Fluorescence Resonance Energy Transfer Systems in Supramolecular Macrocyclic Chemistry
Abstract
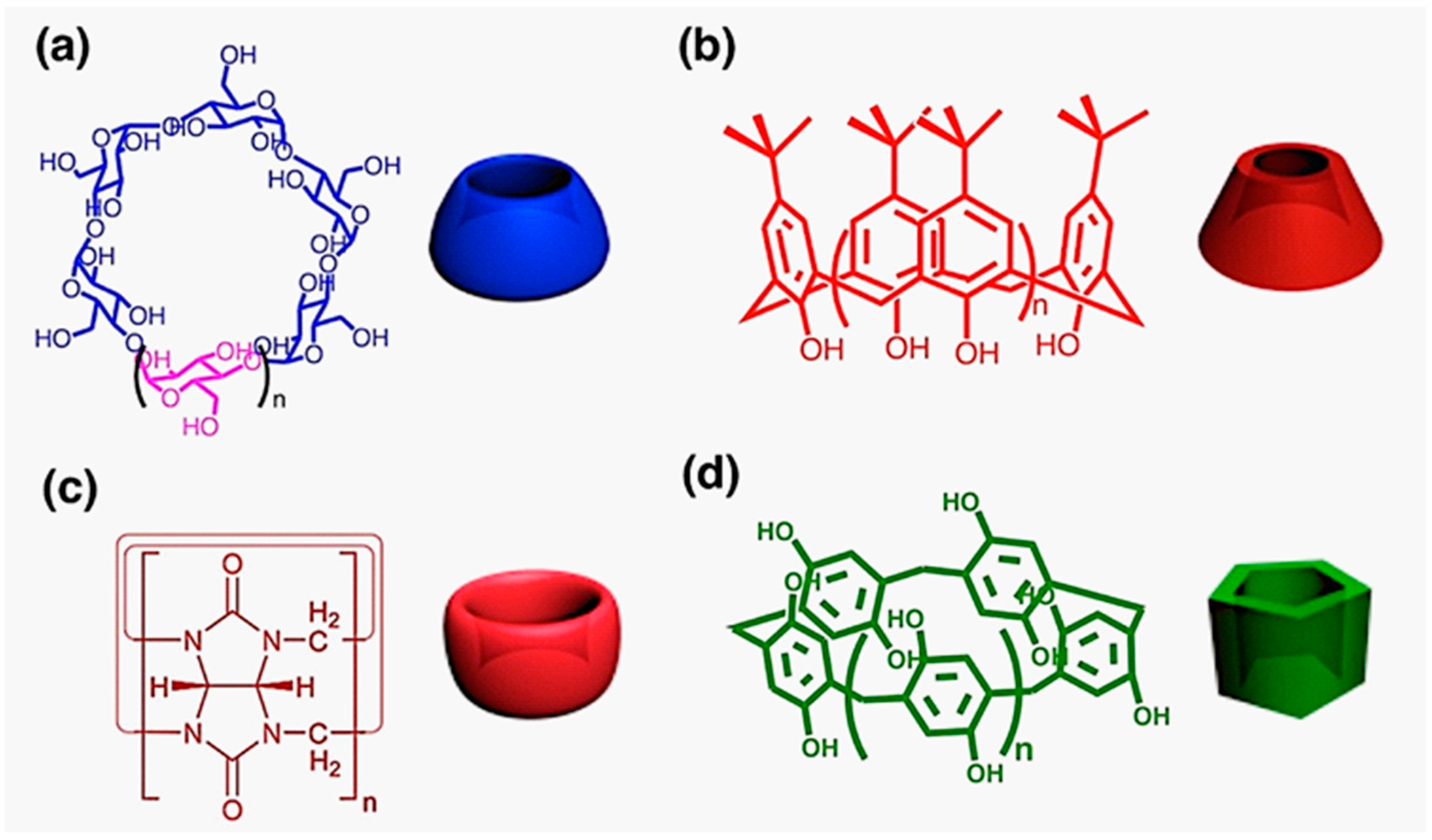
:1. Introduction
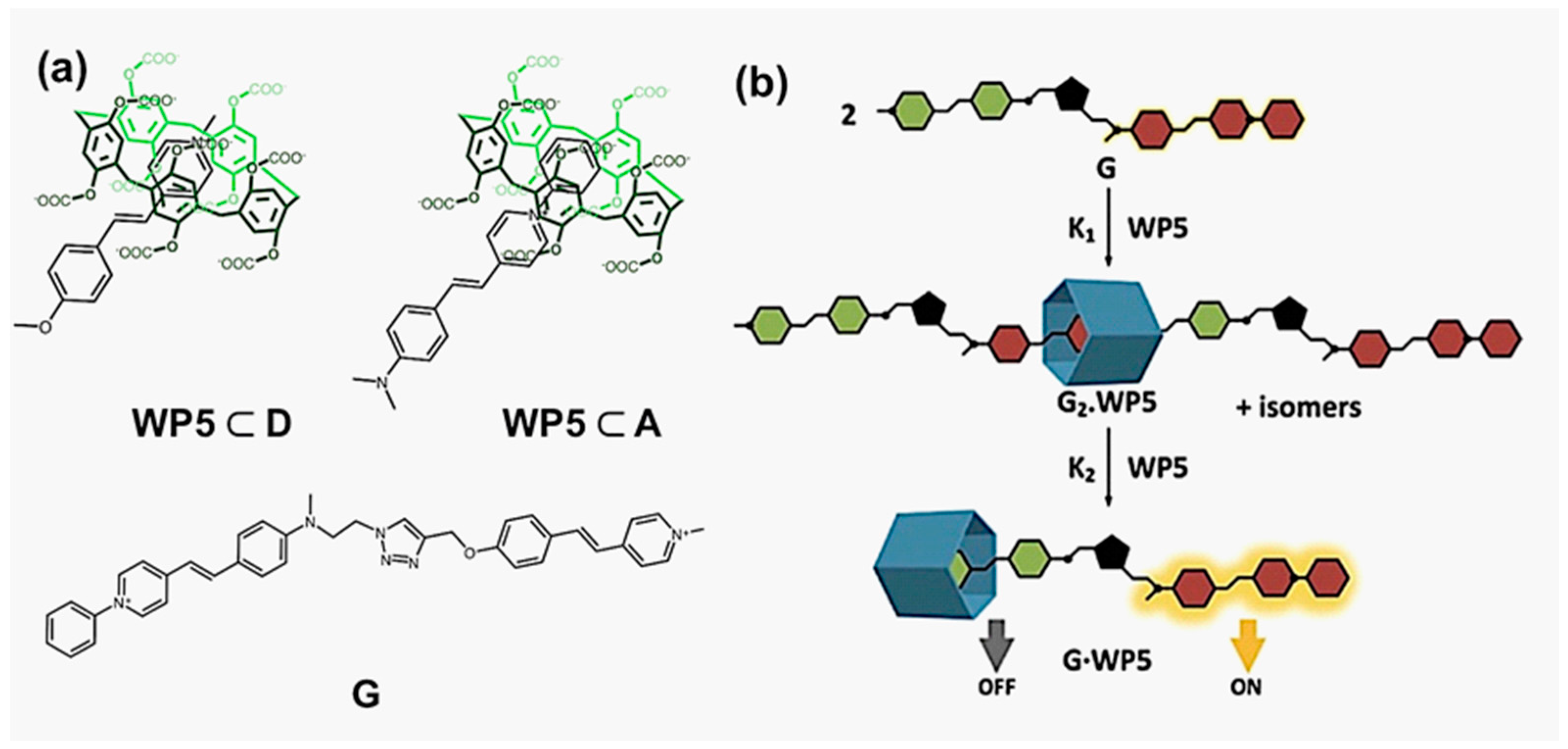
2. Basic Studies for FRET Systems Based on Host-Guest Chemistry
3. Applications of FRET Systems Based on Host-Guest Chemistry
3.1. Protein Assembly
3.2. Enzyme Assays
