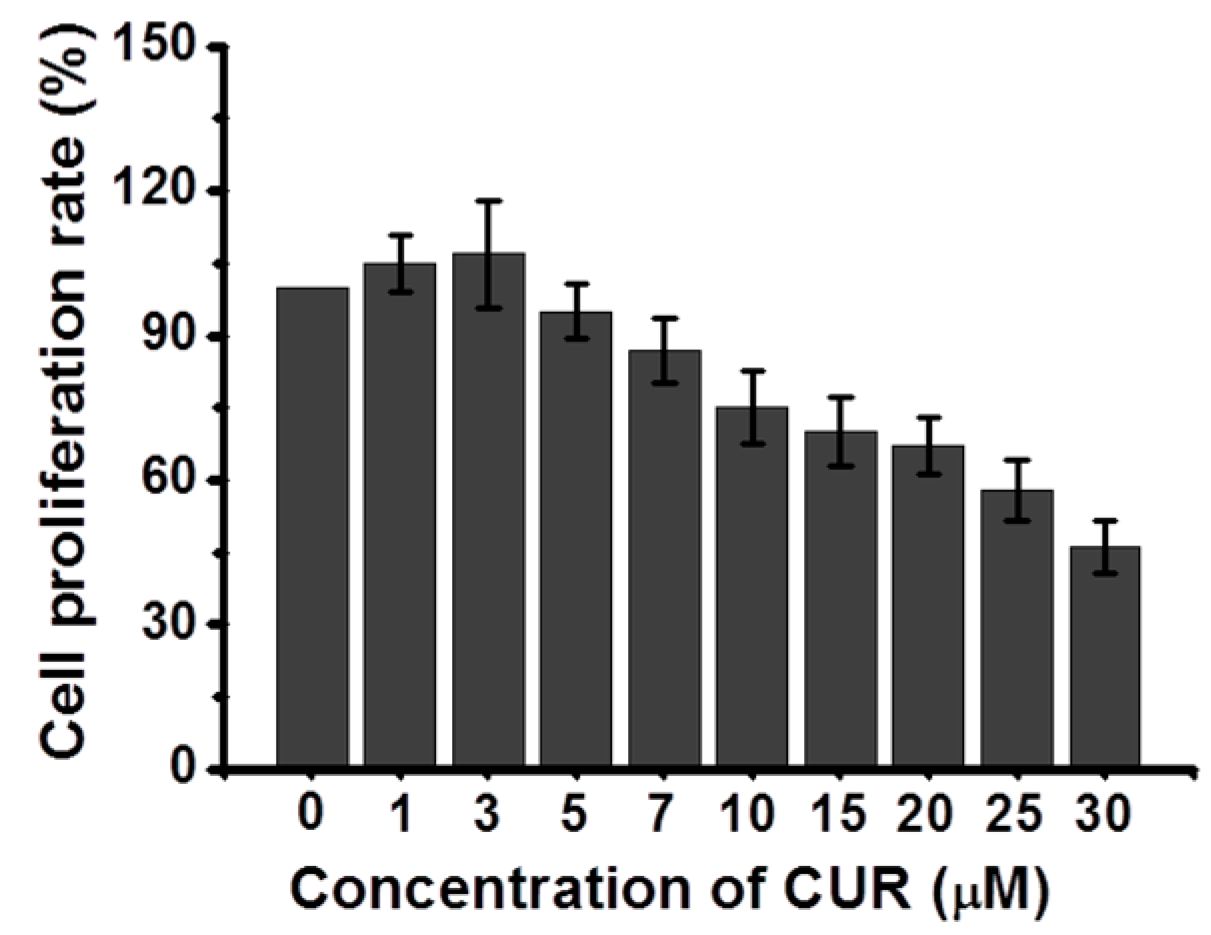
2.4. Cytotoxicity of CUR on L-929 Cells
2.5. Examination of the Inclusion Complex between β-CD and CUR
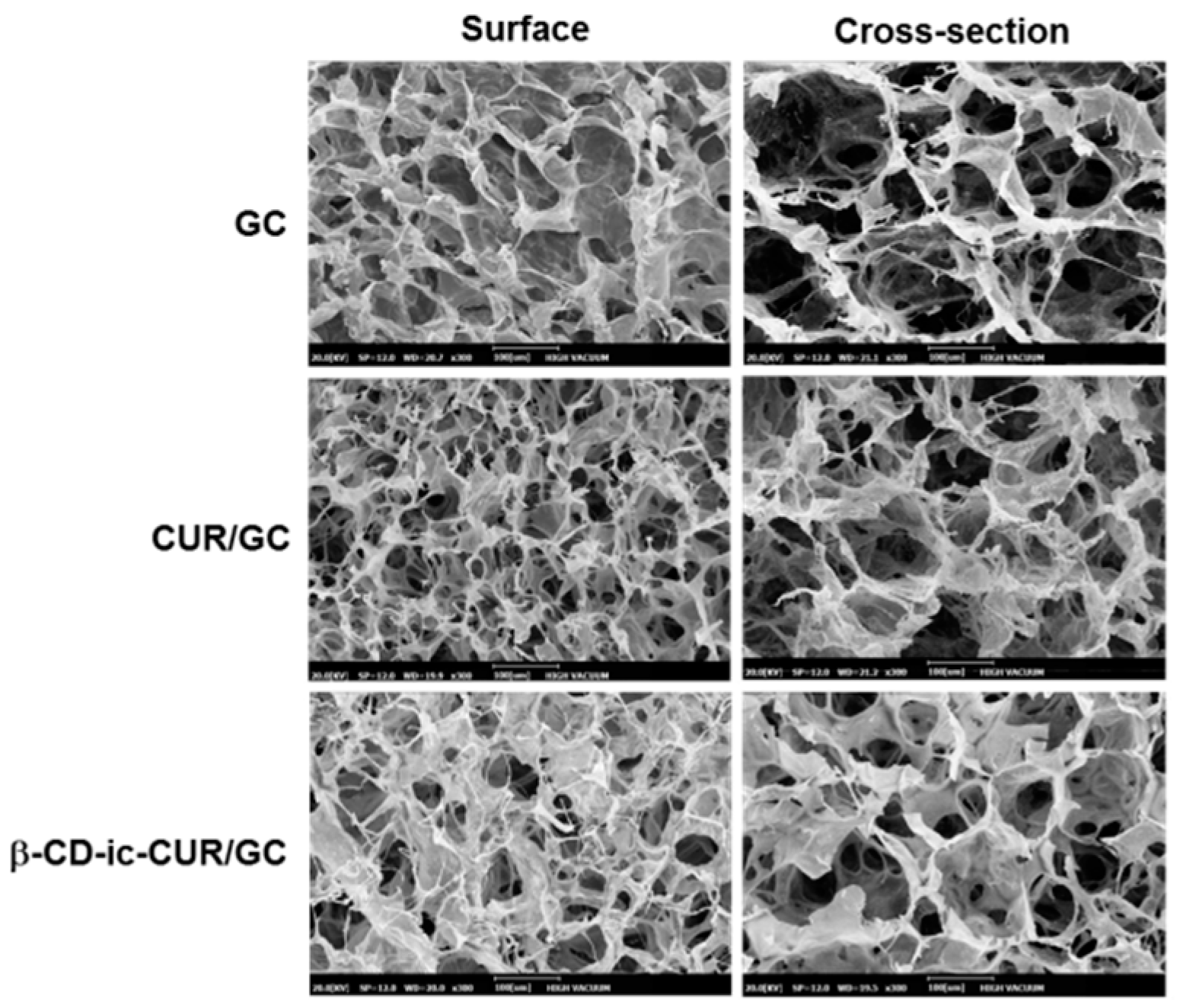
2.6. Surface and Cross-Sectional Morphologies
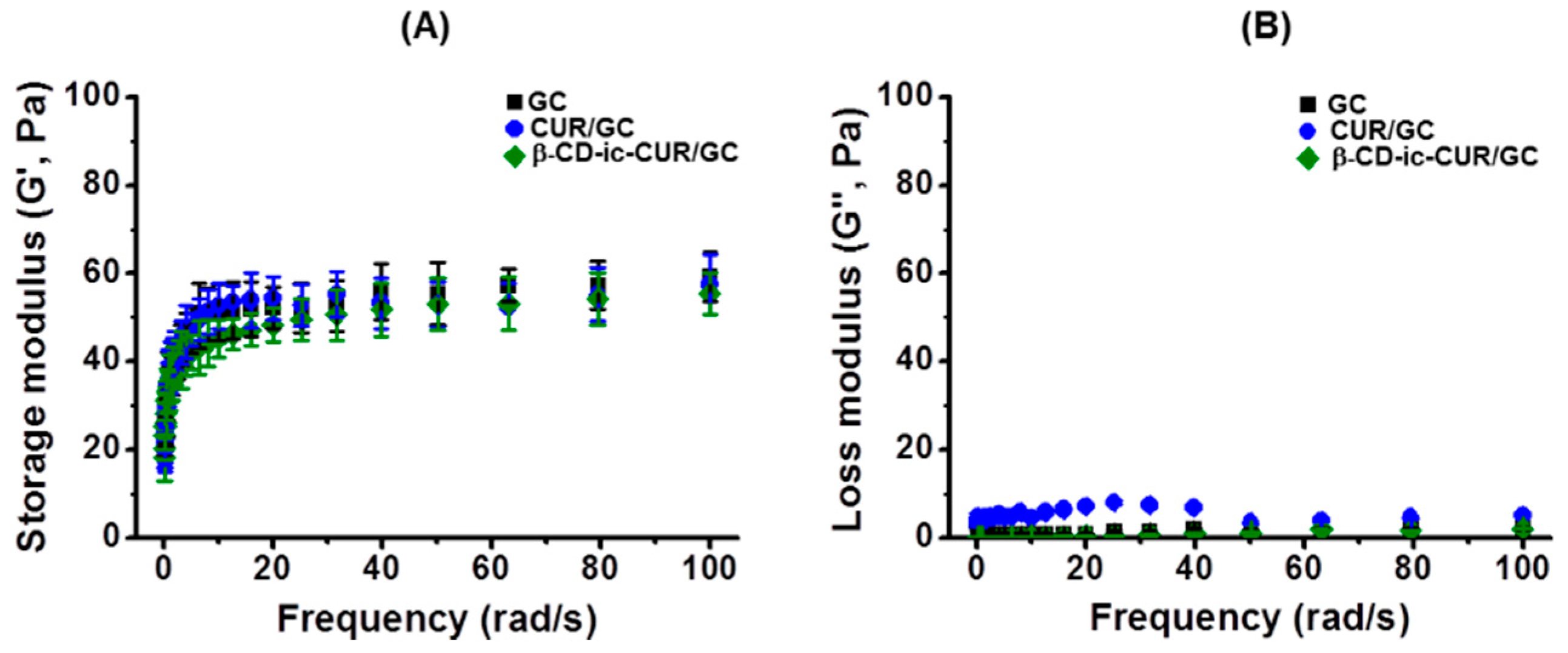
2.7. Storage and Loss Moduli
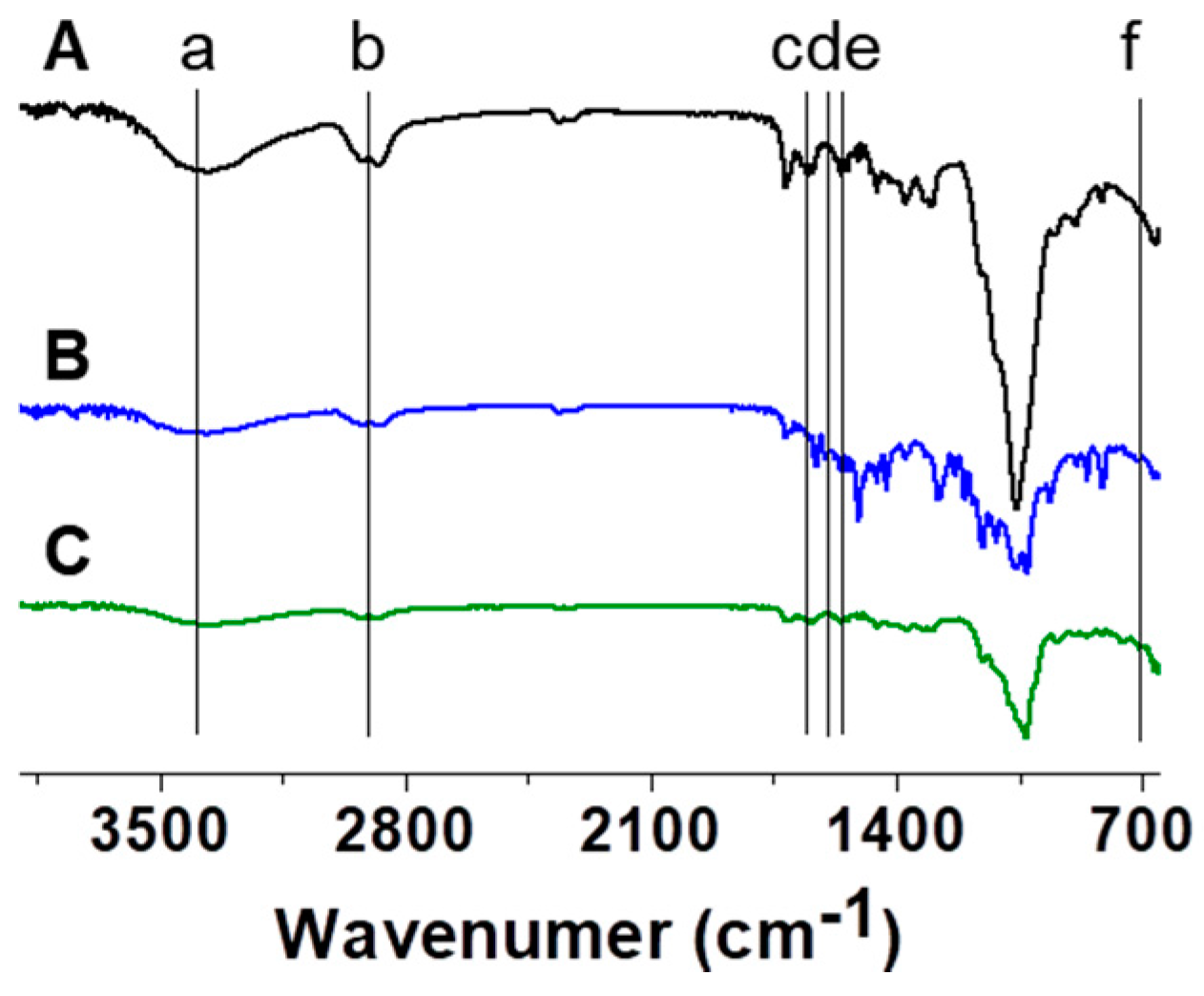
2.8. Fourier-Transform Infrared Spectroscopic (FT-IR) Analysis
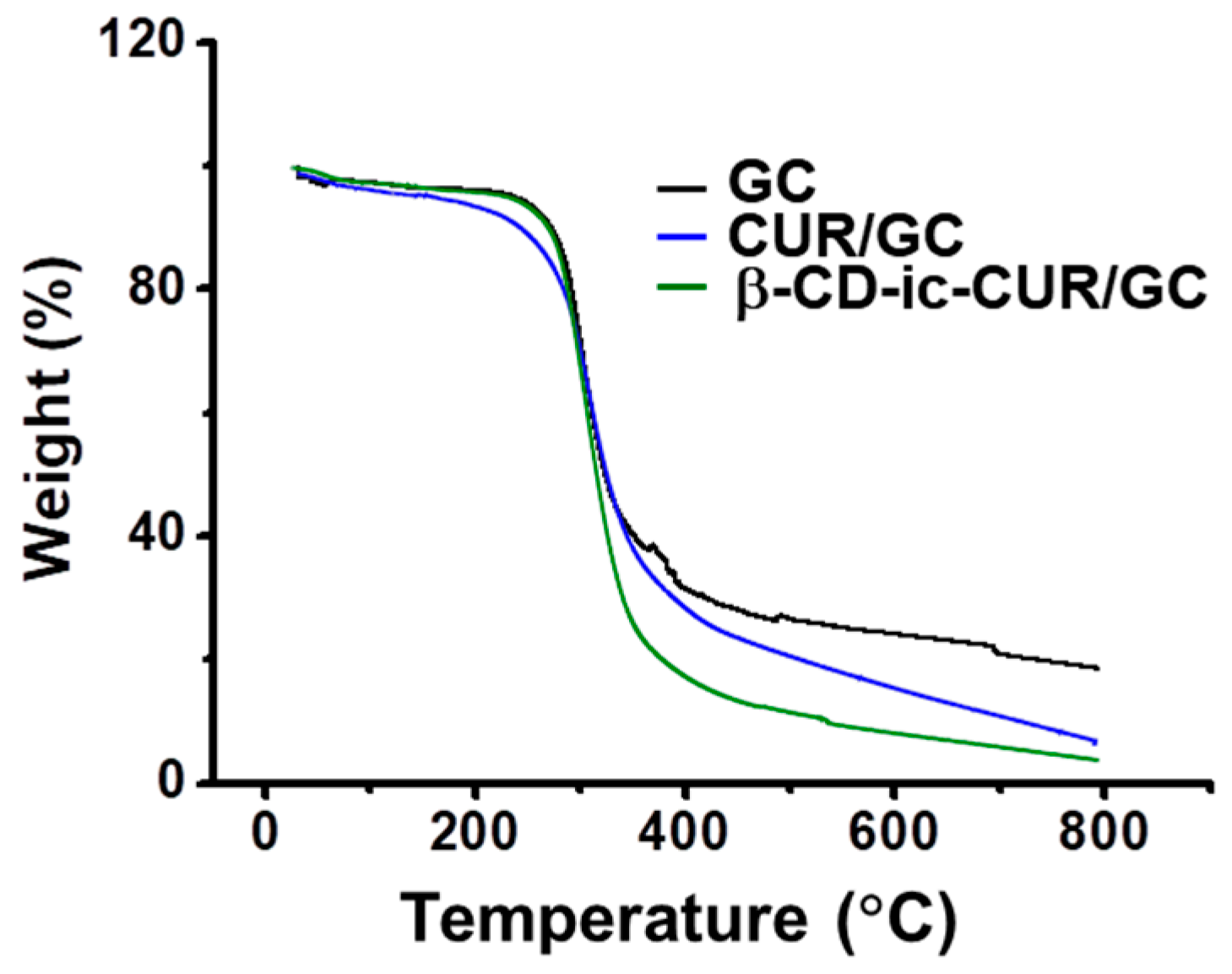
2.9. Thermal Gravimetric Analysis (TGA)
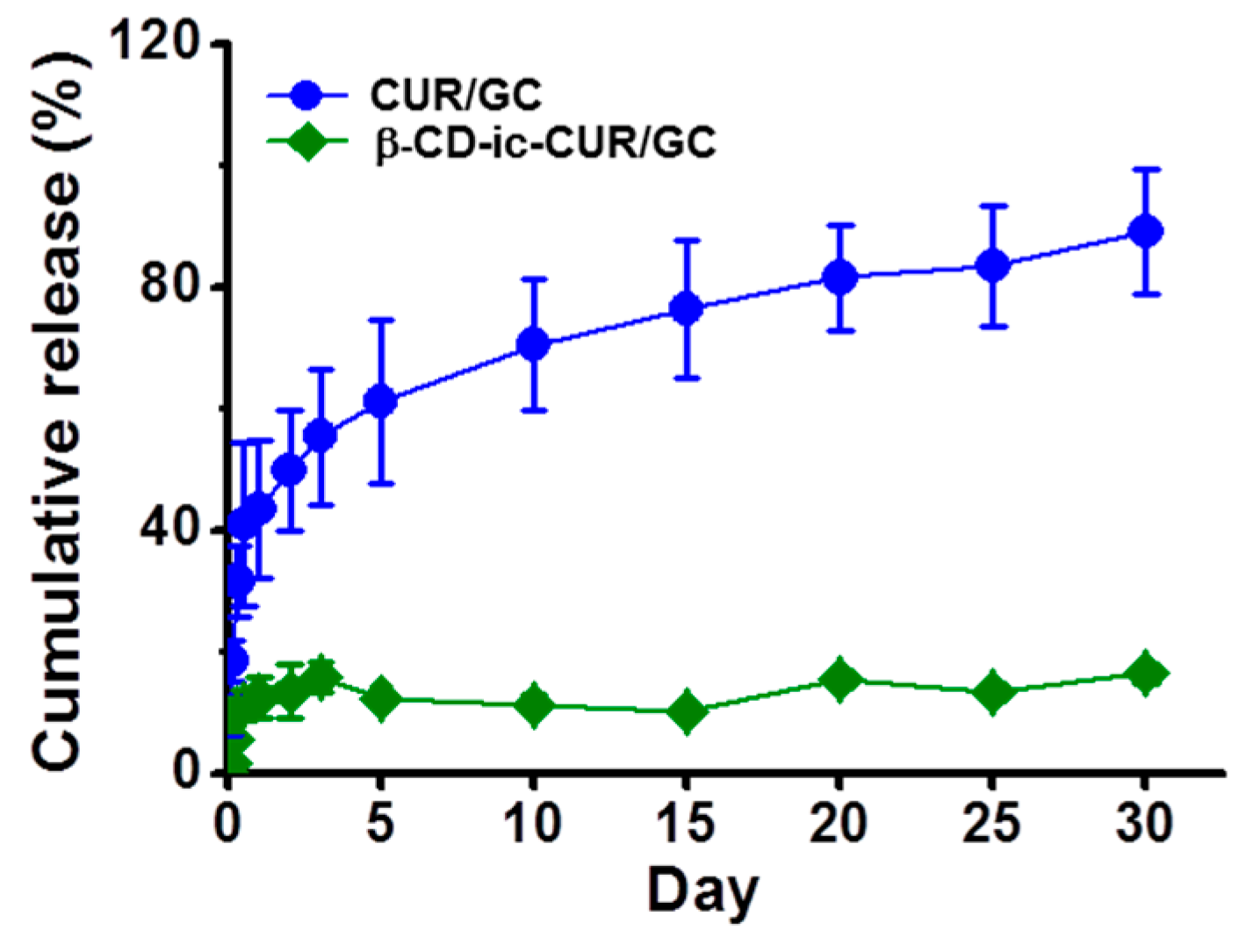
2.10. Release Behavior of CUR
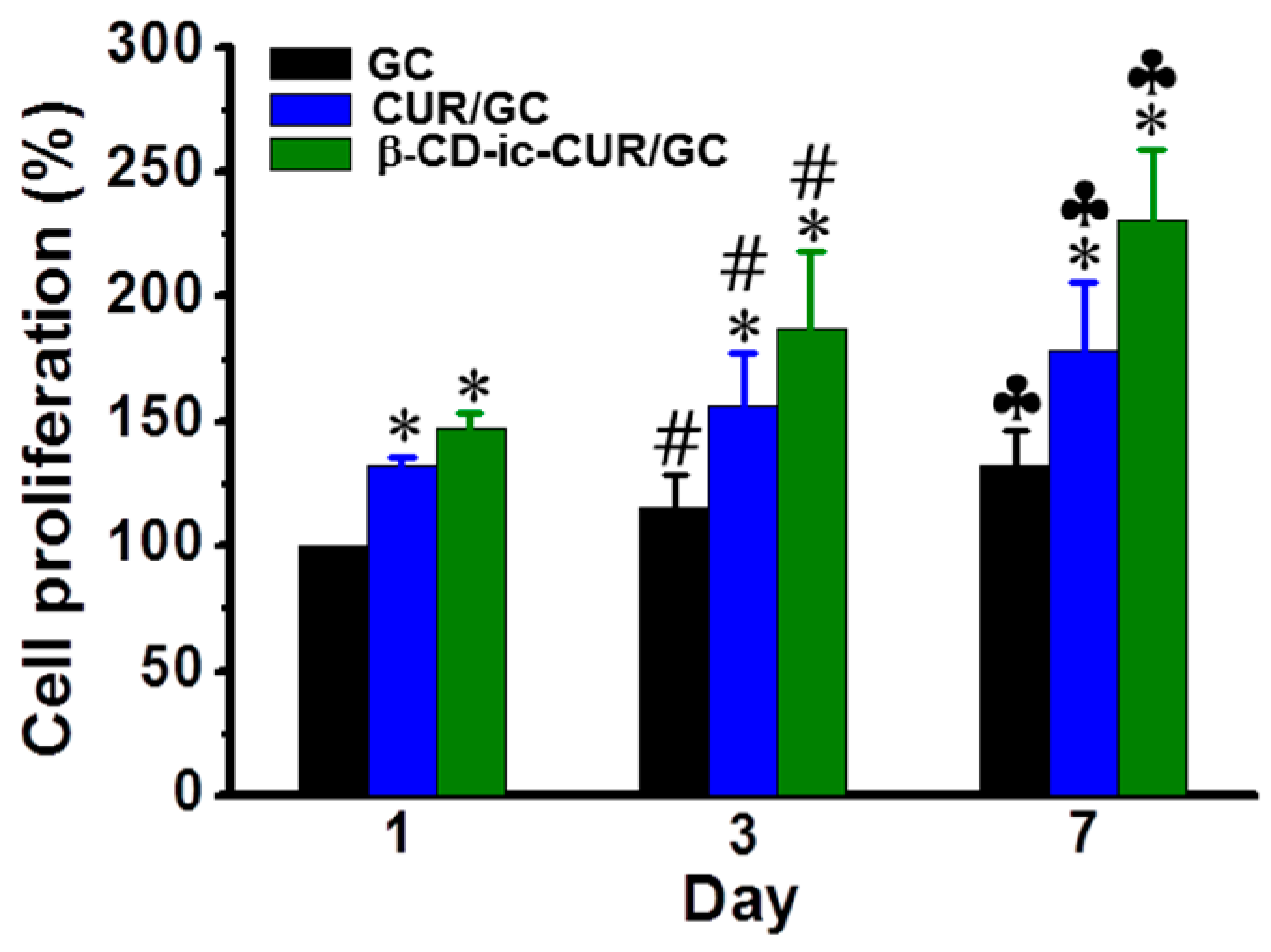
2.11. Proliferation Rate of L-929 Cells
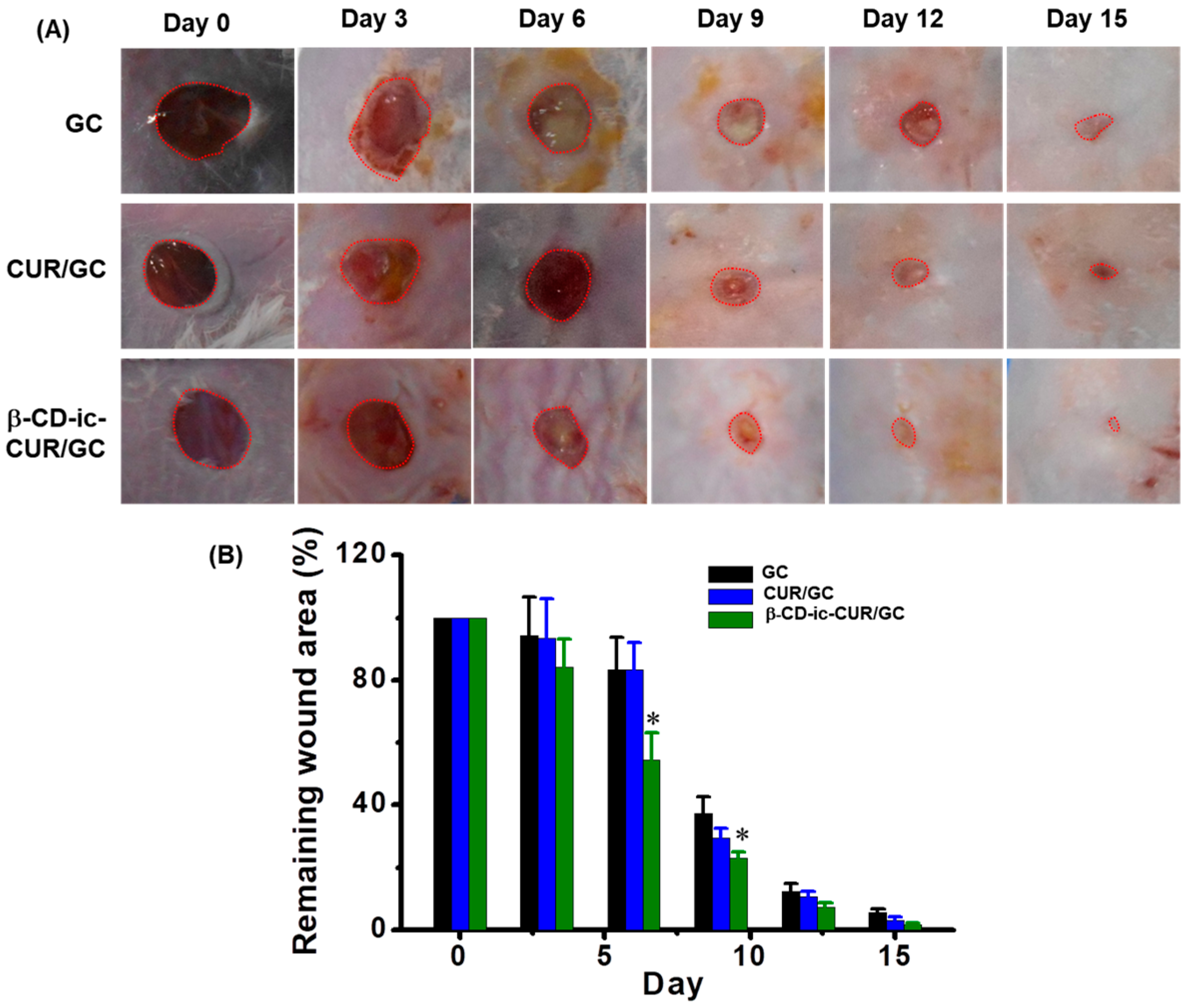
2.12. In Vivo Wound-Healing Analysis
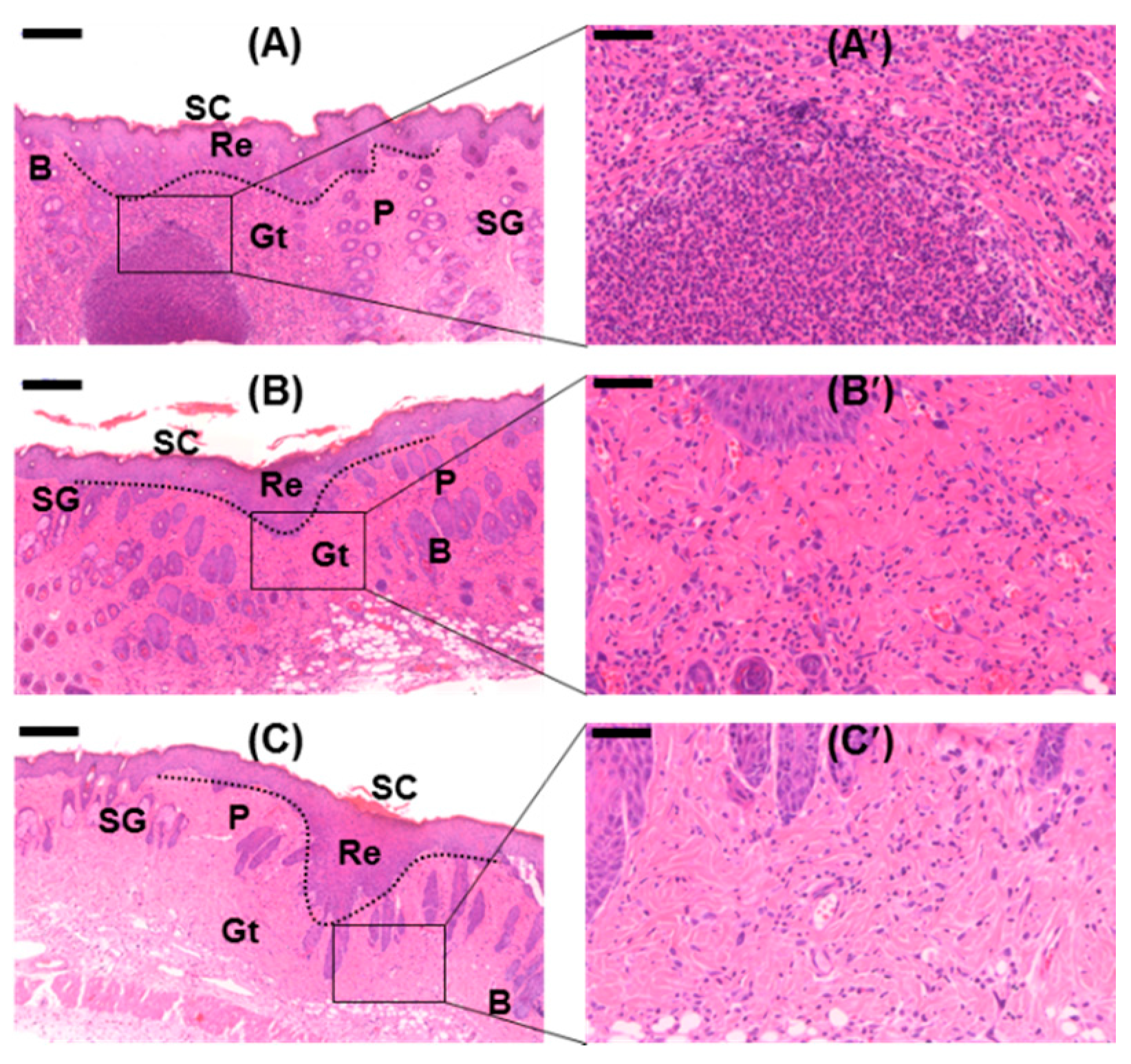
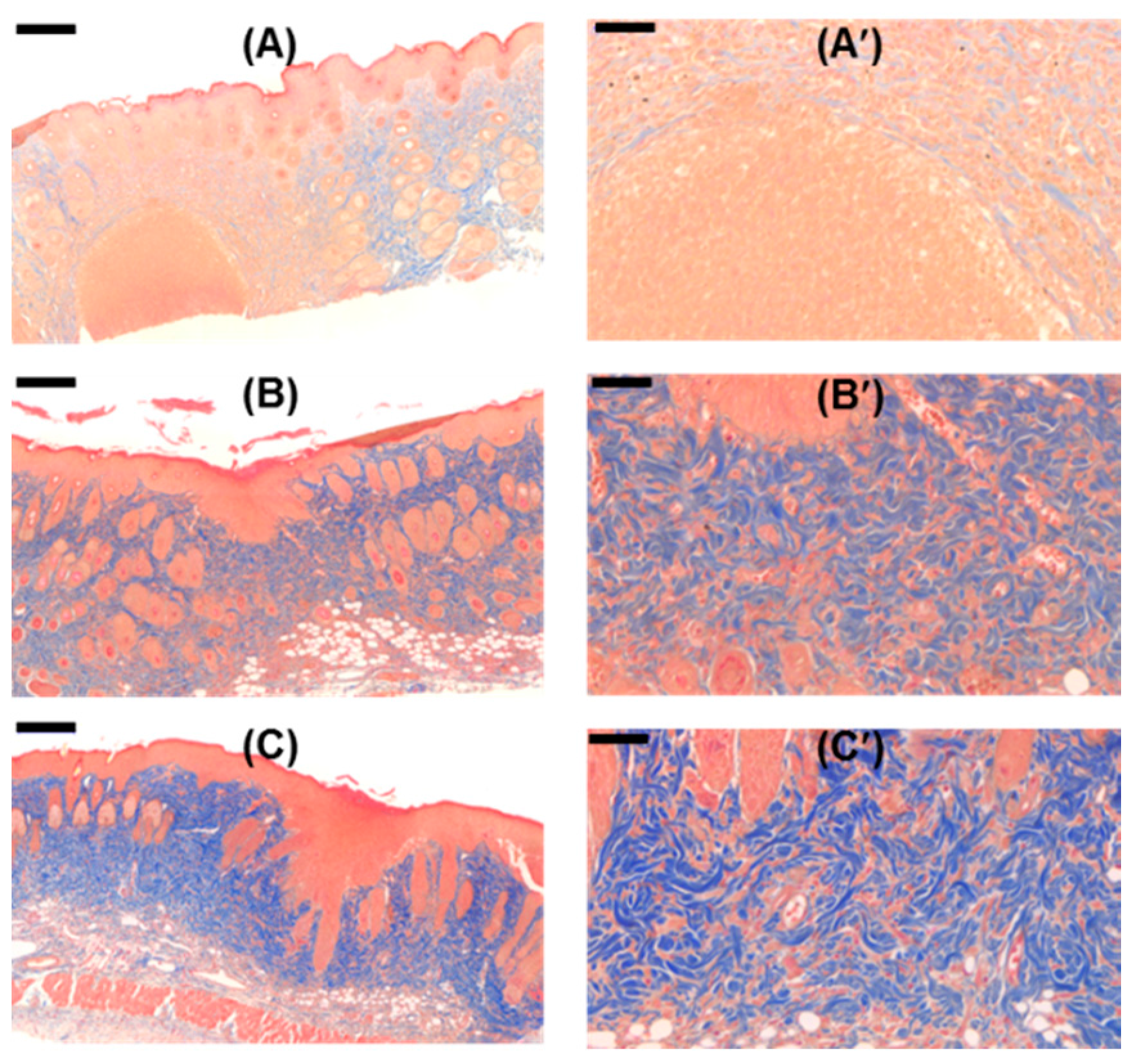
2.13. Histological Analyses
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Preparation of GM-Conjugated GC (GM-GC)
4.3. Preparation and Characterization of Visible Light-Cured GC Hydrogel (GC) as a Function of Concentration
4.4. Formation of Inclusion Complex between β-CD and CUR (β-CD-ic-CUR)
4.5. Analysis of CUR Cytotoxicity
4.6. Preparation of CUR and β-CD-ic-CUR-Loaded GC Hydrogels (CUR/GC and β-CD-ic-CUR/GC)
4.7. Scanning Electron Microscopy (SEM)
4.8. Rheological Analysis
4.9. FTIR Analysis
4.10. TGA Measurement
4.11. Analysis of CUR Release
4.12. In Vitro Cell Proliferation
