1. Introduction
Anoectochilus formosanus Hayata, also known as the Taiwan jewel orchid (2
n = 2
x = 24), is a small terrestrial orchid with high medicinal value grown in Taiwan and other countries [
1]. This plant is also called “The King of Medicine” or “The Tiger of Medicine” because of its multiple pharmaceutical effects, including protection of liver, anti-inflammatory, treatment of chest and abdominal pains, diabetes, nephritis, fever, cardiovascular diseases, and cancer prevention [
2,
3,
4]. Due to their high economical value, wild Taiwan jewel orchids have been harvested excessively and were threatened. In spite of the importance of medical applications, there are only two reliable tissue culture protocols for mass propagation of this species, mainly via asymbiotic germination and multiple shoot induction from nodal stems, respectively [
5,
6]. In this present study, we use nodal stems as explants to induce polyplidy via shoot induction. Otherwise, based on our knowledge, limited success was found in studying the physiology, genetics, and plant breeding of this species.
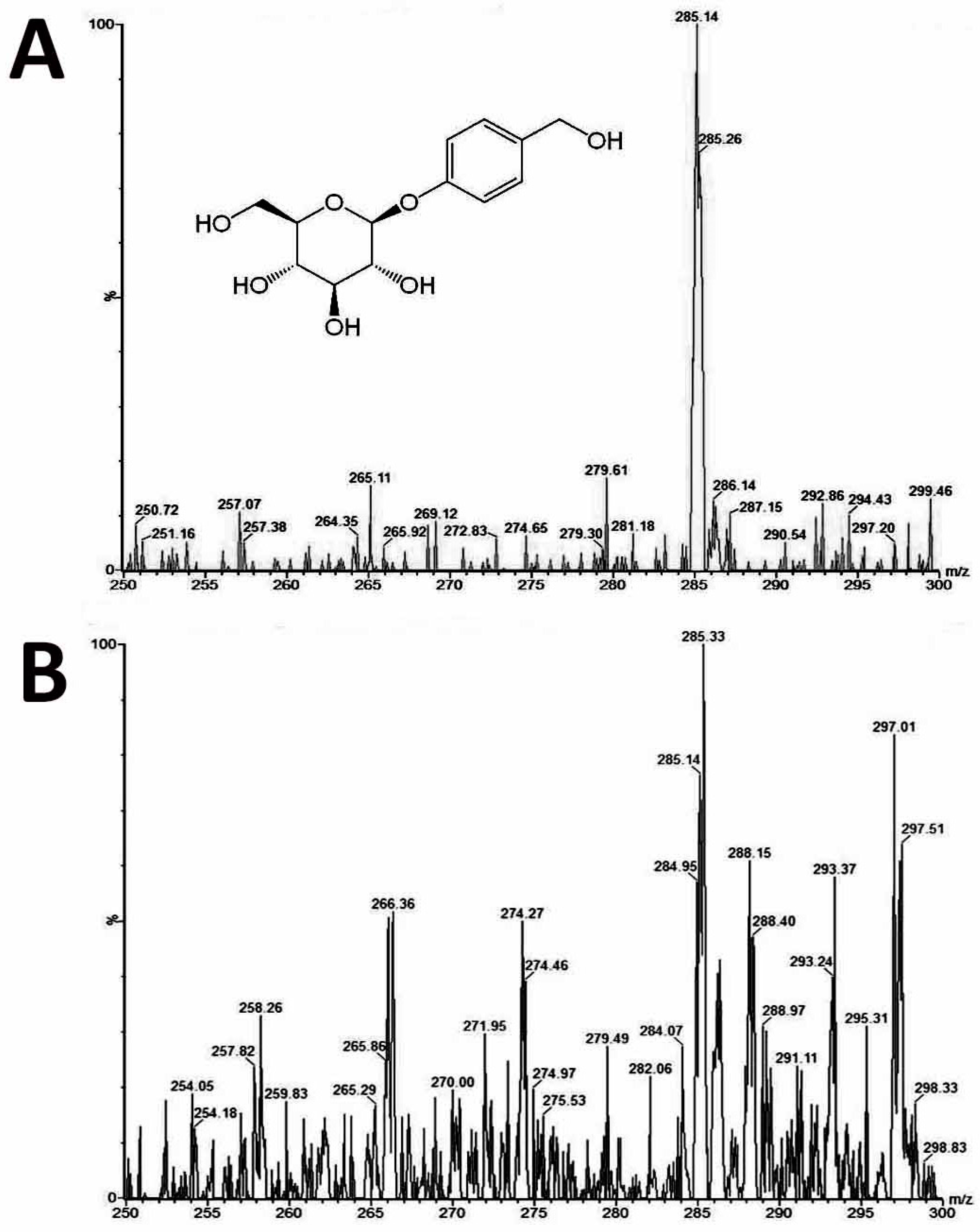
Gastrodin is an effective bioactive compound found in
A. formosanus and other medicinal orchids [
7]. It can promote the secretion of brain-derived neurotrophic factor and thus has considerable effects in clinical treatment of central nervous system disorders, especially the recovery of neurological function and to protect neural cells against injury [
7,
8]. Flavonoids are plant secondary metabolites, and they have beneficial functions in human health, i.e., the prevention and treatment of different pathologies via a protection from the attack by reactive oxygen species (ROS) or other mechanisms [
9]. It has been reported that flavonoid glycosides and their derivatives of