3.3. Diagnosis
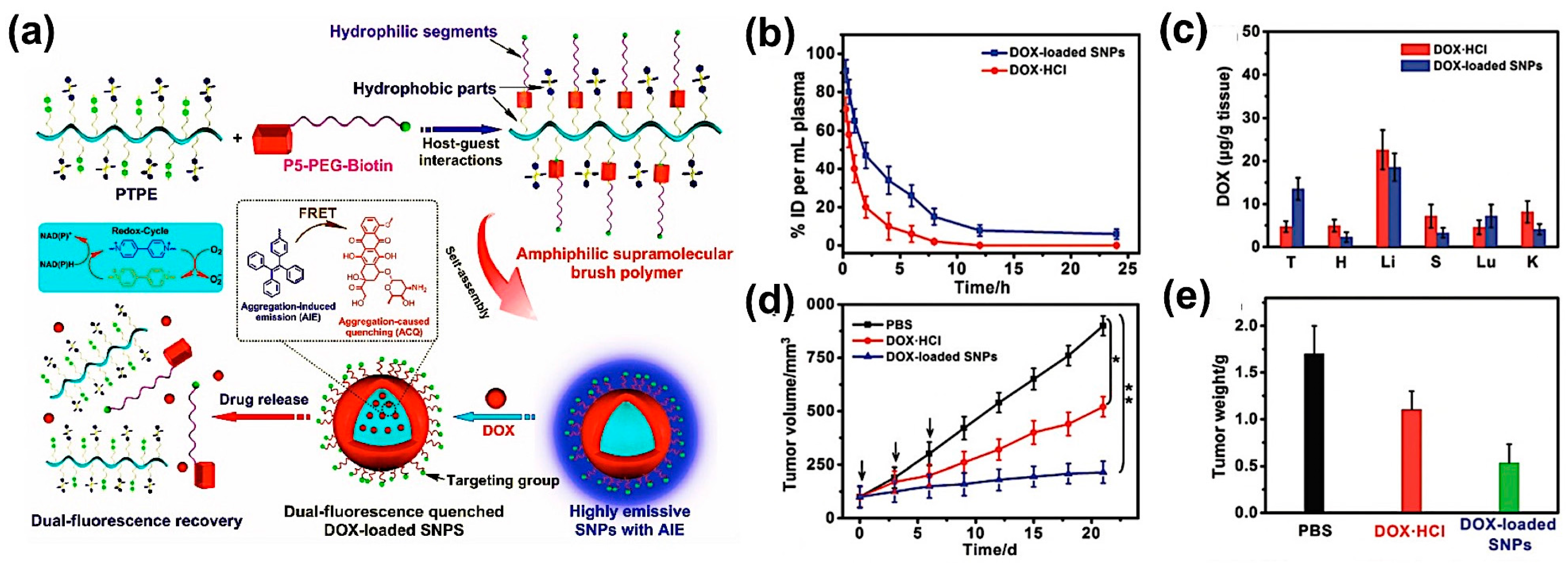
3.4. Drug Delivery Monitoring
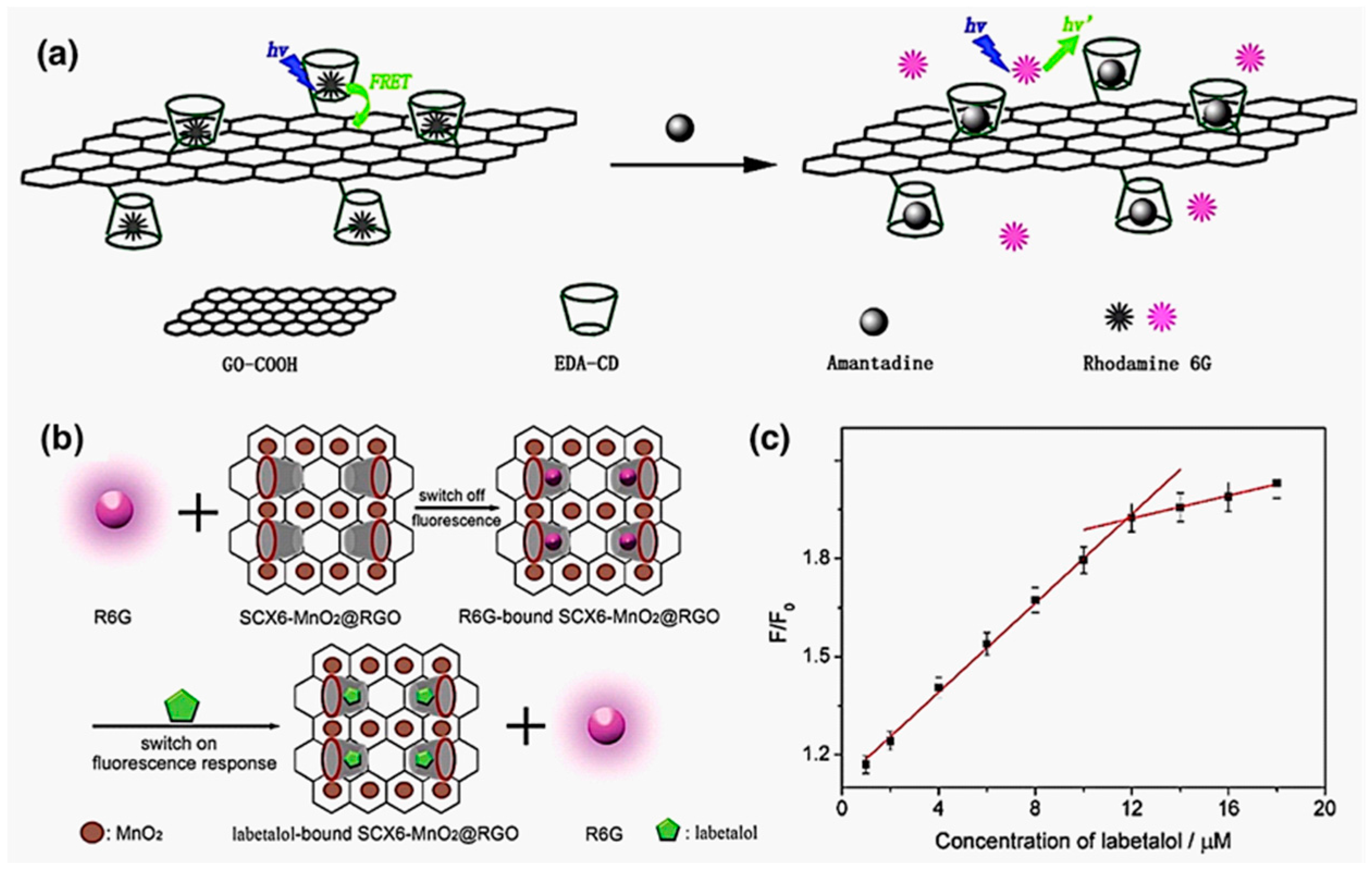
3.5. Sensing
3.6. Photosynthesis Mimicking
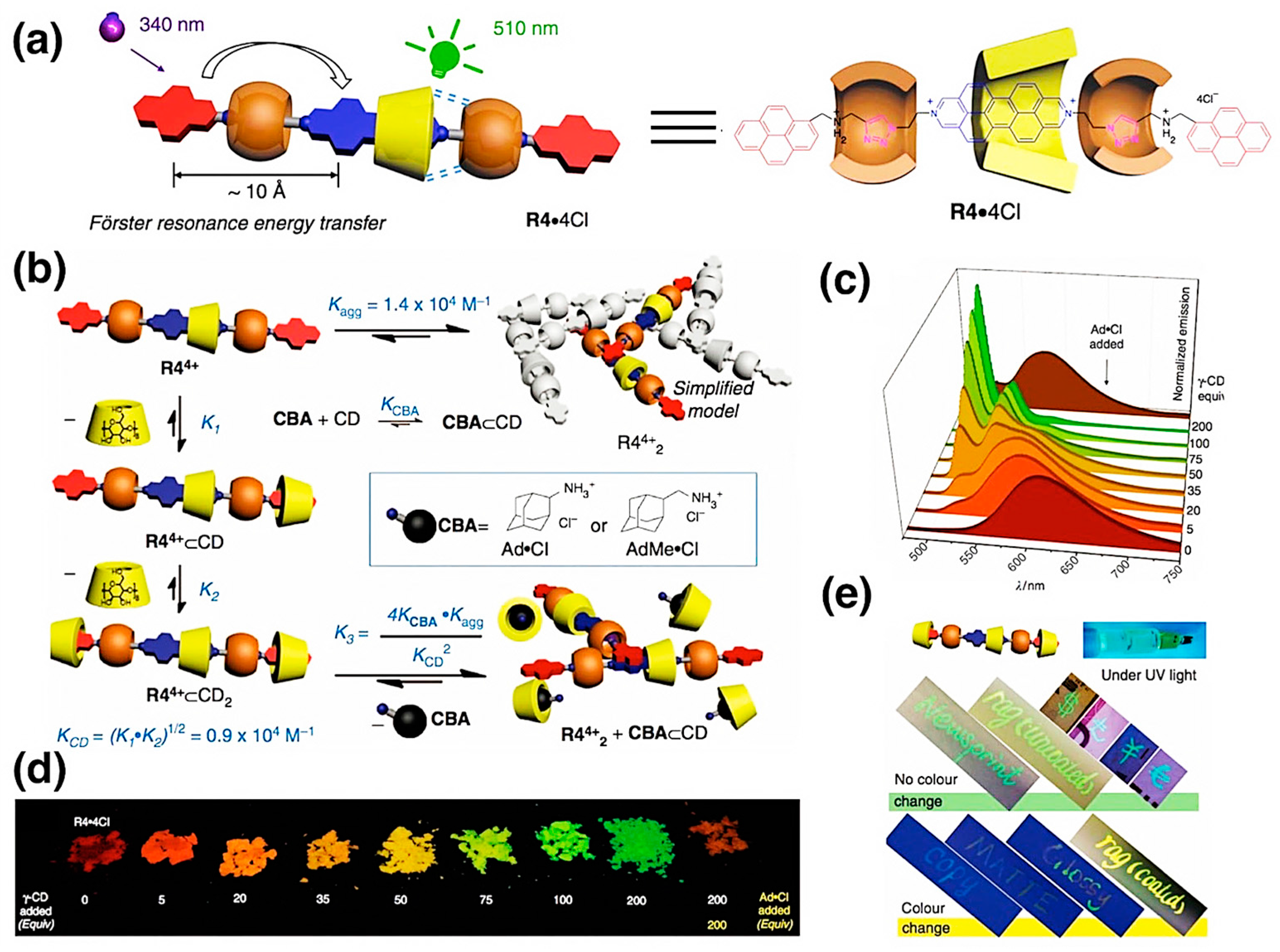
3.7. Chemical Encryption
4. Conclusions and Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lehn, J.-M. Supramolecular chemistry. Science 1993, 260, 1762–1763. [Google Scholar] [CrossRef] [PubMed]
- Pease, A.R.; Jeppesen, J.O.; Stoddart, J.F.; Luo, Y.; Collier, C.P.; Heath, J.R. Switching devices based on interlocked molecules. Acc. Chem. Res. 2000, 34, 433–444. [Google Scholar] [CrossRef]
- Bonifazi, D.; Mohnani, S.; Llanes-Pallas, A. Supramolecular chemistry at interfaces: Molecular recognition on nanopatterned porous surfaces. Chem.-Eur. J. 2009, 15, 7004–7025. [Google Scholar] [CrossRef] [PubMed]
- Amabilino, D.B.; Smith, D.K.; Steed, J.W. Supramolecular materials. Chem. Soc. Rev. 2017, 46, 2404–2420. [Google Scholar] [CrossRef] [PubMed]
- Nobel prize. Available online: http://www.nobelprize.org/nobel_prizes/chemistry/laureates/2016 (accessed on 28 September 2017).
