3.1. Yield Components and Wine Chemical Composition
Components of yield, as the mean of 2010 and 2011 vintages, of five training systems are presented in
Table 2. The training system did not show any effect on grape ripening. Significant differences regarding shoot density were noted, with Parral and GDC differing by up to 30%. Parral was the training system with a higher crop yield, followed by GDC, VSP, SH, and AC. Berry weight at harvest did not vary among systems, but the Parral system had a greatest cluster weight because the highest number of berries per cluster, followed by GDC system. Generally, increases in yield due to training systems tended to result from increases in cluster per vine [
15]. However, in our study increases in yield were due to increases in berries per cluster and therefore cluster weight, where the Parral, followed by the GDC, training systems showed the highest values.
The values (mean and standard deviation from 2010 and 2011 vintages) of classical chemical parameters of Albariño wines are shown in
Table 3. The training system did not show any effect on chemical composition of the wines. In general, berry maturity was not altered by training system in the Cabernet sauvignon from the wet region of China [
31,
32]. Similar results were observed in cv. Viognier when it was compared to VSP, Smart Dyson, and GDC training systems, but Fragasso et al. [
16] observed higher Primitivo grape ripeness when it was grown with the Lira training system versus bilateral Guyot and four rays.
3.2. Volatile Composition of Albariño Wines
A total of 35 compounds were identified and quantified in Albariño wines from different training systems using GC-MS, classified into nine groups (alcohols, C
6-compounds, ethyl esters, acetates, terpenes+C
13-norisoprenoids, volatile phenols, fatty acids, lactones and carbonyl compounds) according to their functional groups and metabolic formation (
Table 4 and
Table 5).
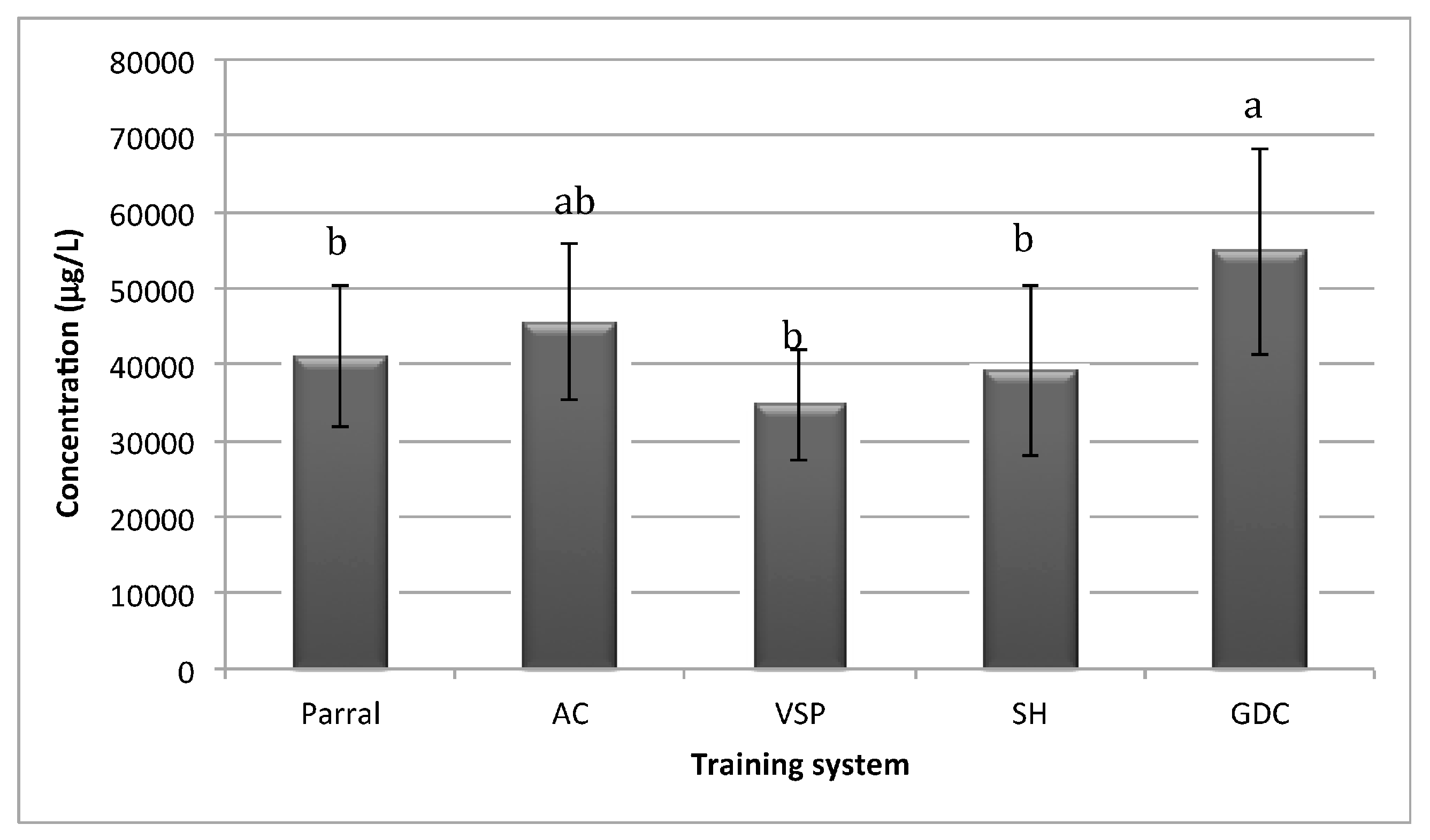
Figure 1 shows the total concentration of volatile compounds of Albariño wines from different training systems. According to the quantitative data shown in