A. formosanus resulted in strong antioxidant properties that could prevent oxidative stress in human systems and thus have potential usages in cancer chemoprevention [
10,
11]. Polyploidy induction is a powerful technique to produce enhanced agronomic characteristic for plant breeding and altered genotypes for genetic studies [
12,
13]. In addition, ploidy manipulations can provide a rapid means or a promising parameter to realize enhanced production of phyto-pharmaceuticals [
14,
15]. In this present study, an efficient and reliable system for inducing tetraploidy in the Taiwan jewel orchid was established. The tetraploids were selected for determination of gastrodin and total flavonoid to test the effect of the ploidy level on the production of bioactive constituents in this species.
3. Discussion
Dealing with polyploidy induction, the explant type together with its resulting regeneration pathway could be crucial factors for the successful induction rate and also the purity of the polyploids [
13]. Therefore, for inducing of polyploidy in medicinal plants, various explant types have been used, including apical buds, callus, cotyledonary nodes, leaf explants, seeds, shoots, shoot tips, and zygotic embryos [
13,
16,
17,
18]. In this present study, the nodal-stem explants of
A. formosanus performed well in the polyploidy induction via a three-day treatment of colchicine in the liquid medium. Here, we suggest that this process could increase the exposure area of the shoot bud to the antimitotic agent and thus may reduce chimera.
TDZ was proposed as a multidimensional plant growth regulator that has both auxin and cytokinin effects and could induce a diverse array of in vitro morphogenesis [
19,
20,
21,
22,
23]. Ket et al. reported that TDZ could induce multiple shoot proliferation of
A. formosanus [
6]. Therefore, we suggest that TDZ was beneficial for polyploidy induction in
