Purification of Flavonoids from Chinese Bayberry (Morella rubra Sieb. et Zucc.) Fruit Extracts and α-Glucosidase Inhibitory Activities of Different Fractionations
Abstract
:1. Introduction
2. Results and Discussion
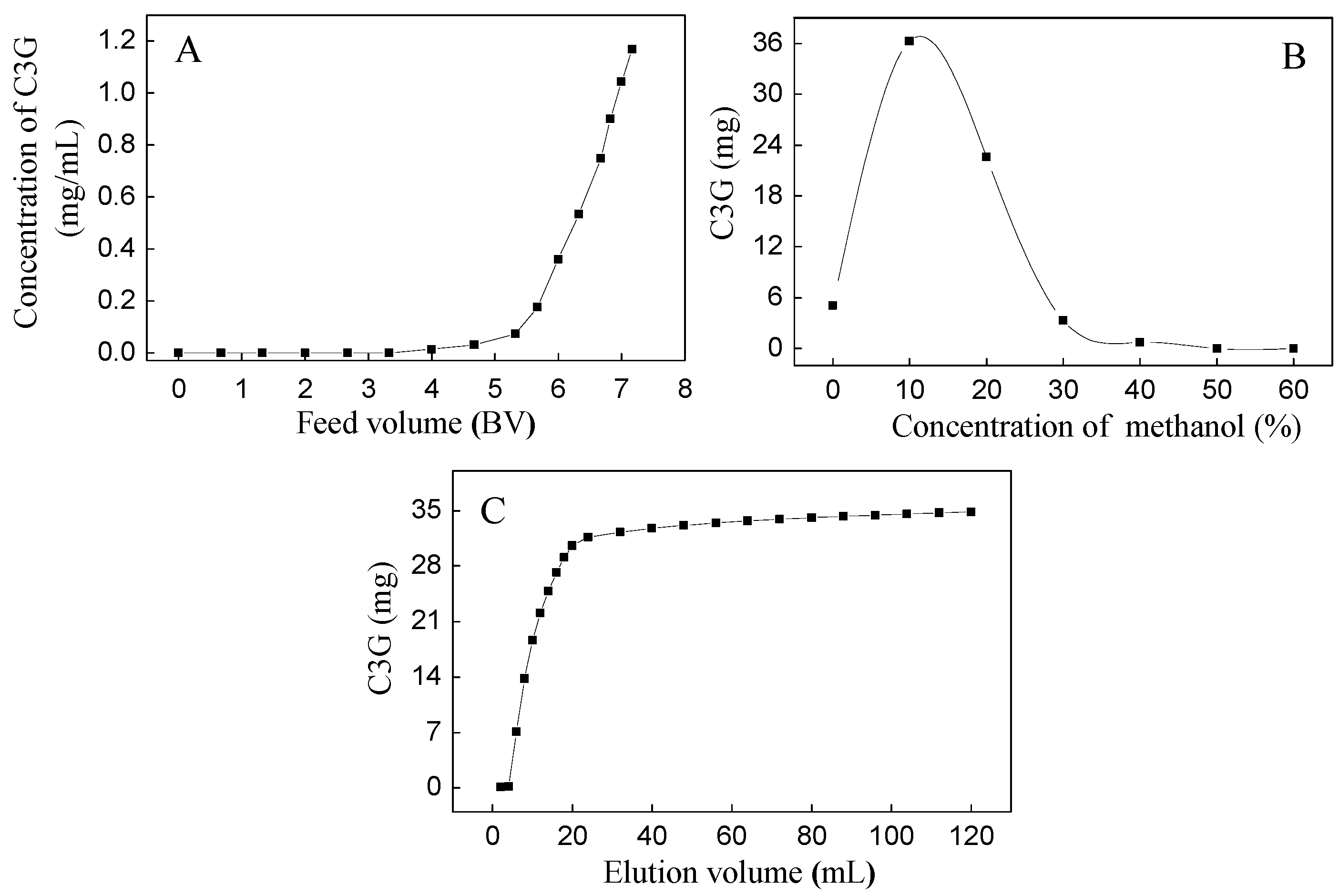
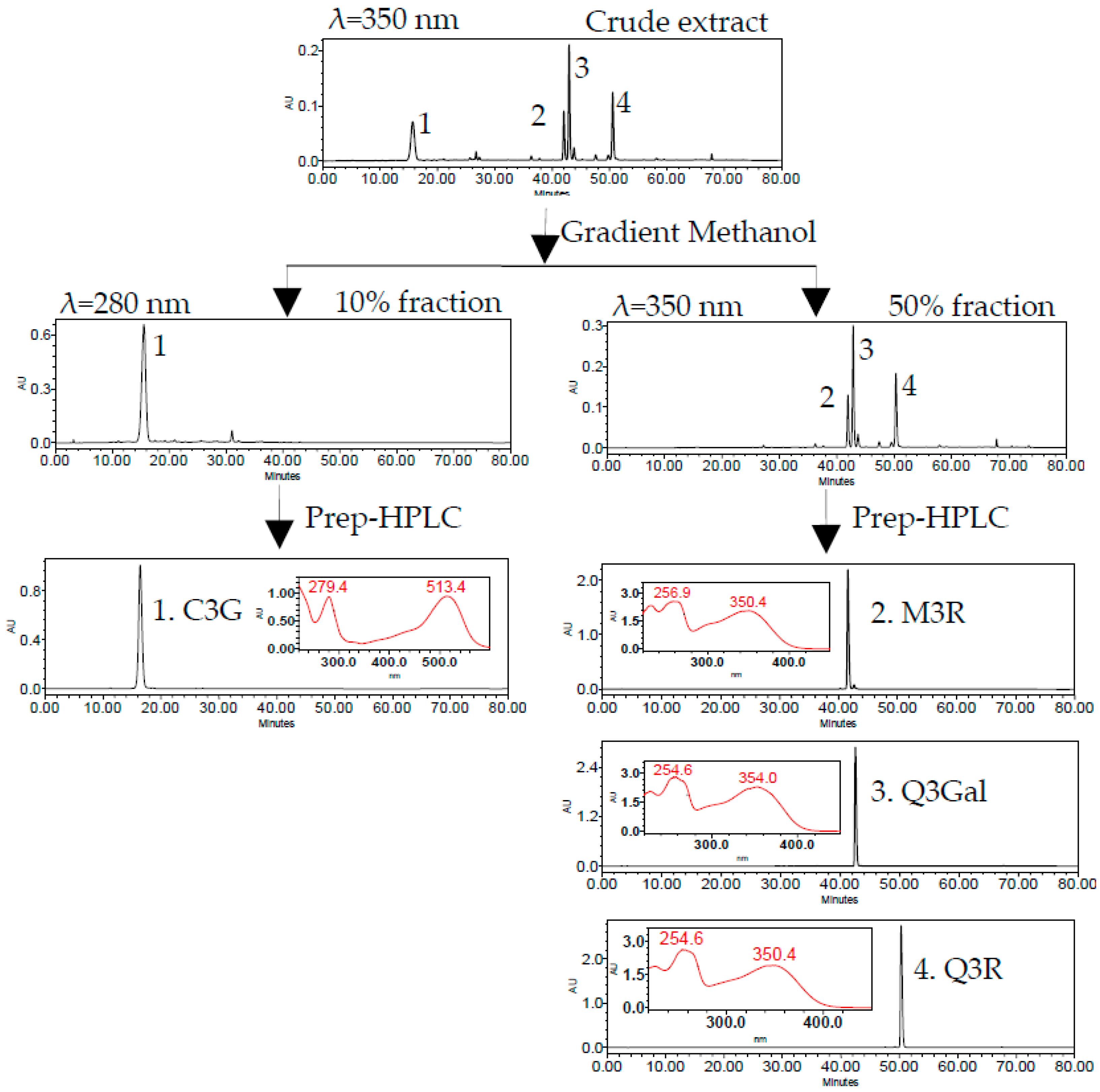
2.1. Purification of Four Flavonoid Glycosides from BQ Pulp
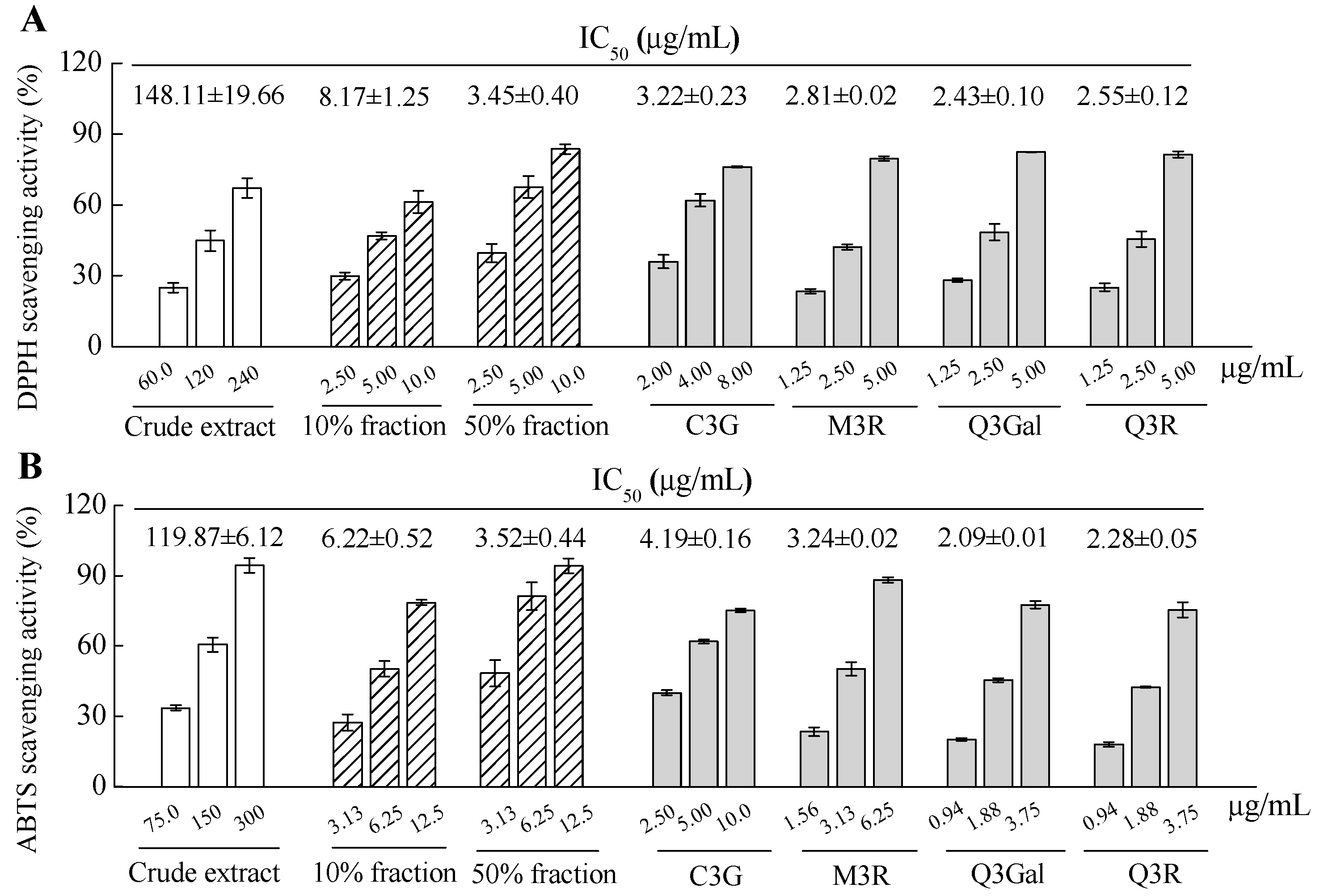
2.2. Antioxidant Activities Assay
2.3. α-Glucosidase Inhibitory Activity of Different Fractions from BQ Pulp
2.4. α-Glucosidase Inhibitory Activity of Six Bayberry Cultivars
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Fruit Materials
3.3. HPLC Analysis of Four Flavonoid Glycosides
3.4. Preparation of the Crude Extract
3.5. Dynamic Adsorption and Desorption Tests
3.6. Prep-HPLC Purification
3.7. LC-MS Analysis
3.8. Antioxidant Activity
3.9. Inhibition of α-Glucosidase Activity
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2014, 37, 81–90. [Google Scholar]
- Bischoff, H. Pharmacology of α-glucosidase inhibition. Eur. J. Clin. Investig. 1994, 24, 3–10. [Google Scholar]
- Baron, A.D. Postprandial hyperglycaemia and alpha-glucosidase inhibitors. Diabetes Res. Clin. Pract. 1998, 40, S51–S55. [Google Scholar] [CrossRef]