4.13. In Vivo Wound-Healing Study
4.14. Histological Analysis
4.15. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vyas, K.S.; Vasconez, H.C. Wound healing: Biologics, skin substitutes, biomembranes and scaffolds. Healthcare 2014, 2, 356–400. [Google Scholar] [CrossRef] [PubMed]
- Dabiri, G.; Damstetter, E.; Phillips, T. Choosing a wound dressing based on common wound characteristics. Adv. Wound Care 2016, 5, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Han, G.; Ceilley, R. Chronic wound healing: A review of current management and treatments. Adv. Ther. 2017, 24, 599–610. [Google Scholar] [CrossRef] [PubMed]
- Jones, V.; Grey, J.E.; Harding, K.G. Wound healing. BMJ 2006, 332, 777–780. [Google Scholar] [CrossRef] [PubMed]
- Ifkovits, J.L.; Burdick, J.A. Review: Photopolymerizable and degradable biomaterials for tissue engineering applications. Tissue Eng. 2007, 13, 2369–2385. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Ito, Y. Visible light-curable polymers for biomedical applications. Sci. China Chem. 2014, 57, 510–521. [Google Scholar] [CrossRef]
- Baier, L.J.; Bivens, K.A.; Patrick, C.W., Jr.; Schmidt, C.E. Photocrosslinked hyaluronic acid hydrogels: Natural, biodegradable tissue engineering scaffolds. Biotechnol. Bioeng. 2003, 82, 578–589. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.H.; Seo, D.I.; Lee, D.