Ni(II) Complexes with Schiff Base Ligands: Preparation, Characterization, DNA/Protein Interaction and Cytotoxicity Studies
Abstract
:1. Introduction
2. Results and Discussion
2.1. Spectroscopic Characterization
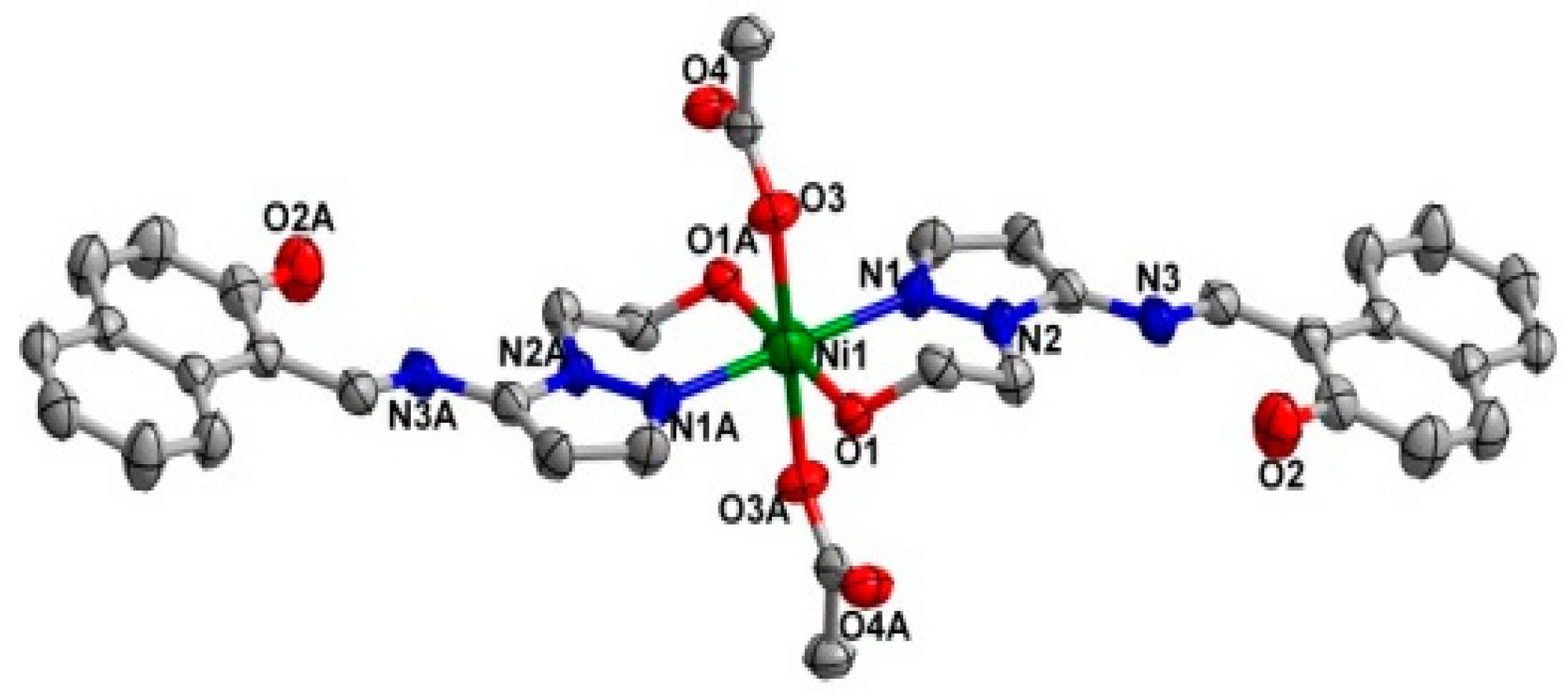
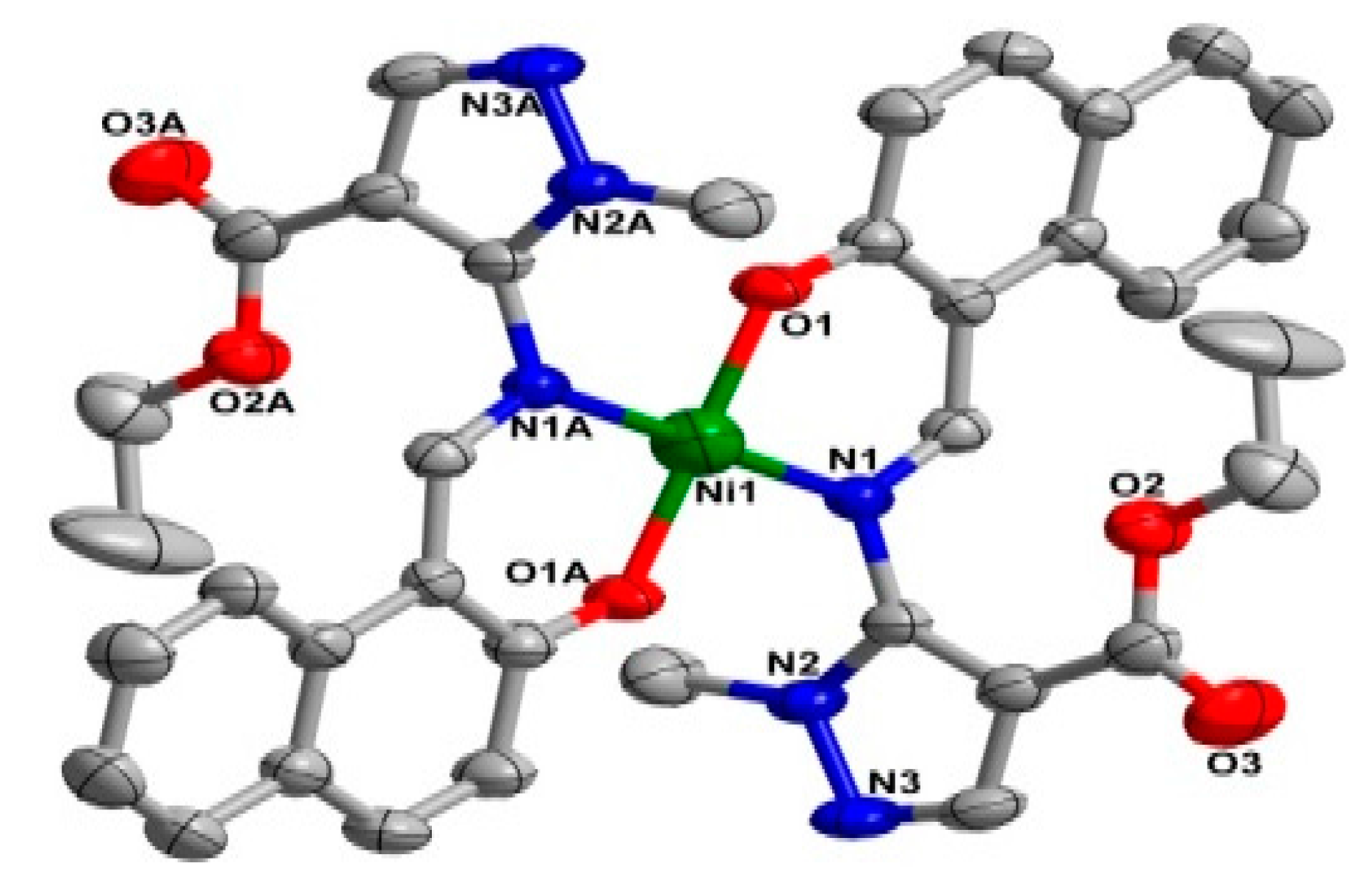
2.2. Crystal Structure
2.3. DNA Binding Studies
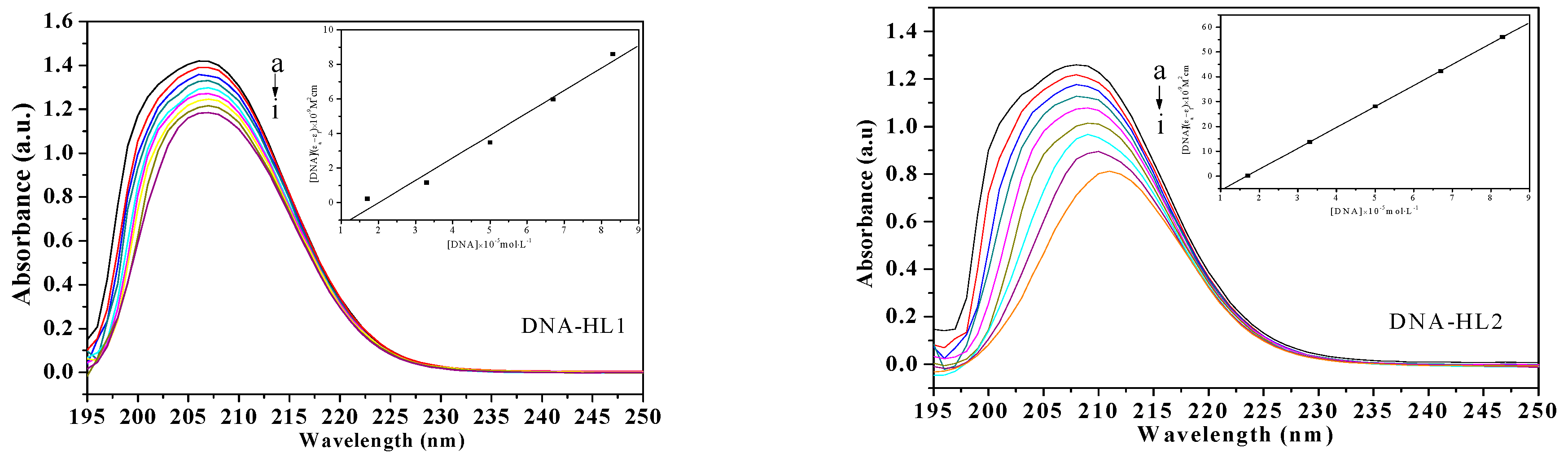
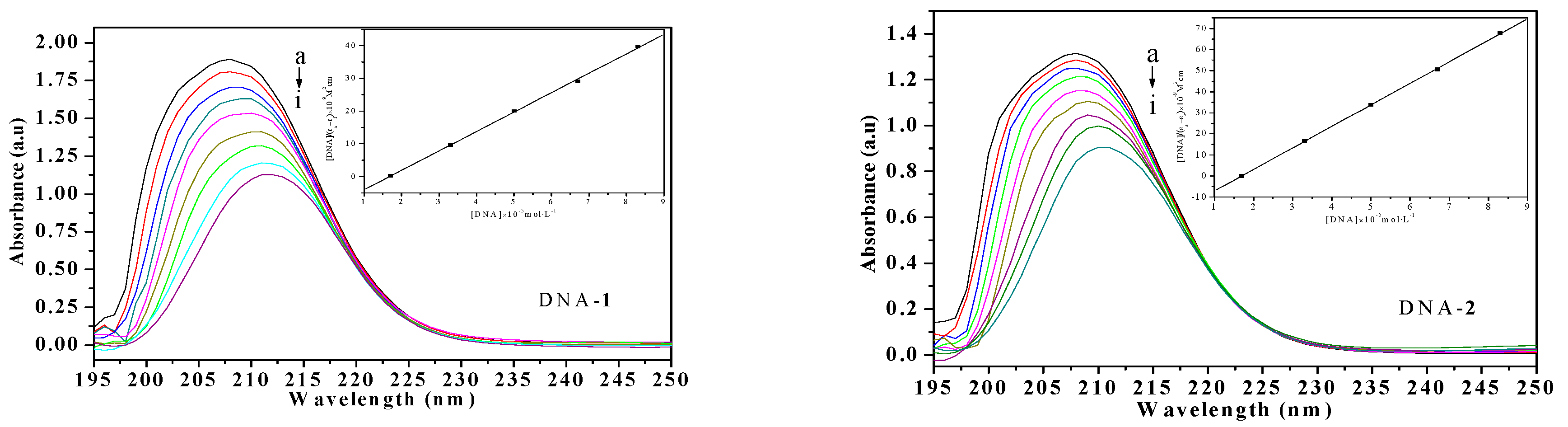
2.3.1. Electronic Absorption Spectral Studies
2.3.2. Circular Dichroic Spectral Studies
2.3.3. Fluorescence Spectroscopic Studies
2.4. Bovine Serum Albumin Binding Studies
2.4.1. UV Absorption Spectra of Bovine Serum Albumin
2.4.2. Circular Dichroism Spectra of BSA
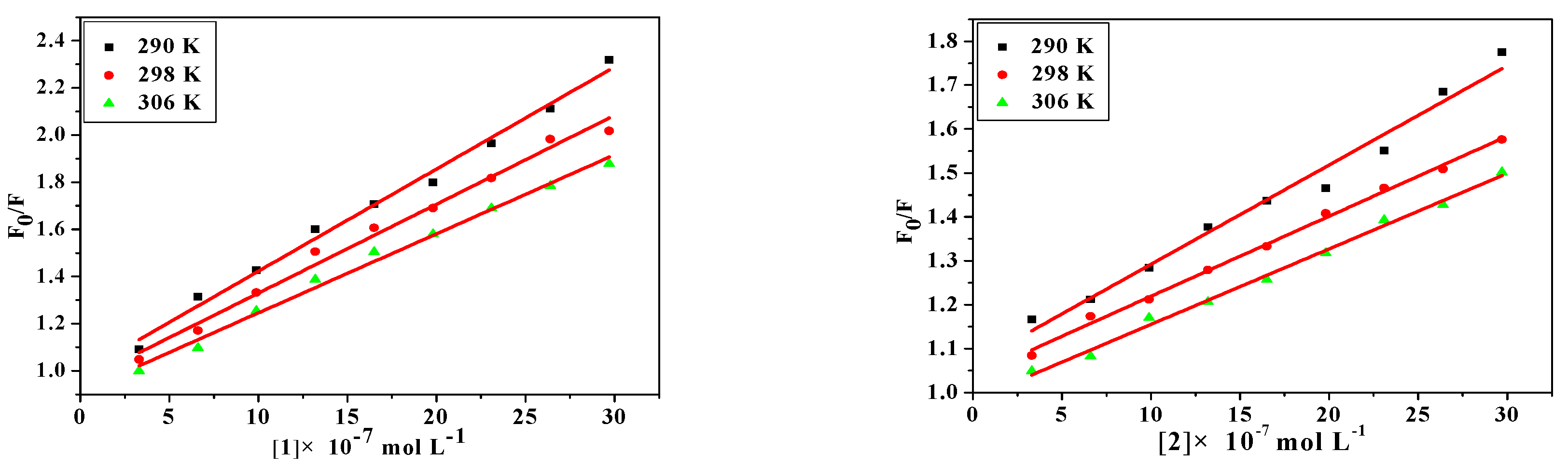
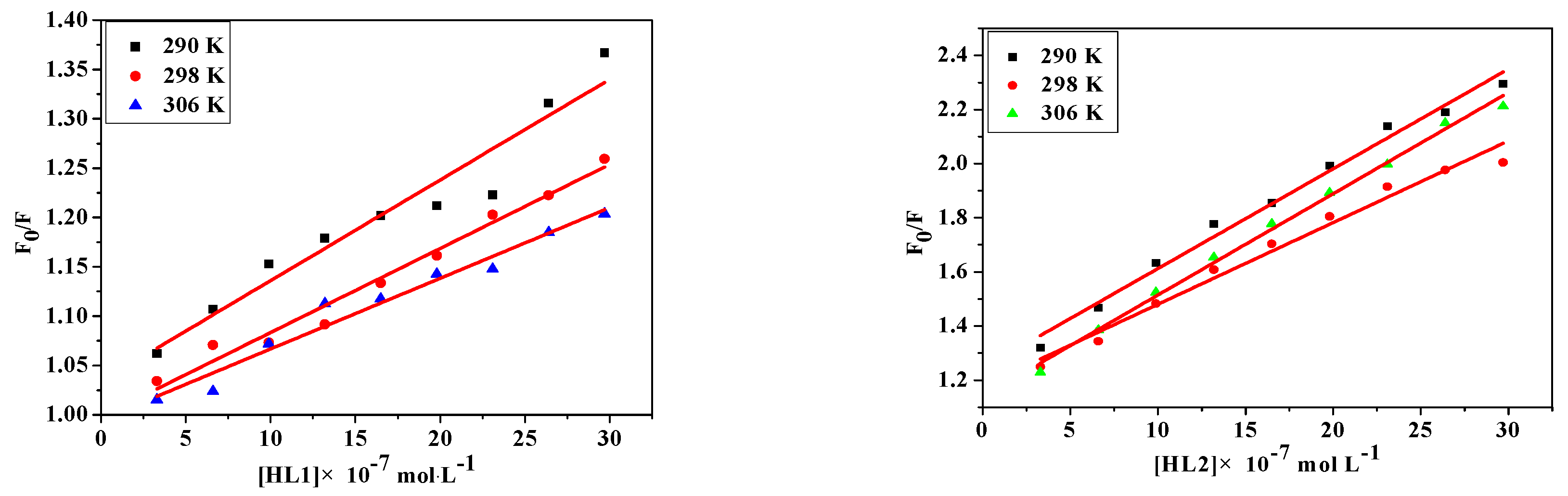
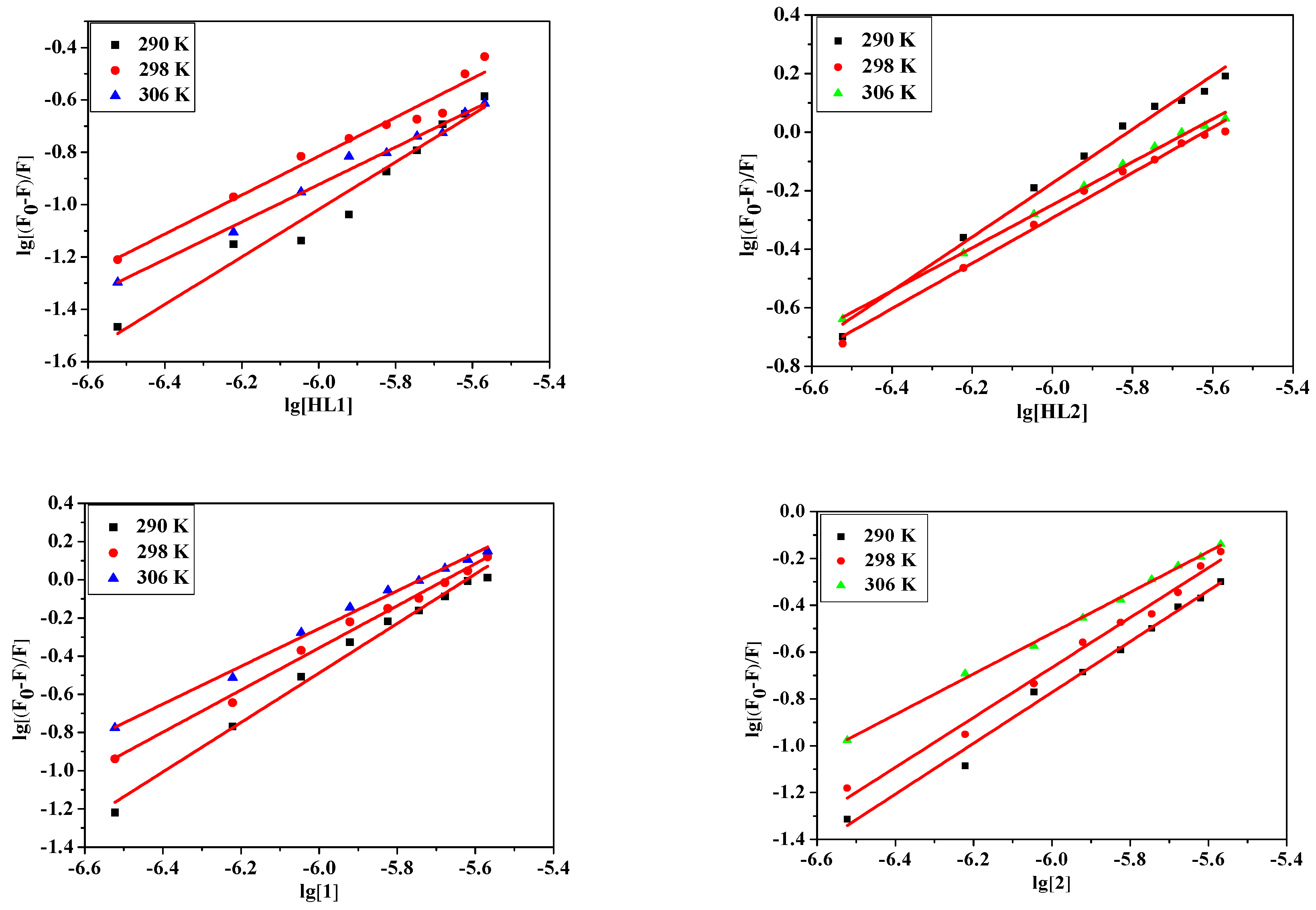
2.4.3. Fluorescence Quenching of Bovine Serum Albumin
2.5. Determination of Thermodynamic Parameters
2.6. In Vitro Studies
2.6.1. Methyl Thiazolyl Tetrazolium (MTT) Assay
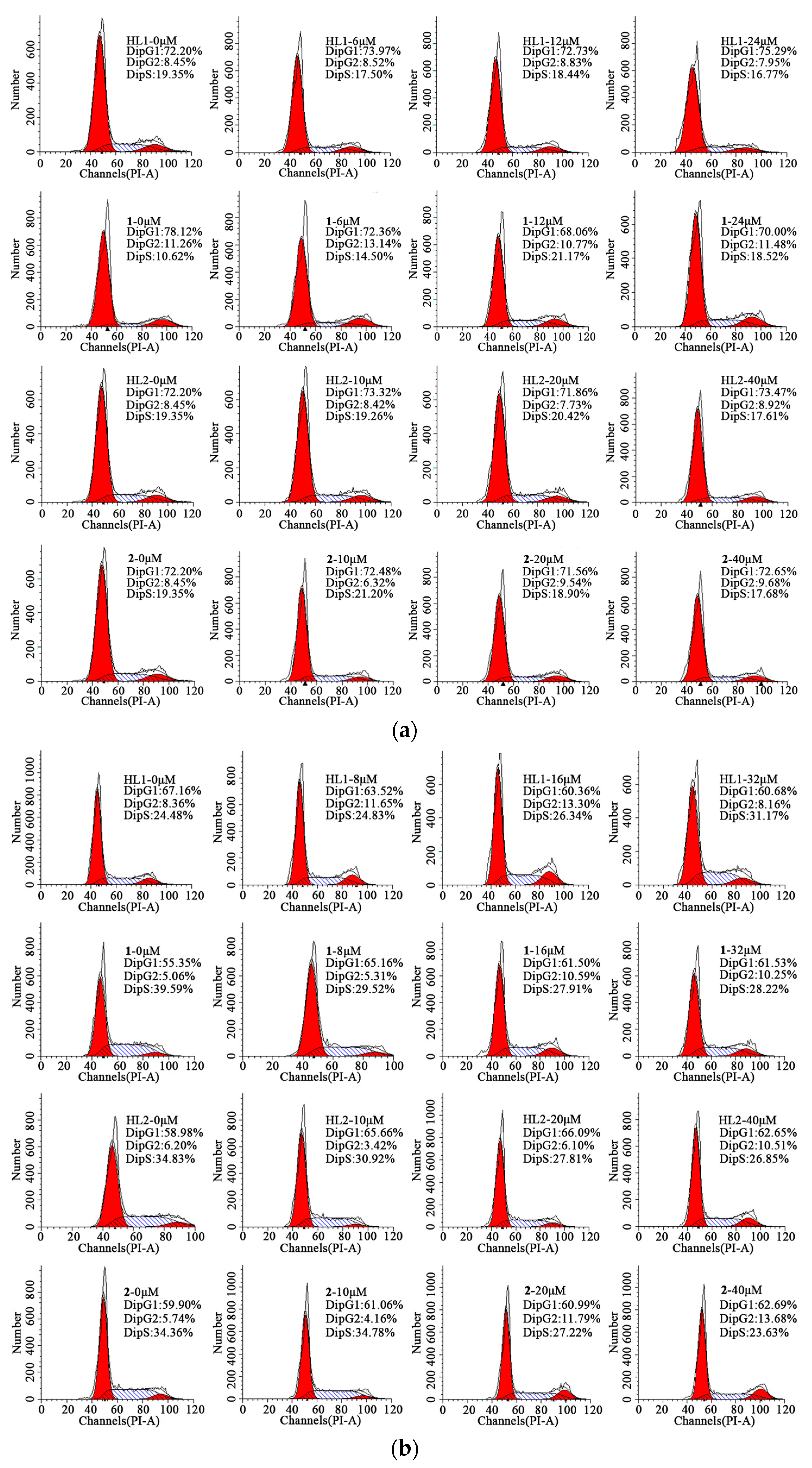
2.6.2. Cell Cycle Arrest
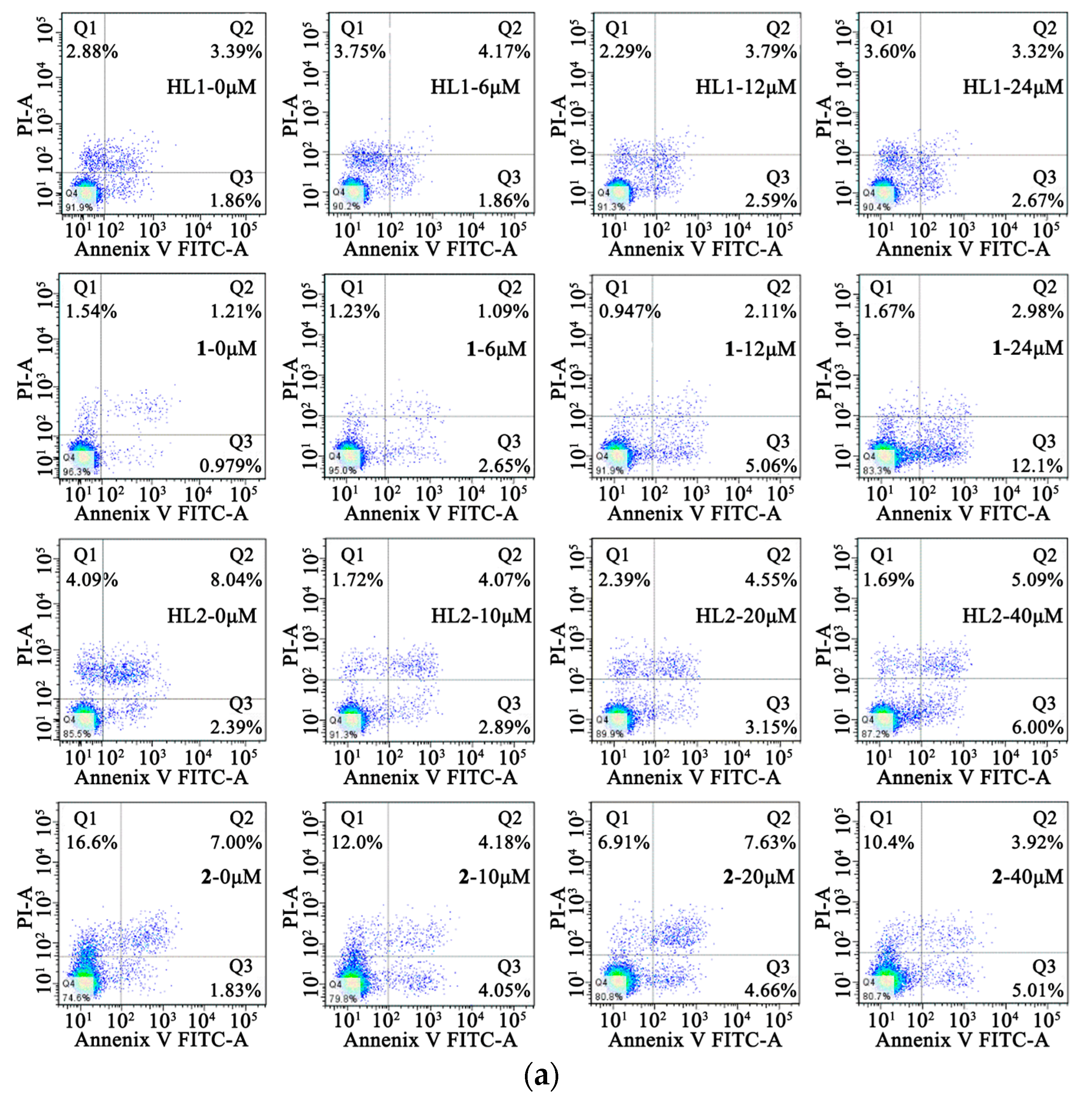
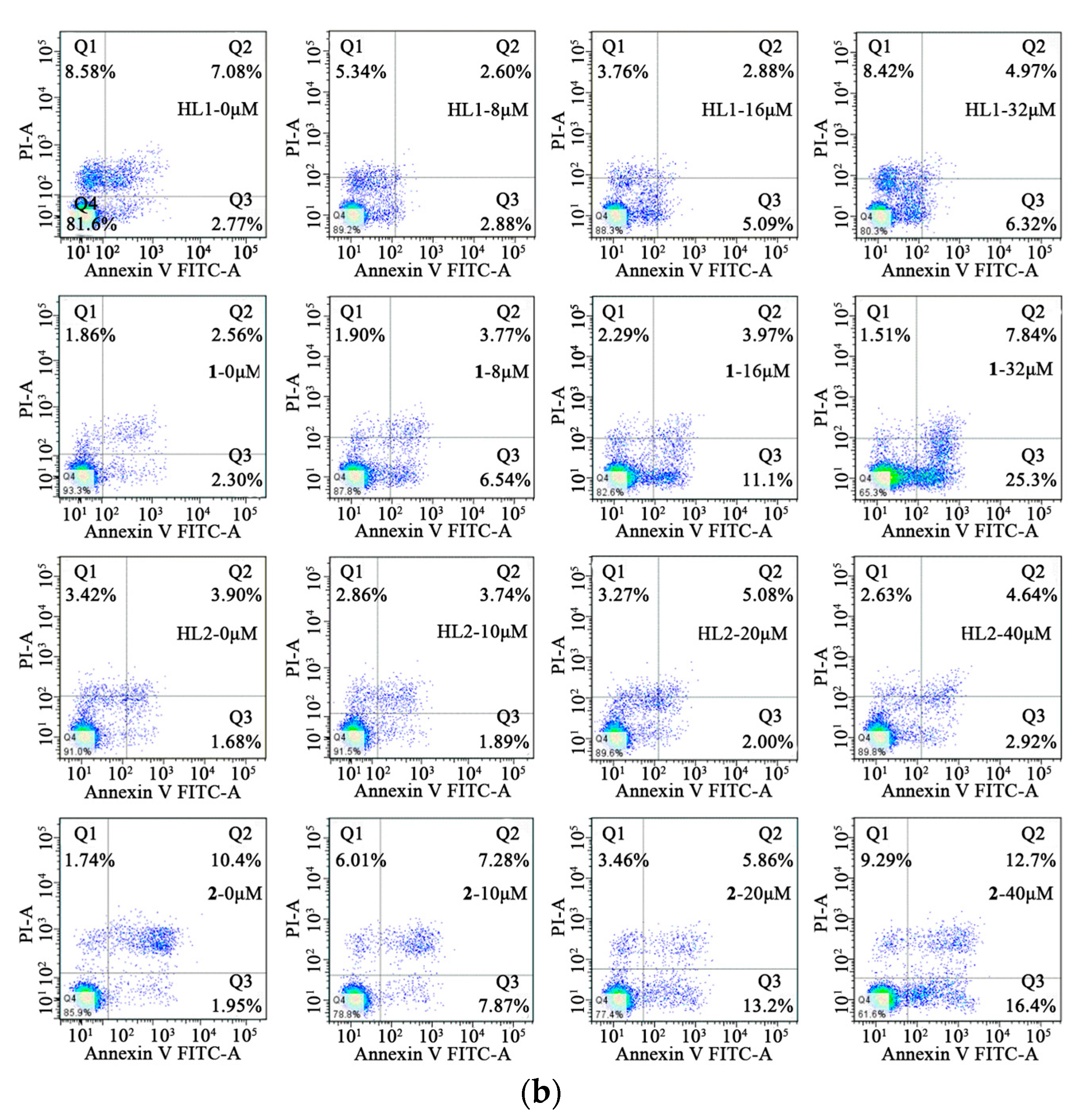
2.6.3. Cell Apoptosis
3. Materials and Methods
3.1. General Information
3.2. Chemistry
3.2.1. Synthesis of the Ligands HL1 and HL2
3.2.2. Synthesis of Ni(HL1)2(OAc)2 (1)
3.2.3. Synthesis of [Ni(L2)2](2)
3.3. Crystal Structure Determination and Refinement
3.4. Stability Studies
3.5. DNA Binding Experiments
3.5.1. UV-Vis Spectra
3.5.2. Circular Dichroism
3.5.3. Competitive Binding Experiments by Fluorescence Spectral Analysis
3.6. Bovine Serum Albumin Binding Studies
3.6.1. UV-Vis Spectra
3.6.2. Circular Dichroism
3.6.3. Fluorescence Spectra
3.7. MTT Assay
3.8. Cell Cycle Arrest
3.9. Apoptosis Study by Flow Cytometry
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Barceló-Oliver, M.; García-Raso, Á.; Terrón, A.; Molins, E.; Prieto, M.J.; Moreno, V.; Martínez, J.; Lladó, V.; López’, I.; Gutiérrez, A. Synthesis and mass spectroscopy kinetics of a novel ternary copper(II) complex with cytotoxic activity against cancer cells. J. Inorg. Biochem. 2007, 101, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, B.; Van Camp, L.; Krigas, T. Inhibition of cell division in Escherichia coli by electrolysis products from a platinum electrode. Nature 1965, 205, 698–699. [Google Scholar] [CrossRef] [PubMed]
- Kelland, L. The resurgence of platinum-based cancer chemotherapy. Nat. Rev. Cancer 2007, 7, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Suntharalingam, K.; Johnstone, T.C.; Bruno, P.M.; Lin, W.; Hemann, M.T.; Lippard, S.J. Bidentate ligands on osmium(VI) nitrido complexes control intracellular targeting and cell death pathways. J. Am. Chem. Soc. 2013, 135, 14060–14063. [Google Scholar] [CrossRef] [PubMed]