- Fujisawa, T.; Ikegami, H.; Inoue, K.; Kawabata, Y.; Ogihara, T. Effect of two α-glucosidase inhibitors, voglibose and acarbose, on postprandial hyperglycemia correlates with subjective abdominal symptoms. Metabolism 2005, 54, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Laar, F.A.V.D.; Lucassen, P.L.; Akkermans, R.P.; Lisdonk, E.H.V.D.; Rutten, G.E.; Weel, C.V. α-Glucosidase inhibitors for patients with type 2 diabetes results from a Cochrane systematic review and meta-analysis. Diabetes Care 2005, 28, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Madar, Z. The effect of acarbose and miglitol (BAY-M-1099) on postprandial glucose levels following ingestion of various sources of starch by nondiabetic and streptozotocin-induced diabetic rats. J. Nutr. 1989, 119, 2023–2029. [Google Scholar] [PubMed]
- Zhou, S.H.; Fang, Z.X.; Yuan, L.; Chen, J.C.; Liu, D.H.; Ye, X.Q. Phenolics and antioxidant properties of bayberry (Myrica rubra Sieb. et Zucc.) pomace. Food Chem. 2009, 112, 394–399. [Google Scholar] [CrossRef]
- Fang, Z.; Zhang, M.; Wang, L. HPLC-DAD-ESIMS analysis of phenolic compounds in bayberries (Myrica rubra Sieb. et Zucc.). Food Chem. 2007, 100, 845–852. [Google Scholar] [CrossRef]