A. formosanus because of its enhancement of the plantlet survival in the presence of colchicine which severely retarded growth of the explants.
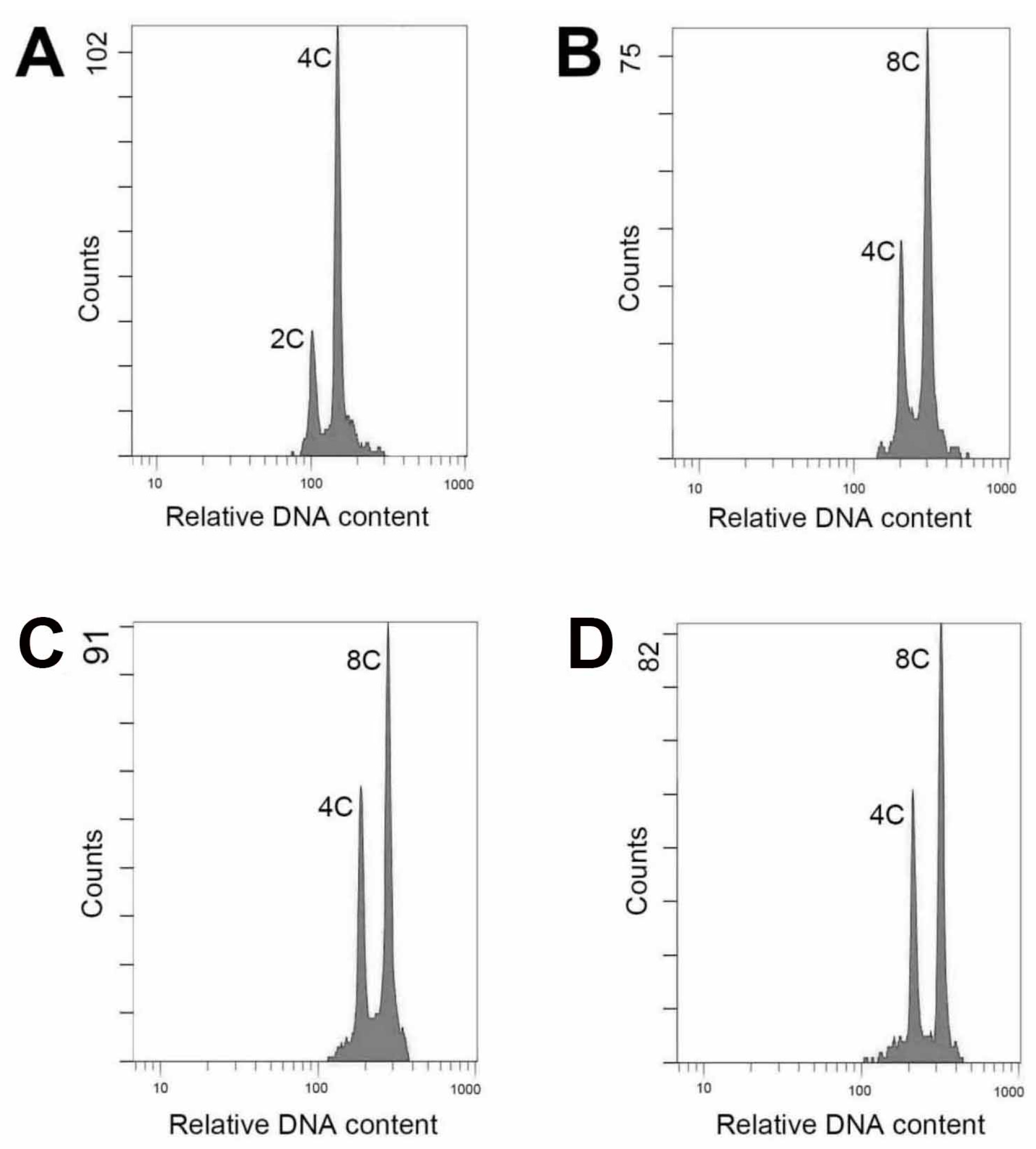
The ploidy level of plants could be analyzed using several protocols, including chromosome counting, flow cytometric analysis, morphological observation and evaluation of anatomical parameters [
18,
24,
25,
26,
27]. It has been reported that flow cytometry is an efficient and reliable protocol and was used to evaluate polyploidy in 63% of previous related publications [
13]. In this study, there was a satisfactory performance when evaluated ploidy level using flow cytometry. The technique is efficient and the materials for testing comprise young leaf tissues could be obtained easily and without killing the parent plant.
It was reported that ploidy could affect many morphological and fitness traits, including stomatal size, flower size, seed weight, and biomass [
28,
29]. In this study, similar marked differences were found in several agronomic traits, including biomass, plant size, leaf shape, and stomatal morphology. In polyploidy selection, the increased leaf size has been considered as one of the remarkable changes in morphology by the ploidy level [
30]. The tetraploid of
Lolium cultivars had a faster leaf elongation rate and consequently longer mature cells than did the diploid [
31]. In
A. formosanus, the tetraploid had a significantly higher leaf width than did the diploid. Consequently, the ratio of leaf length to width in the tetraploid was lower down to approximately 0.95. Therefore, the leaf shape was dramatically changed by the ploidy level, i.e., the leaf shape of the diploid was ovate, but the tetraploid had a cordate leaf. Here we suggest that leaf size and leaf shape could be a reliable indicator for identification of polyploidy.