Figure 1, total concentration of volatile compounds ranged from 34.42 mg/L to 54.81 mg/L. Wines from the GDC training system had the highest concentration of total volatile compounds followed by AC, while VSP had the lowest. In terms of total volatile composition, the GDC training was found to be statistically different from the other training systems (
p < 0.05). These results suggest that the chosen training system could affect the wine volatile composition.
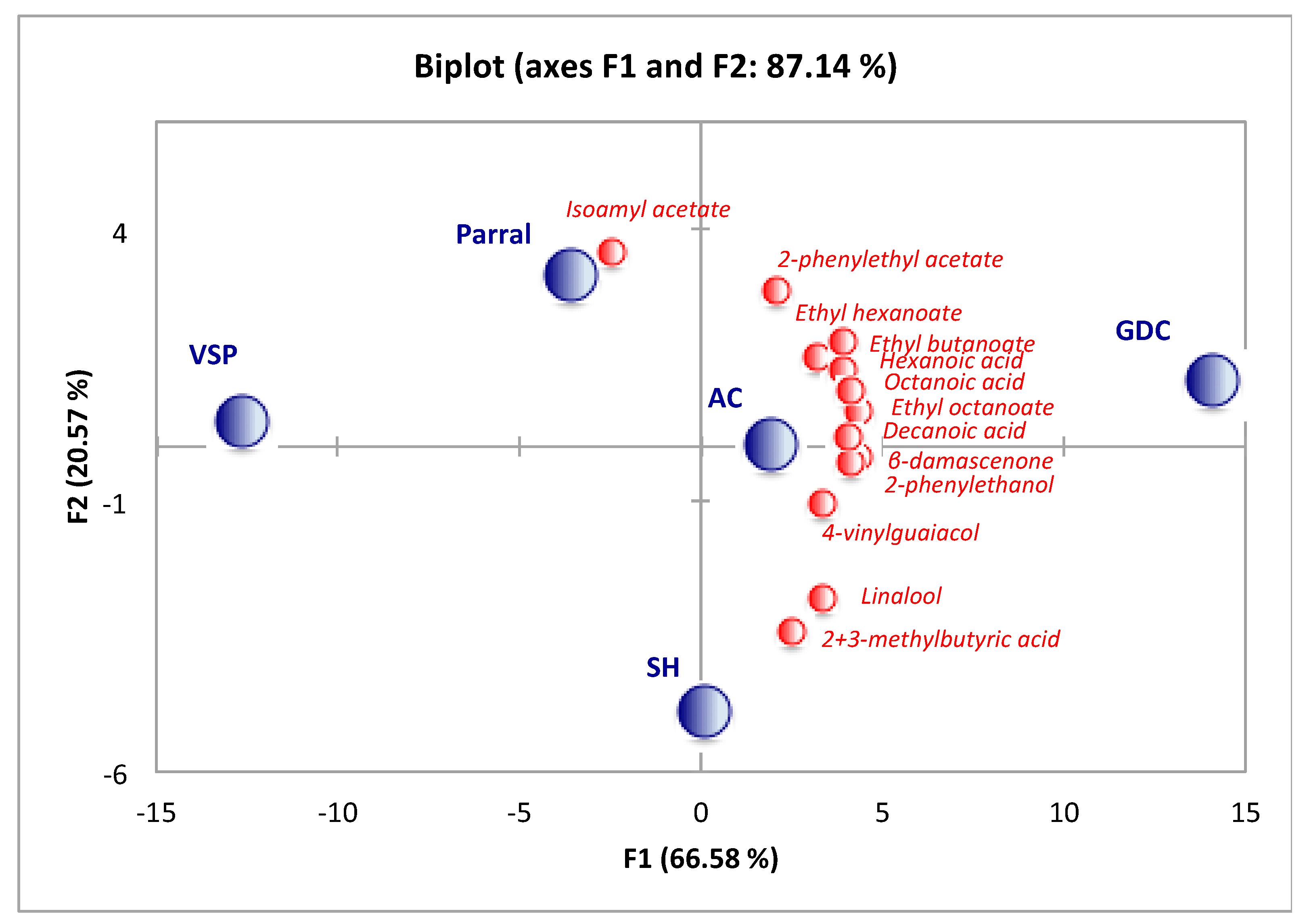
In order to analyze these differences in total volatile composition for wines from different training systems, a comparison of each chemical group was performed. The majority of volatiles were alcohols, ethyl esters, acetates, and fatty acids. The ANOVA analysis demonstrated that only the groups of alcohols, ethyl esters, terpenes+C
13-norisoprenoids, and lactones exhibited statistical differences amongst wines from different training systems (
Table 4). The GDC training system had the significantly highest total concentration of alcohols, ethyl esters, and lactones (
p < 0.05), while SH had the highest concentration of terpenes and C
13-norisoprenoids.
Albariño wines are known for being rich in ethyl esters, creating a fruity characteristic [
22]. Ethyl esters are formed during the process of fermentation and are related to the lipid metabolism, except diethyl succinate and diethyl malate. Ethyl esters are among the most important compounds in wine [
33] and their formation depends on fermentation practices, yeast strain, temperature of fermentation, aeration, and solids content of the must [
34]. In the present study, all the wines were made with grapes harvested to the same ripening stages and under the same fermentation condition, so the differences between the samples can probably be explained by the different yield components caused by the training systems. Six ethyl esters were identified and quantified in wine samples and the total concentrations were between 1558 μg/L and 2569 μg/L. Wines from GDC showed the highest content of ethyl esters and SH had the lowest. Among ethyl esters, the most abundant were ethyl hexanoate, ethyl butanoate (both with fruity and strawberry aromas), and ethyl octanoate (apple, sweetish aroma). Ethyl hexanoate, ethyl butanoate, and diethyl malate were found in significantly high concentrations for wines from the GDC training system (
