Overview of Antagonists Used for Determining the Mechanisms of Action Employed by Potential Vasodilators with Their Suggested Signaling Pathways
Abstract
:1. Introduction
2. Types of Blood Vessels
3. Signaling Mechanisms Involved in Vasodilation Studies
4. Endothelium-Derived Relaxing Factors (EDRFs)
4.1. Nitric Oxide (NO)
4.2. Prostacyclin (PGI2)
5. Enzyme-Linked NO pathway
Soluble Guanylyl Cyclase (sGC)
6. G-Protein-Coupled Receptors
6.1. β-Adrenergic Receptors
6.2. Muscarinic Receptors (M3)
7. Vasoconstriction-Dominated Receptors
8. Channel-Linked Receptors
8.1. Potassium Channels
8.1.1. Calcium-Dependent Potassium Channels (Kca)
8.1.2. ATP-Sensitive Potassium Channels (KATP)
8.1.3. Voltage-Dependent Potassium Channels (Kv) and Inwardly Rectifier Potassium Channels (Kir)
8.2. Calcium Channels
8.2.1. Voltage-Operated Calcium Channels (VOCC)
8.2.2. Store-Operated Calcium Channels (SOCC)
9. List of Antagonists and Its Receptor
10. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Yang, Y.; Zhang, Z.; Li, S.; Ye, X.; Li, X.; He, K. Synergy effects of herb extracts: pharmacokinetics and pharmacodynamic basis. Fitoterapia 2014, 92, 133–147. [Google Scholar] [CrossRef] [PubMed]
- Lenfant, C.; Chobanian, A.V.; Jones, D.W.; Roccella, E.J. Seventh report of the Joint National Committee on the Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7): resetting the hypertension sails. Hypertension 2003, 41, 1178–1179. [Google Scholar] [CrossRef] [PubMed]
- Whalen, K. Lippincott Illustrated Reviews: Pharmacology, 6th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2014; pp. 225–240. [Google Scholar]
- Yildiz, O.; Gul, H.; Seyrek, M. Pharmacology of Arterial Grafts for Coronary Artery Bypass Surgery; Intech Open Access Publisher: Rijeka, Croatia, 2013. [Google Scholar]
- Rameshrad, M.; Babaei, H.; Azarmi, Y.; Fouladia, D.F. Rat aorta as a pharmacological tool for in vitro and in vivo studies. Life Sci. 2016, 145, 190–204. [Google Scholar] [CrossRef] [PubMed]
- Ameer, O.Z.; Salman, I.M.; Siddiqui, M.J.; Yam, M.F.; Sriramaneni, R.N.; Mohamed, A.J.; Sadikun, A.; Ismail, Z.; Shah, A.M.; Asmawi, M.Z. Pharmacological mechanisms underlying the vascular activities of Loranthus ferrugineus Roxb. in rat thoracic aorta. J. Ethnopharmacol. 2010, 127, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Li, R.W.; Yang, C.; Shan, L.; Zhang, Z.; Wang, Y.; Kwan, Y.W.; Lee, S.M.; Hoi, M.P.; Chan, S.W.; Cheung, A.C.; et al. Relaxation effect of a novel Danshensu/tetramethylpyrazine derivative on rat mesenteric arteries. Eur. J. Pharmacol. 2015, 761, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Tsao, C.M.; Chen, S.J.; Tsou, M.Y.; Wu, C.C. Effect of propofol on vascular reactivity in thoracic aortas from rats with endotoxemia. J. Chin. Med. Assoc. 2012, 75, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Davis, B.; Rahman, A.; Arner, A. AMP-activated kinase relaxes agonist induced contractions in the mouse aorta via effects on PKC signaling and inhibits NO-induced relaxation. Eur. J. Pharmacol. 2012, 695, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Csanyi, G.; Gajda, M.; Franczyk-Zarow, M.; Kostogrys, R.; Gwozdz, P.; Mateuszuk, L.; Sternak, M.; Wojcik, L.; Zalewska, T.; Walski, M.; et al. Functional alterations in endothelial NO, PGI(2) and EDHF pathways in aorta in ApoE/LDLR-/- mice. Prostaglandins Other Lipid Mediat. 2012, 98, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.Y.; Lee, Y.J.; Rhyu, M.R. Black cohosh (Cimicifuga racemosa) relaxes the isolated rat thoracic aorta through endothelium-dependent and -independent mechanisms. J. Ethnopharmacol. 2011, 138, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Hernandez, J.M.; Ramirez-Lee, M.A.; Rosas-Hernandez, H.; Salazar-Garcia, S.; Maldonado-Ortega, D.A.; Gonzalez, F.J.; Gonzalez, C. Single-walled carbon nanotubes (SWCNTs) induce vasodilation in isolated rat aortic rings. Toxicol. In Vitro 2015, 29, 657–662. [Google Scholar] [CrossRef] [PubMed]
- Capettini, L.S.; Cortes, S.F.; Lemos, V.S. Relative contribution of eNOS and nNOS to endothelium-dependent vasodilation in the mouse aorta. Eur. J. Pharmacol. 2010, 643, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Kim, H.I.; Park, S.H.; Lee, M.J.; Jun, J.Y.; Kim, H.L.; Chung, J.H.; Yeum, C.H. Endothelium-dependent vasodilation by ferulic acid in aorta from chronic renal hypertensive rats. Kidney Res. Clin. Pract. 2012, 31, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Ziberna, L.; Lunder, M.; Tramer, F.; Drevensek, G.; Passamonti, S. The endothelial plasma membrane transporter bilitranslocase mediates rat aortic vasodilation induced by anthocyanins. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Koon, C.M.; Fong, S.; Wat, E.; Wang, Y.P.; Wing-Shing Cheung, D.; Bik-San Lau, C.; Leung, P.C.; Sun, H.D.; Zhao, Q.S.; Fung, K.P. Mechanisms of the dilator action of the Erigerontis Herba on rat aorta. J. Ethnopharmacol. 2014, 155, 1561–1567. [Google Scholar] [CrossRef] [PubMed]
- Bertin, R.; Chen, Z.; Martinez-Vazquez, M.; Garcia-Argaez, A.; Froldi, G. Vasodilation and radical-scavenging activity of imperatorin and selected coumarinic and flavonoid compounds from genus Casimiroa. Phytomedicine 2014, 21, 586–594. [Google Scholar] [CrossRef] [PubMed]
- Lindsey, S.H.; Liu, L.; Chappell, M.C. Vasodilation by GPER in mesenteric arteries involves both endothelial nitric oxide and smooth muscle cAMP signaling. Steroids 2014, 81, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Shou, Q.; Pan, Y.; Xu, X.; Xu, J.; Wang, D.; Ling, Y.; Chen, M. Salvianolic acid B possesses vasodilation potential through NO and its related signals in rabbit thoracic aortic rings. Eur. J. Pharmacol. 2012, 697, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, B.; Shinn, C.; Sellke, F.W.; Clements, R.T. Rottlerin-induced BKCa channel activation impairs specific contractile responses and promotes vasodilation. Ann. Thorac. Surg. 2015, 99, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Yamawaki, H.; Tsubaki, N.; Mukohda, M.; Okada, M.; Hara, Y. Omentin, a novel adipokine, induces vasodilation in rat isolated blood vessels. Biochem. Biophys. Res. Commun. 2010, 393, 668–672. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Wang, X.; Dai, Y.; Kong, L.; Wang, F.; Xu, H.; Lu, D.; Song, J.; Hou, Z. (+/−)-Praeruptorin A enantiomers exert distinct relaxant effects on isolated rat aorta rings dependent on endothelium and nitric oxide synthesis. Chem. Biol. Interact. 2010, 186, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Celotto, A.C.; Restini, C.B.; Capellini, V.K.; Bendhack, L.M.; Evora, P.R. Acidosis induces relaxation mediated by nitric oxide and potassium channels in rat thoracic aorta. Eur. J. Pharmacol. 2011, 656, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Mori, A.; Suzuki, S.; Sakamoto, K.; Nakahara, T.; Ishii, K. Vasodilation of retinal arterioles induced by activation of BKCa channels is attenuated in diabetic rats. Eur. J. Pharmacol. 2011, 669, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Tsounapi, P.; Saito, M.; Kitatani, K.; Dimitriadis, F.; Ohmasa, F.; Shimizu, S.; Kinoshita, Y.; Takenaka, A.; Satoh, K. Fasudil improves the endothelial dysfunction in the aorta of spontaneously hypertensive rats. Eur. J. Pharmacol. 2012, 691, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Silva, B.R.; Pernomian, L.; Grando, M.D.; Amaral, J.H.; Tanus-Santos, J.E.; Bendhack, L.M. Hydrogen peroxide modulates phenylephrine-induced contractile response in renal hypertensive rat aorta. Eur. J. Pharmacol. 2013, 721, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Silva, B.R.; Pernomian, L.; Grando, M.D.; Bendhack, L.M. Phenylephrine activates eNOS Ser 1177 phosphorylation and nitric oxide signaling in renal hypertensive rat aorta. Eur. J. Pharmacol. 2014, 738, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Nakabayashi, S.; Nagaoka, T.; Tani, T.; Sogawa, K.; Hein, T.W.; Kuo, L.; Yoshida, A. Retinal arteriolar responses to acute severe elevation in systemic blood pressure in cats: Role of endothelium-derived factors. Exp. Eye Res. 2012, 103, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Guven, G.; Seyrek, M.; Vural, I.M.; Cehreli, Z.C.; Yildiz, O. Vasodilatory effect of hydroxyethyl methacrylate and triethylene glycol dimethacrylate in rat aorta through calcium antagonistic action. J. Endod. 2011, 37, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Hong da, H.; Son, Y.K.; Na, S.H.; Jung, W.K.; Bae, Y.M.; Seo, E.Y.; Kim, S.J.; Choi, I.W.; Park, W.S. Cilostazol induces vasodilation through the activation of Ca2+-activated K+ channels in aortic smooth muscle. Vascul. Pharmacol. 2015, 70, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Kamkaew, N.; Scholfield, C.N.; Ingkaninan, K.; Maneesai, P.; Parkington, H.C.; Tare, M.; Chootip, K. Bacopa monnieri and its constituents is hypotensive in anaesthetized rats and vasodilator in various artery types. J. Ethnopharmacol. 2011, 137, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Ng, C.F.; Koon, C.M.; Cheung, D.W.; Lam, M.Y.; Leung, P.C.; Lau, C.B.; Fung, K.P. The anti-hypertensive effect of Danshen (Salvia miltiorrhiza) and Gegen (Pueraria lobata) formula in rats and its underlying mechanisms of vasorelaxation. J. Ethnopharmacol. 2011, 137, 1366–1372. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.N.; Wen, J.F.; Wang, T.T.; Kang, D.G.; Lee, H.S.; Cho, K.W. Vasodilatory effects of ethanol extract of Radix Paeoniae Rubra and its mechanism of action in the rat aorta. J. Ethnopharmacol. 2012, 142, 188–193. [Google Scholar] [CrossRef] [PubMed]
