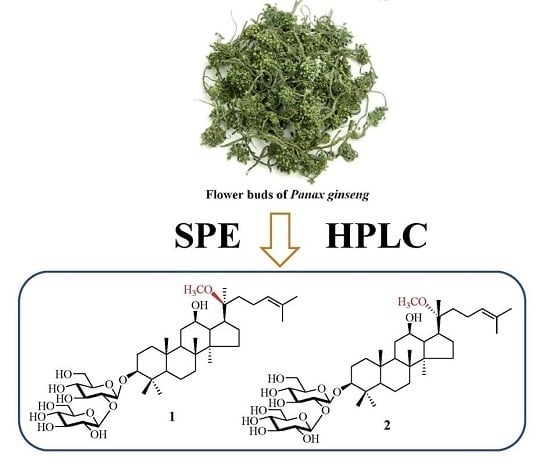
A Strategy for Simultaneous Isolation of Less Polar Ginsenosides, Including a Pair of New 20-Methoxyl Isomers, from Flower Buds of Panax ginseng
Abstract
:1. Introduction
2. Results and Discussion
2.1. Purification of the Five Less Polar Ginsenosides from the Flower Buds of Panax Ginseng
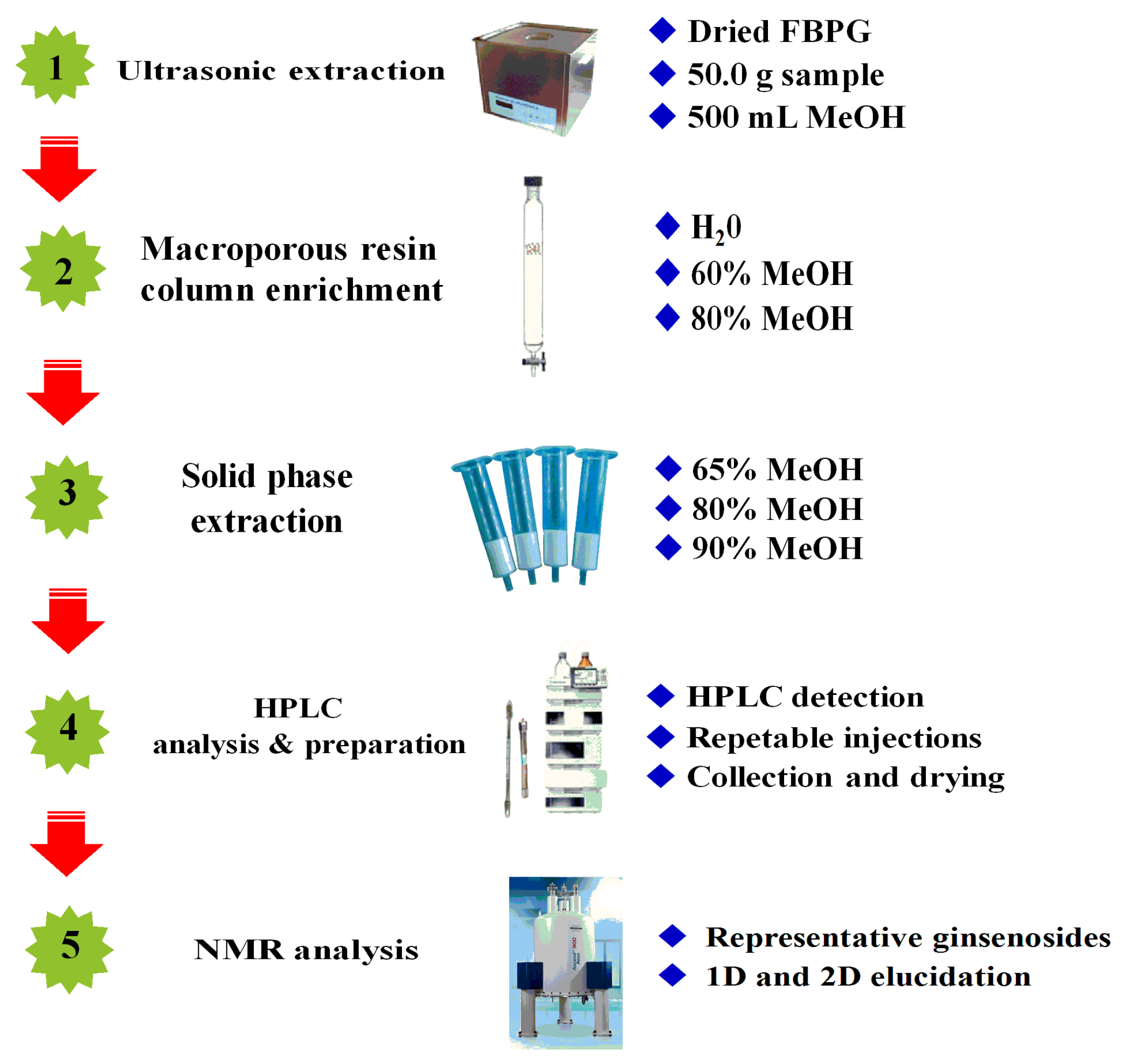
2.1.1. Preparation of Crude Ginsenoside Extracts
2.1.2. Optimization of Solid Phase Extraction Conditions
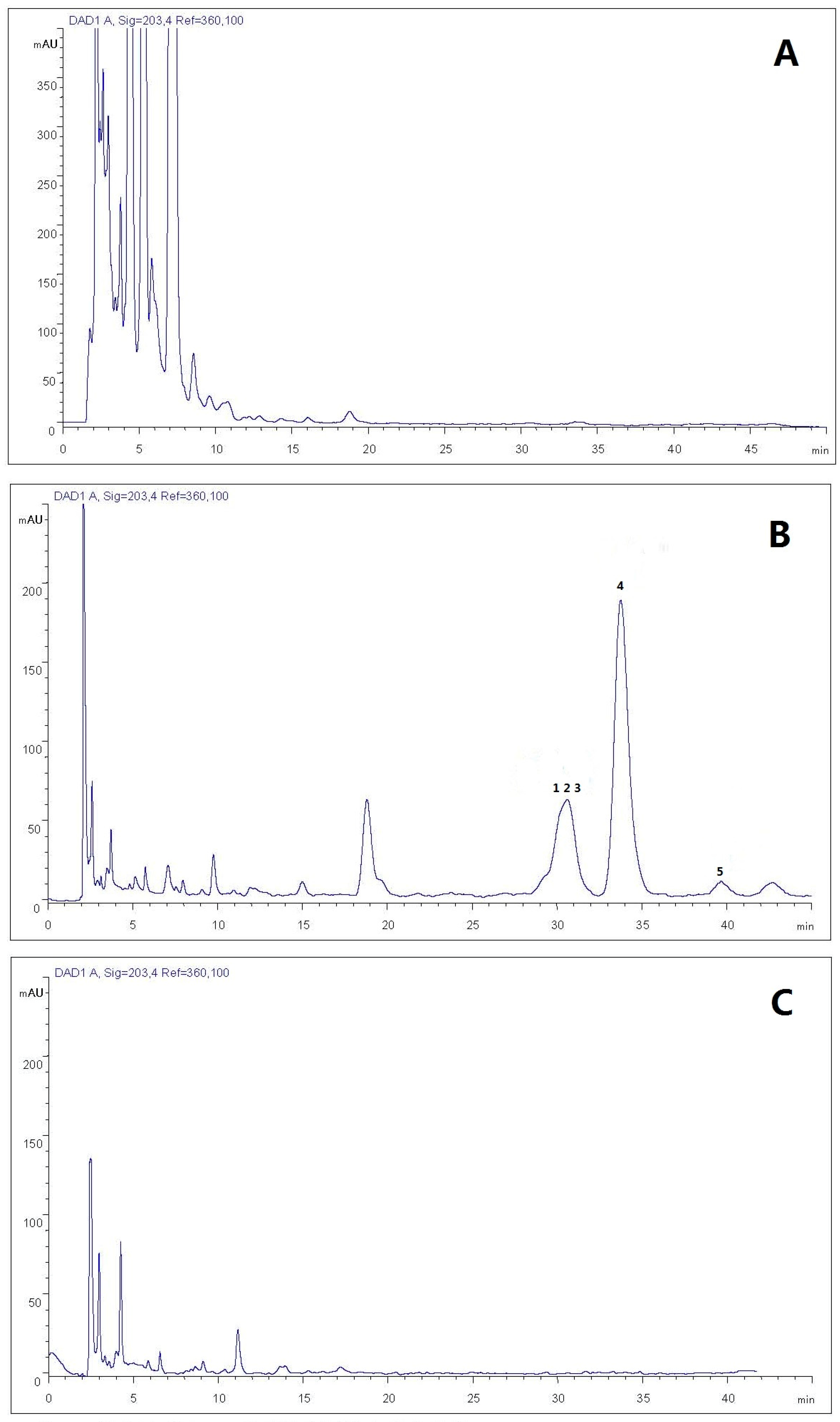
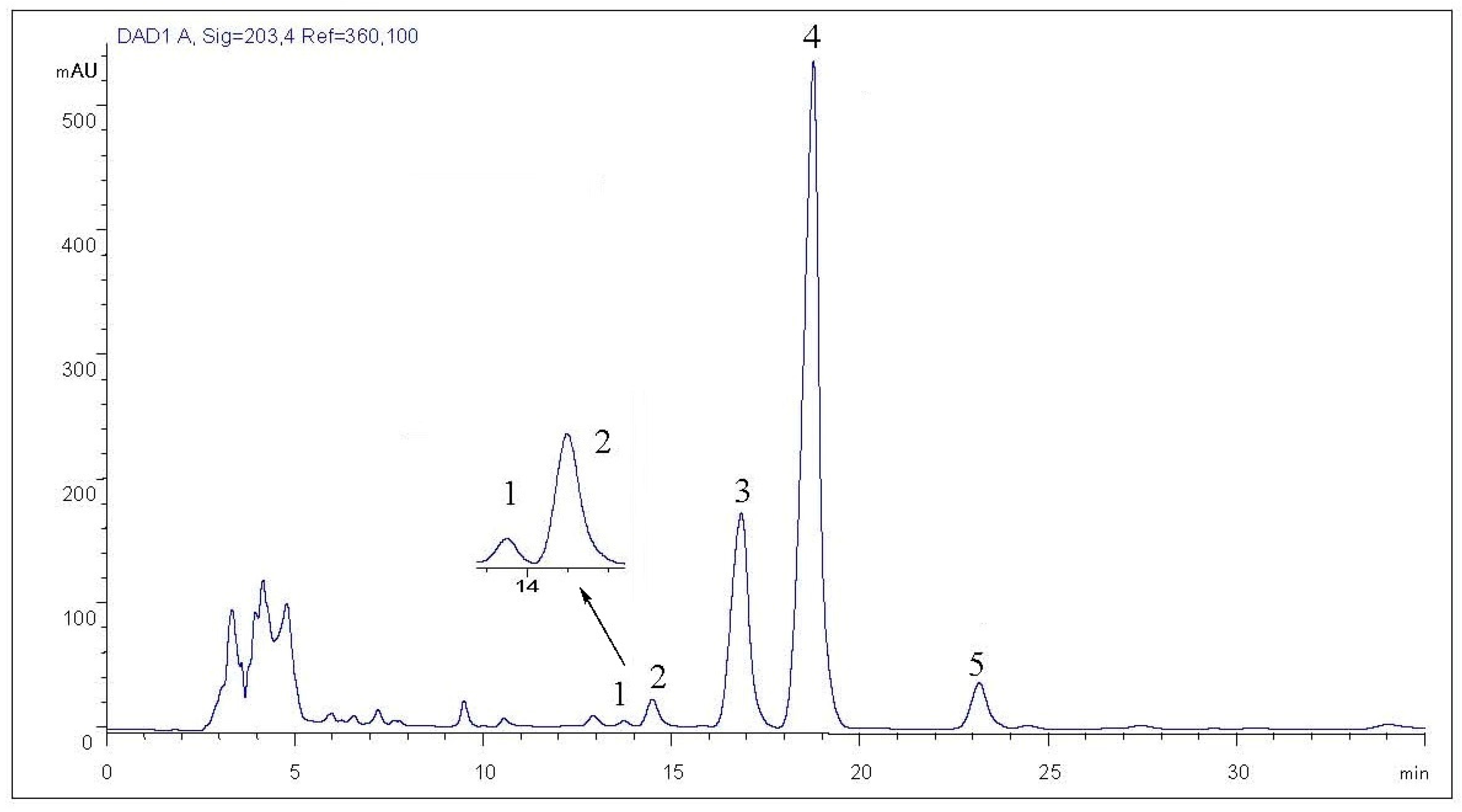
2.1.3. Establishment of HPLC Isolation Conditions
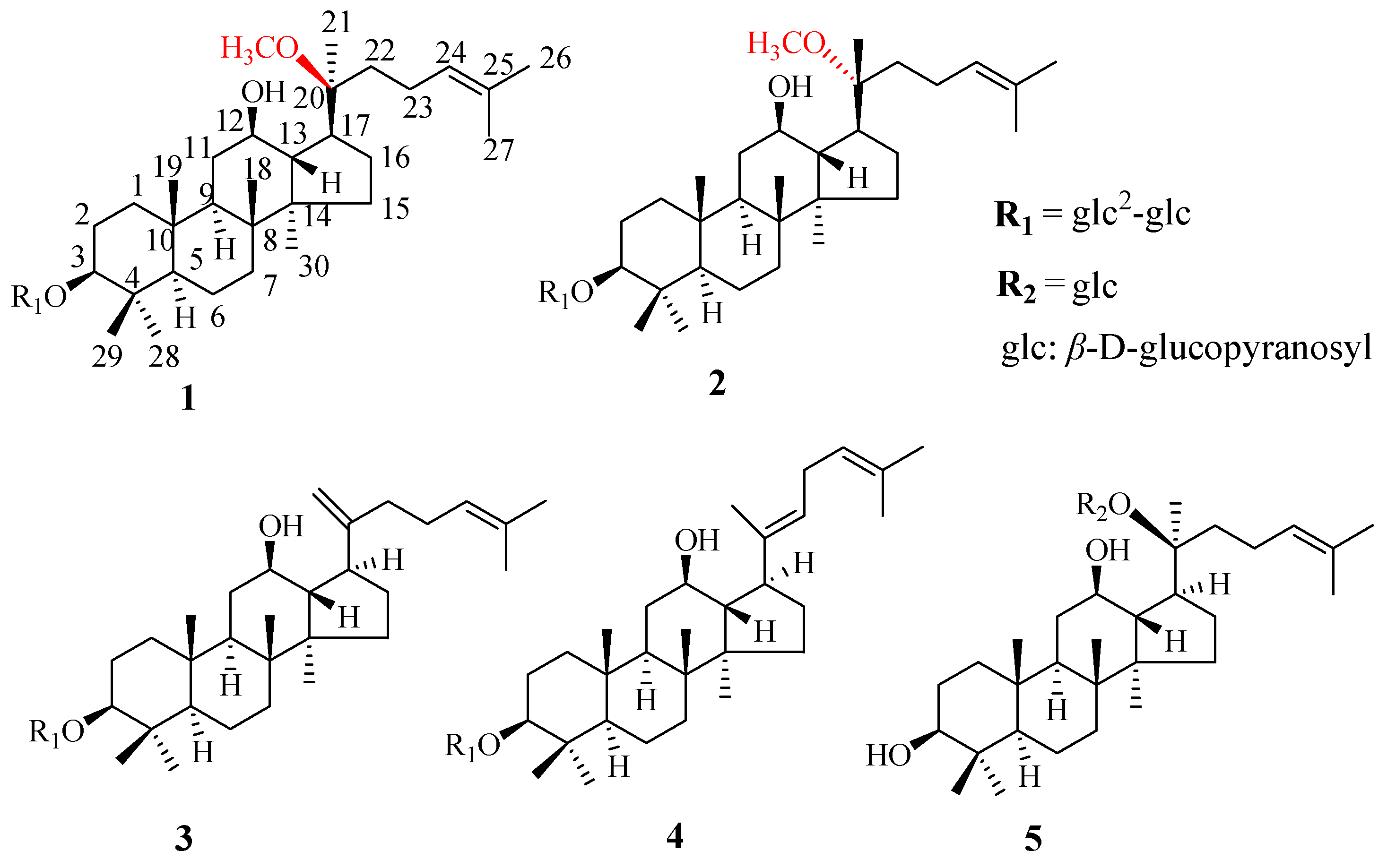
2.2. Identification of the Isolated Ginsenosides
2.3. Method Validation of Quantification
2.4. Quantitative Analysis of the Five Less Polar Ginsenosides in FBPG
3. Materials and Methods
3.1. General
3.2. Plant
3.3. Sample Preparation
3.4. HPLC Analysis and Preparation
3.5. Structural Characterization of New Ginsenosides
3.6. Acid Hydrolysis
3.7. Validation
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| FBPG | Flower buds of Panax ginseng |
| HMBC | Heteronuclear multiple bond correlation |
| HSQC | Heteronuclear singular quantum correlation |
| HR-ESIMS | High resolution electrospray ionization mass spectrometry |
| LC-MS | Liquid chromatography coupled to mass spectrometry |
| LOD | Limits of detection |
| LOQ | Limits of quantification |
| NMR | Nuclear magnetic resonance |
| NOE | Nuclear overhauser effect |
| ROESY | Rotating frame overhauser effect spectroscopy |
| RSD | Relative standard deviation |
| RP-HPLC | Reversed-phase high-performance liquid chromatography |
| S/N | Signal-to-noise |
References
- Christensen, L.P. Ginsenosides chemistry, biosynthesis, analysis, and potential health effects. Adv. Food Nutr. Res. 2009, 55, 1–99. [Google Scholar]