- Bao, J.S.; Cai, Y.Z.; Sun, M.; Wang, G.Y.; Corke, H. Anthocyanins flavonols and free radical scavenging activity of Chinese bayberry Myrica rubra extracts and their color properties and stability. J. Agric. Food Chem. 2005, 53, 2327–2332. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.D.; Huang, H.Z.; Xu, C.J.; Li, X.; Chen, K.S. Biological activities of extracts from Chinese bayberry (Myrica rubra Sieb. et Zucc.): A review. Plant Foods Hum. Nutr. 2013, 68, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.D.; Zhang, B.; Zhang, J.K.; Xu, C.J.; Wu, Y.L.; Li, X.; Chen, K.S. Cyanidin-3-glucoside-rich extract from Chinese bayberry fruit protects pancreatic β cells and ameliorates hyperglycemia in streptozotocin-induced diabetic mice. J. Med. Food 2012, 15, 288–298. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.N.; Huang, H.Z.; Zhao, X.Y.; Lv, Q.; Sun, C.D.; Li, X.; Chen, K.S. Effects of flavonoids-rich Chinese bayberry (Myrica rubra Sieb. et Zucc.) pulp extracts on glucose consumption in human HepG2 cells. J. Funct. Foods 2015, 14, 144–153. [Google Scholar] [CrossRef]
- Zhang, X.N.; Huang, H.Z.; Zhang, Q.L.; Fan, F.J.; Xu, C.J.; Sun, C.D.; Li, X.; Chen, K.S. Phytochemical characterization of chinese bayberry (Myrica rubra Sieb. et Zucc.) of 17 cultivars and their antioxidant properties. Int. J. Mol. Sci. 2015, 16, 12467–12481. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Kang, M.X.; Xie, Q.P.; Xu, B.; Sun, C.D.; Chen, K.S.; Wu, Y.L. Anthocyanins from Chinese bayberry extract protect β cells from oxidative stress-mediated injury via HO-1 upregulation. J. Agric. Food Chem. 2011, 59, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Min, X.; Zou, T.; Ling, W.; Zhong, R.; Zhang, W. Cyanidin 3-glucoside attenuates obesity-associated insulin resistance and hepatic steatosis in high-fat diet-fed and db/db mice via the transcription factor foxo1. J. Nutr. Biochem. 2012, 23, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, R.; Nishimura, N.; Hoshino, H.; Isa, Y.; Kadowaki, M.; Ichi, T.; Tanaka, A.; Nishiumi, S.; Fukuda, I.; Ashida, H.; et al. Cyanidin 3-glucoside ameliorates hyperglycemia and insulin sensitivity due to downregulation of retinol binding protein 4 expression in diabetic mice. Biochem. Pharmacol. 2007, 74, 1619–1627. [Google Scholar] [CrossRef] [PubMed]
- Scazzocchio, B.; Varì, R.; Filesi, C.; D’Archivio, M.; Santangelo, C.; Giovannini, C. Cyanidin-3-O-β-glucoside and protocatechuic acid exert insulin-like effects by upregulating PPARγ activity in human omental adipocytes. Diabetes 2011, 60, 2234–2244. [Google Scholar] [CrossRef] [PubMed]