- Qu, Z.; Zhang, J.; Gao, W.; Chen, H.; Guo, H.; Wang, T.; Li, H.; Liu, C. Vasorelaxant effects of Cerebralcare Granule® are mediated by NO/cGMP pathway, potassium channel opening and calcium channel blockade in isolated rat thoracic aorta. J. Ethnopharmacol. 2014, 155, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Koon, C.M.; Chan, J.Y.; Lau, K.M.; Kwan, Y.W.; Fung, K.P. Involvements of calcium channel and potassium channel in Danshen and Gegen decoction induced vasodilation in porcine coronary LAD artery. Phytomedicine 2012, 19, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Li, S.; Wang, P.; Yan, S.; Hu, L.; Pan, X.; Yang, C.; Leung, G.P. Endothelium-dependent and -independent relaxation of rat aorta induced by extract of Schizophyllum commune. Phytomedicine 2014, 21, 1230–1236. [Google Scholar] [CrossRef] [PubMed]
- Khanna, V.; Jain, M.; Barthwal, M.K.; Kalita, D.; Boruah, J.J.; Das, S.P.; Islam, N.S.; Ramasarma, T.; Dikshit, M. Vasomodulatory effect of novel peroxovanadate compounds on rat aorta: Role of rho kinase and nitric oxide/cGMP pathway. Pharmacol. Res. 2011, 64, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Cameron, M.S.; Nobata, S.; Takei, Y.; Donald, J.A. Vasodilatory effects of homologous adrenomedullin 2 and adrenomedullin 5 on isolated blood vessels of two species of eel. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2015, 179, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Tom, E.N.; Girard, C.; Dimo, T.; Mbafor, J.T.; Berthelot, A.; Demougeot, C. Vasorelaxant effects of extracts of the stem bark of Terminalia superba Engler & Diels (Combretaceae). J. Ethnopharmacol. 2010, 127, 335–340. [Google Scholar] [PubMed]
- Neubauer, R.; Wolkart, G.; Opelt, M.; Schwarzenegger, C.; Hofinger, M.; Neubauer, A.; Kollau, A.; Schmidt, K.; Schrammel, A.; Mayer, B. Aldehyde dehydrogenase-independent bioactivation of nitroglycerin in porcine and bovine blood vessels. Biochem. Pharmacol. 2015, 93, 440–448. [Google Scholar] [CrossRef] [PubMed]
- Ok, S.H.; Kwon, S.C.; Yeol Han, J.; Yu, J.; Shin, I.W.; Lee, H.K.; Chung, Y.K.; Choi, M.J.; Sohn, J.T. Mepivacaine-induced contraction involves increased calcium sensitization mediated via Rho kinase and protein kinase C in endothelium-denuded rat aorta. Eur. J. Pharmacol. 2014, 723, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, K.; Matsumoto, T.; Kamata, K.; Kobayashi, T. Angiotensin II type 2 receptor-dependent increase in nitric oxide synthase activity in the endothelium of db/db mice is mediated via a MEK pathway. Pharmacol. Res. 2012, 66, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Machado, N.T.; Maciel, P.M.; Alustau, M.C.; Queiroz, T.M.; Furtado, F.F.; Assis, V.L.; Veras, R.C.; Araújo, I.G.; Athayde-Filho, P.F.; Medeiros, I.A. Nitric oxide as a target for the hypotensive and vasorelaxing effects induced by (Z)-ethyl 12-nitrooxy-octadec-9-enoate in rats. Eur. J. Pharm. Sci. 2014, 62, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Celotto, A.C.; Capellini, V.K.; Restini, C.B.; Baldo, C.F.; Bendhack, L.M.; Evora, P.R. Extracellular alkalinization induces endothelium-derived nitric oxide dependent relaxation in rat thoracic aorta. Nitric Oxide 2010, 23, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Bonaventura, D.; de Lima, R.G.; da Silva, R.S.; Bendhack, L.M. NO donors-relaxation is impaired in aorta from hypertensive rats due to a reduced involvement of K+ channels and sarcoplasmic reticulum Ca2+-ATPase. Life Sci. 2011, 89, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Senejoux, F.; Girard, C.; Aisa, H.A.; Bakri, M.; Kerram, P.; Berthelot, A.; Bevalot, F.; Demougeot, C. Vasorelaxant and hypotensive effects of a hydroalcoholic extract from the fruits of Nitraria sibirica Pall. (Nitrariaceae). J. Ethnopharmacol. 2012, 141, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Leo, C.; Joshi, A.; Hart, J.; Woodman, O. Endothelium-dependent nitroxyl-mediated relaxation is resistant to superoxide anion scavenging and preserved in diabetic rat aorta. Pharmacol. Res. 2012, 66, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Perusquia, M.; Espinoza, J.; de la Pena, A. Mifepristone (RU 486) induces vasodilation and inhibits platelet aggregation: nongenomic and genomic action to cause hemorrhage. Contraception 2011, 84, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Leal, C.M.; Pereira, S.L.; Kummerle, A.E.; Leal, D.M.; Tesch, R.; de Sant'Anna, C.M.; Fraga, C.A.; Barreiro, E.J.; Sudo, R.T.; Zapata-Sudo, G. Antihypertensive profile of 2-thienyl-3,4-methylenedioxybenzoylhydrazone is mediated by activation of the A2A adenosine receptor. Eur. J. Med. Chem. 2012, 55, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, W.; Edvinsson, L.; Xu, C.B. Apolipoprotein B of low-density lipoprotein impairs nitric oxide-mediated endothelium-dependent relaxation in rat mesenteric arteries. Eur. J. Pharmacol. 2014, 725, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, M.; Shigekuni, Y.; Obi, T.; Shiraishi, M.; Miyamoto, A.; Yamasaki, H.; Etoh, T.; Iwai, S. Nitric oxide-dependent hypotensive effects of wax gourd juice. J. Ethnopharmacol. 2011, 138, 404–407. [Google Scholar] [CrossRef] [PubMed]
- Rios, M.Y.; López-Martínez, S.; López-Vallejo, F.; Medina-Franco, J.L.; Villalobos-Molina, R.; Ibarra-Barajas, M.; Navarrete-Vazquez, G.; Hidalgo-Figueroa, S.; Hernández-Abreu, O.; Estrada-Soto, S. Vasorelaxant activity of some structurally related triterpenic acids from Phoradendron reichenbachianum (Viscaceae) mainly by NO production: Ex vivo and in silico studies. Fitoterapia 2012, 83, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Jerez, S.; Sierra, L.; Scacchi, F.; Peral de Bruno, M. Hypercholesterolemia modifies angiotensin II desensitisation and cross talk between α1-adrenoceptor and angiotensin AT(1) receptor in rabbit aorta. Eur. J. Pharmacol. 2010, 635, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Pagan, R.M.; Prieto, D.; Hernandez, M.; Correa, C.; Garcia-Sacristan, A.; Benedito, S.; Martinez, A.C. Regulation of NO-dependent acetylcholine relaxation by K+ channels and the Na+-K+ ATPase pump in porcine internal mammary artery. Eur. J. Pharmacol. 2010, 641, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Sauvaget, F.; Mallem, M.Y.; Bucas, V.; Gogny, M.; Desfontis, J.-C.; Noireaud, J. Positive influence of AT 1 receptor antagonism upon the impaired celiprolol-induced vasodilatation in aorta from spontaneously hypertensive rats. Eur. J. Pharmacol. 2010, 644, 169–175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shim, H.S.; Ok, S.H.; Lee, S.H.; Kwon, S.C.; Sohn, J.T. Protein kinases participate in the contraction in response to levobupivacaine in the rat aorta. Eur. J. Pharmacol. 2012, 677, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Novakovic, A.; Marinko, M.; Vranic, A.; Jankovic, G.; Milojevic, P.; Stojanovic, I.; Nenezic, D.; Ugresic, N.; Kanjuh, V.; Yang, Q.; et al. Mechanisms underlying the vasorelaxation of human internal mammary artery induced by (−)-epicatechin. Eur. J. Pharmacol. 2015, 762, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chen, G.P.; Li, L.; Wang, K.J.; Zhang, B.Q.; Hu, S.J. Dual effects of sodium aescinate on vascular tension in rat thoracic aorta. Microvasc. Res. 2010, 79, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Yu, Y.; Sun, X.; Wang, B. Exendin-4 directly improves endothelial dysfunction in isolated aortas from obese rats through the cAMP or AMPK–eNOS pathways. Diabetes Res. Clin. Pract. 2012, 97, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, S.M.; Ximenes, C.F.; de Batista, P.R.; Simoes, F.V.; Coser, P.H.; Sena, G.C.; Podratz, P.L.; de Souza, L.N.; Vassallo, D.V.; Graceli, J.B.; et al. Tributyltin contributes in reducing the vascular reactivity to phenylephrine in isolated aortic rings from female rats. Toxicol. Lett. 2014, 225, 378–385. [Google Scholar] [CrossRef] [PubMed]
- Bankar, G.R.; Nayak, P.G.; Bansal, P.; Paul, P.; Pai, K.S.; Singla, R.K.; Bhat, V.G. Vasorelaxant and antihypertensive effect of Cocos nucifera Linn. endocarp on isolated rat thoracic aorta and DOCA salt-induced hypertensive rats. J. Ethnopharmacol. 2011, 134, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.F.; Liu, L.M.; Liu, Y.Y.; Liu, J.; Yan, L.; Pan, C.S.; Wang, M.X.; Wang, C.S.; Fan, J.Y.; Gao, Y.S.; et al. Inhibitory effect of rhynchophylline on contraction of cerebral arterioles to endothelin 1: role of rho kinase. J. Ethnopharmacol. 2014, 155, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Tao, L.; Hu, H.S.; Shen, X.C. Endothelium-dependent vasodilatation effects of the essential oil from Fructus alpiniae zerumbet (EOFAZ) on rat thoracic aortic rings in vitro. Phytomedicine 2013, 20, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Kalea, A.Z.; Clark, K.; Schuschke, D.A.; Kristo, A.S.; Klimis-Zacas, D.J. Dietary enrichment with wild blueberries (Vaccinium angustifolium) affects the vascular reactivity in the aorta of young spontaneously hypertensive rats. J. Nutr. Biochem. 2010, 21, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.H.; Li, Q.; Wu, M.Y.; Guo, D.J.; Chen, H.L.; Chen, S.L.; Seto, S.W.; Au, A.L.; Poon, C.C.; Leung, G.P.; et al. Formononetin, an isoflavone, relaxes rat isolated aorta through endothelium-dependent and endothelium-independent pathways. J. Nutr. Biochem. 2010, 21, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.H.; Leung, G.P.; Leung, S.W.; Vanhoutte, P.M.; Man, R.Y. Genistein enhances relaxation of the spontaneously hypertensive rat aorta by transactivation of epidermal growth factor receptor following binding to membrane estrogen receptors-α and activation of a G protein-coupled, endothelial nitric oxide synthase-dependent pathway. Pharmacol. Res. 2011, 63, 181–189. [Google Scholar] [PubMed]
