Arctigenin Inhibits Lung Metastasis of Colorectal Cancer by Regulating Cell Viability and Metastatic Phenotypes
Abstract
:1. Introduction
2. Results
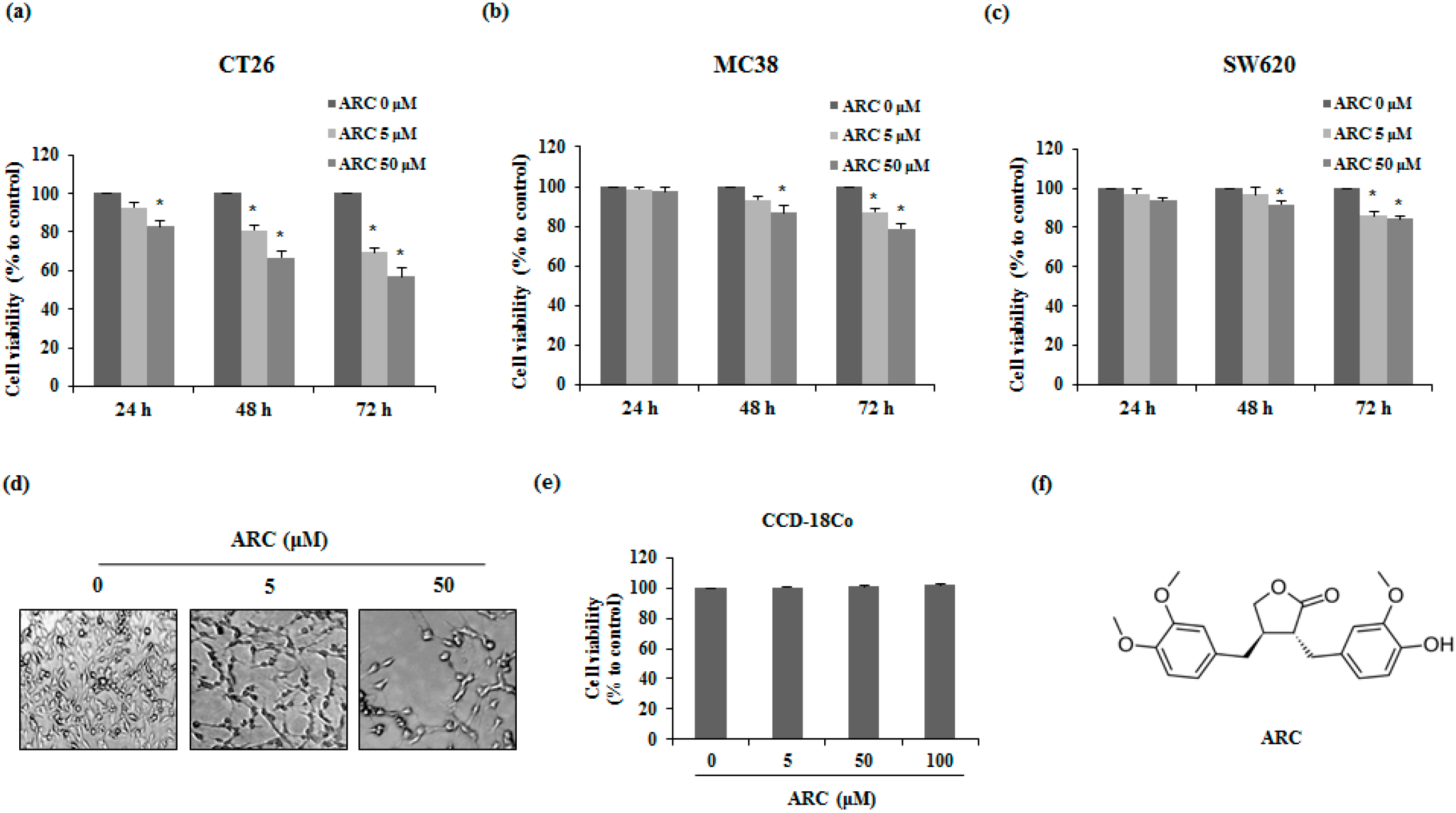
2.1. ARC Induces Cell Death of Colon Cancer Cells
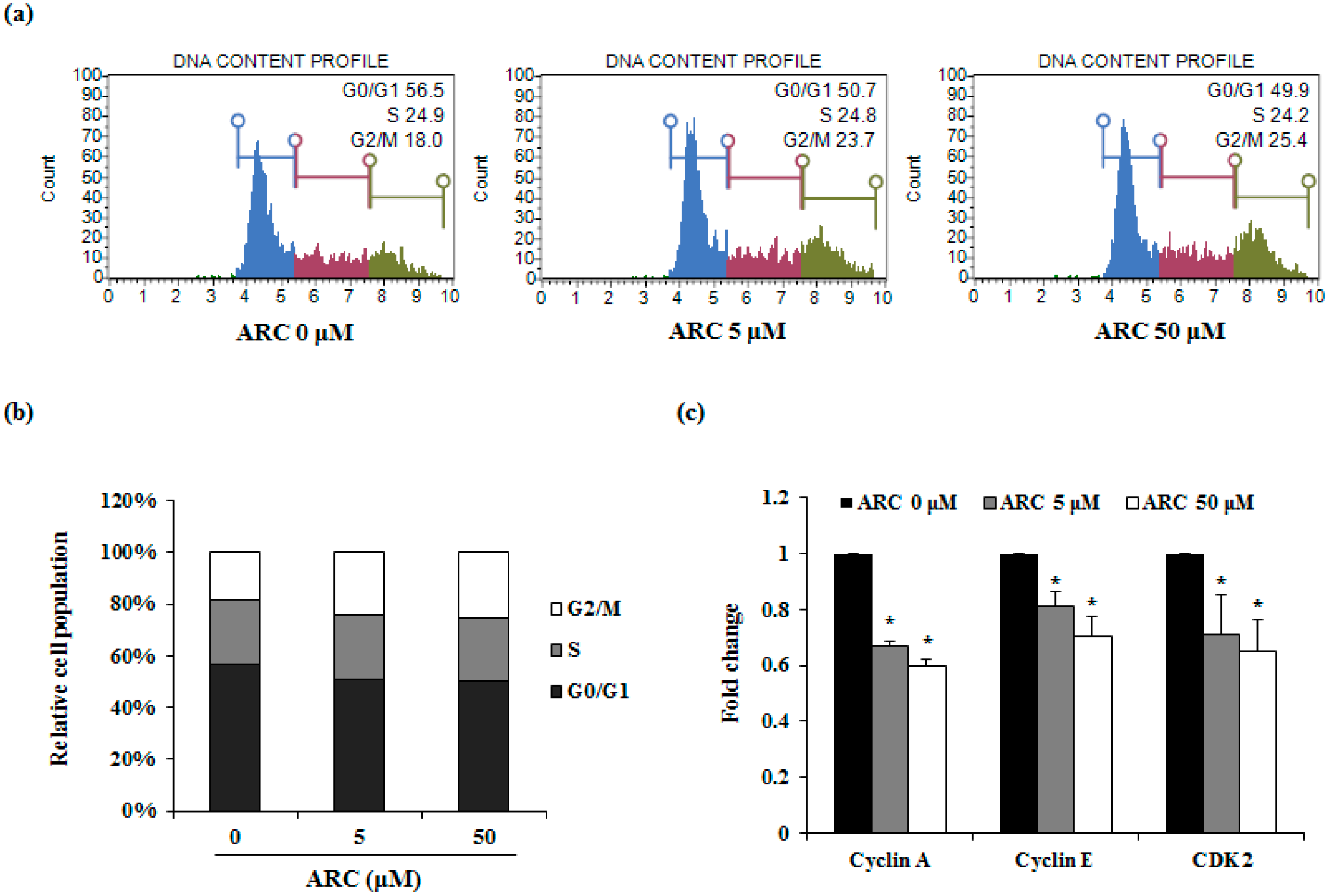
2.2. ARC Increases Cell Cycle Arrest in G2/M1 Phase and Induces Apoptosis in Colon Cancer Cells
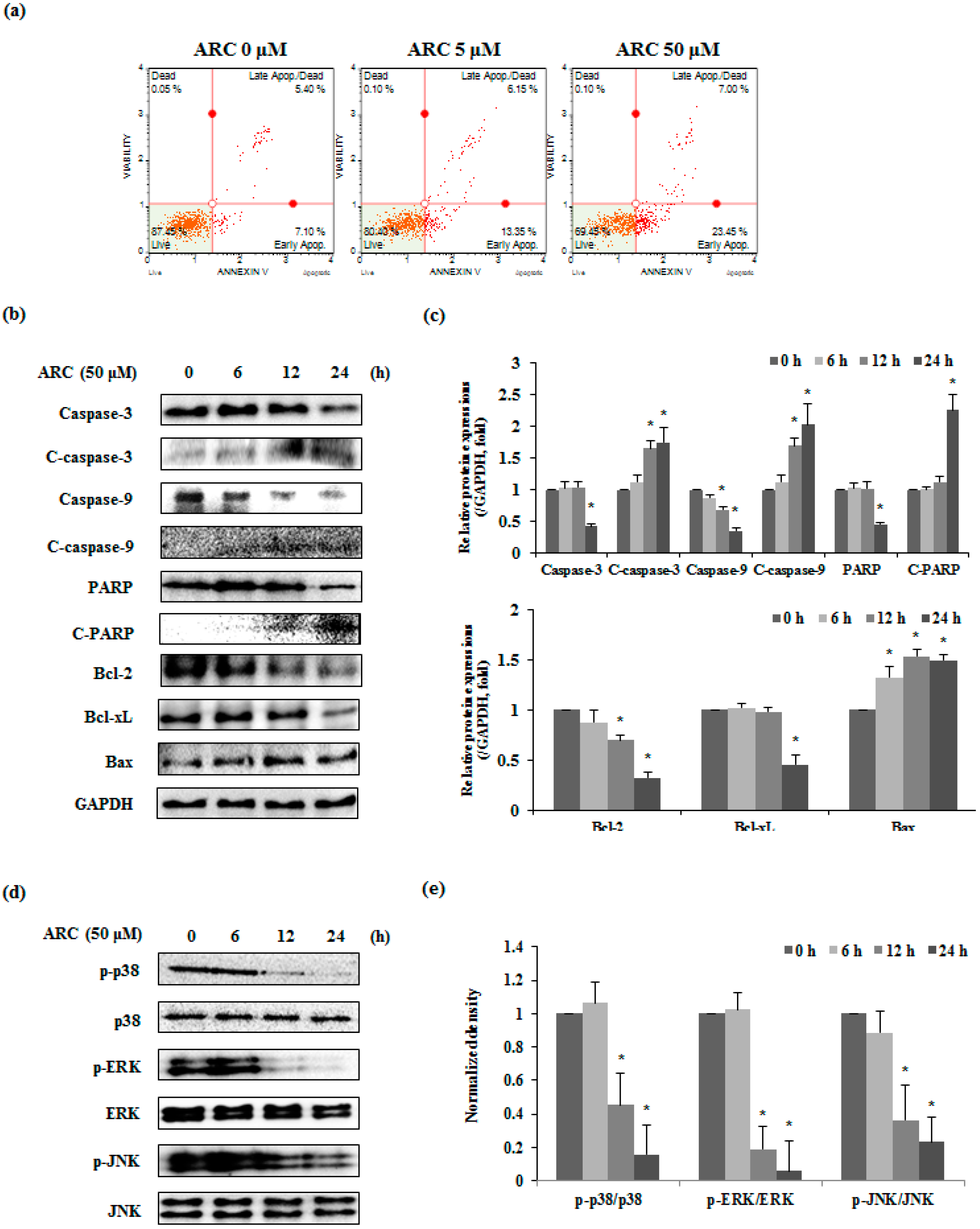
2.3. ARC Induces Apoptosis via MAPKs in Colon Cancer Cells
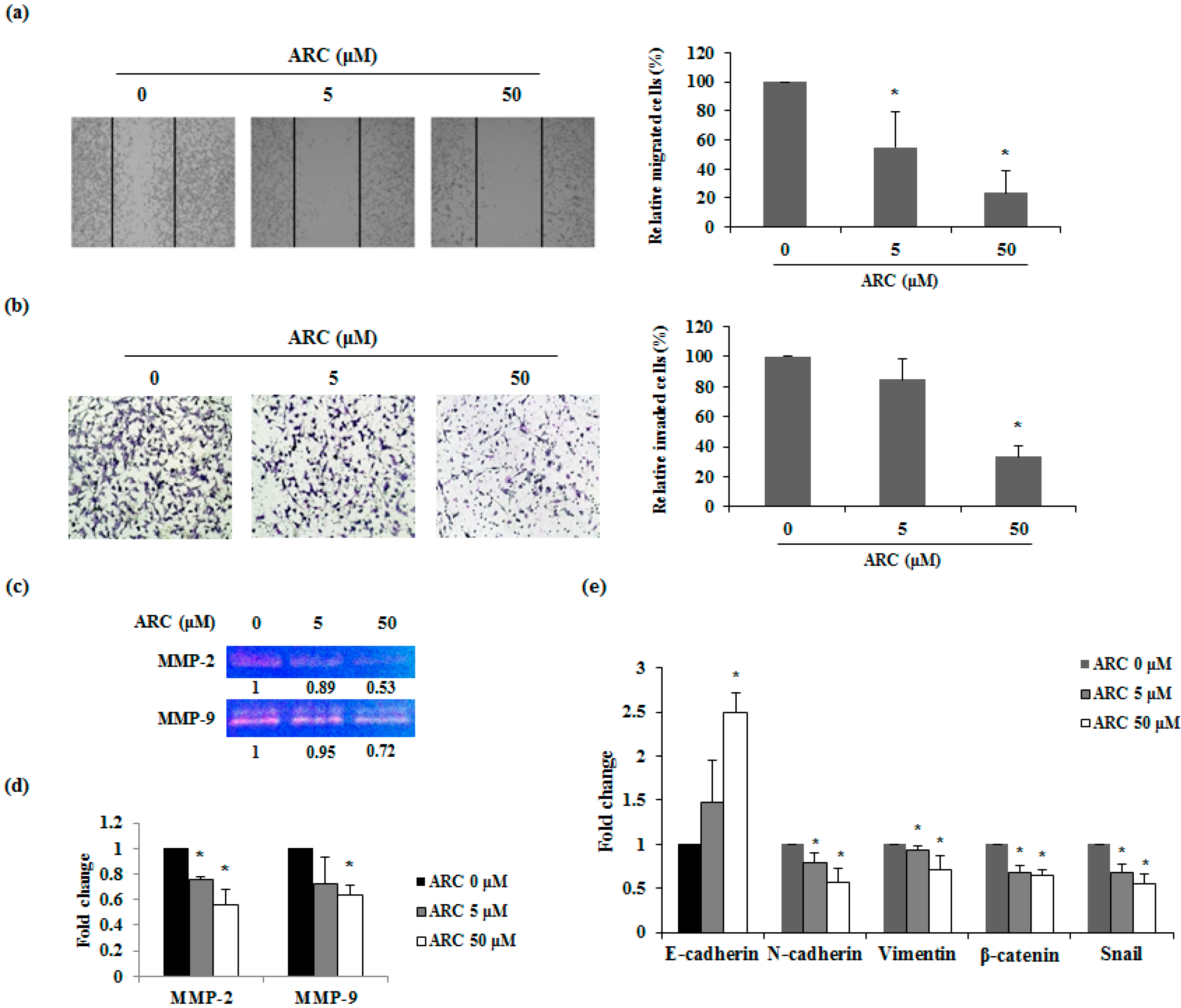
2.4. ARC Inhibits the Migration and Invasion Ability and Regulates EMT Transition of CT26 Cells
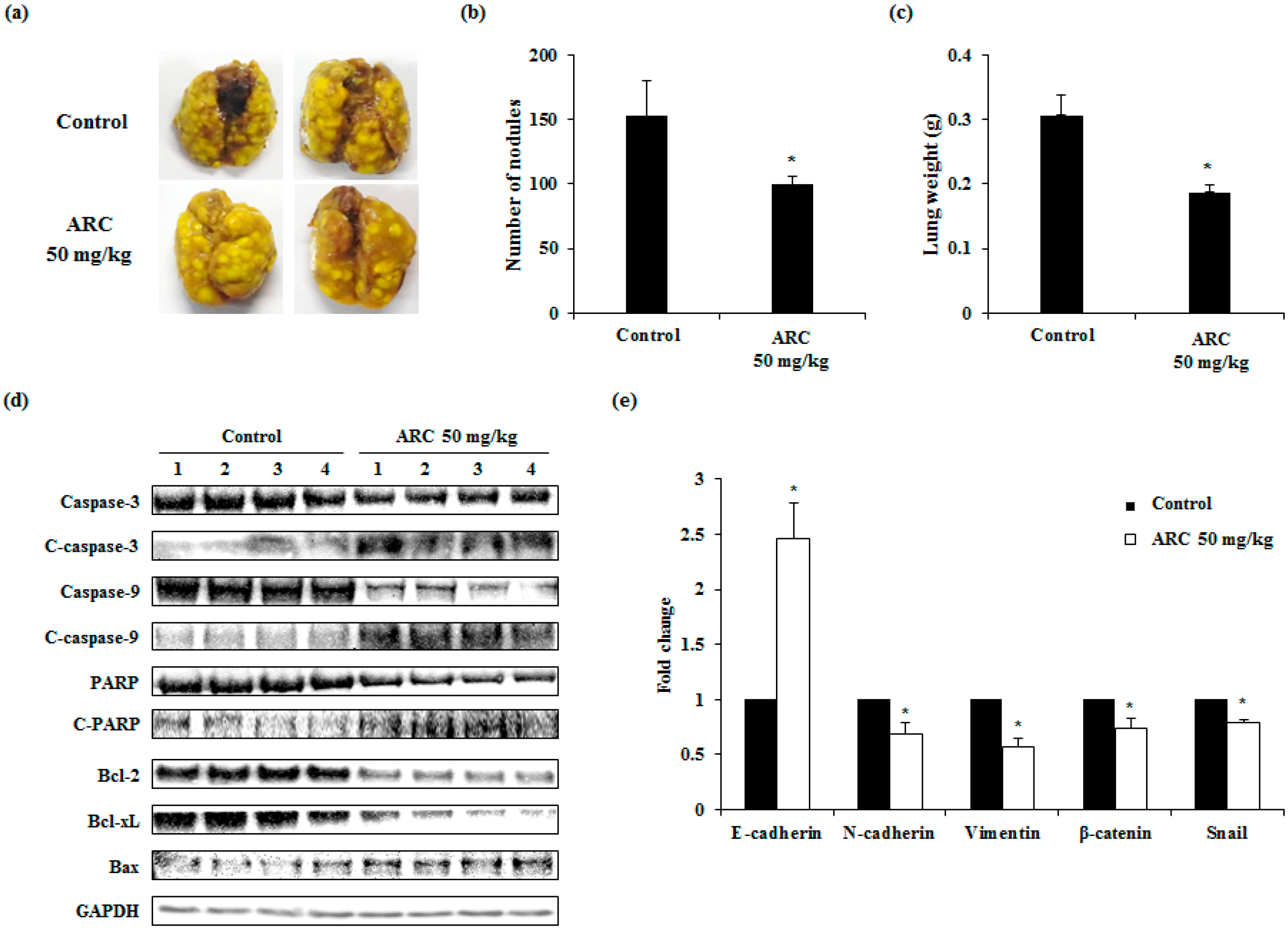
2.5. ARC Ameliorates Lung Metastasis of CT26 Cells in an Experimental Metastasis Model
3. Discussion
4. Experimental Section
4.1. Antibodies and Reagents
4.2. Cell Culture
4.3. Animals
4.4. Assays of Cell Viability
4.5. Annexin V Assay
4.6. Western Blot Analysis
4.7. Cell Cycle Analysis
4.8. Real-Time RT-PCR
4.9. Wound Healing Assay
4.10. Invasion Assay
4.11. Gelatin Zymography
4.12. Experimental Lung Metastasis Model
4.13. Statistical Analyses
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sun, W.J.; Sha, Z.F.; Gao, H. Determination of arctiin and arctigenin in Fructus Arctii by reverse-phase HPLC. Yao Xue Xue Bao 1992, 27, 549–551. [Google Scholar] [PubMed]