- Hu, J.; Liu, S. Engineering responsive polymer building blocks with host-guest molecular recognition for functional applications. Acc. Chem. Res. 2014, 47, 2084–2095. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Nalluri, S.K.M.; Stoddart, J.F. Surveying macrocyclic chemistry: From flexible crown ethers to rigid cyclophanes. Chem. Soc. Rev. 2017, 46, 2459–2478. [Google Scholar] [CrossRef] [PubMed]
- Qu, D.H.; Wang, Q.C.; Zhang, Q.W.; Ma, X.; Tian, H. Photoresponsive host-guest functional systems. Chem. Rev. 2015, 115, 7543–7588. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.W.; Sun, Y.L.; Song, N. Switchable host-guest systems on surfaces. Acc. Chem. Res. 2014, 47, 1950–1960. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.D.; Steed, J.W. Gels with sense: Supramolecular materials that respond to heat, light and sound. Chem. Soc. Rev. 2016, 45, 6546–6596. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; McGonigal, P.R.; Schneebeli, S.T.; Li, H.; Vermeulen, N.A.; Ke, C.; Stoddart, J.F. An artificial molecular pump. Nat. Nanotechnol. 2015, 10, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Klajn, R.; Stoddart, J.F.; Grzybowski, B.A. Nanoparticles functionalised with reversible molecular and supramolecular switches. Chem. Soc. Rev. 2010, 39, 2203–2237. [Google Scholar] [CrossRef] [PubMed]
- Del Valle, E.M.M. Cyclodextrins and their uses: A review. Process Biochem. 2004, 39, 1033–1046. [Google Scholar] [CrossRef]
- Harada, A. Cyclodextrin-based molecular machines. Acc. Chem. Res. 2001, 34, 456–464. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.S.; Liu, Y. Calixarene-based supramolecular polymerization in solution. Chem. Soc. Rev. 2012, 41, 5907–5921. [Google Scholar] [CrossRef] [PubMed]
- Sansone, F.; Baldini, L.; Casnati, A.; Ungaro, R. Calixarenes: From biomimetic receptors to multivalent ligands for biomolecular recognition. New J. Chem. 2010, 34, 2715. [Google Scholar] [CrossRef]
- Lee, J.W.; Samal, S.; Selvapalam, N.; Kim, H.-J.; Kim, K. Cucurbituril homologues and derivatives: New opportunities in supramolecular chemistry. Acc. Chem. Res. 2003, 36, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Lagona, J.; Mukhopadhyay, P.; Chakrabarti, S.; Isaacs, L. The cucurbit[n]uril family. Angew. Chem. Int. Ed. 2005, 44, 4844–4870. [Google Scholar] [CrossRef] [PubMed]
- Cragg, P.J.; Sharma, K. Pillar[5]arenes: Fascinating cyclophanes with a bright future. Chem. Soc. Rev. 2012, 41, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Sathiyajith, C.; Shaikh, R.R.; Han, Q.; Zhang, Y.; Meguellati, K.; Yang, Y.W. Biological and related applications of pillar[n]arenes. Chem. Commun. 2017, 53, 677–696. [Google Scholar] [CrossRef] [PubMed]
- Strutt, N.L.; Zhang, H.; Schneebeli, S.T.; Stoddart, J.F. Functionalizing pillar[n]arenes. Acc. Chem. Res. 2014, 47, 2631–2642. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ping, G.; Li, C. Efficient complexation between pillar[5]arenes and neutral guests: From host-guest chemistry to functional materials. Chem. Commun. 2016, 52, 9858–9872. [Google Scholar] [CrossRef] [PubMed]
- Dong, Z.; Luo, Q.; Liu, J. Artificial enzymes based on supramolecular scaffolds. Chem. Soc. Rev. 2012, 41, 7890–7908. [Google Scholar] [CrossRef] [PubMed]
- Song, N.; Yang, Y.W. Molecular and supramolecular switches on mesoporous silica nanoparticles. Chem. Soc. Rev. 2015, 44, 3474–3504. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.-L.; Li, H.; Qiu, Y.-C.; Chen, D.-X.; Wang, X.; Pan, R.-Y.; Wang, Y.; Zhang, S.X.-A.; Wang, B.; Yang, Y.-W. Stimuli-responsive metal–organic frameworks gated by pillar[5]arene supramolecular switches. Chem. Sci. 2015, 6, 1640–1644. [Google Scholar] [CrossRef]
- Li, Z.; Barnes, J.C.; Bosoy, A.; Stoddart, J.F.; Zink, J.I. Mesoporous silica nanoparticles in biomedical applications. Chem. Soc. Rev. 2012, 41, 2590–2605. [Google Scholar] [CrossRef] [PubMed]