- Santini, C.; Pellei, M.; Gandin, V.; Porchia, M.; Tisato, F.; Marzano, C. Advances in copper complexes as anticancer agents. Chem. Rev. 2014, 114, 815–862. [Google Scholar] [CrossRef] [PubMed]
- Kate, A.N.; Kumbhar, A.A.; Khan, A.A.; Joshi, P.V.; Puranik, V.G. Monitoring cellular uptake and cytotoxicity of copper(II) complex using a fluorescent anthracene thiosemicarbazone ligand. Bioconj. Chem. 2013, 25, 102–114. [Google Scholar] [CrossRef] [PubMed]
- Gaynor, D.; Griffith, D.M. The prevalence of metal-based drugs as therapeutic or diagnostic agents: Beyond platinum. Dalton Trans. 2012, 41, 13239–13257. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Jia, L.; Xu, J.; Zhu, T.; Xu, Z.; Chen, R.; Ma, T.; Wang, Y.; Wu, W. Syntheses, crystal structures, anticancer activities of three reduce Schiff base ligand based transition metal complexes. J. Mol. Struct. 2016, 1106, 366–372. [Google Scholar] [CrossRef]
- Inam, A.; Siddiqui, S.M.; Macedo, T.S.; Moreira, D.R.M.; Leite, A.C.L.; Soares, M.B.P.; Azam, A. Design, synthesis and biological evaluation of 3-[4-(7-chloro-quinolin-4-yl)-piperazin-1-yl]-propionic acid hydrazones as antiprotozoal agents. Eur. J. Med. Chem. 2014, 75, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Júnior, W.B.; Alexandre-Moreira, M.S.; Alves, M.A.; Perez-Rebolledo, A.; Parrilha, G.L.; Castellano, E.E.; Piro, O.E.; Barreiro, E.J.; Lima, L.M.; Beraldo, H. Analgesic and anti-inflammatory activities of salicylaldehyde 2-chlorobenzoyl hydrazone (H2LASSBio-466), salicylaldehyde 4-chlorobenzoyl hydrazone (H2LASSBio-1064) and their zinc(II) complexes. Molecules 2011, 16, 6902–6915. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muregi, F.W.; Ishih, A. Next-generation antimalarial drugs: Hybrid molecules as a new strategy in drug design. Drug Dev. Res. 2010, 71, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, G.G.; Zayed, E.M.; Hindy, A.M. Coordination behavior of new bis Schiff base ligand derived from 2-furan carboxaldehyde and propane-1,3-diamine. Spectroscopic, thermal, anticancer and antibacterial activity studies. Spectrochim. Acta Part A 2015, 145, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Pfaltz, A.; Lancaster, J.R., Jr. The Bioinorganic Chemistry of Nickel; VCH: New York, NY, USA, 1988. [Google Scholar]
- Alexiou, M.; Tsivikas, I.; Dendrinou-Samara, C.; Pantazaki, A.A.; Trikalitis, P.; Lalioti, N.; Kyriakidis, D.A.; Kessissoglou, D.P. High nuclearity nickel compounds with three, four or five metal atoms showing antibacterial activity. J. Inorg. Biochem. 2003, 93, 256–264. [Google Scholar] [CrossRef]