- Li, K.K.; Gong, X.J. A review on the medicinal potentials of Panax ginseng saponins in diabetes mellitus. RSC Adv. 2015, 5, 47353–47366. [Google Scholar] [CrossRef]
- Wong, A.S.T.; Che, C.M.; Leung, K.W. Recent advances in ginseng as cancer therapeutics: A functional and mechanistic. Nat. Prod. Rep. 2015, 32, 256–272. [Google Scholar] [CrossRef]
- Park, J.D.; Rhee, D.K.; Lee, Y.H. Biological activities and chemistry of saponins from Panax ginseng C. A. Meyer. Phytochem. Rev. 2005, 4, 159–175. [Google Scholar] [CrossRef]
- Li, K.K.; Yao, C.M.; Yang, X.W. New dammarane-type triterpene saponins from the stems and leaves of Panax ginseng and their cytotoxicity on HL-60 cells. Planta Med. 2012, 78, 189–192. [Google Scholar] [CrossRef] [PubMed]
- Li, K.K.; Yang, X.B.; Yang, X.W.; Liu, J.X.; Gong, X.J. New triterpenoids from the stems and leaves of Panax ginseng. Fitoterapia 2012, 83, 1030–1035. [Google Scholar] [CrossRef] [PubMed]
- Li, K.K.; Yang, X.W. Advances of chemical constituents in the stems and leaves of Panax ginseng. Zhongguo Xian Dai Zhong Yao 2012, 14, 47–50. (in Chinese). [Google Scholar]
- Yang, W.Z.; Hu, Y.; Wu, W.Y.; Ye, M.; Guo, D.A. Saponins in the genus Panax L. (Araliaceae): A systematic review of their chemical diversity. Phytochemistry 2014, 106, 7–24. [Google Scholar] [CrossRef] [PubMed]
- Attele, A.S.; Wu, J.A.; Yuan, C.S. Ginseng pharmacology: Multiple constituents and multiple actions. Biochem. Pharmacol. 1999, 58, 1685–1693. [Google Scholar] [CrossRef]
- Leung, K.W.; Wong, A.S.T. Pharmacology of ginsenosides: A literature review. Chin. Med. 2010, 5, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, L.; Zhao, Y.Q.; Liang, X.J. Current evaluation of the millennium phytomedicine—Ginseng (II): Collected chemical entities, modern pharmacology, and clinical applications emanated from traditional Chinese medicine. Curr. Med. Chem. 2009, 16, 2924–2942. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Kong, K.F.; Wu, H.; Maricic, N.; Ramalingam, B.; Priestap, H.; Schneper, L.; Quirke, J.M.E.; Høiby, N.; Mathee, K. Panax ginseng has anti-infective activity against opportunistic pathogen Pseudomonas aeruginosa by inhibiting quorum sensing, a bacterial communication process critical for establishing infection. Phytomedicine 2010, 17, 1040–1046. [Google Scholar] [CrossRef] [PubMed]
- Li, R.J.; Ma, Y.F.; Zhai, L.J.; Lu, Y.S.; Chi, X.Q.; Wu, J.S.; Hu, S.H. Enhanced immune response to foot-and-mouth disease vaccine by oral administration of ginseng stem-leaf saponins. Immunopharmacol. Immunotoxicol. 2016, 38, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.; Song, G.Y.; Kim, J.A.; Hyun, J.H.; Kang, H.K.; Kim, Y.H. Dammarane-type saponins from the flower buds of Panax ginseng and their effects on human leukemia cells. Bioorg. Med. Chem. Lett. 2010, 20, 309–314. [Google Scholar] [PubMed]
- Wang, W.; Zhao, Y.Q.; Rayburn, E.R.; Hill, D.L.; Wang, H.; Zhang, R.W. In vitro anti-cancer activity and structure-activity relationships of natural products isolated from fruits of Panax ginseng. Cancer Chemother. Pharmacol. 2007, 59, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Lu, Z.Y.; Teng, Y.R.; Guo, Y.Y.; Liu, S.Y. Structural characterization of ginsenosides from flower buds of Panax ginseng by RRLC-Q-TOF MS. J. Chromatogr. Sci. 2016, 54, 136–143. [Google Scholar] [PubMed]
- Sugimoto, S.; Nakamura, S.; Matsuda, H.; Kitagawa, N.; Yoshikawa, M. Chemical constituents from seeds of Panax ginseng: Structure of new dammarane-type triterpene ketone, panaxadione, and HPLC comparisons of seeds and flesh. Chem. Pharm. Bull. 2009, 57, 283–287. [Google Scholar] [CrossRef] [PubMed]