-W.; Bhang, S.H.; Park, K.S.; Jang, G.; Kim, C.H.; Chun, H.J. Preparation and evaluation of visible-light cured glycol chitosan hydrogel dressing containing dual growth factors for accelerated wound healing. J. Ind. Eng. Chem. 2017, 53, 360–370. [Google Scholar] [CrossRef]
- Kamoun, E.A.; Chen, X.; Eldin, M.S.M.; Kenawy, E.-R.S. Crosslinked poly(vinyl alcohol) hydrogels for wound dressing applications: A review of remarkably blended polymers. Arab. J. Chem. 2015, 8, 1–14. [Google Scholar] [CrossRef]
- Jeon, D.-W.; Ahn, W.S.; You, S.J.; Chae, G.T.; Shim, Y.B.; Chun, H.J. Induction of classical activation of macrophage in vitro by water soluble chitin. Appl. Surf. Sci. 2012, 262, 134–139. [Google Scholar] [CrossRef]
- Hajimiri, M.; Shavverdi, S.; Esfandiari, M.A.; Larijani, B.; Atyabi, F.; Rajabiani, A.; Dehpour, A.R.; Amini, M.; Dinarvand, R. Preparation of hydrogel embedded polymer-growth factor conjugated nanoparticles as a diabetic wound dressing. Drug Dev. Ind. Pharm. 2016, 42, 707–719. [Google Scholar] [CrossRef] [PubMed]
- Akbik, D.; Ghadiri, M.; Chrzanowski, W.; Rohanizadeh, R. Curcumin as a wound healing agent. Life Sci. 2014, 116, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Prasad, S.; Tyagi, A.K.; Aggarwal, B.B. Recent developments in delivery, bioavailability, absorption and metabolism of curcumin: The golden pigment from golden spice. Cancer Res. Treat. 2014, 46, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Hoare, T.R.; Kohane, D.S. Hydrogels in drug delivery: Progress and challenges. Polymer 2008, 49, 1993–2007. [Google Scholar] [CrossRef]