- Kurtaran, R.; Yıldırım, L.T.; Azaz, A.D.; Namli, H.; Atakol, O. Synthesis, characterization, crystal structure and biological activity of a novel heterotetranuclear complex: [NiLPb (SCN)2(DMF)(H2O)]2, bis-{[μ-N,N′-bis(salicylidene)-1,3-propanediaminato-aqua-nickel(II)](thiocyanato)(μ-thiocyanato)(μ-N,N′-dimethylformamide) lead(II)}. J. Inorg. Biochem. 2005, 99, 1937–1944. [Google Scholar] [PubMed]
- Luo, W.; Meng, X.; Sun, X.; Xiao, F.; Shen, J.; Zhou, Y.; Cheng, G.; Ji, Z. Synthesis, crystal structure and bioactivity of a novel linear trinuclear nickel(II) complex. Inorg. Chem. Commun. 2007, 10, 1351–1354. [Google Scholar] [CrossRef]
- Afrasiabi, Z.; Sinn, E.; Lin, W.; Ma, Y.; Campana, C.; Padhye, S. Nickel(II) complexes of naphthaquinone thiosemicarbazone and semicarbazone: Synthesis, structure, spectroscopy, and biological activity. J. Inorg. Biochem. 2005, 99, 1526–1531. [Google Scholar] [CrossRef] [PubMed]
- Buschini, A.; Pinelli, S.; Pellacani, C.; Giordani, F.; Ferrari, M.B.; Bisceglie, F.; Giannetto, M.; Pelosi, G.; Tarasconi, P. Synthesis, characterization and deepening in the comprehension of the biological action mechanisms of a new nickel complex with antiproliferative activity. J. Inorg. Biochem. 2009, 103, 666–677. [Google Scholar] [CrossRef] [PubMed]
- Kalaivani, P.; Saranya, S.; Poornima, P.; Prabhakaran, R.; Dallemer, F.; Vijaya Padma, V.; Natarajan, K. Biological evaluation of new nickel(II) metallates: Synthesis, DNA/protein binding and mitochondrial mediated apoptosis in human lung cancer cells (A549) via ROS hypergeneration and depletion of cellular antioxidant pool. Eur. J. Med. Chem. 2014, 82, 584–599. [Google Scholar] [CrossRef] [PubMed]
- Raj, P.; Singh, A.; Singh, A.; Singh, N. Syntheses and Photophysical properties of Schiff-Base Ni(II) Complexes: Application for Sustainable Antibacterial activity and Cytotoxicity. ACS Sustain. Chem. Eng. 2017, 5, 6070–6080. [Google Scholar] [CrossRef]
- Prabhakaran, R.; Jayabalakrishnan, C.; Krishnan, V.; Pasumpon, K.; Sukanya, D.; Bertagnolli, H.; Natarajan, K. Preparation, spectral characterization, electrochemistry, EXAFS, antibacterial and catalytic activity of new ruthenium(III) complexes containing ONS donor ligands with triphenylphosphine/arsine. Appl. Organomet. Chem. 2006, 20, 203–213. [Google Scholar] [CrossRef]
- Krishnamoorthy, P.; Sathyadevi, P.; Butorac, R.R.; Cowley, A.H.; Bhuvanesh, N.S.; Dharmaraj, N. Copper(I) and nickel(II) complexes with 1:1 vs. 1:2 coordination of ferrocenyl hydrazone ligands: Do the geometry and composition of complexes affect DNA binding/cleavage, protein binding, antioxidant and cytotoxic activities? Dalton Trans. 2012, 41, 4423–4436. [Google Scholar] [CrossRef] [PubMed]