- Eid, H.M.; Martineau, L.C.; Saleem, A.; Muhammad, A.; Vallerand, D.; Benhaddou-Andaloussi, A.; Nistor, L.; Afshar, A.; Arnason, J.T.; Haddad, P.S. Stimulation of AMPK-activated protein kinase and enhancement of basal glucose uptake in muscle cells by quercetin and quercetin glycosides, active principles of the antidiabetic medicinal plant Vaccinium vitis-idaea. Mol. Nutr. Food Res. 2010, 54, 991–1003. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Yoshitomi, H.; Liu, T.; Zhou, B.; Sun, W.; Qin, L.; Guo, X.; Huang, L.; Wu, L.; Gao, M. Isoquercitrin activates the AMPK-activated protein kinase (AMPK) signal pathway in rat H4IIE cells. BMC Complement. Altern. Med. 2014, 14, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Schieber, A.; Keller, P.; Carle, R. Determination of phenolic acids and flavonoids of apple and pear by high-performance liquid chromatography. J. Chromatogr. A 2001, 910, 265–273. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cationdecolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Li, T.; Zhang, X.D.; Song, Y.W.; Liu, J.W. A microplate-based screening method for alpha-glucosidase inhibitors. J. Clin. Pharmacol. Ther. 2005, 10, 1128–1134. [Google Scholar]
- Zhang, J.; Zhao, S.; Yin, P.; Yan, L.; Han, J.; Shi, L.; Zhou, X.; Liu, Y.; Ma, C. α-Glucosidase inhibitory activity of polyphenols from the burs of Castanea mollissima Blume. Molecules 2014, 19, 8373–8386. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds 1–4 are available from the authors.