- Lee, J.Y.; Kim, C.J. Arctigenin, a phenylpropanoid dibenzylbutyrolactone lignan, inhibits type I-IV allergic inflammation and pro-inflammatory enzymes. Arch. Pharm. Res. 2010, 33, 947–957. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Zhu, F.; Zhao, Z.; Liu, C.; Luo, L.; Yin, Z. Arctigenin enhances chemosensitivity of cancer cells to cisplatin through inhibition of the STAT3 signaling pathway. J. Cell. Biochem. 2011, 112, 2837–2849. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Li, G.; Lü, C.; Xu, H.; Yin, Z. Arctigenin promotes degradation of inducible nitric oxide synthase through CHIP-associated proteasome pathway and suppresses its enzyme activity. Int. Immunopharmacol. 2012, 14, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.C.; Liang, Y.; Tian, Y.; Hu, G.R. Arctigenin induces apoptosis in colon cancer cells through ROS/p38MAPK pathway. J. BUON 2016, 21, 87–94. [Google Scholar] [PubMed]
- Madka, V.; Rao, C.V. Anti-inflammatory phytochemicals for chemoprevention of colon cancer. Curr. Cancer Drug Target. 2013, 13, 542–557. [Google Scholar] [CrossRef]
- Debrabander, J.; Shay, J.W.; Wang, W. Therapeutics Targeting Truncated Adenomatous Polyposis Coli (APC) Proteins. U.S. Patent WO 2015038644, 19 March 2015. [Google Scholar]