- Tysoe, S.A.; Morgan, R.J.; Baker, A.D.; Strekas, T.C. Spectroscopic investigation of differential binding modes of Δ- and Λ-Ru (bpy)2(ppz)2+ with calf thymus DNA. J. Phys. Chem. 1993, 97, 1707–1711. [Google Scholar] [CrossRef]
- Rajarajeswari, C.; Ganeshpandian, M.; Palaniandavar, M.; Riyasdeen, A.; Akbarsha, M.A. Mixed ligand copper(II) complexes of 1,10-phenanthroline with tridentate phenolate/pyridyl/(benz) imidazolyl Schiff base ligands: Covalent vs. non-covalent DNA binding, DNA cleavage and cytotoxicity. J. Inorg. Biochem. 2014, 140, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Niu, M.; Hong, M.; Cheng, S.; Dou, J. Effect of structure and composition of nickel(II) complexes with salicylidene Schiff base ligands on their DNA/protein interaction and cytotoxicity. J. Inorg. Biochem. 2014, 137, 101–108. [Google Scholar]
- Schäfer, S.; Ott, I.; Gust, R.; Sheldrick, W.S. Influence of the Polypyridyl (pp) Ligand Size on the DNA Binding Properties, Cytotoxicity and Cellular Uptake of Organoruthenium(II) Complexes of the Type [(η6-C6Me6)Ru(L)(pp)]n+ [L = Cl, n = 1; L = (NH2)2CS, n = 2]. Eur. J. Inorg. Chem. 2007, 3034–3046. [Google Scholar] [CrossRef]
- Richards, A.D.; Rodger, A. Synthetic metallomolecules as agents for the control of DNA structure. Chem. Soc. Rev. 2007, 36, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Wang, X.; Zhu, Y.; Li, Y.; Guo, Z. Selective guanosine binding and cytotoxicity of a benzimidazole derived dinickel complex. J. Inorg. Biochem. 2007, 101, 1894–1902. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Guo, J.; Zhao, N.; Wang, J. Study of interaction between kaempferol—Eu3+ complex and DNA with the use of the Neutral Red dye as a fluorescence probe. Sens. Actuators B 2010, 144, 239–246. [Google Scholar] [CrossRef]
- Qiu, B.; Guo, L.; Chen, Z.; Chi, Y.; Zhang, L.; Chen, G. Synthesis of N-4-butylamine acridone and its use as fluorescent probe for ctDNA. Biosens. Bioelectron. 2009, 24, 1281–1285. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, J.; Vicente, C.; de Haro, C.; Bautista, D. Novel bis-C,N-cyclometalated iridium(III) thiosemicarbazide antitumor complexes: Interactions with human serum albumin and DNA, and Inhibition of cathepsin B. Inorg. Chem. 2013, 52, 974–982. [Google Scholar] [CrossRef] [PubMed]
- Haghighi, F.H.; Hadadzadeh, H.; Darabi, F.; Jannesari, Z.; Ebrahimi, M.; Khayamian, T.; Salimi, M.; Rudbari, H.A. Polypyridyl Ni(II) complex, [Ni(tppz)2]2+: Structure, DNA- and BSA binding and molecular modeling. Polyhedron 2013, 65, 16–30. [Google Scholar] [CrossRef]