- Zhang, Y.; Chen, Q.; Sun, Z.; Han, J.; Wang, L.; Zheng, L. Impaired capsaicin-induced relaxation in diabetic mesenteric arteries. J. Diabetes Complications 2015, 29, 747–754. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.P.; Lu, J.F.; Zhang, G.L.; Li, X.Y.; Peng, H.Y.; Lu, Y.; Zhao, L.; Ye, Z.G.; Bruce, I.C.; Xia, Q.; et al. Endothelium-dependent and -independent vasorelaxant actions and mechanisms induced by total flavonoids of Elsholtzia splendens in rat aortas. Environ. Toxicol. Pharmacol. 2014, 38, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.J.; Bao, J.X.; Xu, J.W.; Murad, F.; Bian, K. Vascular dilation by paeonol—A mechanism study. Vascul. Pharmacol. 2010, 53, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Perez, T.; Lopez, R.M.; Lopez, P.; Castillo, C.; Castillo, E.F. Lack of heterologous receptor desensitization induced by angiotensin II type 1 receptor activation in isolated normal rat thoracic aorta. Vascul. Pharmacol. 2011, 54, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.J.; Wang, L.; Zhou, G.Y.; Yu, X.L.; Zhang, Y.H.; Hu, N.; Li, Q.Q.; Chen, C.; Qing, C.; Liu, Y.T.; et al. Scutellarin attenuates endothelium-dependent aasodilation impairment induced by hypoxia reoxygenation, through regulating the PKG signaling pathway in rat coronary artery. Chin. J. Nat. Med. 2015, 13, 264–273. [Google Scholar] [CrossRef]
- Araujo, A.V.; Ferezin, C.Z.; Rodrigues, G.J.; Lunardi, C.N.; Vercesi, J.A.; Grando, M.D.; Bonaventura, D.; Bendhack, L.M. Prostacyclin, not only nitric oxide, is a mediator of the vasorelaxation induced by acetylcholine in aortas from rats submitted to cecal ligation and perforation (CLP). Vascul. Pharmacol. 2011, 54, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Shen, M.; Zhao, L.; Wu, R.X.; Yue, S.Q.; Pei, J.M. The vasorelaxing effect of resveratrol on abdominal aorta from rats and its underlying mechanisms. Vascul. Pharmacol. 2013, 58, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Choi, H.C. Acetylcholine-induced AMP-activated protein kinase activation attenuates vasoconstriction through an LKB1-dependent mechanism in rat aorta. Vascul. Pharmacol. 2013, 59, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Perez-Aso, M.; Flacco, N.; Carpena, N.; Montesinos, M.C.; D'Ocon, P.; Ivorra, M.D. β-Adrenoceptors differentially regulate vascular tone and angiogenesis of rat aorta via ERK1/2 and p38. Vascul. Pharmacol. 2014, 61, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, I.; Damjanovic, M.; Dominguez, A.P.; Haas, E. Inhibition of activated ERK1/2 and JNKs improves vascular function in mouse aortae in the absence of nitric oxide. Eur. J. Pharmacol. 2011, 658, 22–27. [Google Scholar] [CrossRef] [PubMed]
- El-Gowelli, H.M.; El-Gowilly, S.M.; Elsalakawy, L.K.; El-Mas, M.M. Nitric oxide synthase/K+ channel cascade triggers the adenosine A2B receptor-sensitive renal vasodilation in female rats. Eur. J. Pharmacol. 2013, 702, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Hou, N.; Han, F.; Guo, Y.; Hui, Z.; Du, G.; Zhang, Y. Effect of high free fatty acids on the anti-contractile response of perivascular adipose tissue in rat aorta. J. Mol. Cell. Cardiol. 2013, 63, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Gallo, L.C.; Davel, A.P.; Xavier, F.E.; Rossoni, L.V. Time-dependent increases in ouabain-sensitive Na+, K+-ATPase activity in aortas from diabetic rats: The role of prostanoids and protein kinase C. Life Sci. 2010, 87, 302–308. [Google Scholar] [CrossRef] [PubMed]
- Horta, C.; Rezende, B.; Oliveira-Mendes, B.; Carmo, A.; Capettini, L.; Silva, J.; Gomes, M.; Chávez-Olórtegui, C.; Bravo, C.; Lemos, V. ADP is a vasodilator component from Lasiodora sp. mygalomorph spider venom. Toxicon 2013, 72, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, K.; Kobayashi, T.; Takenouchi, Y.; Matsumoto, T.; Kamata, K. Angiotensin II causes endothelial dysfunction via the GRK2/Akt/eNOS pathway in aortas from a murine type 2 diabetic model. Pharmacol. Res. 2011, 64, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.N.; Wen, J.F.; Li, X.; Kang, D.G.; Lee, H.S.; Cho, K.W. The mechanism of vasorelaxation induced by ethanol extract of Sophora flavescens in rat aorta. J. Ethnopharmacol. 2011, 137, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Gaete, P.S.; Lillo, M.A.; Ardiles, N.M.; Perez, F.R.; Figueroa, X.F. Ca2+-activated K+ channels of small and intermediate conductance control eNOS activation through NAD(P)H oxidase. Free Radic. Biol. Med. 2012, 52, 860–870. [Google Scholar] [CrossRef] [PubMed]
- Silveira, E.A.; Siman, F.D.; de Oliveira Faria, T.; Vescovi, M.V.; Furieri, L.B.; Lizardo, J.H.; Stefanon, I.; Padilha, A.S.; Vassallo, D.V. Low-dose chronic lead exposure increases systolic arterial pressure and vascular reactivity of rat aortas. Free Radic. Biol. Med. 2014, 67, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Dongmo, A.B.; Nkeng-Efouet, P.A.; Devkota, K.P.; Wegener, J.W.; Sewald, N.; Wagner, H.; Vierling, W. Tetra-acetylajugasterone a new constituent of Vitex cienkowskii with vasorelaxant activity. Phytomedicine 2014, 21, 787–792. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Sun, C.; Tilley, S.L.; Mustafa, S.J. Mechanisms underlying uridine adenosine tetraphosphate-induced vascular contraction in mouse aorta: Role of thromboxane and purinergic receptors. Vascul. Pharmacol. 2015, 73, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Ling, W.C.; Lau, Y.S.; Murugan, D.D.; Vanhoutte, P.M.; Mustafa, M.R. Sodium nitrite causes relaxation of the isolated rat aorta: By stimulating both endothelial NO synthase and activating soluble guanylyl cyclase in vascular smooth muscle. Vascul. Pharmacol. 2015, 74, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Al-Nakkash, L.; Martin, J.B.; Petty, D.; Lynch, S.M.; Hamrick, C.; Lucy, D.; Robinson, J.; Peterson, A.; Rubin, L.J.; Broderick, T.L. Dietary genistein induces sex-dependent effects on murine body weight, serum profiles, and vascular function of thoracic aortae. Gend. Med. 2012, 9, 295–308. [Google Scholar] [CrossRef] [PubMed]
- Sélley, E.; Molnár, G.A.; Kun, S.; Szijártó, I.A.; Laczy, B.; Kovács, T.; Fülöp, F.; Wittmann, I. Complex vasoactivity of liraglutide. Contribution of three gasotransmitters. Artery Res. 2015, 11, 1–9. [Google Scholar] [CrossRef]
- Choi, S.; Jung, W.S.; Cho, N.S.; Ryu, K.H.; Jun, J.Y.; Shin, B.C.; Chung, J.H.; Yeum, C.H. Mechanisms of phytoestrogen biochanin A-induced vasorelaxation in renovascular hypertensive rats. Kidney Res. Clin. Pract. 2014, 33, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Ntchapda, F.; Talla, E.; Sakava, P.; Tanzi, F.; Fohouo, F.-N.T.; Tanyi, J.M.; Dimo, T. Nitric oxide-dependent vasodilation and Ca 2+ signalling induced by erythrodiol in rat aorta. Asian Pac. J. Trop. Dis. 2015, 5, S214–S223. [Google Scholar] [CrossRef]
- Nsuadi Manga, F.; El Khattabi, C.; Fontaine, J.; Berkenboom, G.; Duez, P.; Noyon, C.; van Antwerpen, P.; Lami Nzunzu, J.; Pochet, S. Vasorelaxant and antihypertensive effects of methanolic extracts from Hymenocardia acida Tul. J. Ethnopharmacol. 2013, 146, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Paredes-Carbajal, M.C.; Monsalvo, I.; Hernandez-Diaz, C.; Regla, I.; Demare, P.; Mascher, D. Effects of ranolazine on vasomotor responses of rat aortic rings. Arch. Med. Res. 2013, 44, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Kim, H.Y.; Cui, H.Z.; Cho, K.W.; Kang, D.G.; Lee, H.S. Water extract of Zanthoxylum piperitum induces vascular relaxation via endothelium-dependent NO-cGMP signaling. J. Ethnopharmacol. 2010, 129, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.N.; Wen, J.F.; Kim, H.Y.; Kang, D.G.; Lee, H.S.; Cho, K.W. Vascular relaxation by ethanol extract of Xanthoceras sorbifolia via Akt- and SOCE-eNOS-cGMP pathways. J. Ethnopharmacol. 2010, 132, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Oh, H.; Li, X.; Cho, K.W.; Kang, D.G.; Lee, H.S. Ethanol extract of seeds of Oenothera odorata induces vasorelaxation via endothelium-dependent NO-cGMP signaling through activation of Akt-eNOS-sGC pathway. J. Ethnopharmacol. 2011, 133, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Nsuadi Manga, F.; El Khattabi, C.; Fontaine, J.; Berkenboom, G.; Duez, P.; Lami Nzunzu, J.; Pochet, S. Vascular effects and antioxidant activity of two Combretum species from Democratic Republic of Congo. J. Ethnopharmacol. 2012, 142, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Bernardes, M.J.; de Carvalho, F.S.; Lima Silveira, L.; de Paula, J.R.; Bara, M.T.; Garrote, C.F.; Pedrino, G.R.; Rocha, M.L. Hypotensive effect of Aspidosperma subincanum Mart. in rats and its mechanism of vasorelaxation in isolated arteries. J. Ethnopharmacol. 2013, 145, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Senejoux, F.; Demougeot, C.; Cuciureanu, M.; Miron, A.; Cuciureanu, R.; Berthelot, A.; Girard-Thernier, C. Vasorelaxant effects and mechanisms of action of Heracleum sphondylium L. (Apiaceae) in rat thoracic aorta. J. Ethnopharmacol. 2013, 147, 536–539. [Google Scholar] [CrossRef] [PubMed]