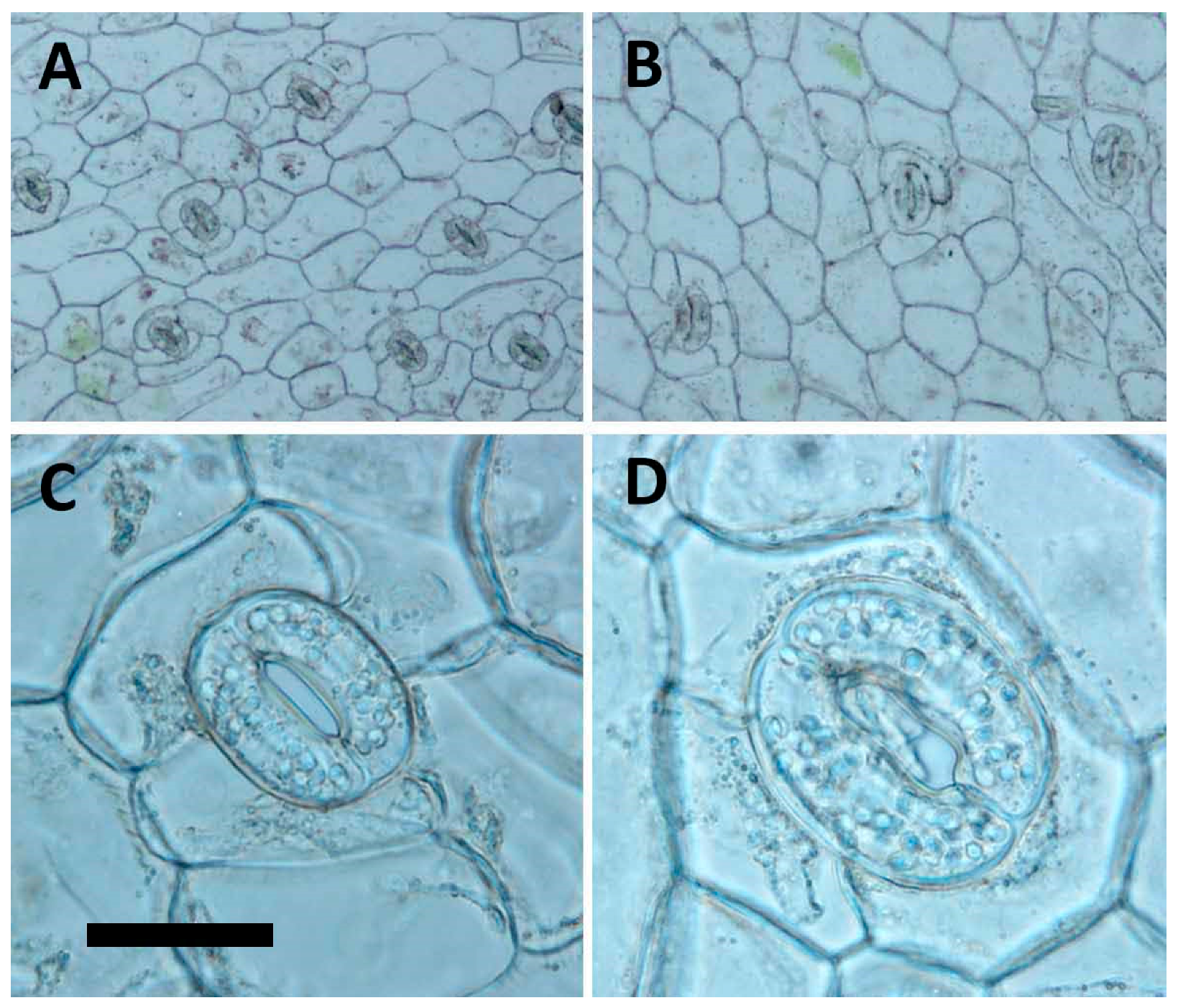
The morphology of stomata had been documented as a reliable indicator for polyploidy selection [
30,
32]. In
A. formosanus, the tetraploid had a dramatically change in frequency, size, the ratio of length to width, and number of chloroplasts of stomata, and therefore the stomatal morphology, could be a reliable selection indicator for polyploidy inducing in the future test.
In theory, the regeneration pathway without the intervention of callus is an efficient method of obtaining regenerants and could reduce unnecessary somaclonal variations [
23,
33]. In the present study, the 4
x plants of
A. formosanus showed a high stability in ploidy level as their tenth generation of clonal regenerants derived from nodal stem cuttings still maintained the polyploidy (2
n = 4
x) after five years of subculturing.