- Zhao, L.; Liu, R.; Zhao, X.; Yang, B.; Gao, C.; Hao, X.; Wu, Y. New strategy for the evaluation of CdTe quantum dot toxicity targeted to bovine serum albumin. Sci. Total Environ. 2009, 407, 5019–5023. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Liu, P.; Zhou, X.; Zhang, X.; Fang, T.; Liu, P.; Min, X.; Li, X. Thermodynamic and conformational investigation of the influence of CdTe QDs size on the toxic interaction with BSA. J. Photochem. Photobiol. A 2012, 230, 23–30. [Google Scholar] [CrossRef]
- Xiao, Q.; Huang, S.; Qi, Z.; Zhou, B.; He, Z.; Liu, Y. Conformation, thermodynamics and stoichiometry of HSA adsorbed to colloidal CdSe/ZnS quantum dots. Biochim. Biophys. Acta 2008, 1784, 1020–1027. [Google Scholar] [CrossRef] [PubMed]
- Pan, B.F.; Gao, F.; Ao, L.M. Investigation of interactions between dendrimer-coated magnetite nanoparticles and bovine serum albumin. J. Magn. Magn. Mater. 2005, 293, 252–258. [Google Scholar] [CrossRef]
- Sułkowska, A. Interaction of drugs with bovine and human serum albumin. J. Mol. Struct. 2002, 614, 227–232. [Google Scholar] [CrossRef]
- Jana, S.K.; Seth, S.K.; Puschmann, H.; Hossain, M.; Dalai, S. Synthesis and X-ray structure of a new zinc(II) coordination polymer: Interaction with DNA and double stranded RNA and elucidation of the molecular aspects of the binding to bovine serum albumin. RSC Adv. 2014, 4, 57855–57868. [Google Scholar] [CrossRef]
- Rajendiran, V.; Karthik, R.; Palaniandavar, M.; Stoeckli-Evans, H.; Periasamy, V.S.; Akbarsha, M.A.; Srinag, B.S.; Krishnamurthy, H. Mixed-ligand copper(II)-phenolate complexes: Effect of coligand on enhanced DNA and protein binding, DNA cleavage, and anticancer activity. Inorg. Chem. 2007, 46, 8208–8221. [Google Scholar] [CrossRef] [PubMed]
- Gelamo, E.L.; Tabak, M. Spectroscopic studies on the interaction of bovine (BSA) and human (HSA) serum albumins with ionic surfactants. Spectrochim. Acta Part A 2000, 56, 2255–2271. [Google Scholar] [CrossRef]
- Ware, W.R. Oxygen quenching of fluorescence in solution: An experimental study of the diffusion process. J. Phys. Chem. 1962, 66, 455–458. [Google Scholar] [CrossRef]
- Leckband, D. Measuring the forces that control protein interactions. Annu. Rev. Biophys. Biomol. Struct. 2000, 29, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Tong, J.; Tian, F.; Li, Q.; Li, L.; Xiang, C.; Liu, Y.; DAI, J.; Jiang, F. Probing the adverse temperature dependence in the static fluorescence quenching of BSA induced by a novel anticancer hydrazine. Photochem. Photobiol. Sci. 2012, 11, 1868–1879. [Google Scholar] [CrossRef] [PubMed]