- Cuinas, A.; Elies, J.; Orallo, F.; Campos-Toimil, M. Cyclic AMP relaxation of rat aortic smooth muscle is mediated in part by decrease of depletion of intracellular Ca2+ stores and inhibition of capacitative calcium entry. Vascul. Pharmacol. 2013, 58, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Morales, V.; Cuinas, A.; Elies, J.; Campos-Toimil, M. PKA and Epac activation mediates cAMP-induced vasorelaxation by increasing endothelial NO production. Vascul. Pharmacol. 2014, 60, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Ng, E.S.; Kwan, Y.W.; Lau, C.B.; Cheung, D.W.; Koon, J.C.; Zhang, Z.; Zuo, Z.; Leung, P.C.; Fung, K.P.; et al. Cerebral vasodilator properties of Danshen and Gegen: A study of their combined efficacy and mechanisms of actions. Phytomedicine 2014, 21, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Ng, H.K.; Poh, T.F.; Lam, S.K.; Hoe, S.Z. Potassium channel openers and prostacyclin play a crucial role in mediating the vasorelaxant activity of Gynura procumbens. BMC Complement. Altern. Med. 2013, 13, 188. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-Q.; Liu, Y.-Y.; Li, Y.-W.; Li, L.; Cui, Z.-Q. Effects of total alkaloids in Buxus microphylla leaves on aorta smooth muscle of rats and their mechanisms. Chin. Herbal Med. 2012, 4, 136–141. [Google Scholar]
- Potje, S.R.; Munhoz, F.C.; Perassa, L.A.; Graton, M.E.; Pereira, A.A.; Nakamune, A.C.; da Silva, R.S.; Bendhack, L.M.; Sumida, D.H.; Antoniali, C. Mechanisms underlying the hypotensive and vasodilator effects of Ru(terpy)(bdq)NO]3+, a nitric oxide donor, differ between normotensive and spontaneously hypertensive rats. Eur. J. Pharmacol. 2014, 741, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Tikoo, K.; Patel, G.; Kumar, S.; Karpe, P.A.; Sanghavi, M.; Malek, V.; Srinivasan, K. Tissue specific up regulation of ACE2 in rabbit model of atherosclerosis by atorvastatin: Role of epigenetic histone modifications. Biochem. Pharmacol. 2015, 93, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhang, Y.; Meng, Z. The vasodilator mechanisms of sodium metabisulfite on precontracted isolated aortic rings in rats: signal transduction pathways and ion channels. Food Chem. Toxicol. 2012, 50, 3114–3119. [Google Scholar] [CrossRef] [PubMed]
- Senejoux, F.; Girard, C.; Kerram, P.; Aisa, H.A.; Berthelot, A.; Bevalot, F.; Demougeot, C. Mechanisms of vasorelaxation induced by Ziziphora clinopodioides Lam. (Lamiaceae) extract in rat thoracic aorta. J. Ethnopharmacol. 2010, 132, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, F.S.; Silva, A.C.; Martins, I.R.; Correia, A.C.; Basilio, I.J.; Agra, M.F.; Bhattacharyya, J.; Silva, B.A. Vasorelaxant action of the total alkaloid fraction obtained from Solanum paludosum Moric. (Solanaceae) involves NO/cGMP/PKG pathway and potassium channels. J. Ethnopharmacol. 2012, 141, 895–900. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Choi, Y.W.; Yun, J.W.; Bae, J.U.; Seo, K.W.; Lee, S.J.; Park, S.Y.; Kim, C.D. Gomisin J from Schisandra chinensis induces vascular relaxation via activation of endothelial nitric oxide synthase. Vascul. Pharmacol. 2012, 57, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Prieto, C.F.; Pulido-Olmo, H.; Ruiz-Hurtado, G.; Gil-Ortega, M.; Aranguez, I.; Rubio, M.A.; Ruiz-Gayo, M.; Somoza, B.; Fernandez-Alfonso, M.S. Mild caloric restriction reduces blood pressure and activates endothelial AMPK-PI3K-Akt-eNOS pathway in obese Zucker rats. Vascul. Pharmacol. 2015, 65–66, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, S.; Kannan, K.; Pule Addison, M.; Darzi, S.A.; Singh, V.; Singh, T.U.; Thangamalai, R.; Dash, J.R.; Parida, S.; Debroy, B.; et al. Combined treatment with atorvastatin and imipenem improves survival and vascular functions in mouse model of sepsis. Vascul. Pharmacol. 2015, 71, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Masszi, G.; Novak, A.; Tarszabo, R.; Horvath, E.M.; Buday, A.; Ruisanchez, E.; Tokes, A.M.; Sara, L.; Benko, R.; Nadasy, G.L.; et al. Effects of vitamin D3 derivative—Calcitriol on pharmacological reactivity of aortic rings in a rodent PCOS model. Pharmacol. Rep. 2013, 65, 476–483. [Google Scholar] [CrossRef]
- Kazmierczak, P.A.; Dobaczewski, M.P.; Przygodzki, T.; Carsky, J.; Watala, C. β-Resorcylidene aminoguanidine (RAG) dilates coronary arteries in an endothelium-independent manner. Pharmacol. Rep. 2015, 67, 631–635. [Google Scholar] [CrossRef] [PubMed]
- Munin, J.; Quezada, E.; Cuinas, A.; Campos-Toimil, M.; Uriarte, E.; Santana, L.; Vina, D. Synthesis, biological evaluation and structure-activity relationships of new phthalazinedione derivatives with vasorelaxant activity. Eur. J. Med. Chem. 2014, 82, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Meyer, M.R.; Barton, M.; Prossnitz, E.R. Functional heterogeneity of NADPH oxidase-mediated contractions to endothelin with vascular aging. Life Sci. 2014, 118, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Maia-Joca, R.P.; Joca, H.C.; Ribeiro, F.J.; do Nascimento, R.V.; Silva-Alves, K.S.; Cruz, J.S.; Coelho-de-Souza, A.N.; Leal-Cardoso, J.H. Investigation of terpinen-4-ol effects on vascular smooth muscle relaxation. Life Sci. 2014, 115, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Kamiya, T.; Nagaoka, T.; Omae, T.; Yoshioka, T.; Ono, S.; Tanano, I.; Yoshida, A. Role of Ca2+-dependent and Ca2+-sensitive mechanisms in sphingosine 1-phosphate-induced constriction of isolated porcine retinal arterioles in vitro. Exp. Eye Res. 2014, 121, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Fajemiroye, J.O.; Amaral, N.O.; da Silva, E.F.; Galdino, P.M.; de Oliveira, T.S.; Ghedini, P.C.; Zjawiony, J.K.; Costa, E.A.; Pedrino, G.R.; Menegatti, R. Hypotensive and antihypertensive potential of 4-[(1-phenyl-1H-pyrazol-4-yl) methyl]1-piperazine carboxylic acid ethyl ester: A piperazine derivative. Life Sci. 2014, 112, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Tom, E.N.; Girard-Thernier, C.; Martin, H.; Dimo, T.; Alvergnas, M.; Nappey, M.; Berthelot, A.; Demougeot, C. Treatment with an extract of Terminalia superba Engler & Diels decreases blood pressure and improves endothelial function in spontaneously hypertensive rats. J. Ethnopharmacol. 2014, 151, 372–379. [Google Scholar] [PubMed]
- Boonla, O.; Kukongviriyapan, U.; Pakdeechote, P.; Kukongviriyapan, V.; Pannangpetch, P.; Prachaney, P.; Greenwald, S.E. Curcumin improves endothelial dysfunction and vascular remodeling in 2K-1C hypertensive rats by raising nitric oxide availability and reducing oxidative stress. Nitric Oxide 2014, 42, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Inoue, Y.; Nakahara, K.; Maruyama, K.; Suzuki, Y.; Hayashi, Y.; Kangawa, K.; Murakami, N. Central and peripheral des-acyl ghrelin regulates body temperature in rats. Biochem. Biophys. Res. Commun. 2013, 430, 278–283. [Google Scholar] [CrossRef] [PubMed]
- Pereira, S.L.; Kummerle, A.E.; Fraga, C.A.; Barreiro, E.J.; Rocha Nde, N.; Ferraz, E.B.; do Nascimento, J.H.; Sudo, R.T.; Zapata-Sudo, G. A novel Ca2+ channel antagonist reverses cardiac hypertrophy and pulmonary arteriolar remodeling in experimental pulmonary hypertension. Eur. J. Pharmacol. 2013, 702, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Priestley, J.R.; Buelow, M.W.; McEwen, S.T.; Weinberg, B.D.; Delaney, M.; Balus, S.F.; Hoeppner, C.; Dondlinger, L.; Lombard, J.H. Reduced angiotensin II levels cause generalized vascular dysfunction via oxidant stress in hamster cheek pouch arterioles. Microvasc. Res. 2013, 89, 134–145. [Google Scholar] [CrossRef] [PubMed]
- Babacanoglu, C.; Yildirim, N.; Sadi, G.; Pektas, M.B.; Akar, F. Resveratrol prevents high-fructose corn syrup-induced vascular insulin resistance and dysfunction in rats. Food Chem. Toxicol. 2013, 60, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Sinagra, T.; Tamburella, A.; Urso, V.; Siarkos, I.; Drago, F.; Bucolo, C.; Salomone, S. Reversible inhibition of vasoconstriction by thiazolidinediones related to PI3K/Akt inhibition in vascular smooth muscle cells. Biochem. Pharmacol. 2013, 85, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Brito, T.S.; Lima, F.J.; Aragao, K.S.; de Siqueira, R.J.; Sousa, P.J.; Maia, J.G.; Filho, J.D.; Lahlou, S.; Magalhaes, P.J. The vasorelaxant effects of 1-nitro-2-phenylethane involve stimulation of the soluble guanylate cyclase-cGMP pathway. Biochem. Pharmacol. 2013, 85, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Simões, M.R.; Furieri, L.B.; Forechi, L.; Baldo, M.P.; Rodrigues, S.L.; Salaices, M.; Vassallo, D.V.; Mill, J.G. High salt intake does not produce additional impairment in the coronary artery relaxation of spontaneously hypertensive aged rats. Food Chem. Toxicol. 2013, 58, 193–197. [Google Scholar] [CrossRef] [PubMed]