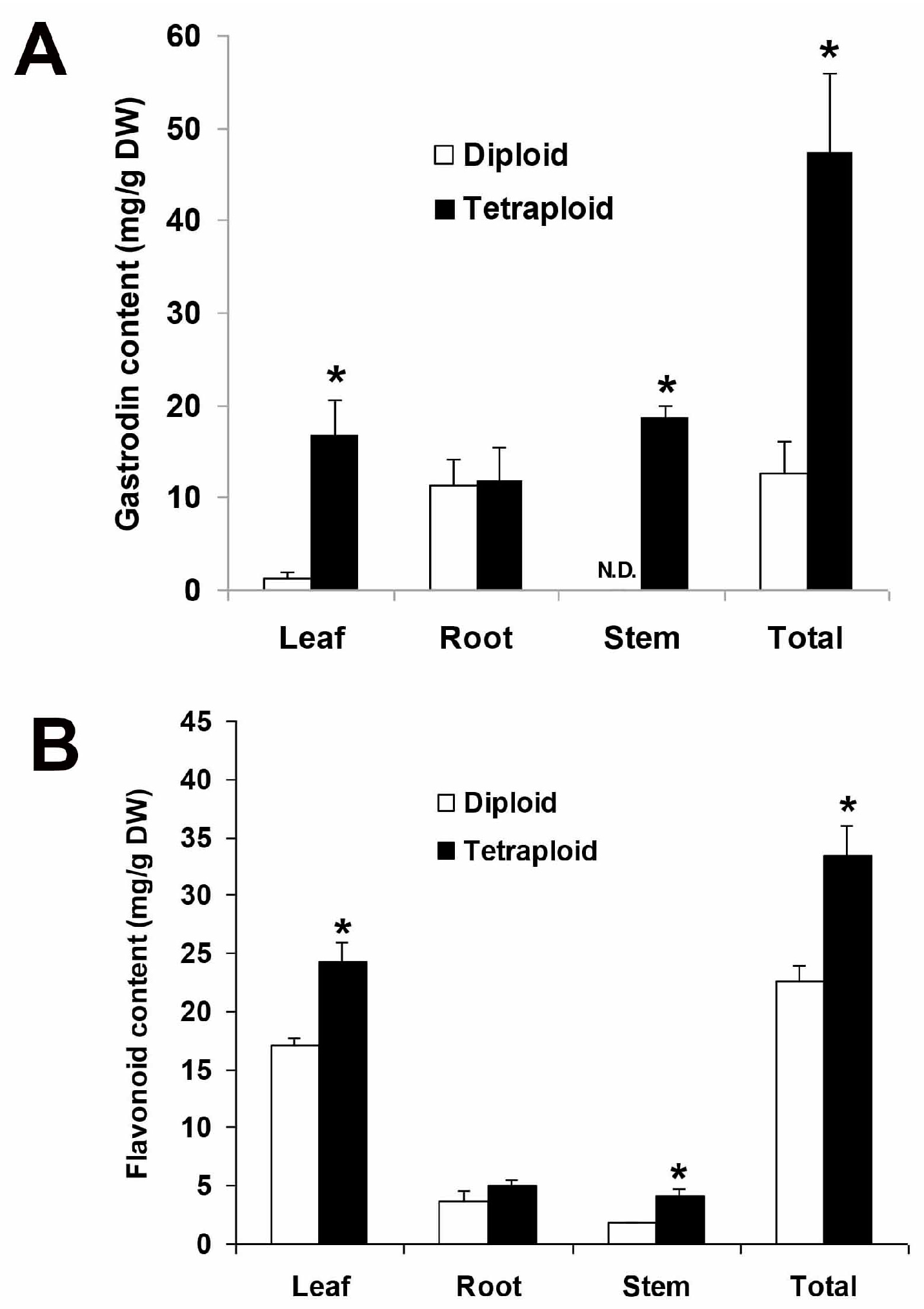
Tetraploidy could enhance the production of bioactive compounds via heritable genetic variation or overexpression of relative biosynthesis pathways, e.g., the tropane biosynthetic pathway for scopolamine production in
Hyoscyamus muticus [
34,
35]. The tetraploid of
A. formosanus had a significantly higher content of gastrodin in the leaf, stem, and the whole plants. Therefore, here we suggest that the tetraploid may possess a higher activity of phenolic glycoside biosynthesis and consequently could accumulate a higher content of gastrodin. In previous reports, there were marked differences in flavonoid accumulation in relation to ploidy level [
35,
36]. A similar result was also found in this present study, the tetraploid had a significantly higher total content of flavonoids than the diploid in