- Benkö, Z.; Burck, S.; Gudat, D.; Nieger, M.; Nyulászi, L.; Shore, N. Pyrido-annellated diazaphospholenes and phospholenium ions. Dalton Trans. 2008, 4937–4945. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, A.; Vaidya, S.; Venkatasai, V.S.; Jayapal, P.; Srivastava, A.K.; Shanmugam, M.; Shanmugam, M. Synthesis and characterization of 3d and 4f metal complexes of Schiff base ligands. Polyhedron 2013, 66, 87–96. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Phase annealing in SHELX-90: Direct methods for larger structures. Acta Crystallogr. Sect. A Found. Crystallogr. 1990, 46, 467–473. [Google Scholar] [CrossRef]
- Sheldrick, G.M. The SHELX Homepage. Available online: http://shelx.uni-ac.gwdg.de/SHELX/ (accessed on 15 June 2015).
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. Sect. A Found. Crystallogr. 2008, 64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Brandenburg, K.; Putz, H. DIAMOND, Program for Crystal and Molecular Structure Visualization. Available online: http://www.crystalimpact.com/diamond/ (accessed on 6 May 2017).
- Chand, D.K.; Schneider, H.J.; Bencini, A.; Bianchi, A.; Giorgi, C.; Ciattini, S.; Valtancoli, B. Affinity and nuclease activity of macrocyclic polyamines and their Cu(II) complexes. Chem. Eur. J. 2015, 6, 4001–4008. [Google Scholar] [CrossRef]
- Reichmann, M.E.; Rice, S.A.; Thomas, C.A.; Doty, P. A further examination of the molecular weight and size of desoxypentose nucleic acid. J. Am. Chem. Soc. 1954, 76, 3047–3053. [Google Scholar] [CrossRef]
- Saleh, M.; Soliman, H.; Elmatbouli, M. Loop-mediated isothermal amplification as an emerging technology for detection of Yersinia ruckeri the causative agent of enteric red mouth disease in fish. BMC Vet. Res. 2008, 4, 1184–1191. [Google Scholar] [CrossRef] [PubMed]
- Joseph, R.L.; Lakowicz, R. Principles of Fluorescence Spectroscopy; Kluwer Academic/Plenum Publishers: New York, NY, USA, 1999. [Google Scholar]
- Hu, Y.J.; Liu, Y.; Wang, J.B.; Xiao, X.H.; Qu, S.S. Study of the interaction between monoammonium glycyrrhizinate and bovine serum albumin. J. Pharm. Biomed. Anal. 2004, 36, 915–919. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.C.; Yang, J.S.; Lu, H.F.; Ip, S.W.; Lo, C.; Wu, C.C.; Lin, J.P.; Tang, N.Y.; Chung, J.G.; Chou, M.J.; Teng, Y.H.; Chen, D.R. Quercetin-mediated cell cycle arrest and apoptosis involving activation of a caspase cascade through the mitochondrial pathway in human breast cancer MCF-7 cells. Arch. Pharm. Res. 2010, 33, 1181–1191. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |

| Complex | 1 | 2 |
|---|---|---|
| Empirical formula | C36H36NiN6O8 | C36H32NiN6O6 |
| Formula weight (M) | 739.42 | 703.39 |