- Roberts, R.E.; Allen, S.; Chang, A.P.; Henderson, H.; Hobson, G.C.; Karania, B.; Morgan, K.N.; Pek, A.S.; Raghvani, K.; Shee, C.Y.; et al. Distinct mechanisms of relaxation to bioactive components from chamomile species in porcine isolated blood vessels. Toxicol. Appl. Pharmacol. 2013, 272, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Centeno, J.M.; Marrachelli, V.G.; Miranda, L.; Castello-Ruiz, M.; Burguete, M.C.; Jover-Mengual, T.; Salom, J.B.; Torregrosa, G.; Miranda, F.J.; Alborch, E. Involvement of prostacyclin and potassium channels in the diabetes-induced hyporeactivity of the rabbit carotid artery to B-type natriuretic peptide. Eur. J. Pharmacol. 2013, 701, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Oh, K.S.; Oh, B.K.; Park, C.H.; Seo, H.W.; Kang, N.S.; Lee, J.H.; Lee, J.S.; Ho Lee, B. Cardiovascular effects of a novel selective Rho kinase inhibitor, 2-(1H-indazole-5-yl)amino-4-methoxy-6-piperazino triazine (DW1865). Eur. J. Pharmacol. 2013, 702, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Kriska, T.; Cepura, C.; Siangjong, L.; Wan, T.C.; Auchampach, J.A.; Shaish, A.; Haratz, D.; Kumar, G.; Falck, J.R.; Gauthier, K.M.; et al. Effect of human 15-lipoxygenase-1 metabolites on vascular function in mouse mesenteric arteries and hearts. Prostaglandins Other Lipid Mediat. 2013, 106, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Nofal, Z.M.; Srour, A.M.; El-Eraky, W.I.; Saleh, D.O.; Girgis, A.S. Rational design, synthesis and QSAR study of vasorelaxant active 3-pyridinecarbonitriles incorporating 1H-benzimidazol-2-yl function. Eur. J. Med. Chem. 2013, 63, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.T.; Zhou, G.H.; Kho, J.H.; Sun, Y.Y.; Wen, J.F.; Kang, D.G.; Lee, H.S.; Cho, K.W.; Jin, S.N. Vasorelaxant action of an ethylacetate fraction of Euphorbia humifusa involves NO-cGMP pathway and potassium channels. J. Ethnopharmacol. 2013, 148, 655–663. [Google Scholar] [CrossRef] [PubMed]
- Gortan Cappellari, G.; Losurdo, P.; Mazzucco, S.; Panizon, E.; Jevnicar, M.; Macaluso, L.; Fabris, B.; Barazzoni, R.; Biolo, G.; Carretta, R.; et al. Treatment with n-3 polyunsaturated fatty acids reverses endothelial dysfunction and oxidative stress in experimental menopause. J. Nutr. Biochem. 2013, 24, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Martelli, A.; Testai, L.; Breschi, M.C.; Lawson, K.; McKay, N.G.; Miceli, F.; Taglialatela, M.; Calderone, V. Vasorelaxation by hydrogen sulphide involves activation of Kv7 potassium channels. Pharmacol. Res. 2013, 70, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Sukumaran, S.V.; Singh, T.U.; Parida, S.; Narasimha Reddy Ch, E.; Thangamalai, R.; Kandasamy, K.; Singh, V.; Mishra, S.K. TRPV4 channel activation leads to endothelium-dependent relaxation mediated by nitric oxide and endothelium-derived hyperpolarizing factor in rat pulmonary artery. Pharmacol. Res. 2013, 78, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Chen, R.; Liu, P.; Gu, Y. Docosahexaenoic acid attenuates hypoxic pulmonary vasoconstriction by activating the large conductance Ca2+-activated K+ currents in pulmonary artery smooth muscle cells. Pulm. Pharmacol. Ther. 2014, 28, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Cekic, E.G.; Soydan, G.; Guler, S.; Babaoglu, M.O.; Tuncer, M. Propranolol-induced relaxation in the rat basilar artery. Vascul. Pharmacol. 2013, 58, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Dalaklioglu, S.; Tasatargil, A.; Kale, S.; Tanriover, G.; Dilmac, S.; Erin, N. Metastatic breast carcinoma induces vascular endothelial dysfunction in Balb-c mice: Role of the tumor necrosis factor-α and NADPH oxidase. Vascul. Pharmacol. 2013, 59, 103–111. [Google Scholar] [CrossRef] [PubMed]
- El-Kashef, D.H.; El-Agamy, D.S.; Gamil, N.M. Protective effects of hydrogen sulfide against high glucose induced-endothelial dysfunction: An in vitro study. J. Taibah Univ. Sci. 2013, 7, 97–104. [Google Scholar] [CrossRef]
- Senbel, A.M.; Omar, A.G.; Abdel-Moneim, L.M.; Mohamed, H.F.; Daabees, T.T. Evaluation of l-arginine on kidney function and vascular reactivity following ischemic injury in rats: protective effects and potential interactions. Pharmacol. Rep. 2014, 66, 976–983. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Yang, H.G.; Yuan, T.Y.; Zhao, Y.; Du, G.H. Rho kinase inhibition activity of pinocembrin in rat aortic rings contracted by angiotensin II. Chin. J. Nat. Med. 2013, 11, 258–263. [Google Scholar] [CrossRef] [PubMed]
- McCormick, C.; Jones, R.L.; Kennedy, S.; Wadsworth, R.M. Activation of prostanoid EP receptors by prostacyclin analogues in rabbit iliac artery: implications for anti-restenotic potential. Eur. J. Pharmacol. 2010, 641, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Takebayashi, K.; Sohma, R.; Aso, Y.; Inukai, T. Effects of retinol binding protein-4 on vascular endothelial cells. Biochem. Biophys. Res. Commun. 2011, 408, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, Y.; Kuwabara, M.; Tanaka-Totoribe, N.; Kanai, T.; Nakamura, E.; Gamoh, S.; Suzuki, A.; Asada, Y.; Hisa, H.; Yamamoto, R. The defective protein level of myosin light chain phosphatase (MLCP) in the isolated saphenous vein, as a vascular conduit in coronary artery bypass grafting (CABG), harvested from patients with diabetes mellitus (DM). Biochem. Biophys. Res. Commun. 2011, 412, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Giustarini, D.; Tsikas, D.; Rossi, R. Study of the effect of thiols on the vasodilatory potency of S-nitrosothiols by using a modified aortic ring assay. Toxicol. Appl. Pharmacol. 2011, 256, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Su, L.Y.; Lee, R.M.; Gao, Y.J. Alterations in perivascular adipose tissue structure and function in hypertension. Eur. J. Pharmacol. 2011, 656, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Sapa, J.; Kubacka, M. The possible mechanism of hypotensive activity of some pyrrolidin-2-one derivatives with antagonist properties at α1-adrenoceptors. Eur. J. Pharmacol. 2011, 673, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Lamarre, N.S.; Parry, T.; Tallarida, R.J. On the quantitation of an agonist with dual but opposing components of action: Application to vascular endothelial relaxation. Eur. J. Pharmacol. 2011, 670, 204–207. [Google Scholar] [CrossRef] [PubMed]
- Van Drongelen, J.; Pertijs, J.; Wouterse, A.; Hermsen, R.; Sweep, F.C.; Lotgering, F.K.; Smits, P.; Spaanderman, M.E. Contribution of different local vascular responses to mid-gestational vasodilation. Am. J. Obstet. Gynecol. 2011, 205, 155.e12–155.e17. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, T.; Anraku, T.; Matsuzawa, S. Ibudilast, a mixed PDE3/4 inhibitor, causes a selective and nitric oxide/cGMP-independent relaxation of the intracranial vertebrobasilar artery. Eur. J. Pharmacol. 2011, 650, 605–611. [Google Scholar] [CrossRef] [PubMed]
- Fiorim, J.; Ribeiro, R.F., Jr.; Azevedo, B.F.; Simoes, M.R.; Padilha, A.S.; Stefanon, I.; Alonso, M.J.; Salaices, M.; Vassallo, D.V. Activation of K+ channels and Na+/K+ ATPase prevents aortic endothelial dysfunction in 7-day lead-treated rats. Toxicol. Appl. Pharmacol. 2012, 262, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Agbor, L.N.; Walsh, M.T.; Boberg, J.R.; Walker, M.K. Elevated blood pressure in cytochrome P4501A1 knockout mice is associated with reduced vasodilation to omega-3 polyunsaturated fatty acids. Toxicol. Appl. Pharmacol. 2012, 264, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Zapata-Sudo, G.; Pontes, L.B.; da Silva, J.S.; Lima, L.M.; Nunes, I.K.; Barreiro, E.J.; Sudo, R.T. Benzenesulfonamide attenuates monocrotaline-induced pulmonary arterial hypertension in a rat model. Eur. J. Pharmacol. 2012, 690, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Sara, L.; Antal, P.; Masszi, G.; Buday, A.; Horvath, E.M.; Hamar, P.; Monos, E.; Nadasy, G.L.; Varbiro, S. Arteriolar insulin resistance in a rat model of polycystic ovary syndrome. Fertil. Steril. 2012, 97, 462–468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alvarez-Medina, D.I.; Hernandez, A.; Orozco, C. Endothelial hyperpolarizing factor increases acetylcholine-induced vasodilatation in pulmonary hypertensive broilers arterial rings. Res. Vet. Sci. 2012, 92, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.B.; O, W.S.; Tang, F. Adrenomedullin inhibits norepinephrine-induced contraction of rat seminal vesicle. Urology 2012, 80, 224.e1–224.e5. [Google Scholar] [CrossRef] [PubMed]
- Meng, Z.; Yang, Z.; Li, J.; Zhang, Q. The vasorelaxant effect and its mechanisms of sodium bisulfite as a sulfur dioxide donor. Chemosphere 2012, 89, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Zhang, X.; Wang, T.; Hu, J. Botany, traditional uses, phytochemistry and pharmacology of Apocynum venetum L. (Luobuma): A review. J. Ethnopharmacol. 2012, 141, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Quintela, A.M.; Jimenez, R.; Gomez-Guzman, M.; Zarzuelo, M.J.; Galindo, P.; Sanchez, M.; Vargas, F.; Cogolludo, A.; Tamargo, J.; Perez-Vizcaino, F.; et al. Activation of peroxisome proliferator-activated receptor-β/-δ (PPARβ/δ) prevents endothelial dysfunction in type 1 diabetic rats. Free Radic. Biol. Med. 2012, 53, 730–741. [Google Scholar] [CrossRef] [PubMed]