A. formosanus.
In conclusion, an efficient and reliable system for polyploidy induction of A. formosanus was successfully established in this study. The resulting tetraploids showed a significant promotion on various agronomic traits and an alternation on organ shape. In addition, the tetraploid produced significantly higher contents of bioactive compounds, including total flavonoid and gastrodin.
4. Materials and Methods
4.1. Plant Materials
In vitro-grown clonal Anoectochilus formosanus Hayata plants (2n = 2x = 24) from Winpower Technology Co. (Kaohsiung, Taiwan) were used as donor plants in this study. These plants were maintained in vitro for approximately 3 months, and each plant had approximately 3–4 nodes and 8–10 leaves.
4.2. Subculture of the In Vitro–Grown Plants
The in vitro-grown plants were removed the leaves and cut into nodal-stem segments, then propagated on the basal medium (BM) containing full strength medium Murashige and Skoog (1962) medium (MS) [
37] salt and vitamins, 30 g/L sucrose and 9 g/L agar. The pH of the media was adjusted to 5.8 with 1M KOH or HCl prior to autoclaving for 20 min at 121 °C. The culture containers were 600-mL flasks, and each contained 100 ml of the medium. The cultures were incubated in a growth chamber with a 16/8 h (light/dark) photoperiod at an irradiance of 42–55 μmol m
−2 s
−1 (daylight fluorescent tubes FL-20BR/18, 18 W, (China Electric Co., Taipei, Taiwan)) and a temperature of 25 ± 2 °C. The subculture period is approximately six weeks.
4.3. Induction of Polyploidy
The nodal-stem explants (the leaves were removed and each contained three nodes) were used to test the effects of colchicine on polyploidy induction. Various concentrations of colchicine (i.e., 0, 100, 250, 500, and 1000 mg/L) were added to liquid BM for induction of polyploidy. Following three days of induction, the cultures were transferred onto three different media, including hormone-free 0.5 mg/L BA and 0.5 mg/L TDZ-containing solid BM that was devoid of colchicine, to induce shoot development. Following an additional six weeks, all the cultures were transferred to solid BM supplemented with 0.5 mg/L 1H-Indole-3-butanoic acid (indole-3-butyric acid, IBA) to obtain plantlets. The light and temperature conditions were the same as mention above. Four replicates (each contains five nodal-stem explants) were performed in each treatment.
4.4. Flow Cytometric Analysis
The young leaves of the nodal-stem-derived plantlets, including the wild type and putative polyploids (first generation), were used for identification of the polyploidy. The analysis was performed using the flow cytometer (CytomicsTM FC500, Beckman Coulter Inc., Indianapolis, IN, USA). The fresh leaves (each was 2 × 2 cm2) were chopped with a razor blade in a 6 cm glass dish containing 500 μL of the extracting buffer (containing 10 mL/L Triton X-100, 6.3 g/L Na2SO3 and 50 mL/L 1 M Tris-Hcl) and filtered through a 30 μm nylon mesh. The released nuclei were stained with 10 μL/L of propidium iodide (No. AVK250, Strong Biotech Corp., Taipei, Taiwan) for further analysis. For each sample, approximately 2000 nuclei were analyzed.
4.5. Analysis of Stability of Polyploidy
Ploidy level and morphological characteristics (stomata density, length of stomata, width of stomata, and leaf shape) of tenth generation (five years) of clonal plants that derived from in vitro nodal stem cultures and seedlings obtained via self-pollination of 4x plants (first generation) were evaluated to prove the purity and stability of their polyploidy. Young leaves, lateral shoots and root tips from 1.5-year-old plants were harvested separately for flow cytometric analysis.