- Chitragari, G.; Sumpio, J.; Sumpio, B.E. Indian spices for the management of diabetic foot complications. Angiology 2013, 1, 114. [Google Scholar] [CrossRef]
- Scharstuhl, A.; Mutsaers, H.A.M.; Pennings, S.W.C.; Szarek, W.A.; Russel, F.G.M.; Wagener, F.A.D.T.G. Curcumin-induced fibroblast apoptosis and in vitro wound contraction are regulated by antioxidants and heme oxygenase: Implications for scar formation. J. Cell Mol. Med. 2009, 13, 712–725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yadav, V.R.; Suresh, S.; Devi, K.; Yadav, S. Effect of cyclodextrin complexation of curcumin on its solubility and antiangiogenic and anti-inflammatory activity in rat colitis model. AAPS PharmSciTech 2009, 10, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Mansur, A.A.P.; Mansur, H.S. Quantum dot/glycol chitosan fluorescent nanoconjugates. Nanoscale Res. Lett. 2015, 10, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Mohan, P.R.K.; Sreelakshmi, G.; Muraleedharan, C.V.; Joseph, R. Water soluble complexes of curcumin with cyclodextrins: Characterization by FT-Raman spectroscopy. Vib. Spectrosc. 2012, 62, 77–84. [Google Scholar] [CrossRef]