p < 0.05) (
Table 5).
Higher alcohols are synthetized by yeast during alcoholic fermentation [
34]. In this study, nine alcohols were identified and quantified with total concentration from 18.95 mg/L and 33.20 mg/L. Wines from GDC showed the highest content of alcohols and VSP had the lowest (
Table 5). Three compounds, 2+3-methyl-1-butanol and 2-phenylethanol, had the highest concentrations in all wines, but only 2-phenylethanol showed significant differences among training systems with the highest concentration in the GDC system (
p < 0.05). Moreover, one other alcohol, 3-methyl-1-pentanol, was shown to be the significantly highest in the GDC system.
In terms of total concentration of terpenes together with C
13-norisoprenoids, the SH training system showed the highest value at 69 μg/L, while the lowest concentration was for the SVP system at 38 μg/L. Three terpenes (linalool, hotrienol and α-terpineol) and one C
13-norisoprenoid (β-damascenone) were identified and quantified in wines, but only hotrienol showed significant differences among training samples, with the highest value being obtained for SH (
p < 0.05). Linalool and α-terpineol exhibited higher concentrations for SH, as did β-damascenone for GDC. Terpenes and C
13-norisoprenoides are varietal compounds responsible for the aromatic quality of the wine, bringing fruity, citrus, and floral nuances [
33,
34,
35].
Several viticultural parameters, including sunlight exposure, have been shown to influence terpene and norisoprenoid concentrations [
12,
36,
37,
38] and lower fruit temperature favors the accumulation of volatile compounds with lower molecular weights and boiling points [
13]. Concentrations of TDN, vitispirane, hydroxy-TDN, ionols, actinidols, grasshopper ketone, and vomifoliol were significantly higher in sun-exposed Chenin blanc and white Riesling grapes compared to shaded grapes of the same varieties. However, β-damascenone concentrations did not appear to be influenced by sunlight levels [
36]. The Scott-Henry training system generates a vertically divided canopy increasing the effective canopy surface area and reducing its density. Canopy division also improves light exposure [
13].