4.6. Analysis of Stomata and Agronomic Traits
The fully expanded leaves from the third node of nine-month-old in vitro plantlets (tenth generation) were used to compare the difference in stomata between the 2x plants to the 4x plants. Data of stomata density and length and width of stomata were collected using Olympus BX41 microscope (Tokyo, Japan) at magnifications of 100×, 400× and 400×, respectively. The epidermis of leaves was peeled for analysis of stomata density at an area of 500 × 500 μm2 (at a magnification of 100×). The pictures were taken at a magnification of 100× or 400× with DP20 microscope camera (Olympus, Tokyo, Japan) and application software (DP2-BSW-E, Olympus, Tokyo, Japan). The nine-month-old plantlets (tenth generation) were also used to analyse differences in agronomic traits, including dry weight, fresh weight, shoot length, shoot diameter, leaf length, leaf width, length/width ratio of leave, and leaf shape.
4.7. Acclimatization of Regenerated Plants
Nine-month-old plantlets (tenth generation) with well-developed shoots and roots were transplanted into 3-inch pots with peat moss and vermiculite (1:1) for acclimatization. The plants were incubated for acclimatization in a growth chamber with a 16/8 h (light/dark) photoperiod and a temperature of 25 ± 2 °C. A commercial powder fertilizer (Hyponex No. 5, N-P-K = 30-10-10, Hyponex Co., Marysville, OH, USA) was applied together with the irrigation at one-week intervals.
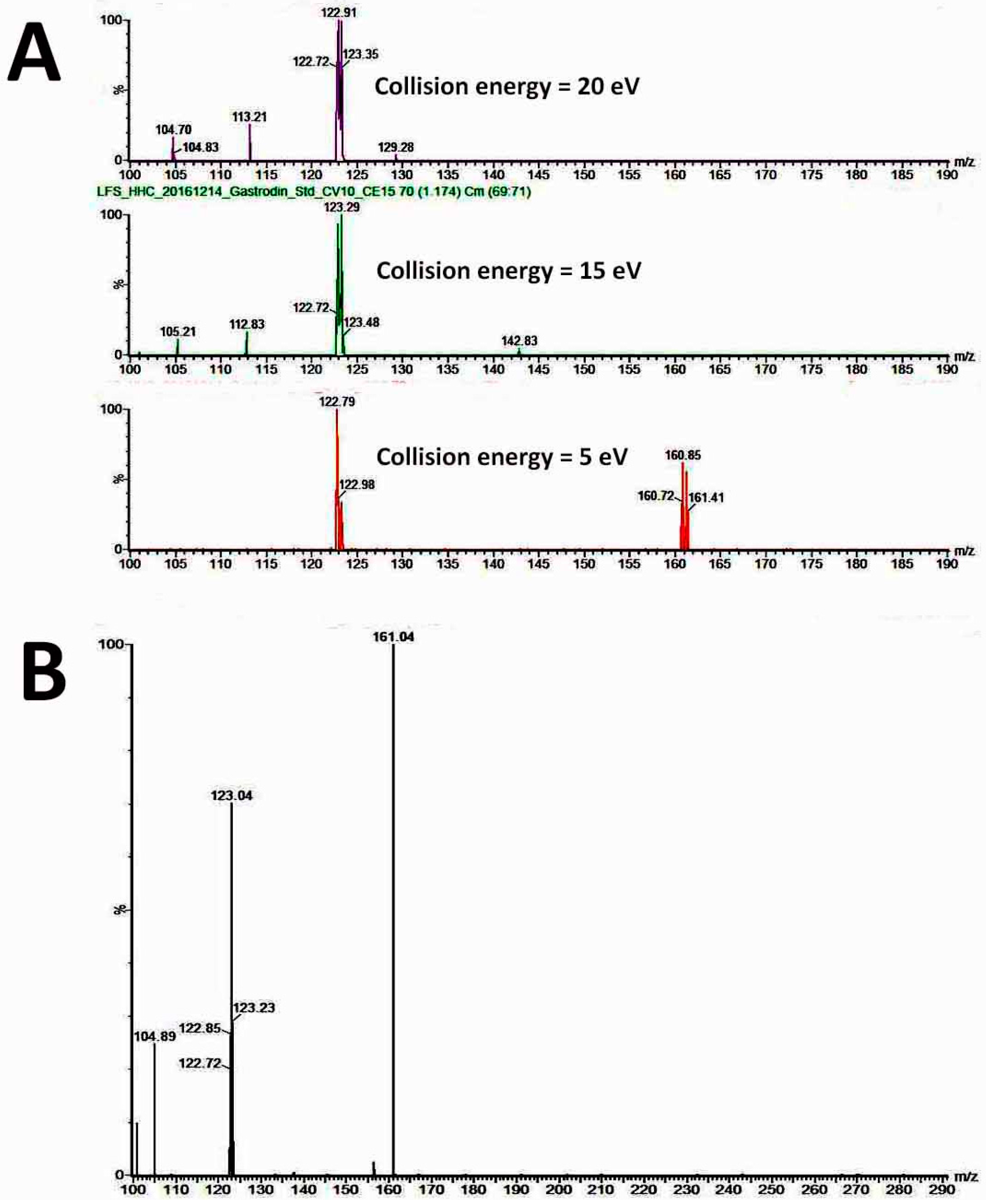
4.8. Determination of the Gastrodin Content
Leaves, roots and stems from 1.5-year-old plants of diploids and tetraploids (tenth generation) were harvested separately for analysis of the gastrodin content (mg/g DW). The tissues were dried and pulverised into powder, and extracted by distilled water (1 mL for 100 mg of fresh sample) for one day and then sonicated for 20 min. All extracts were centrifuged at 13,000 rpm for 10 min, and the supernatants were filtered by 0.22 μm PES syringe filters to remove the residues. The crude extracts were condensed to 5% of initial volume in a CentriVap Centrifugal Vacuum Concentrators (Labconco Corp., Kansas City, MO, USA). The fresh samples were collected and dried in the oven at 70 °C for calculating the ratio of dry and fresh weight. The system used for analysis of gastrodin was by under ultra-performance liquid chromatography (UPLC) system (ACQUITY UPLC, Waters Corp., Milford, CT, USA) with ACQUITY UPLC BEH C18 column (particle size 1.7 μm, 2.1 × 100 mm). The flow rate was at 