- Ko, S.K.; Cho, O.S.; Bae, H.M.; Im, B.O.; Lee, O.H.; Lee, B.Y. Quantitative analysis of ginsenosides composition in flower buds of various ginseng plants. J. Korean Soc. Appl. Biol. Chem. 2011, 54, 154–157. [Google Scholar] [CrossRef]
- Yoshikawa, M.; Sugimoto, S.; Nakamura, S.; Sakumae, H.; Matsuda, H. Medicinal flowers. XVI. New dammarane-type triterpene tetraglycosides and gastroprotective principles from flower buds of Panax ginseng. Chem. Pharm. Bull. 2007, 55, 1034–1038. [Google Scholar] [CrossRef] [PubMed]
- Tung, N.H.; Song, G.Y.; Nhiem, N.X.; Ding, Y.; Tai, B.H.; Jin, L.G.; Lim, C.M.; Hyun, J.W.; Park, C.J.; Kang, H.K.; et al. Dammarane-type saponins from the flower buds of Panax ginseng and their intracellular radical scavenging capacity. J. Agric. Food Chem. 2010, 58, 868–874. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.; Song, S.B.; Tung, N.H.; Kim, K.E.; Kim, Y.H. Inhibition of TNF-α-mediated NF-κB transcriptional activity by dammarane-type ginsenosides from steamed flower buds of Panax ginseng in HepG2 and SK-Hep1 cells. Biomol. Ther. 2014, 22, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.Z.; Ye, M.; Qiao, X.; Liu, C.F.; Miao, W.J.; Bo, T.; Tao, H.Y.; Guo, D.A. A strategy for efficient discovery of new natural compounds by integrating orthogonal column chromatography and liquid chromatography/mass spectrometry analysis: Its application in Panax ginseng, Panax quinquefolium and Panax notoginseng to characterize 437 potential new ginsenosides. Anal. Chim. Acta 2012, 739, 56–66. [Google Scholar] [PubMed]
- Qiu, S.; Yang, W.Z.; Shi, X.J.; Yao, C.L.; Yang, M.; Liu, X.; Jiang, B.H.; Wu, W.Y.; Guo, D.A. A green protocol for efficient discovery of novel natural compounds: Characterization of new ginsenosides from the stems and leaves of Panax ginseng as a case study. Anal. Chim. Acta 2015, 893, 65–76. [Google Scholar] [CrossRef] [PubMed]