- Lee, E.; Jin, D.; Lee, B.B.; Kim, Y.; Han, J.; Shim, Y.M.; Kim, D.H. Negative effect of cyclin D1 overexpression on recurrence-free survival in stage II-IIIA lung adenocarcinoma and its expression modulation by vorinostat in vitro. BMC Cancer 2015, 15, 982. [Google Scholar] [CrossRef] [PubMed]
- Abbas, T.; Dutta, A. P21 in cancer: intricate networks and multiple activities. Nat. Rev. Cancer 2009, 9, 400–414. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Li, Q.; Wang, W.; Wang, Y.; Fu, X.; Wang, W.; Fan, L.; Yan, W. Apoptosis-related protein-1 acts as a tumor suppressor in cholangiocarcinoma cells by inducing cell cycle arrest via downregulation of cyclin-dependent kinase subunits. Oncol. Rep. 2016, 35, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; An, Y.; Lu, X.; Zhong, H.; Zhu, Y.; Wu, Y.; Ma, F.; Yang, J.; Liu, Y.; Zhou, Z.; et al. A Novel Naphthalimide Compound restores p53 function in Non-Small-Cell Lung Cancer by Reorganizing the Bak-Bcl-xL Complex and Triggering Transcriptional Regulation. J. Biol. Chem. 2016, 291, 4211–4225. [Google Scholar] [CrossRef] [PubMed]
- Airiau, K.; Prouzet-Mauléon, V.; Rousseau, B.; Pigneux, A.; Jeanneteau, M.; Giraudon, M.; Allou, K.; Dubus, P.; Belloc, F.; Mahon, F.X. Synergistic cooperation between ABT-263 and MEK1/2 inhibitor: Effect on apoptosis and proliferation of acute myeloid leukemia cells. Oncotarget 2016, 7, 845–859. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, S.; Wang, L.; Wang, R.; Chen, S.; Pan, B.; Sun, Y.; Chen, H. Prognostic value of Bcl-2 expression in patients with non-small-cell lung cancer: A meta-analysis and systemic review. Onco Targets Ther. 2015, 8, 3361–3369. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Investig. 2009, 119, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Leonard, G.D.; Brenner, B.; Kemeny, N.E. Neoadjuvant chemotherapy before liver resection for patients with unresectable liver metastases from colorectal carcinoma. J. Clin. Oncol. 2005, 23, 2038–2048. [Google Scholar] [CrossRef] [PubMed]
- Vervoort, S.J.; Lourenço, A.R.; van Boxtel, R.; Coffer, R.J. SOX4 mediates TGF-β-induced expression of mesenchymal markers during mammary cell epithelial to mesenchymal transition. PLoS ONE 2013, 8, e53238. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Zhu, Q.; Zhang, Y.; Song, W.; Wilson, M.J.; Liu, P. Dual-Functions of miR-373 and miR-520c by Differently Regulating the Activities of MMP-2 and MMP-9. J. Cell. Physiol. 2015, 230, 1862–1870. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Lan, F.; Wang, X.; Yu, Y.; Ouyang, X.; Zheng, F.; Han, J.; Lin, Y.; Xie, Y.; Xie, F. IL-1β-induced activation of p38 promotes metastasis in gastric adenocarcinoma via upregulation of AP-1/c-FOS, MMP-2 and MMP-9. Mol. Cancer 2014, 13, 18. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.H.; Kee, J.Y.; Park, J.; Kim, H.L.; Jeong, M.Y.; Kim, D.S.; Jeon, Y.D.; Jung, Y.; Youn, D.H.; Kang, J.W.; et al. Arctigenin inhibits adipogenesis by inducing AMPK activation and reduces weight gain in high-fat diet-induced obese mice. J. Cell. Biochem. 2016, 117, 2067–2077. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Cao, S.; Zhou, H.; Hua, L.; Zhang, S.; Cao, J. Mechanism of Arctigenin-Induced Specific Cytotoxicity against Human Hepatocellular Carcinoma Cell Lines: Hep G2 and SMMC7721. PLoS ONE 2015, 10, e0125727. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Qi, C.; Sun, X.; Ma, X.; Zhang, H.; Hu, L.; Yuan, J.; Yu, Q. Arctigenin preferentially induces tumor cell death under glucose deprivation by inhibiting cellular energy metabolism. Biochem. Pharmacol. 2012, 84, 468–476. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.J.; Kuo, P.L.; Hsu, Y.C.; Huang, Y.F.; Tsai, E.M.; Hsu, Y.L. Arctigenin, a dietary phytoestrogen, induces apoptosis of estrogen receptor-negative breast cancer cells through the ROS/p38 MAPK pathway and epigenetic regulation. Free Radic. Biol. Med. 2014, 67, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.H.; Lee, H.J.; Kang, K.; Jho, E.H.; Kim, C.Y.; Baturen, D.; Tunsag, J.; Nho, C.W. Lignans inhibit cell growth via regulation of Wnt/β-catenin signaling. Food Chem. Toxicol. 2010, 48, 2247–2252. [Google Scholar] [CrossRef] [PubMed]