- Del Bo, C.; Kristo, A.S.; Kalea, A.Z.; Ciappellano, S.; Riso, P.; Porrini, M.; Klimis-Zacas, D. The temporal effect of a wild blueberry (Vaccinium angustifolium)-enriched diet on vasomotor tone in the Sprague-Dawley rat. Nutr. Metab. Cardiovasc. Dis. 2012, 22, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Jerez, S.; Sierra, L.; de Bruno, M.P. 17-Octadecynoic acid improves contractile response to angiotensin II by releasing vasocontrictor prostaglandins. Prostaglandins Other Lipid Mediat. 2012, 97, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Szasz, T.; Tostes, R.C.; Webb, R.C. Impaired β-adrenoceptor-induced relaxation in small mesenteric arteries from DOCA-salt hypertensive rats is due to reduced K(Ca) channel activity. Pharmacol. Res. 2012, 65, 537–545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, C.; Huo, Y.; An, X.; Singh, G.; Chen, M.; Yang, Z.; Pu, J.; Li, J. Panax notoginseng and its components decreased hypertension via stimulation of endothelial-dependent vessel dilatation. Vascul. Pharmacol. 2012, 56, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Aloysius, U.I.; Achike, F.I.; Mustafa, M.R. Mechanisms underlining gender differences in Phenylephrine contraction of normoglycaemic and short-term Streptozotocin-induced diabetic WKY rat aorta. Vascul. Pharmacol. 2012, 57, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Pelham, C.J.; Ketsawatsomkron, P.; Groh, S.; Grobe, J.L.; de Lange, W.J.; Ibeawuchi, S.R.; Keen, H.L.; Weatherford, E.T.; Faraci, F.M.; Sigmund, C.D. Cullin-3 regulates vascular smooth muscle function and arterial blood pressure via PPARgamma and RhoA/Rho-kinase. Cell Metab. 2012, 16, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Beleznai, T.; Bagi, Z. Activation of hexosamine pathway impairs nitric oxide (NO)-dependent arteriolar dilations by increased protein O-GlcNAcylation. Vascul. Pharmacol. 2012, 56, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Shi, L.; Yu, X.; Sun, J.; Zhang, H.; Wang, S.; Fang, L.; Du, G. Differential effects of Rho-kinase inhibitor and angiotensin II type-1 receptor antagonist on the vascular function in hypertensive rats induced by chronic l-NAME treatment. Acta Pharm. Sin. B 2012, 2, 450–458. [Google Scholar] [CrossRef]
- Haines, R.J.; Corbin, K.D.; Pendleton, L.C.; Meininger, C.J.; Eichler, D.C. Insulin transcriptionally regulates argininosuccinate synthase to maintain vascular endothelial function. Biochem. Biophys. Res. Commun. 2012, 421, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Davel, A.P.; Lemos, M.; Pastro, L.M.; Pedro, S.C.; de Andre, P.A.; Hebeda, C.; Farsky, S.H.; Saldiva, P.H.; Rossoni, L.V. Endothelial dysfunction in the pulmonary artery induced by concentrated fine particulate matter exposure is associated with local but not systemic inflammation. Toxicology 2012, 295, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Toba, H.; Tojo, C.; Wang, J.; Noda, K.; Kobara, M.; Nakata, T. Telmisartan inhibits vascular dysfunction and inflammation via activation of peroxisome proliferator-activated receptor-gamma in subtotal nephrectomized rat. Eur. J. Pharmacol. 2012, 685, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Inoue, N.; Fuchikami, C.; Tajima, K.; Hashino, A.; Fukui, H.; Noda, K.; Oka, M. Blockade of voltage-gated calcium channel Cav1.2 and α1-adrenoceptors increases vertebral artery blood flow induced by the antivertigo agent difenidol. Eur. J. Pharmacol. 2012, 689, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Sathishkumar, K.; Elkins, R.; Yallampalli, U.; Balakrishnan, M.; Yallampalli, C. Fetal programming of adult hypertension in female rat offspring exposed to androgens in utero. Early Hum. Dev. 2011, 87, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Malakul, W.; Thirawarapan, S.; Ingkaninan, K.; Sawasdee, P. Effects of Kaempferia parviflora Wall. Ex Baker on endothelial dysfunction in streptozotocin-induced diabetic rats. J. Ethnopharmacol. 2011, 133, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Malakul, W.; Ingkaninan, K.; Sawasdee, P.; Woodman, O.L. The ethanolic extract of Kaempferia parviflora reduces ischaemic injury in rat isolated hearts. J. Ethnopharmacol. 2011, 137, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Bertin, R.; Garcia-Argaez, A.; Martinez-Vazquez, M.; Froldi, G. Age-dependent vasorelaxation of Casimiroa edulis and Casimiroa pubescens extracts in rat caudal artery in vitro. J. Ethnopharmacol. 2011, 137, 934–936. [Google Scholar] [CrossRef] [PubMed]
- Witting, P.K.; Song, C.; Hsu, K.; Hua, S.; Parry, S.N.; Aran, R.; Geczy, C.; Freedman, S.B. The acute-phase protein serum amyloid A induces endothelial dysfunction that is inhibited by high-density lipoprotein. Free Radic. Biol. Med. 2011, 51, 1390–1398. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, H.; Okamoto, R.; Zaima, K.; Hirasawa, Y.; Ismail, I.S.; Lajis, N.H.; Morita, H. New vasorelaxant indole alkaloids, villocarines A–D from Uncaria villosa. Bioorg. Med. Chem. 2011, 19, 4075–4079. [Google Scholar] [CrossRef] [PubMed]
- Morita, H.; Zaima, K.; Koga, I.; Saito, A.; Tamamoto, H.; Okazaki, H.; Kaneda, T.; Hashimoto, T.; Asakawa, Y. Vasorelaxant effects of macrocyclic bis(bibenzyls) from liverworts. Bioorg. Med. Chem. 2011, 19, 4051–4056. [Google Scholar] [CrossRef] [PubMed]
- Monroy-Ruiz, J.; Sevilla, M.A.; Carron, R.; Montero, M.J. Astaxanthin-enriched-diet reduces blood pressure and improves cardiovascular parameters in spontaneously hypertensive rats. Pharmacol. Res. 2011, 63, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Siarkos, I.; Urso, V.; Sinagra, T.; Drago, F.; Salomone, S. Endothelium-dependent vasomotor effects of telmisartan in isolated rat femoral arteries. Pharmacol. Res. 2011, 63, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Wojcicka, G.; Jamroz-Wisniewska, A.; Atanasova, P.; Chaldakov, G.N.; Chylinska-Kula, B.; Beltowski, J. Differential effects of statins on endogenous H2S formation in perivascular adipose tissue. Pharmacol. Res. 2011, 63, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Marrachelli, V.G.; Miranda, F.J.; Centeno, J.M.; Miranda, I.; Castello-Ruiz, M.; Burguete, M.C.; Jover-Mengual, T.; Salom, J.B.; Torregrosa, G.; Alborch, E. Mechanisms underlying the diabetes-induced hyporeactivity of the rabbit carotid artery to atrial natriuretic peptide. Pharmacol. Res. 2011, 63, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Sciorati, C.; Miglietta, D.; Buono, R.; Pisa, V.; Cattaneo, D.; Azzoni, E.; Brunelli, S.; Clementi, E. A dual acting compound releasing nitric oxide (NO) and ibuprofen, NCX 320, shows significant therapeutic effects in a mouse model of muscular dystrophy. Pharmacol. Res. 2011, 64, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Cantu-Medellin, N.; Vitturi, D.A.; Rodriguez, C.; Murphy, S.; Dorman, S.; Shiva, S.; Zhou, Y.; Jia, Y.; Palmer, A.F.; Patel, R.P. Effects of T- and R-state stabilization on deoxyhemoglobin-nitrite reactions and stimulation of nitric oxide signaling. Nitric Oxide 2011, 25, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Pereira, A.D.C.; Ford, P.C.; da Silva, R.S.; Bendhack, L.M. Ruthenium-nitrite complex as pro-drug releases NO in a tissue and enzyme-dependent way. Nitric Oxide 2011, 24, 192–198. [Google Scholar] [CrossRef] [PubMed]
- Mei, Y.; Jin, H.; Tian, W.; Wang, H.; Wang, H.; Zhao, Y.; Zhang, Z.; Meng, F. Urantide alleviates monocrotaline induced pulmonary arterial hypertension in Wistar rats. Pulm. Pharmacol. Ther. 2011, 24, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Chen, Y.; Sun, L.; Fu, G.; Guo, L. Vasodilatation produced by fasudil mesylate in vivo and in vitro. Vascul. Pharmacol. 2011, 55, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, T.; Sheu, J.-R. Cardiovascular pharmacological actions of rutaecarpine, a quinazolinocarboline alkaloid isolated from Evodia rutaecarpa. J. Exp. Clin. Med. 2011, 3, 63–69. [Google Scholar] [CrossRef]
- Choi, H.; Tostes, R.C.; Webb, R.C. Mitochondrial aldehyde dehydrogenase prevents ROS-induced vascular contraction in angiotensin-II hypertensive mice. J. Am. Soc. Hypertens. 2011, 5, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Medei, E.; Lima-Leopoldo, A.P.; Pereira-Junior, P.P.; Leopoldo, A.S.; Campos, D.H.S.; Raimundo, J.M.; Sudo, R.T.; Zapata-Sudo, G.; Bruder-Nascimento, T.; Cordellini, S. Could a high-fat diet rich in unsaturated fatty acids impair the cardiovascular system? Can. J. Cardiol. 2010, 26, 542–548. [Google Scholar] [CrossRef]
- Wang, J.; Dong, M.Q.; Liu, M.L.; Xu, D.Q.; Luo, Y.; Zhang, B.; Liu, L.L.; Xu, M.; Zhao, P.T.; Gao, Y.Q.; et al. Tanshinone IIA modulates pulmonary vascular response to agonist and hypoxia primarily via inhibiting Ca2+ influx and release in normal and hypoxic pulmonary hypertension rats. Eur. J. Pharmacol. 2010, 640, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.; Xu, X.; Zhang, L.; Wu, H.; Peng, L. Identification of a nitric oxide-dependent hypotensive effect of anticoagulation factor II from the venom of Agkistrodon acutus. Biochem. Pharmacol. 2010, 79, 498–506. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.P.; Li, L.; Yang, Y.; Fu, M.; Yao, L.; Wu, T.; Zhang, X.Q.; Hu, S.J. Chronic inhibition of farnesyl pyrophosphate synthase improves endothelial function in spontaneously hypertensive rats. Biochem. Pharmacol. 2010, 80, 1684–1689. [Google Scholar] [CrossRef] [PubMed]