- Li, K.K.; Xu, F.; Gong, X.J. Isolation, purification and quantification of ginsenoside F5 and F3 isomeric compounds from crude extracts of flower buds of Panax ginseng. Molecules 2016, 21, 315. [Google Scholar] [CrossRef] [PubMed]
- Corbit, R.M.; Ferreira, J.F.; Ebbs, S.D.; Murphy, L.L. Simplified extraction of ginsenosides from American ginseng (Panax quinquefolius L.) for high–performance liquid chromatography-ultraviolet analysis. J. Agric. Food Chem. 2005, 53, 9867–9873. [Google Scholar] [CrossRef] [PubMed]
- Mason, T.J.; Paniwnyk, L.; Lorimer, J.P. The uses of ultrasound in food technology. Ultrason. Sonochem. 1996, 3, S253–S260. [Google Scholar] [CrossRef]
- Wang, C.Z.; Wu, J.A.; McEntee, E.; Yuan, C.S. Saponins composition in American ginseng leaf and berry assayed by high-performance liquid chromatography. J. Agric. Food Chem. 2006, 54, 2261–2266. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.N.; Ha, Y.W.; Shin, H.; Son, S.H.; Wu, S.J.; Kim, Y.S. Simultaneous quantification of 14 ginsenosides in Panax ginseng C.A. Meyer (Korean red ginseng) by HPLC-ELSD and its application to quality control. J. Pharm. Biomed. Anal. 2007, 45, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.W.; Li, K.K.; Zhou, Q.L. 20(S)-ginsenoside-Rf2, a novel triterpenoid saponin from stems and leaves of Panax ginseng. Zhong Cao Yao 2015, 46, 3137–3145. [Google Scholar]
- Yang, J.L.; Ha, T.K.; Dhodary, B.; Kim, K.H.; Park, J.; Lee, C.H.; Kim, Y.C.; Oh, W.K. Dammarane triterpenes as potential SIRT1 activators from the leaves of Panax ginseng. J. Nat. Prod. 2014, 77, 1615–1623. [Google Scholar] [CrossRef] [PubMed]