- Song, N.; Chen, D.X.; Qiu, Y.C.; Yang, X.Y.; Xu, B.; Tian, W.; Yang, Y.W. Stimuli-responsive blue fluorescent supramolecular polymers based on a pillar[5]arene tetramer. Chem. Commun. 2014, 50, 8231–8234. [Google Scholar] [CrossRef] [PubMed]
- Song, N.; Chen, D.X.; Xia, M.C.; Qiu, X.L.; Ma, K.; Xu, B.; Tian, W.; Yang, Y.W. Supramolecular assembly-induced yellow emission of 9,10-distyrylanthracene bridged bis(pillar[5]arene)s. Chem. Commun. 2015, 51, 5526–5529. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.; Kim, K.; Ogoshi, T.; Yao, W.; Gibb, B.C. The aqueous supramolecular chemistry of cucurbit[n]urils, pillar[n]arenes and deep-cavity cavitands. Chem. Soc. Rev. 2017, 46, 2479–2496. [Google Scholar] [CrossRef] [PubMed]
- Ni, X.L.; Chen, S.; Yang, Y.; Tao, Z. Facile cucurbit[8]uril-based supramolecular approach to fabricate tunable luminescent materials in aqueous solution. J. Am. Chem. Soc. 2016, 138, 6177–6183. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.W.; Li, D.; Li, X.; White, P.B.; Mecinovic, J.; Ma, X.; Agren, H.; Nolte, R.J.; Tian, H. Multicolour photoluminescence including white-light emission by a single host-guest complex. J. Am. Chem. Soc. 2016, 138, 13541–13550. [Google Scholar] [CrossRef] [PubMed]
- Medintz, I.L.; Clapp, A.R.; Mattoussi, H.; Goldman, E.R.; Fisher, B.; Mauro, J.M. Self-assembled nanoscale biosensors based on quantum dot fret donors. Nat. Mater. 2003, 2, 630–638. [Google Scholar] [CrossRef] [PubMed]
- Sapsford, K.E.; Berti, L.; Medintz, I.L. Materials for fluorescence resonance energy transfer analysis: Beyond traditional donor-acceptor combinations. Angew. Chem. Int. Ed. 2006, 45, 4562–4589. [Google Scholar] [CrossRef] [PubMed]
- Scholes, G.D. Long-range resonance energy transfer in molecular systems. Annu. Rev. Phys. Chem. 2003, 54, 57–87. [Google Scholar] [CrossRef] [PubMed]
- Tsien, R.Y. The green fluorescent protein. Annu. Rev. Biochem. 1998, 67, 509–544. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.A.; Dey, D.; Chakraborty, S.; Saha, J.; Roy, A.D.; Chakraborty, S.; Debnath, P.; Bhattacharjee, D. Fluorescence Resonance Energy Transfer (FRET) sensor. Sci. Lett. J. 2015, 4, 119. [Google Scholar]
- Ray, P.C.; Fan, Z.; Crouch, R.A.; Sinha, S.S.; Pramanik, A. Nanoscopic optical rulers beyond the fret distance limit: Fundamentals and applications. Chem. Soc. Rev. 2014, 43, 6370–6404. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.D.; Dang, D.T.; van Dongen, J.L.; Brunsveld, L. Protein dimerization induced by supramolecular interactions with cucurbit[8]uril. Angew. Chem. Int. Ed. 2010, 49, 895–898. [Google Scholar] [CrossRef]
- Uhlenheuer, D.A.; Young, J.F.; Nguyen, H.D.; Scheepstra, M.; Brunsveld, L. Cucurbit[8]uril induced heterodimerization of methylviologen and naphthalene functionalized proteins. Chem. Commun. 2011, 47, 6798–6800. [Google Scholar] [CrossRef]
- Hossain, M.A.; Mihara, H.; Ueno, A. Novel peptides bearing pyrene and coumarin units with or without β-cyclodextrin in their side chains exhibit intramolecular fluorescence resonance energy transfer. J. Am. Chem. Soc. 2003, 125, 11178–11179. [Google Scholar] [CrossRef]
- Yu, G.; Wu, D.; Li, Y.; Zhang, Z.; Shao, L.; Zhou, J.; Hu, Q.; Tang, G.; Huang, F. A pillar[5]arene-based [2]rotaxane lights up mitochondria. Chem. Sci. 2016, 7, 3017–3024. [Google Scholar] [CrossRef]
- Yu, G.; Zhao, R.; Wu, D.; Zhang, F.; Shao, L.; Zhou, J.; Yang, J.; Tang, G.; Chen, X.; Huang, F. Pillar[5]arene-based amphiphilic supramolecular brush copolymer: Fabrication, controllable self-assembly and application in self-imaging targeted drug delivery. Polym. Chem. 2016, 7, 6178–6188. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Wu, S.; Zeng, F.; Yu, C. Cyclodextrin supramolecular complex as a water-soluble ratiometric sensor for ferric ion sensing. Langmuir 2010, 26, 4529–4534. [Google Scholar] [CrossRef] [PubMed]
- Sha, J.; Song, Y.; Liu, B.; Lü, C. Host-guest-recognition-based polymer brush-functionalized mesoporous silica nanoparticles loaded with conjugated polymers: A facile fret-based ratiometric probe for Hg2+. Micropor. Mesopor. Mat. 2015, 218, 137–143. [Google Scholar] [CrossRef]