- Romero, M.; Jimenez, R.; Hurtado, B.; Moreno, J.M.; Rodriguez-Gomez, I.; Lopez-Sepulveda, R.; Zarzuelo, A.; Perez-Vizcaino, F.; Tamargo, J.; Vargas, F.; et al. Lack of beneficial metabolic effects of quercetin in adult spontaneously hypertensive rats. Eur. J. Pharmacol. 2010, 627, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Subramani, J.; Leo, M.D.; Kathirvel, K.; Arunadevi, R.; Singh, T.U.; Prakash, V.R.; Mishra, S.K. Essential role of nitric oxide in sepsis-induced impairment of endothelium-derived hyperpolarizing factor-mediated relaxation in rat pulmonary artery. Eur. J. Pharmacol. 2010, 630, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.T.; Wang, Y.; Deng, X.L.; Dong, M.Q.; Zhao, L.M.; Wang, Y.W. Daidzein relaxes rat cerebral basilar artery via activation of large-conductance Ca2+-activated K+ channels in vascular smooth muscle cells. Eur. J. Pharmacol. 2010, 630, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Singh, T.U.; Kathirvel, K.; Choudhury, S.; Garg, S.K.; Mishra, S.K. Eicosapentaenoic acid-induced endothelium-dependent and -independent relaxation of sheep pulmonary artery. Eur. J. Pharmacol. 2010, 636, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Enkhjargal, B.; Hashimoto, M.; Sakai, Y.; Shido, O. Characterization of vasoconstrictor-induced relaxation in the cerebral basilar artery. Eur. J. Pharmacol. 2010, 637, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, A.; Recio, P.; Orensanz, L.M.; Bustamante, S.; Navarro-Dorado, J.; Climent, B.; Benedito, S.; Garcia-Sacristan, A.; Prieto, D.; Hernandez, M. Mechanisms involved in the effects of endothelin-1 in pig prostatic small arteries. Eur. J. Pharmacol. 2010, 640, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Bayram, Z.; Golbasi, I.; Ozdem, S.S. The role of nitric oxide and potassium channels in the effect of adrenomedullin in human internal thoracic arteries. Regul. Pept. 2010, 161, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Franca-Silva, M.S.; Luciano, M.N.; Ribeiro, T.P.; Silva, J.S.; Santos, A.F.; Franca, K.C.; Nakao, L.S.; Athayde-Filho, P.F.; Braga, V.A.; Medeiros, I.A. The 2-nitrate-1,3-dibuthoxypropan, a new nitric oxide donor, induces vasorelaxation in mesenteric arteries of the rat. Eur. J. Pharmacol. 2012, 690, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Perusquia, M.; Espinoza, J.; Montano, L.M.; Stallone, J.N. Regional differences in the vasorelaxing effects of testosterone and its 5-reduced metabolites in the canine vasculature. Vascul. Pharmacol. 2012, 56, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Arai, H.; Zaima, K.; Mitsuta, E.; Tamamoto, H.; Saito, A.; Hirasawa, Y.; Rahman, A.; Kusumawati, I.; Zaini, N.C.; Morita, H. Alstiphyllanines I–O, ajmaline type alkaloids from Alstonia macrophylla showing vasorelaxant activity. Bioorg. Med. Chem. 2012, 20, 3454–3459. [Google Scholar] [CrossRef] [PubMed]
- Marrachelli, V.G.; Centeno, J.M.; Miranda, I.; Castello-Ruiz, M.; Burguete, M.C.; Jover-Mengual, T.; Salom, J.B.; Torregrosa, G.; Miranda, F.J.; Alborch, E. Diabetes impairs the atrial natriuretic peptide relaxant action mediated by potassium channels and prostacyclin in the rabbit renal artery. Pharmacol. Res. 2012, 66, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Fu, Y.; Ge, Y.; Juncos, L.A.; Reckelhoff, J.F.; Liu, R. The vasodilatory effect of testosterone on renal afferent arterioles. Gend. Med. 2012, 9, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Sodhi, K.; Puri, N.; Inoue, K.; Falck, J.R.; Schwartzman, M.L.; Abraham, N.G. EET agonist prevents adiposity and vascular dysfunction in rats fed a high fat diet via a decrease in Bach 1 and an increase in HO-1 levels. Prostaglandins Other Lipid Mediat. 2012, 98, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Lesniewski, L.A.; Zigler, M.C.; Durrant, J.R.; Donato, A.J.; Seals, D.R. Sustained activation of AMPK ameliorates age-associated vascular endothelial dysfunction via a nitric oxide-independent mechanism. Mech. Ageing Dev. 2012, 133, 368–371. [Google Scholar] [CrossRef] [PubMed]
- Shuto, H.; Tominaga, K.; Yamauchi, A.; Ikeda, M.; Kusaba, K.; Mitsunaga, D.; Hirabara, Y.; Egawa, T.; Takano, Y.; Kataoka, Y. The statins fluvastatin and pravastatin exert anti-flushing effects by improving vasomotor dysfunction through nitric oxide-mediated mechanisms in ovariectomized animals. Eur. J. Pharmacol. 2011, 651, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Toba, H.; Morishita, M.; Tojo, C.; Nakano, A.; Oshima, Y.; Kojima, Y.; Yoshida, M.; Nakashima, K.; Wang, J.; Kobara, M.; et al. Recombinant human erythropoietin ameliorated endothelial dysfunction and macrophage infiltration by increasing nitric oxide in hypertensive 5/6 nephrectomized rat aorta. Eur. J. Pharmacol. 2011, 656, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Villalon, A.L.; Fernandez, N.; Monge, L.; Dieguez, G. Coronary response to diadenosine tetraphosphate after ischemia-reperfusion in the isolated rat heart. Eur. J. Pharmacol. 2011, 660, 394–401. [Google Scholar] [CrossRef] [PubMed]
- Medeiros, M.A.; Pinho, J.F.; De-Lira, D.P.; Barbosa-Filho, J.M.; Araujo, D.A.; Cortes, S.F.; Lemos, V.S.; Cruz, J.S. Curine, a bisbenzylisoquinoline alkaloid, blocks l-type Ca2+ channels and decreases intracellular Ca2+ transients in A7r5 cells. Eur. J. Pharmacol. 2011, 669, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.B.; Jin, H.F.; Tang, C.S.; Du, J.B. The biological effect of endogenous sulfur dioxide in the cardiovascular system. Eur. J. Pharmacol. 2011, 670, 1–6. [Google Scholar] [CrossRef] [PubMed]
- de Buys Roessingh, A.; Fouquet, V.; Aigrain, Y.; Mercier, J.-C.; de Lagausie, P.; Dinh-Xuan, A.T. Nitric oxide activity through guanylate cyclase and phosphodiesterase modulation is impaired in fetal lambs with congenital diaphragmatic hernia. J. Pediatr. Surg. 2011, 46, 1516–1522. [Google Scholar] [CrossRef] [PubMed]
- Takir, S.; Uydes-Dogan, B.S.; Ozdemir, O. Retina evokes biphasic relaxations in retinal artery unrelated to endothelium, K(V), K(ATP), K(Ca) channels and methyl palmitate. Microvasc. Res. 2011, 81, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Dasiewicz, P.J.; Conlon, J.M.; Anderson, W.G. Cardiovascular and vasoconstrictive actions of skate bradykinin in the little skate, Leucoraja erinacea (Elasmobranchii). Gen. Comp. Endocrinol. 2011, 174, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Nicol, L.; Harouki, N.; Monteil, C.; Wecker, D.; Debunne, M.; Bauer, F.; Lallemand, F.; Richard, V.; Thuillez, C.; et al. Improvement of left ventricular diastolic function induced by β-blockade: A comparison between nebivolol and metoprolol. J. Mol. Cell. Cardiol. 2011, 51, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.T.; Ng, C.H.; Tsang, S.Y.; Huang, Y.; Chen, Z.Y. Relative contribution of individual oxidized components in ox-LDL to inhibition on endothelium-dependent relaxation in rat aorta. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Chakkarwar, V.A. Fenofibrate attenuates nicotine-induced vascular endothelial dysfunction in the rat. Vascul. Pharmacol. 2011, 55, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Bertinaria, M.; Guglielmo, S.; Rolando, B.; Giorgis, M.; Aragno, C.; Fruttero, R.; Gasco, A.; Parapini, S.; Taramelli, D.; Martins, Y.C.; et al. Amodiaquine analogues containing NO-donor substructures: synthesis and their preliminary evaluation as potential tools in the treatment of cerebral malaria. Eur. J. Med. Chem. 2011, 46, 1757–1767. [Google Scholar] [CrossRef] [PubMed]
- Parlar, A.; Can, C.; Erol, A.; Ulker, S. Posttransplantation therapeutic rapamycin concentration protects nitric oxide-related vascular endothelial function: Comparative effects in rat thoracic aorta and coronary endothelial cell culture. Transpl. Proc. 2010, 42, 1923–1930. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.L.; Dai, Z.K.; Lin, R.J.; Chu, K.S.; Chen, I.J.; Wu, J.R.; Wu, B.N. Baicalin, a flavonoid from Scutellaria baicalensis Georgi, activates large-conductance Ca2+-activated K+ channels via cyclic nucleotide-dependent protein kinases in mesenteric artery. Phytomedicine 2010, 17, 760–770. [Google Scholar] [CrossRef] [PubMed]
- Marrachelli, V.G.; Miranda, F.J.; Centeno, J.M.; Salom, J.B.; Torregrosa, G.; Jover-Mengual, T.; Perez, A.M.; Moro, M.A.; Alborch, E. Role of NO-synthases and cyclooxygenases in the hyperreactivity of male rabbit carotid artery to testosterone under experimental diabetes. Pharmacol. Res. 2010, 61, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Novella, S.; Dantas, A.P.; Segarra, G.; Novensa, L.; Bueno, C.; Heras, M.; Hermenegildo, C.; Medina, P. Gathering of aging and estrogen withdrawal in vascular dysfunction of senescent accelerated mice. Exp. Gerontol. 2010, 45, 868–874. [Google Scholar] [CrossRef] [PubMed]
- Raffetto, J.D.; Qiao, X.; Beauregard, K.G.; Khalil, R.A. Estrogen receptor-mediated enhancement of venous relaxation in female rat: implications in sex-related differences in varicose veins. J. Vasc. Surg. 2010, 51, 972–981. [Google Scholar] [CrossRef] [PubMed]
- Khoo, N.K.; White, C.R.; Pozzo-Miller, L.; Zhou, F.; Constance, C.; Inoue, T.; Patel, R.P.; Parks, D.A. Dietary flavonoid quercetin stimulates vasorelaxation in aortic vessels. Free Radic. Biol. Med. 2010, 49, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Zhao, R.; Zhang, Z.; Wang, J. Changes in vasoreactivity of rat large- and medium-sized arteries induced by hyperthyroidism. Exp. Toxicol. Pathol. 2010, 62, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Seto, S.W.; Lam, T.Y.; Or, P.M.; Lee, W.Y.; Au, A.L.; Poon, C.C.; Li, R.W.; Chan, S.W.; Yeung, J.H.; Leung, G.P.; et al. Folic acid consumption reduces resistin level and restores blunted acetylcholine-induced aortic relaxation in obese/diabetic mice. J. Nutr. Biochem. 2010, 21, 872–880. [Google Scholar] [CrossRef] [PubMed]
- Lolli, M.L.; Rolando, B.; Tosco, P.; Chaurasia, S.; di Stilo, A.; Lazzarato, L.; Gorassini, E.; Ferracini, R.; Oliaro-Bosso, S.; Fruttero, R.; et al. Synthesis and preliminary pharmacological characterisation of a new class of nitrogen-containing bisphosphonates (N-BPs). Bioorg. Med. Chem. 2010, 18, 2428–2438. [Google Scholar] [CrossRef] [PubMed]