The lactones group, represented by only one compound, γ-butyrolactone, showed concentrations between 91 μg/L and 253 μg/L. The highest value was reached in the GDC training system (
p < 0.05). γ-butyrolactone is a volatile compound found in wine and produced during fermentation and is responsible for a cheesy aroma [
29]. This compound reached higher concentrations (>3.7 mg/L) in Loureira and Albariño wines from Galicia [
33].
Other individual compounds showed significant differences among wines from different training systems. From the acetates, 2-phenylethyl acetate reached concentrations between 331.5 μg/L and 499.6 μg/L (
Table 5). The highest concentration was observed in wine from the GDC system. From the fatty acids, two compounds, hexanoic acid and octanoic acid, which creates a sweaty and cheese aroma, showed significant differences among the wines. The highest concentrations were also found in the GDC training system.
In summary, of the 35 volatile compounds identified and quantified in this study, ten showed significant differences among wines from five different training systems, where the highest concentrations were observed in the GDC system. GDC reached higher yields, however, the quality of the wine did not change. Yields can be increased with the appropriate training system without detrimentally impacting the fruit quality [
15]. GDC increased the fruiting areas and has a split canopy that allows more sun penetration. Canopy division has been reported to increase canopy volume and surface area compared to non-divided VSP systems in previous studies [
39]. Zoecklein et al. [
13] also showed that, despite a significantly increased crop yield in cv. Viognier, the fruit glycosides and free volatiles were greater in the divided canopies with Smart Dyson and GDC, than in VSP.
3.3. Odor Active Values for Albariño Wines
The odor activity values (
OAV) of each individual compound were calculated.
OAV is used as an indicator for the potential sensorial contribution of each compound to the overall aroma of the wine. Compounds with
OAV > 1 are considered as having a dynamic contribution to the wine aroma [
40]. However, the wine aroma is a result of the synergic effect and interactions of all present chemical compounds.
Table 6 lists the odorant activity values (
OAV), odor threshold, and odor descriptor for the aromatic compounds where
OAV > 1. From the 35 identified and quantified volatile compounds, 14 had an
OAV > 1. The compounds that showed the highest
OAV were β-damascenone (
OAV > 151) and isoamyl acetate (
OAV > 74), both with fruity characteristics of baked apple and banana, respectively. These results align with the findings of Falqué et al. [
25], demonstrating that isoamyl acetate had high
OAV and a positive effect on the aroma of Albariño wines. Vilanova et al. [
33] found the same results when studying the volatile composition of white wines from Galicia. In this study, higher
OAV values were reached by β-damascenone and isoamyl acetate for Albariño wines.
From the ethyl esters, the content of ethyl hexanoate was higher than their odor threshold in all wines analyzed, with the highest value found in GDC, with an
OAV of 12 (
Table 6). Other ethyl esters, including ethyl butanoate and ethyl octanoate, also showed values above their odor threshold. In both compounds, the highest
OAV was exhibited for the GDC system, at 2.8 and 1.1, respectively (
Table 6). These results are in accordance with those published by Zoecklein et al. [
13], who concluded that wines from the GDC system had stronger fruity characteristics.
Among the higher alcohols, 2-phenylethanol contributed positively to the wine aroma with a rose aroma [
1]. In our study, an
OAV > 1 was found for GDC, AC, and SH wines, with the highest value (1.5) obtained from the GDC system (
Table 6).
Five volatile acids showed
OAV values > 1, where octanoic, hexanoic, and decanoic acids had the highest value in the GDC system. Falqué et al. [
25] also found that octanoic and hexanoic acids were the most important acids in the Albariño wines. Vilanova et al. [
33] showed hexanoic acid having a high
OAV in Albariño wines from Galicia.
Vinylphenol, with a total concentration above 100 μg/L, is known to be found in large quantities in Albariño wines [
23,
25]. In the present study, 4-vinylguaiacol had an
OAV between 1.4 for VSP and 2.9 for GDC. The major positive
OAV units were found in the GDC training system, at 381.2, followed by Parral at 326.5, Arch-Cane at 309.9, and Scott Henry at 307.2; the smallest value was found in the VSP training system at 293.7.