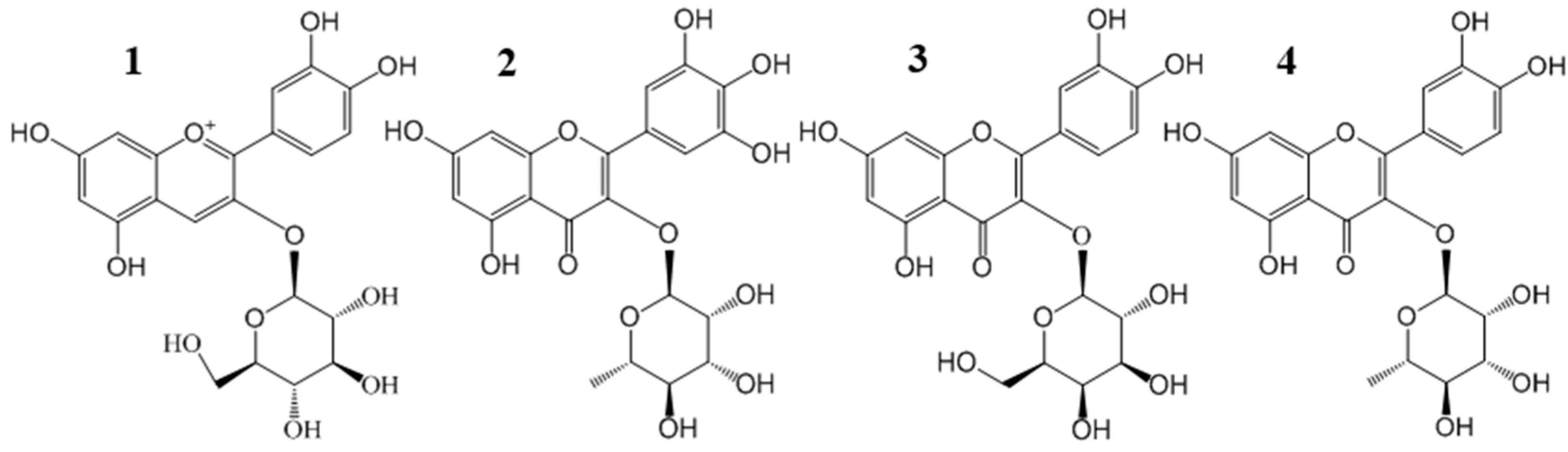

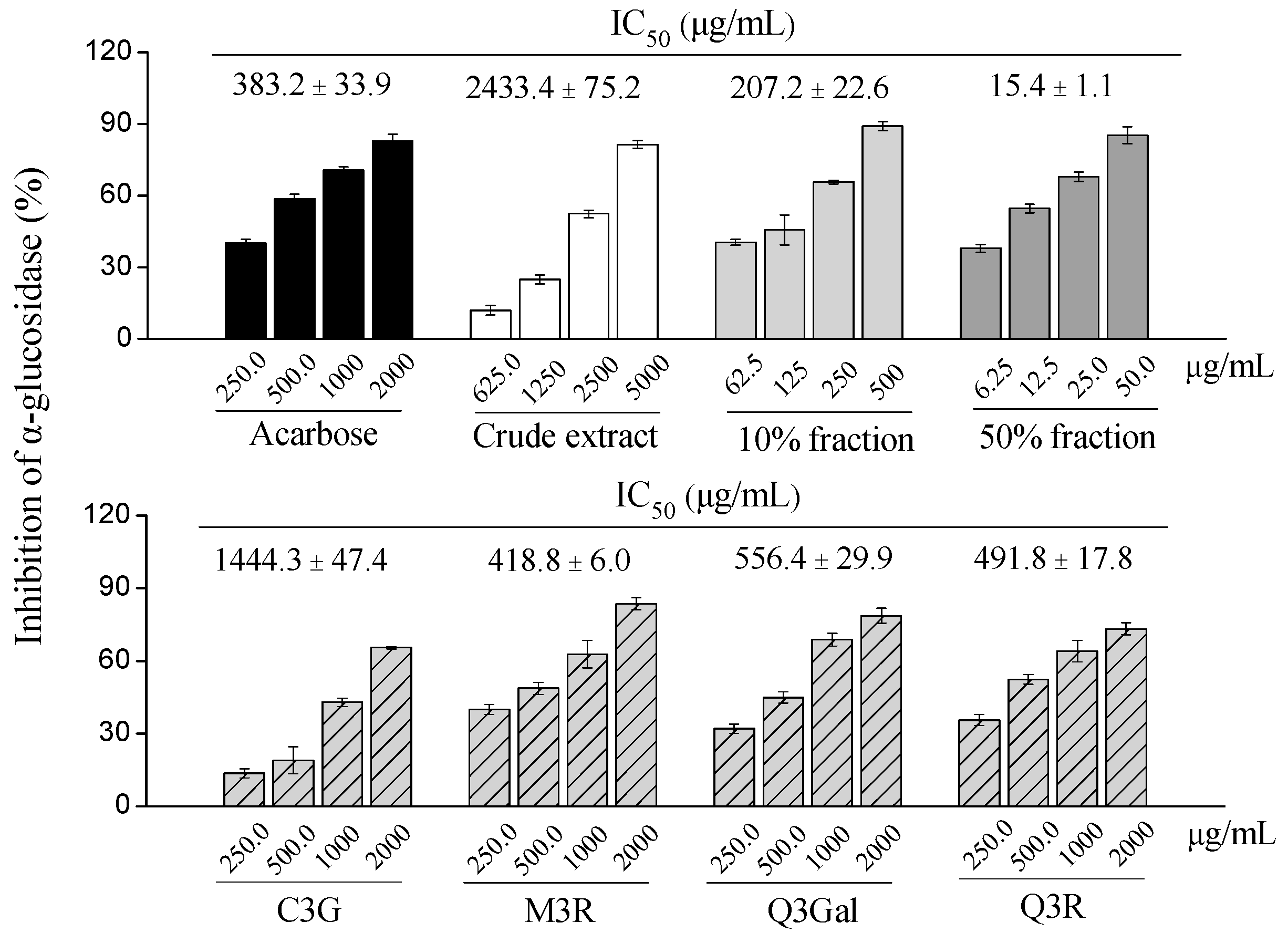
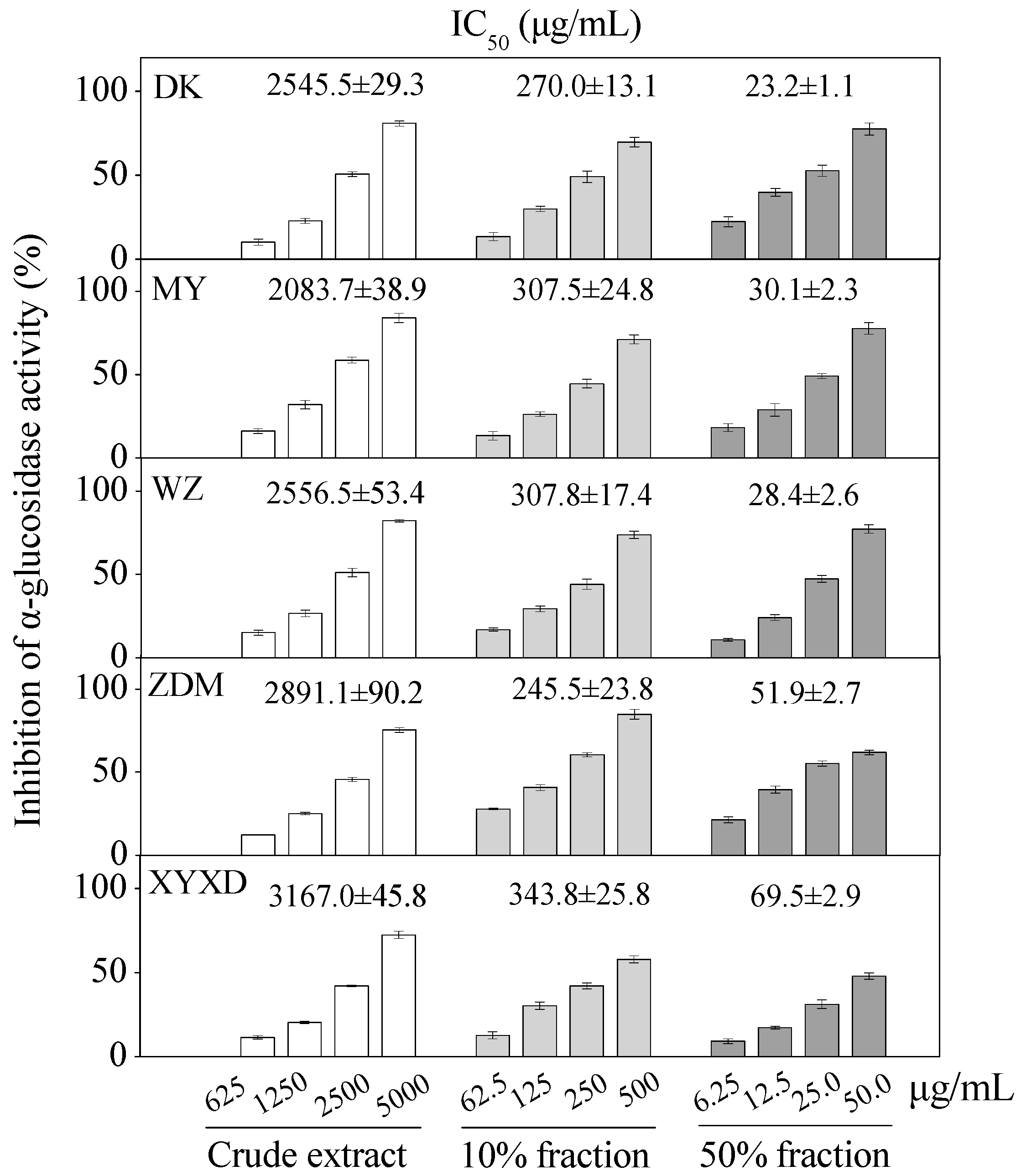
| Compounds | Purification Step | Purity (%) | Recovery (%) | Yield (mg) |
|---|---|---|---|---|
| C3G | ① Crude extract | 0.74 | / | / |
| ② SPE column | 54.12 | 90.75 | 620.4 1 | |
| ③ Prep-HPLC | 99.17 | 78.18 | 102.4 2 | |
| M3R | ① Crude extract | 0.02 | / | / |
| ② SPE column | 2.42 | 72.24 | 298.5 1 | |
| ③ Prep-HPLC | 90.38 | 71.58 | 4.6 3 | |
| Q3Gal | ① Crude extract | 0.06 | / | / |
| ② SPE column | 7.75 | 77.11 | 298.5 1 | |
| ③ Prep-HPLC | 94.57 | 79.32 | 15.6 3 | |
| Q3R | ① Crude extract | 0.03 | / | / |
| ② SPE column | 3.84 | 76.42 | 298.5 1 | |