| Temperature (K) | 293 (2) | 293 (2) |
| Crystal system | Monoclinic | Monoclinic |
| Space group | P21/n | P21/c |
| a (Å) | 14.0548 (5) | 12.6852 (8) |
| b (Å) | 7.9911 (2) | 11.0140 (6) |
| c (Å) | 15.2876 (6) | 11.8301 (8) |
| α (°) | 90.00 | 90.00 |
| β (°) | 105.902 (4) | 100.008 (6) |
| γ (°) | 90.00 | 90.00 |
| Volume (Å3) | 1651.30 (10) | 1627.69 (17) |
| Z | 2 | 2 |
| Calculated density (Mg/m3) | 1.487 | 1.435 |
| Absorption coefficient (mm−1) | 0.65 | 0.65 |
| F(000) | 772.0 | 732.0 |
| θ range for data collection | 3.8 to 24 | 3.7 to 24.7 |
| Limiting indices | −17 ≤ h ≤ 17 | −15 ≤ h ≤ 15 |
| −9 ≤ k ≤ 9 | −13 ≤ k ≤ 13 | |
| −19 ≤ l ≤ 18 | −14 ≤ l ≤ 14 | |
| Reflections collected | 8684 | 16088 |
| Independent reflections | 3374 [Rint = 0.0436] | 3325 [Rint = 0.0907] |
| Data/restraints/parameters | 3374/2/237 | 3325/0/225 |
| Goodness-of-fit on F2 | 1.031 | 1.062 |
| Final R indices [I > 2σ(I)] | R1 = 0.0478 | R1 = 0.0528 |
| ωR2 = 0.0957 | ωR2 = 0.1206 | |
| R indices (all data) | R1 = 0.0804 | R1 = 0.0832 |
| ωR2 = 0.1119 | ωR2 = 0.1429 |
| Ni1–O1 | 2.052 (2) | O3–Ni1–N1A | 90.12 (9) |
| Ni1–O3 | 2.0632 (2) | O3A–Ni1–N1A | 89.88 (9) |
| Ni1–N1 | 2.094 (2) | O1A–Ni1–N1 | 89.83 (9) |
| O1A–Ni1–O3A | 89.43 (8) | O1A–Ni1–N1A | 90.17 (9) |
| O1–Ni1–O3A | 90.57 (8) |
| Ni1–O1 | 1.822 (2) | O1–Ni1–N1 | 92.13 (10) |
| Ni1–N1 | 1.891 (2) | O1–Ni1–N1A | 87.87 (10) |
| System (%) | [ML]:[BSA] 0:1 | [ML]:[BSA] 0.5:1 | [ML]:[BSA] 1:1 |
|---|---|---|---|
| BSA-HL1 | 51.16 | 48.57 | 46.88 |
| BSA-1 | 51.16 | 41.67 | 38.14 |
| BSA-HL2 | 51.16 | 44.38 | 42.23 |
| BSA-2 | 51.16 | 44.49 | 40.15 |
| System | T (K) | KSV (×105 L·mol−1) | kq (×1013 L·mol−1·S−1) | R a | SD b |
|---|---|---|---|---|---|
| BSA-HL1 | 290 | 3.36 | 3.36 | 0.9319 | 0.00320 |
| 298 | 2.81 | 2.81 | 0.9771 | 0.00152 | |
| 306 | 2.36 | 2.36 | 0.9621 | 0.00165 | |
| BSA-1 | 290 | 14.31 | 14.31 | 0.9908 | 0.00488 |
| 298 | 12.44 | 12.44 | 0.9999 | 0.00078 | |
| 306 | 11.06 | 11.06 | 0.9915 | 0.00362 | |
| BSA-HL2 | 290 | 11.93 | 11.93 | 0.9275 | 0.01174 |
| 298 | 9.96 | 9.96 | 0.9791 | 0.00514 | |
| 306 | 4.12 | 4.12 | 0.9998 | 0.00079 | |
| BSA-2 | 290 | 7.47 | 7.47 | 0.9782 | 0.00394 |
| 298 | 6.02 | 6.02 | 0.9962 | 0.00132 | |
| 306 | 5.68 | 5.68 | 0.9997 | 0.00080 |
| System | T (K) | K (L·mol−1) | n | ΔG0 (kJ·mol−1) | ΔH0 (kJ·mol−1) | ΔS0 (J·mol−1·K−1) |
|---|---|---|---|---|---|---|
| BSA-HL1 | 290 | 2.65 × 104 | 0.91 | −24.04 | −112.30 | −304.34 |
| 298 | 4.43 × 103 | 0.74 | −21.61 | |||
| 306 | 2.33 × 103 | 0.71 | −19.17 | |||
| BSA-1 | 290 | 1.93 × 107 | 1.30 | −40.09 | −187.51 | −508.35 |
| 298 | 1.83 × 106 | 1.10 | −36.02 | |||