- Tung, N.H.; Yang, S.Y.; Kim, J.A.; Song, G.Y.; Kim, Y.H. Dammarane-type saponins from the black ginseng. Bull. Korean Chem. Soc. 2010, 31, 3423–3426. [Google Scholar] [CrossRef]
- Li, K.K.; Yang, X.W. A new triterpene natural product from stems and leaves of Panax ginseng. Zhong Cao Yao 2015, 46, 169–173, (in Chinese with English Abstract). [Google Scholar]
- Li, W.F.; Li, Z.N.; Chen, L.R.; Gong, X.J. Synthesis and structural analysis of mono-dodecanoic acid esters of ginsenoside M1. Chin. J. Nat. Med. 2011, 9, 199–203. [Google Scholar]
- Kim, I.W.; Sun, W.S.; Yun, B.S.; Kim, N.R.; Min, D.S.; Kim, S.K. Characterizing a full spectrum of physico-chemical properties of (20S)-and (20R)-ginsenoside Rg3 to be proposed as standard reference materials. J. Ginseng Res. 2013, 37, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Gui, F.J.; Yang, X.W.; Li, L.Y.; Tian, J.M. Simultaneous enantiomer determination of 20(R)- and 20(S)-ginsenoside-Rg2 in rat plasma after intravenous administration using HPLC method. J. Chromatogr. B 2007, 850, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Teng, R.W.; Li, H.Z.; Wang, D.Z.; He, Y.N.; Yang, C.R. NMR complete assignments of three protopanaxodiol monodesmoside. Chin. J. Magn. Reson. 2000, 17, 461–468. [Google Scholar]
- Anufriev, V.P.; Malinovskaya, G.V.; Denisenko, V.A.; Uvaròva, N.I.; Elyakov, G.B.; Kim, S.I.; Baek, N.I. Synthesis of ginsenoside Rg3, a minor constituent of ginseng radix. Carbohydr. Res. 1997, 304, 179–182. [Google Scholar] [CrossRef]
- Tran, Q.L.; Adnyana, I.K.; Tezuka, Y.; Nagaoka, T.; Tran, Q.K.; Kadota, S. Triterpene Saponins from vietnamese ginseng (Panax vietnamensis) and their hepatocytoprotective activity. J. Nat. Prod. 2001, 64, 456–461. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of compounds 1–5 are available from the authors.