- Niknejad, H.; Yazdanpanah, G.; Ahmadiani, A. Induction of apoptosis, stimulation of cell-cycle arrest and inhibition of angiogenesis make human amnion-derived cells promising sources for cell therapy of cancer. Cell Tissue Res. 2016, 363, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Gérard, C.; Goldbeter, A. Temporal self-organization of the cyclin/CDK network driving the mammalian cell cycle. Proc. Natl. Acad. Sci. USA 2009, 106, 21643–21648. [Google Scholar] [CrossRef] [PubMed]
- Ekberg, J.; Holm, C.; Jalili, S.; Richter, J.; Anagnostaki, L.; Landberg, G.; Persson, J.L. Expression of cyclin A1 and cell cycle proteins in hematopoietic cells and acute myeloid leukemia and links to patient outcome. Eur. J. Haematol. 2005, 75, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.H.; Cheng, Y.P.; Chang, I.L.; Chen, H.Y.; Wu, C.C.; Hsieh, C.P. Dodecyl gallate induces apoptosis by upregulating the caspase-dependent apoptotic pathway and inhibiting the expression of anti-apoptotic Bcl-2 family proteins in human osteosarcoma cells. Mol. Med. Rep. 2016, 13, 1495–1500. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.H.; Hu, J.R.; Wang, L.Y.; Hu, Y.J.; Tan, F.Q.; Zhou, H.; Shao, J.Z.; Yang, W.X. The apoptotic function analysis of p53, Apaf1, Caspase3 and Caspase7 during the spermatogenesis of the Chinese fire-bellied newt Cynops orientalis. PLoS ONE 2012, 7, e39920. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Tao, B.; Liu, G.; Chen, G.; Zhu, Q.; Yu, Y.; Yu, Y.; Xiong, H. Thromboxane A2 Receptor Inhibition Suppresses Multiple Myeloma Cell Proliferation by Inducing P38/JNK MAP Kinase Mediated-G2/M Progression Delay and Cell Apoptosis. J. Biol. Chem. 2016, 291, 4779–4792. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.H.; Sui, X.M.; Sui, Y.N.; Zhu, Q.W.; Yan, K.; Wang, L.S.; Wang, F.; Zhou, J.H. BRD4 induces cell migration and invasion in HCC cells through MMP-2 and MMP-9 activation mediated by the Sonic hedgehog signaling pathway. Oncol. Lett. 2015, 10, 2227–2232. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Shen, D.; Li, X.; Shan, X.; Wang, X.; Yan, Q.; Liu, J. Ginsenoside Rg3 inhibits epithelial-mesenchymal transition (EMT) and invasion of lung cancer by down-regulating FUT4. Oncotarget 2016, 7, 1619–1632. [Google Scholar] [PubMed]
- Sample Availability: Samples of the compounds are not available from the authors.
| Gene | Forward (5′-3′) | Reverse (5′-3′) |
|---|---|---|
| MMP-2 | CCCCATGAAGCCTTGTTTACC | TTGTAGGAGGTGCCCTGGAA |
| MMP-9 | AGACCAAGGGTACAGCCTGTTC | GGCACGCTGGAATGATCTAAG |
| E-cadherin | AATGGCGGCAATGCAATCCCAAGA | TGCCACAGACCGATTGTGGAGATA |