- Corazzari, I.; Nisticò, R.; Turci, F.; Faga, M.G.; Franzoso, F.; Tabasso, S.; Magnacca, G. Advanced physico-chemical characterization of chitosan by means of TGA coupled on-line with FTIR and GCMS: Thermal degradation and water adsorption capacity. Polym. Degrad. Stab. 2015, 112, 1–9. [Google Scholar] [CrossRef]
- Williams, R.O., III; Reynolds, T.D.; Cabelka, T.D.; Sykora, M.A.; Mahaguna, V. Investigation of excipient type and level on drug release from controlled release tablets containing HPMC. Pharm. Dev. Technol. 2002, 7, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Gurtner, G.C.; Werner, S.; Barrandon, Y.; Longaker, M.T. Review article wound repair and regeneration. Nature 2008, 453, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Koh, T.J.; DiPietro, L.A. Inflammation and wound healing: The role of the macrophage. Expert Rev. Mol. Med. 2011, 13, e23. [Google Scholar] [CrossRef] [PubMed]
- Sogias, I.A.; Khutoryanskiy, V.V.; Williams, A.C. Exploring the factors affecting the solubility of chitosan in water. Macromol. Chem. Phys. 2010, 211, 426–433. [Google Scholar] [CrossRef]
- Knight, D.K.; Shapka, S.N.; Amsden, B.G. Characterization of glycol chitosan, a potential material for use in biomedical and pharmaceutical applications. ACS Symp. Ser. 2006, 934, 227–242. [Google Scholar]
- Tan, J.; Saltzman, W.M. Influence of synthetic polymers on neutrophil migration in three-dimensional collagen gels. J. Biomed. Mater. Res. 1999, 46, 465–474. [Google Scholar] [CrossRef]