400 uL/min, and a 10-min gradient was used for analysis. Two Mobile phases were used including solvent A contained 2% acetonitrile in water, and solvent B was acetonitrile. The extracts were separated by under the solvent gradient in the following manner: 0.5–99.5% of solvent B (0–4 min), 99.5% of solvent B (4–6 min), 99.5–0.5% of solvent B (6–6.5 min) and 0.5% of solvent B (6.5–10 min). The sample was reconstituted in H2O and injected 10μL into the UPLC system. The UPLC system was coupled online to the Waters Xevo TQ-S triple quadrupole mass spectrometer. The instrument was operated in negative multiple reaction-monitoring mode. The calibration curve was established using gastrodin (≥98% of purity) (SMB00313, Sigma-Aldrich Inc., St. Louis, MO, USA). Data acquisition and processing were performed using MassLynx version 4.1 and TargetLynx software (Waters Corp., Milford, CT, USA). The gastrodin compounds were eluted at 1.16 min (retention time) after injection, and their peak areas were calculated for quantification.
4.9. Determination of the Flavonoid Content
The total flavonoid content (mg/g DW) was evaluated using the aluminum chloride colorimetric method [
38]. Leaves, roots, and stems from 1.5-year-old plants of diploids and tetraploids (tenth generation) were harvested separately for analysis. The tissues were dried and pulverised into powder and extracted by distilled water (1 mL for 100 mg of fresh sample) for one day and then sonicated for 20 min. All extracts were centrifuged at 13,000 rpm for 10 min, and the supernatants were filtered by 0.22 μm PES syringe filters to remove the residues. The crude extracts were used for determination of the flavonoid content. Each reaction contained 100 μL crude extract and 100 μL of 2% AlCl
3·6H
2O solution. After l hour of incubation at room temperature, the absorbance was determined at 415 nm by BioTek Eon microplate spectrophotometer (BioTek Instruments, Inc., Winooski, VT, USA). The calibration curve was established using rutin (0–200 mg/L) as the standard. Total flavonoid content is showed as mg rutin equivalents per g of dry weight.
4.10. Statistical Analysis
The experiment was designed with a randomized complete block design, and each treatment contained at least four replicates. The analysis of bioactive compounds was repeated for three times. Analysis of variance (ANOVA) was used for data evaluation. The significant differences among the treatments were compared using the Duncan multiple range test [
39] with a 0.05 level of probability.