| N-cadherin | TGGAGAACCCCATTGACATT | TGATCCCTCAGGAACTGTCC |
| Snail | TCCAAACCCACTCGGATGTGAAGA | TTGGTGCTTGTGGAGCAAGGACAT |
| β-catenin | ACTGCTGGGACTCTG | TGATGGCGTAGAACAG |
| Vimentin | CGGAAAGTGGAATCCTTGCA | CACATCGATCTGGACATGCTG |
| GAPDH | GACATGCCGCCTGGAGAAAC | AGCCCAGGATGCCCTTTAGT |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, Y.-H.; Kee, J.-Y.; Kim, D.-S.; Mun, J.-g.; Jeong, M.-Y.; Park, S.-H.; Choi, B.-M.; Park, S.-J.; Kim, H.-J.; Um, J.-Y.; et al. Arctigenin Inhibits Lung Metastasis of Colorectal Cancer by Regulating Cell Viability and Metastatic Phenotypes. Molecules 2016, 21, 1135. https://doi.org/10.3390/molecules21091135
Han Y-H, Kee J-Y, Kim D-S, Mun J-g, Jeong M-Y, Park S-H, Choi B-M, Park S-J, Kim H-J, Um J-Y, et al. Arctigenin Inhibits Lung Metastasis of Colorectal Cancer by Regulating Cell Viability and Metastatic Phenotypes. Molecules. 2016; 21(9):1135. https://doi.org/10.3390/molecules21091135
Chicago/Turabian StyleHan, Yo-Han, Ji-Ye Kee, Dae-Seung Kim, Jeong-geon Mun, Mi-Young Jeong, Sang-Hyun Park, Byung-Min Choi, Sung-Joo Park, Hyun-Jung Kim, Jae-Young Um, and et al. 2016. "Arctigenin Inhibits Lung Metastasis of Colorectal Cancer by Regulating Cell Viability and Metastatic Phenotypes" Molecules 21, no. 9: 1135. https://doi.org/10.3390/molecules21091135