- Joe, B.; Vijaykumar, M.; Lokesh, B. Biological properties of curcumin-cellular and molecular mechanisms of action. Crit. Rev. Food Sci. Nutr. 2004, 44, 97–111. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, C.; Das, M.; Sahoo, S. Sustained wound healing activity of curcumin loaded oleic acid based polymeric bandage in a rat model. Mol. Pharm. 2012, 9, 2801–2811. [Google Scholar] [CrossRef] [PubMed]
- Gong, C.Y.; Wu, Q.; Wang, Y.; Zhang, D.D.; Luo, F.; Zhao, X.; Wei, Y.Q.; Qian, Z. A biodegradable hydrogel system containing curcumin encapsulated in micelles for cutaneous wound healing. Biomaterials 2013, 34, 6377–6387. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.S.; Shin, J.; Bhang, S.H.; Shin, J.-Y.; Park, J.; Im, G.-I.; Kim, C.-S.; Kim, B.-S. Enhanced skin wound healing by a sustained release of growth factors contained in platelet-rich plasma. Exp. Mol. Med. 2011, 43, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Sharma, S.; Dhiman, A. Desing of antibiotic containing hydrogel wound dressings: Biomedical properties and histological study of wound healing. Int. J. Pharm. 2013, 457, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Xing, X.; Yang, H.; Jia, Y.; Zhou, T.; Chen, Y.; Ling, Z.; Hu, X. Covalently antibacterial alginate-chitosan hydrogel dressing integrated gelatin microspheres