- Xue, M.; Wei, W.; Su, Y.; Johnson, D.; Heath, J.R. Supramolecular probes for assessing glutamine uptake enable semi-quantitative metabolic models in single cells. J. Am. Chem. Soc. 2016, 138, 3085–3093. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Gao, Y.; Li, Y.; Liu, S.; Zhang, H.; Su, X. A novel fluorescence probing strategy based on mono-[6-(2-aminoethylamino)-6-deoxy]-β-cyclodextin functionalized graphene oxide for the detection of amantadine. Sens. Actuat. B-Chem. 2014, 202, 323–329. [Google Scholar] [CrossRef]
- Ye, H.; Yang, L.; Zhao, G.; Zhang, Y.; Ran, X.; Wu, S.; Zou, S.; Xie, X.; Zhao, H.; Li, C.-P. A fret-based fluorescent approach for labetalol sensing using calix[6]arene functionalized MnO2@graphene as a receptor. RSC Adv. 2016, 6, 79350–79360. [Google Scholar] [CrossRef]
- Villafiorita-Monteoleone, F.; Daita, V.; Quarti, C.; Perdicchia, D.; Del Buttero, P.; Scavia, G.; Zoppo, M.D.; Botta, C. Light harvesting of cdse/cds quantum dots coated with β-cyclodextrin based host–guest species through resonant energy transfer from the guests. RSC Adv. 2014, 4, 28886–28892. [Google Scholar] [CrossRef]
- Meng, L.B.; Li, D.; Xiong, S.; Hu, X.Y.; Wang, L.; Li, G. Fret-capable supramolecular polymers based on a bodipy-bridged pillar[5]arene dimer with bodipy guests for mimicking the light-harvesting system of natural photosynthesis. Chem. Commun. 2015, 51, 4643–4646. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Y.; Han, J.M.; Yan, J.; Ma, Y.; Pei, J. A mechanically interlocked [3]rotaxane as a light-harvesting antenna: Synthesis, characterization, and intramolecular energy transfer. Chem. Eur. J. 2009, 15, 3585–3594. [Google Scholar] [CrossRef] [PubMed]
- Han, J.-M.; Wang, X.-Y.; Zhang, Y.-H.; Liu, C.; Pei, J. Main-chain hyperbranched polyrotaxane: Synthesis, photophysical properties, and energy funnel. Polymer 2012, 53, 3704–3711. [Google Scholar] [CrossRef]
- Ogoshi, T.; Yamafuji, D.; Yamagishi, T.A.; Brouwer, A.M. Forster resonance energy transfer by formation of a mechanically interlocked [2]rotaxane. Chem. Commun. 2013, 49, 5468–5470. [Google Scholar] [CrossRef] [PubMed]
- Bojtár, M.; Szakács, Z.; Hessz, D.; Bazsó, F.L.; Kállay, M.; Kubinyi, M.; Bitter, I. Supramolecular fret modulation by pseudorotaxane formation of a ditopic stilbazolium dye and carboxylato-pillar[5]arene. Dyes Pigments 2016, 133, 415–423. [Google Scholar] [CrossRef]
- Liu, G.; Xu, X.; Chen, Y.; Wu, X.; Wu, H.; Liu, Y. A highly efficient supramolecular photoswitch for singlet oxygen generation in water. Chem. Commun. 2016, 52, 7966–7969. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Jin, W.; Chen, E.; Zhou, J.; Zhou, L.; Wei, S. Drug delivery function of carboxymethyl-β-cyclodextrin modified upconversion nanoparticles for adamantine phthalocyanine and their nir-triggered cancer treatment. Dalton Trans. 2016, 45, 3853–3862. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Gomez-Duran, C.F.A.; Smith, B.D. Fluorescent neuraminidase assay based on supramolecular dye capture after enzymatic cleavage. J. Am. Chem. Soc. 2017, 139, 6390–6395. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Dong, R.; Wang, D.; Zhao, T.; Gao, Y.; Duffy, P.; Zhu, X.; Wang, W. Supramolecular fluorescent nanoparticles constructed via multiple non-covalent interactions for the detection of hydrogen peroxide in cancer cells. Chem.-Eur. J. 2015, 21, 11427–11434. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Ke, C.; Bruns, C.J.; McGonigal, P.R.; Pettman, R.B.; Stoddart, J.F. Tunable solid-state fluorescent materials for supramolecular encryption. Nat. Commun. 2015, 6, 6884. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gangopadhyay, M.; Maity, A.; Dey, A.; Das, A. [2]Pseudorotaxane formation with fret based luminescence response: Demonstration of boolean operations through self-sorting on solid surface. J. Org. Chem. 2016, 81, 8977–8987. [Google Scholar] [CrossRef] [PubMed]