| Position | 1 | 2 | ||
|---|---|---|---|---|
| δH | δC | δH | δC | |
| 1 | 0.87 (1H, m) 1.58 (1H, m) | 39.2 | 0.78 (1H, m) 1.52 (1H, m) | 39.2 |
| 2 | 1.24 (1H, m) 1.86 (1H, m) | 26.3 | 1.29 (1H, m) 1.89 (1H, m) | 26.3 |
| 3 | 3.32 (1H, dd, 12.0, 4.5) | 89.0 | 3.32 (1H, dd, 11.5, 4.0) | 89.0 |
| 4 | - | 39.7 | - | 39.7 |
| 5 | 0.73 (1H, br d, 12.0) | 56.4 | 0.72 (1H, br d, 11.5) | 56.4 |
| 6 | 1.32 (1H, m) 1.47 (1H, dd, 12.0, 4.2) | 18.5 | 1.40 (1H, m) 1.52 (1H, dd, 12.0, 4.2) | 18.5 |
| 7 | 1.22 (1H, m) 1.51 (1H, m) | 35.2 | 1.24 (1H, m) 1.47 (1H, m) | 35.2 |
| 8 | - | 40.0 | - | 40.1 |
| 9 | 1.41 (1H, m) | 50.0 | 1.43 (1H, m) | 50.2 |
| 10 | - | 37.0 | - | 37.0 |
| 11 | 1.38 (1H, m) 2.03 (1H, m) | 31.2 | 1.45 (1H, m) 2.04 (1H, dd, 12.0, 7.0) | 31.4 |
| 12 | 3.76 (1H, dt, 9.5, 5.5) | 70.4 | 3.81 (1H, dt, 10.0, 3.0) | 70.6 |
| 13 | 1.86 (1H, t, 11.0) | 49.0 | 1.83 (1H, t, 10.5) | 49.7 |
| 14 | - | 51.5 | - | 51.8 |
| 15 | 1.05 (1H, t, 11.0) 1.57 (1H, m) | 30.9 | 1.05 (1H, t, 10.0) 1.54 (1H, m) | 31.3 |
| 16 | 1.73 (1H, m) 2.23 (1H, m) | 26.8 | 1.85 (1H, m) 2.22 (1H, m) | 26.8 |
| 17 | 2.42 (1H, td, 9.0) | 50.0 | 2.35 (1H, td, 7.5) | 47.2 |
| 18 | 0.95 (3H, s) | 15.9 | 1.00 (3H, s) | 15.9 |
| 19 | 0.83 (3H, s) | 16.3 | 0.86 (3H, s) | 16.4 |
| 20 | - | 79.9 | - | 80.2 |
| 21 | 1.19 (3H, s) | 21.2 | 1.17 (3H, s) | 18.5 |
| 22 | 1.67 (1H, m) 2.04 (1H, m) | 35.1 | 1.44 (1H, m) 1.66 (1H, m) | 35.7 |
| 23 | 2.05 (1H, m) 2.30 (1H, m) | 22.8 | 2.13 (1H, m) n.d. | 21.7 |
| 24 | 5.25 (1H, t, 6.5) | 125.5 | 5.23 (1H, t, 7.5) | 125.0 |
| 25 | - | 131.3 | - | 131.4 |
| 26 | 1.67 (3H, s) | 25.8 | 1.75 (3H, s) | 25.8 |
| 27 | 1.64 (3H, s) | 17.7 | 1.67 (3H, s) | 17.7 |
| 28 | 1.32 (3H, s) | 28.2 | 1.33 (3H, s) | 28.2 |
| 29 | 1.13 (3H, s) | 16.7 | 1.14 (3H, s) | 16.6 |
| 30 | 0.96 (3H, s) | 17.1 | 0.98 (3H, s) | 17.4 |
| 20-OCH3 | 3.27 (3H, s) | 48.8 | 3.20 (3H, s) | 48.4 |
| 1′ | 4.95 (1H, d, 7.5) | 105.1 | 4.96 (1H, d, 7.5) | 105.2 |
| 2′ | 4.26 (1H, m) | 83.5 | 4.25 (1H, m) | 83.5 |
| 3′ | 4.28 (1H, m) | 78.0 | 4.27 (1H, m) | 78.0 |
| 4′ | 4.18 (1H, t, 10.0) | 71.7 | 4.17 (1H, t, 9.0) | 71.7 |
| 5′ | 3.96 (1H, m) | 78.3 | 3.96 (1H, m) | 78.3 |
| 6′ | 4.50 (1H, m) 4.58 (1H, br d, 11.0) | 62.9 | 4.49 (1H, m) 4.58 (1H, br d, 11.5) | 62.9 |
| 1″ | 5.40 (1H, d, 7.5) | 106.1 | 5.41 (1H, d, 7.5) | 106.1 |
| 2″ | 4.16 (1H, t, 10.0) | 77.2 | 4.16 (1H, t, 9.0) | 77.2 |
| 3″ | 4.33 (1H, m) | 78.4 | 4.34 (1H, m) | 78.4 |
| 4″ | 4.36 (1H, m) | 71.8 | 4.36 (1H, m) | 71.8 |
| 5″ | 3.94 (1H, m) | 78.2 | 3.94 (1H, m) | 78.1 |
| 6″ | 4.32 (1H, m) 4.50 (1H, m) | 62.8 | 4.38 (1H, br d, 11.5) 4.50 (1H, m) | 62.8 |
| Ginsenoside | C-17 | C-20 | C-21 | C-22 |
|---|---|---|---|---|
| 20(R)-methoxyl-ginsenoside Rg3 | 47.2 | 80.2 | 18.5 | 35.7 |
| 20(S)-methoxyl-ginsenoside Rg3 | 50.0 | 79.9 | 21.2 | 35.1 |
| Δ20(R)-20(S)δ | −2.8 | 0.3 | −2.7 | 0.6 |