containing tetracycline hydrochloride for wound healing. Mater. Sci. Eng. C 2017, 70, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, C.; Zhang, Q.; Cheng, Y. Near infrared light-responsive and injectable supramolecular hydrogels for on-demand drug delivery. Chem. Commun. 2016, 52, 978–981. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Oo, N.N.L.; Lee, J.P.; Li, Z.; Loh, X.J. Recent development of synthetic nonviral systems for sustained gene delivery. Drug Discov. Today 2017. [Google Scholar] [CrossRef] [PubMed]
- Wee, C.Y.; Liow, S.S.; Li, Z.; Wu, Y.-L.; Loh, X.J. New poly[(R)-3-hydroxybutyrate-co-4-hydroxybutyrate] (P3HB4HB)-based thermogels. Macromol. Chem. Phys. 2017. [Google Scholar] [CrossRef]
- Liu, X.; Chen, X.; Chua, M.X.; Li, Z.; Loh, X.J.; Wu, Y.-L. Injectable supramolecular hydrogels as delivery agents of Bcl-2 conversion gene for the effective shrinkage of therapeutic resistance tumors. Adv. Healthc. Mater. 2017, 6, 1700159. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-C.; Metters, A.T. Hydrogels in controlled release formulations: Network design and mathematical modeling. Adv. Drug Deliv. Rev. 2006, 58, 1379–1408. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.; Zhang, X.; Shen, Y.; Sebastian, R.; Dickinson, L.E.; Fox-Talbot, K.; Reinblatt, M.; Steenbergen, C.; Harmon, J.W.; Gerecht, S. Dextran hydrogel scaffolds enhance angiogenic responses and promote complete skin regeneration during burn wound healing. Proc. Natl. Acad. Sci. USA 2011, 108, 20976–20981. [Google Scholar] [CrossRef] [PubMed]