| ③ Prep-HPLC | 93.61 | 77.20 | 7.6 3 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yan, S.; Zhang, X.; Wen, X.; Lv, Q.; Xu, C.; Sun, C.; Li, X. Purification of Flavonoids from Chinese Bayberry (Morella rubra Sieb. et Zucc.) Fruit Extracts and α-Glucosidase Inhibitory Activities of Different Fractionations. Molecules 2016, 21, 1148. https://doi.org/10.3390/molecules21091148
Yan S, Zhang X, Wen X, Lv Q, Xu C, Sun C, Li X. Purification of Flavonoids from Chinese Bayberry (Morella rubra Sieb. et Zucc.) Fruit Extracts and α-Glucosidase Inhibitory Activities of Different Fractionations. Molecules. 2016; 21(9):1148. https://doi.org/10.3390/molecules21091148
Chicago/Turabian StyleYan, Shuxia, Xianan Zhang, Xin Wen, Qiang Lv, Changjie Xu, Chongde Sun, and Xian Li. 2016. "Purification of Flavonoids from Chinese Bayberry (Morella rubra Sieb. et Zucc.) Fruit Extracts and α-Glucosidase Inhibitory Activities of Different Fractionations" Molecules 21, no. 9: 1148. https://doi.org/10.3390/molecules21091148
APA StyleYan, S., Zhang, X., Wen, X., Lv, Q., Xu, C., Sun, C., & Li, X. (2016). Purification of Flavonoids from Chinese Bayberry (Morella rubra Sieb. et Zucc.) Fruit Extracts and α-Glucosidase Inhibitory Activities of Different Fractionations. Molecules, 21(9), 1148. https://doi.org/10.3390/molecules21091148