| 20(R)-ginsenoside Rg3 [27] | 50.7 | 73.0 | 22.8 | 43.3 |
| 20(S)-ginsenoside Rg3 | 54.8 | 73.0 | 27.1 | 35.9 |
| Δ20(R)-20(S)δ | −4.1 | 0 | −4.3 | 7.4 |
| 20(R)-ginsenoside Rf2 [27] | 51.8 | 73.4 | 22.9 | 43.6 |
| 20(S)-ginsenoside Rf2 | 54.7 | 72.5 | 27.2 | 36.4 |
| Δ20(R)-20(S)δ | −2.9 | 0.9 | −4.3 | 7.2 |
| 20(R)-ginsenoside Rh2 [27] | 52.2 | 73.4 | 23.0 | 43.7 |
| 20(S)-ginsenoside Rh2 | 54.8 | 72.9 | 26.9 | 35.2 |
| Δ20(R)-20(S)δ | −2.6 | 0.5 | −3.9 | 8.5 |
| Compounds * | Calibration Curve | Correlation Coefficient (R2) | Test Range (μg/mL) | LOD (μg) | LOQ (μg) |
|---|---|---|---|---|---|
| 1 | y = 2359.3x + 65.67 | 0.9998 | 8–80 | 0.086 | 0.262 |
| 2 | y = 2714.9x + 46.11 | 0.9998 | 100–1000 | 0.074 | 0.275 |
| 3 | y = 6665.5x + 270.39 | 0.9998 | 400–1500 | 0.092 | 0.258 |
| 4 | y = 3332.6x + 139.30 | 0.9999 | 1000–5000 | 0.069 | 0.232 |
| 5 | y = 2640.6x + 140.72 | 0.9994 | 100–1000 | 0.088 | 0.263 |
| Compounds | Precision | |||
|---|---|---|---|---|
| Intra-Day (n = 3) | Inter-Day (n = 5) | |||
| Content (μg/mL) | RSD (%) | Content (μg/mL) | RSD (%) | |
| 1 | 80.34 ± 1.02 | 1.51 | 80.58 ± 1.24 | 1.54 |
| 2 | 200.26 ± 1.52 | 0.76 | 200.47 ± 1.82 | 0.91 |
| 3 | 501.72 ± 2.55 | 0.51 | 500.29 ± 2.78 | 0.56 |
| 4 | 1401.89 ± 4.93 | 0.35 | 1401.25 ± 5.78 | 0.41 |
| 5 | 203.57 ± 1.58 | 0.78 | 202.15 ± 2.51 | 1.24 |
| Compounds | Original (μg) | Spiked (μg) | Found (μg) | Recovery (%) | RSD (%) |
|---|---|---|---|---|---|
| 1 | 5.70 | 35.00 | 40.58 ± 0.41 | 97.89 | 1.00 |
| 2 | 106.92 | 150.00 | 256.26 ± 2.08 | 99.38 | 0.81 |
| 3 | 512.03 | 500.00 | 1010.06 ± 5.23 | 99.61 | 0.52 |
| 4 | 3123.73 | 400.00 | 3521.27 ± 5.65 | 99.92 | 0.16 |
| 5 | 240.24 | 200.00 | 439.16 ± 5.36 | 99.54 | 1.22 |
| Compounds | Content (μg/g FBPG) (n = 9) (mean ± SD) |
|---|---|
| 1 | 1.77 ± 0.93 |
| 2 | 33.26 ± 7.46 |
| 3 | 159.31 ± 17.12 |
| 4 | 971.28 ± 28.05 |
| 5 | 74.65 ± 8.42 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.-S.; Li, K.-K.; Xu, F.; Tao, L.; Yang, L.; Chen, S.-X.; Gong, X.-J. A Strategy for Simultaneous Isolation of Less Polar Ginsenosides, Including a Pair of New 20-Methoxyl Isomers, from Flower Buds of Panax ginseng. Molecules 2017, 22, 442. https://doi.org/10.3390/molecules22030442
Li S-S, Li K-K, Xu F, Tao L, Yang L, Chen S-X, Gong X-J. A Strategy for Simultaneous Isolation of Less Polar Ginsenosides, Including a Pair of New 20-Methoxyl Isomers, from Flower Buds of Panax ginseng. Molecules. 2017; 22(3):442. https://doi.org/10.3390/molecules22030442
Chicago/Turabian StyleLi, Sha-Sha, Ke-Ke Li, Fei Xu, Li Tao, Li Yang, Shu-Xiao Chen, and Xiao-Jie Gong. 2017. "A Strategy for Simultaneous Isolation of Less Polar Ginsenosides, Including a Pair of New 20-Methoxyl Isomers, from Flower Buds of Panax ginseng" Molecules 22, no. 3: 442. https://doi.org/10.3390/molecules22030442