- Grana-Suarez, L.; Verboom, W.; Huskens, J. Fluorescent supramolecular nanoparticles signal the loading of electrostatically charged cargo. Chem. Commun. 2016, 52, 2597–2600. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Yan, F.; Huang, Y.; Kong, D.; Ye, Q.; Xu, J.; Chen, L. Rhodamine-based ratiometric fluorescent probes based on excitation energy transfer mechanisms: Construction and applications in ratiometric sensing. RSC Adv. 2016, 6, 50732–50760. [Google Scholar] [CrossRef]
- Lee, Y.H.; Lee, M.H.; Zhang, J.F.; Kim, J.S. Pyrene excimer-based calix[4]arene FRET chemosensor for mercury(II). J. Org. Chem. 2010, 75, 7159–7165. [Google Scholar] [CrossRef] [PubMed]
- Othman, A.B.; Lee, J.W.; Wu, J.-S.; Kim, J.S.; Abidi, R.; Thuery, P.; Strub, J.M.; Van Dorsselaer, A.; Vicens, J. Calix[4]arene-based, Hg2+-induced intramolecular fluorescence resonance energy transfer chemosensor. J. Org. Chem. 2007, 72, 7634–7640. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yan, R.; Huo, Z.; Wang, L.; Zeng, J.; Bao, J.; Wang, X.; Peng, Q.; Li, Y. Fluorescence resonant energy transfer biosensor based on upconversion-luminescent nanoparticles. Angew. Chem. Int. Ed. 2005, 44, 6054–6057. [Google Scholar] [CrossRef] [PubMed]
- Shulov, I.; Rodik, R.V.; Arntz, Y.; Reisch, A.; Kalchenko, V.I.; Klymchenko, A.S. Protein-sized bright fluorogenic nanoparticles based on cross-linked calixarene micelles with cyanine corona. Angew. Chem. Int. Ed. 2016, 55, 15884–15888. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-S.; Kim, J.-H.; Kim, H.; Laquai, F.; Arifin, E.; Lee, J.-K.; Yoo, S.; Sohn, B.-H. Switching off FRET in the hybrid assemblies of diblock copolymer micelles, quantum dots, and dyes by plasmonic nanoparticles. ACS Nano 2012, 6, 5051–5059. [Google Scholar] [CrossRef] [PubMed]
- Gatti, T.; Brambilla, L.; Tommasini, M.; Villafiorita-Monteleone, F.; Botta, C.; Sarritzu, V.; Mura, A.; Bongiovanni, G.; Zoppo, M.D. Near IR to red up-conversion in tetracene/pentacene host/guest cocrystals enhanced by energy transfer from host to guest. J. Phys. Chem. C 2015, 119, 17495–17501. [Google Scholar] [CrossRef]
- Balogh, D.; Garcia, M.A.A.; Albada, H.B.; Willner, I. Programmed synthesis by stimuli-responsive dnazyme-modified mesoporous SiO2 nanoparticles. Angew. Chem. Int. Ed. 2015, 54, 11652–11656. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.H.; Cecconello, A.; Elbaz, J.; Credi, A.; Willner, I. A three-station DNA catenane rotary motor with controlled directionality. Nano Lett. 2013, 13, 2303–2308. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.H.; Cecconello, A.; Qi, X.J.; Wu, N.; Jester, S.S.; Famulok, M.; Matthies, M.; Schmidt, T.L.; Willner, I. Switchable reconfiguration of a seven-ring interlocked DNA catenane nanostructure. Nano Lett. 2015, 15, 7133–7137. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.H.; Qi, X.J.; Cecconello, A.; Jester, S.S.; Famulok, M.; Willner, I. Switchable reconfiguration of an interlocked DNA olympiadane nanostructure. Angew. Chem. Int. Ed. 2014, 53, 7499–7503. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.H.; Willner, I. Stimuli-responsive DNA-functionalized nano-/microcontainers for switchable and controlled release. Angew. Chem. Int. Ed. 2015, 54, 12212–12235. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lou, X.-Y.; Song, N.; Yang, Y.-W. Fluorescence Resonance Energy Transfer Systems in Supramolecular Macrocyclic Chemistry. Molecules 2017, 22, 1640. https://doi.org/10.3390/molecules22101640
Lou X-Y, Song N, Yang Y-W. Fluorescence Resonance Energy Transfer Systems in Supramolecular Macrocyclic Chemistry. Molecules. 2017; 22(10):1640. https://doi.org/10.3390/molecules22101640
Chicago/Turabian StyleLou, Xin-Yue, Nan Song, and Ying-Wei Yang. 2017. "Fluorescence Resonance Energy Transfer Systems in Supramolecular Macrocyclic Chemistry" Molecules 22, no. 10: 1640. https://doi.org/10.3390/molecules22101640