| 306 | 3.33 × 105 | 0.99 | −31.95 | |||
| BSA-HL2 | 290 | 1.77 × 105 | 0.92 | −28.42 | −116.21 | −302.71 |
| 298 | 2.22 × 104 | 0.77 | −26.00 | |||
| 306 | 1.43 × 104 | 0.73 | −23.58 | |||
| BSA-2 | 290 | 5.60 × 105 | 1.09 | −32.87 | −122.24 | −308.16 |
| 298 | 5.35 × 105 | 1.07 | −30.41 | |||
| 306 | 3.97 × 104 | 0.87 | −27.94 |
| HepG 2 | MGC80-3 | T-24 | BEL-7404 | NCI-H460 | SK-OV-3 | A549 | HL-7702 | |
|---|---|---|---|---|---|---|---|---|
| HL1 | 62.82 ± 1.46 | 41.57 ± 0.19 | 53.83 ± 0.33 | 36.00 ± 5.91 | 28.69 ± 0.18 | 50.24 ± 2.65 | 19.74 ± 0.05 | 61.25 ± 0.47 |
| HL2 | 62.23 ± 0.93 | 39.11 ± 0.80 | 53.43 ± 0.09 | 27.04 ± 0.87 | 34.98 ± 0.05 | 55.62 ± 0.07 | 27.63 ± 1.12 | 58.75 ± 2.51 |
| 1 | 94.01 ± 1.22 | 90.07 ± 0.03 | 92.82 ± 0.60 | 74.53 ± 0.17 | 85.13 ± 0.42 | 89.34 ± 1.75 | 76.37 ± 0.48 | 92.48 ± 1.06 |
| 2 | 73.45 ± 0.28 | 59.30 ± 0.31 | 54.63 ± 0.49 | 45.31 ± 0.55 | 31.45 ± 0.41 | 53.76 ± 3.20 | 28.65 ± 5.15 | 65.44 ± 0.64 |
| Cisplatin | 90.59 ± 0.41 | 88.28 ± 0.33 | 80.40 ± 0.63 | 73.99 ± 0.20 | 72.45 ± 0.13 | 68.19 ± 4.43 | 52.80 ± 0.55 | 91.18 ± 0.17 |
| HepG2 | MGC80-3 | T-24 | BEL-7404 | NCI-H460 | SK-OV-3 | A549 | HL-7702 | |
|---|---|---|---|---|---|---|---|---|
| HL1 | 31.17 ± 2.06 | 93.85 ± 1.96 | 36.77 ± 0.09 | >100 | >100 | 45.36 ± 1.17 | >100 | 29.15 ± 0.14 |
| HL2 | 30.59 ± 1.01 | 88.25 ± 3.12 | 38.85 ± 0.14 | >100 | >100 | 39.80 ± 0.74 | >100 | 26.77 ± 1.58 |
| 1 | 13.33 ± 0.11 | 15.73 ± 0.02 | 18.18 ± 0.05 | 24.07 ± 0.12 | 24.84 ± 0.08 | 16.16 ± 0.39 | 13.26 ± 0.20 | 14.95 ± 0.05 |
| 2 | 23.18 ± 0.01 | 37.84 ± 1.53 | 28.84 ± 0.42 | 46.59 ± 0.79 | >100 | 36.80 ± 4.62 | >100 | 23.75 ± 0.91 |
| Cisplatin | 3.93 ± 0.01 | 14.61 ± 0.06 | 17.22 ± 0.13 | 20.41 ± 0.05 | 14.84 ± 0.11 | 10.09 ± 1.11 | 35.29 ± 0.42 | 7.86 ± 0.10 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, H.; Zhang, W.; Yu, Q.; Huang, F.-P.; Bian, H.-D.; Liang, H. Ni(II) Complexes with Schiff Base Ligands: Preparation, Characterization, DNA/Protein Interaction and Cytotoxicity Studies. Molecules 2017, 22, 1772. https://doi.org/10.3390/molecules22101772
Yu H, Zhang W, Yu Q, Huang F-P, Bian H-D, Liang H. Ni(II) Complexes with Schiff Base Ligands: Preparation, Characterization, DNA/Protein Interaction and Cytotoxicity Studies. Molecules. 2017; 22(10):1772. https://doi.org/10.3390/molecules22101772
Chicago/Turabian StyleYu, Hui, Wei Zhang, Qing Yu, Fu-Ping Huang, He-Dong Bian, and Hong Liang. 2017. "Ni(II) Complexes with Schiff Base Ligands: Preparation, Characterization, DNA/Protein Interaction and Cytotoxicity Studies" Molecules 22, no. 10: 1772. https://doi.org/10.3390/molecules22101772