- Roh, H.; Yang, D.H.; Chun, H.J.; Khang, G. Cellular behavior of hepatocyte-like cells from nude mouse bone marrow-derived mesenchymal stem cells on galactosylated poly(d,l-lactic-co-glycolic acid). J. Tissue Eng. Regen. Med. 2015, 9, 819–825. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds GM-conjugated GC, and GC, CUR/GC and β-CD-ic-CUR/GC are available from the authors. |

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoon, S.-J.; Hyun, H.; Lee, D.-W.; Yang, D.H. Visible Light-Cured Glycol Chitosan Hydrogel Containing a Beta-Cyclodextrin-Curcumin Inclusion Complex Improves Wound Healing In Vivo. Molecules 2017, 22, 1513. https://doi.org/10.3390/molecules22091513
Yoon S-J, Hyun H, Lee D-W, Yang DH. Visible Light-Cured Glycol Chitosan Hydrogel Containing a Beta-Cyclodextrin-Curcumin Inclusion Complex Improves Wound Healing In Vivo. Molecules. 2017; 22(9):1513. https://doi.org/10.3390/molecules22091513
Chicago/Turabian StyleYoon, Sun-Jung, Hoon Hyun, Deok-Won Lee, and Dae Hyeok Yang. 2017. "Visible Light-Cured Glycol Chitosan Hydrogel Containing a Beta-Cyclodextrin-Curcumin Inclusion Complex Improves Wound Healing In Vivo" Molecules 22, no. 9: 1513. https://doi.org/10.3390/molecules22091513