- Marrachelli, V.G.; Miranda, F.J.; Centeno, J.M.; Burguete, M.C.; Castello-Ruiz, M.; Jover-Mengual, T.; Perez, A.M.; Salom, J.B.; Torregrosa, G.; Alborch, E. Mechanisms involved in the relaxant action of testosterone in the renal artery from male normoglycemic and diabetic rabbits. Pharmacol. Res. 2010, 61, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Ishida, K.; Nakayama, N.; Taguchi, K.; Kobayashi, T.; Kamata, K. Mechanisms underlying the losartan treatment-induced improvement in the endothelial dysfunction seen in mesenteric arteries from type 2 diabetic rats. Pharmacol. Res. 2010, 62, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Olukman, M.; Orhan, C.E.; Celenk, F.G.; Ulker, S. Apocynin restores endothelial dysfunction in streptozotocin diabetic rats through regulation of nitric oxide synthase and NADPH oxidase expressions. J. Diabetes Complicat. 2010, 24, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.H.; van Breemen, C.; Chung, A.W. Vasomotor dysfunction in the thoracic aorta of Marfan syndrome is associated with accumulation of oxidative stress. Vascul. Pharmacol. 2010, 52, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Liao, W.C.; Hou, M.C.; Wang, G.J.; Yu, K.W.; Lee, F.Y.; Lin, H.C.; Lee, S.D. Sepsis worsening vascular hyporeactivity of the superior mesenteric artery in portal vein-ligated rats. J. Chin. Med. Assoc. 2010, 73, 462–470. [Google Scholar] [CrossRef]
- Dominguez, J.M., 2nd; Prisby, R.D.; Muller-Delp, J.M.; Allen, M.R.; Delp, M.D. Increased nitric oxide-mediated vasodilation of bone resistance arteries is associated with increased trabecular bone volume after endurance training in rats. Bone 2010, 46, 813–819. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.H.; Xu, Q.; Xu, W.H.; Guo, X.H.; Zhang, S.; Chen, Y.D. Mechanisms of protection against diabetes-induced impairment of endothelium-dependent vasorelaxation by Tanshinone IIA. Biochim. Biophys. Acta 2015, 1850, 813–823. [Google Scholar] [CrossRef] [PubMed]
- de Candia, M.; Marini, E.; Zaetta, G.; Cellamare, S.; di Stilo, A.; Altomare, C.D. New organic nitrate-containing benzyloxy isonipecotanilide derivatives with vasodilatory and anti-platelet activity. Eur. J. Pharm. Sci. 2015, 72, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.; Park, M.; hyung Lee, J.; Min, B.S.; Ryoo, S. Endothelial nitric oxide synthase activation through obacunone-dependent arginase inhibition restored impaired endothelial function in ApoE-null mice. Vascul. Pharmacol. 2014, 60, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Kagota, S.; Maruyama, K.; Wakuda, H.; McGuire, J.J.; Yoshikawa, N.; Nakamura, K.; Shinozuka, K. Disturbance of vasodilation via protease-activated receptor 2 in SHRSP.Z-Lepr fa/IzmDmcr rats with metabolic syndrome. Vascul. Pharmacol. 2014, 63, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Kline, L.W.; Karpinski, E. The flavonoid chrysin, an endocrine disrupter, relaxes cholecystokinin- and KCl-induced tension in male guinea pig gallbladder strips through multiple signaling pathways. Steroids 2014, 79, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Salomone, S.; Foresti, R.; Villari, A.; Giurdanella, G.; Drago, F.; Bucolo, C. Regulation of vascular tone in rabbit ophthalmic artery: Cross talk of endogenous and exogenous gas mediators. Biochem. Pharmacol. 2014, 92, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.L.; Yan, L.; Chen, Y.H.; Zeng, G.H.; Zhou, Y.; Chen, H.P.; Peng, W.J.; He, M.; Huang, Q.R. A role for diallyl trisulfide in mitochondrial antioxidative stress contributes to its protective effects against vascular endothelial impairment. Eur. J. Pharmacol. 2014, 725, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Watanabe, S.; Kawamura, R.; Taguchi, K.; Kobayashi, T. Epigallocatechin gallate attenuates ET-1-induced contraction in carotid artery from type 2 diabetic OLETF rat at chronic stage of disease. Life Sci. 2014, 118, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Schrammel, A.; Mussbacher, M.; Wolkart, G.; Stessel, H.; Pail, K.; Winkler, S.; Schweiger, M.; Haemmerle, G.; Al Zoughbi, W.; Hofler, G.; et al. Endothelial dysfunction in adipose triglyceride lipase deficiency. Biochim. Biophys. Acta 2014, 1841, 906–917. [Google Scholar] [CrossRef] [PubMed]
- Kivimäki, A.S.; Siltari, A.; Ehlers, P.I.; Korpela, R.; Vapaatalo, H. Lingonberry juice negates the effects of a high salt diet on vascular function and low-grade inflammation. J. Funct. Foods 2014, 7, 238–245. [Google Scholar] [CrossRef]
- de Sá, L.Z.M.; Castro, P.F.; Lino, F.M.; Bernardes, M.J.; Viegas, J.C.; Dinis, T.C.; Santana, M.J.; Romao, W.; Vaz, B.G.; Lião, L.M. Antioxidant potential and vasodilatory activity of fermented beverages of jabuticaba berry (Myrciaria jaboticaba). J. Funct. Foods 2014, 8, 169–179. [Google Scholar] [CrossRef]
- Zhou, Z.; de Wijs-Meijler, D.; Lankhuizen, I.; Jankowski, J.; Jankowski, V.; Jan Danser, A.H.; Duncker, D.J.; Merkus, D. Blunted coronary vasodilator response to uridine adenosine tetraphosphate in post-infarct remodeled myocardium is due to reduced P1 receptor activation. Pharmacol. Res. 2013, 77, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Gadkari, T.V.; Cortes, N.; Madrasi, K.; Tsoukias, N.M.; Joshi, M.S. Agmatine induced NO dependent rat mesenteric artery relaxation and its impairment in salt-sensitive hypertension. Nitric Oxide 2013, 35, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Mamo, Y.A.; Angus, J.A.; Ziogas, J.; Soeding, P.F.; Wright, C.E. The role of voltage-operated and non-voltage-operated calcium channels in endothelin-induced vasoconstriction of rat cerebral arteries. Eur. J. Pharmacol. 2014, 742, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Hedegaard, E.R.; Nielsen, B.D.; Mogensen, S.; Rembold, C.M.; Frobert, O.; Simonsen, U. Mechanisms involved in increased sensitivity to adenosine A(2A) receptor activation and hypoxia-induced vasodilatation in porcine coronary arteries. Eur. J. Pharmacol. 2014, 723, 216–226. [Google Scholar] [CrossRef] [PubMed]
- Chaothanaphat, N.; Dhumma-Upakorn, P.; Jianmongkol, S. In vitro modulating effects of glutathione on vascular tension and involvement of extracellular calcium. Drug Discov. Ther. 2010, 4, 19–25. [Google Scholar] [PubMed]
- Rodrigues, G.J.; Cicillini, S.A.; Silva, R.S.; Bendhack, L.M. Mechanisms underlying the vascular relaxation induced by a new nitric oxide generator. Nitric Oxide 2011, 25, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Vlasova, M.A.; Tarasova, O.S.; Riikonen, J.; Raula, J.; Lobach, A.S.; Borzykh, A.A.; Smirin, B.V.; Kauppinen, E.I.; Eletskii, A.V.; Herzig, K.H.; et al. Injected nanoparticles: the combination of experimental systems to assess cardiovascular adverse effects. Eur. J. Pharm. Biopharm. 2014, 87, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Laspas, P.; Goloborodko, E.; Sniatecki, J.J.; Kordasz, M.L.; Manicam, C.; Wojnowski, L.; Li, H.; Patzak, A.; Pfeiffer, N.; Gericke, A. Role of nitric oxide synthase isoforms for ophthalmic artery reactivity in mice. Exp. Eye Res. 2014, 127, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sakakibara, K.; Feng, G.G.; Li, J.; Akahori, T.; Yasuda, Y.; Nakamura, E.; Hatakeyama, N.; Fujiwara, Y.; Kinoshita, H. Kynurenine causes vasodilation and hypotension induced by activation of KCNQ-encoded voltage-dependent K+ channels. J. Pharmacol. Sci. 2015, 129, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Zarzuelo, M.J.; Lopez-Sepulveda, R.; Sanchez, M.; Romero, M.; Gomez-Guzman, M.; Ungvary, Z.; Perez-Vizcaino, F.; Jimenez, R.; Duarte, J. SIRT1 inhibits NADPH oxidase activation and protects endothelial function in the rat aorta: Implications for vascular aging. Biochem. Pharmacol. 2013, 85, 1288–1296. [Google Scholar] [CrossRef] [PubMed]
- Kurtel, H.; Rodrigues, S.F.; Yilmaz, C.E.; Yildirim, A.; Granger, D.N. Impaired vasomotor function induced by the combination of hypertension and hypercholesterolemia. J. Am. Soc. Hypertens. 2013, 7, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Suyama, A.; Tanaka, M.; Matsui, T. Ferulic acid enhances the vasorelaxant effect of epigallocatechin gallate in tumor necrosis factor-α-induced inflammatory rat aorta. J. Nutr. Biochem. 2014, 25, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Kumar, A.; Jaggi, A.S.; Singh, N. Efficacy of Cilostazol a selective phosphodiesterase-3 inhibitor in rat model of Streptozotocin diabetes induced vascular dementia. Pharmacol. Biochem. Behav. 2015, 135, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Ying, Z.; Xie, X.; Chen, M.; Yi, K.; Rajagopalan, S. α-lipoic acid activates eNOS through activation of PI3-kinase/Akt signaling pathway. Vascul. Pharmacol. 2015, 64, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Soloviev, A.; Zholos, A.; Ivanova, I.; Novokhatska, T.; Tishkin, S.; Raevska, A.; Stroyuk, A.; Yefanov, V. Plasmonic gold nanoparticles possess the ability to open potassium channels in rat thoracic aorta smooth muscles in a remote control manner. Vascul. Pharmacol. 2015, 72, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Ignarro, L.J.; Buga, G.M.; Wood, K.S.; Byrns, R.E.; Chaudhuri, G. Endothelium-derived relaxing factor produced and released from artery and vein is nitric oxide. Proc. Natl. Acad. Sci. USA 1987, 84, 9265–9269. [Google Scholar] [CrossRef] [PubMed]
- Moncada, S.; Korbut, R.; Bunting, S.; Vane, J.R. Prostacyclin is a circulating hormone. Nature 1978, 273, 767–768. [Google Scholar] [CrossRef] [PubMed]
- Palmer, R.M.; Ferrige, A.G.; Moncada, S. Nitric oxide release accounts for the biological activity of endothelium-derived relaxing factor. Nature 1987, 327, 524–526. [Google Scholar] [CrossRef] [PubMed]
- Wang, R. Hydrogen sulfide: A new EDRF. Kidney Int. 2009, 76, 700–704. [Google Scholar] [CrossRef] [PubMed]
- Quillon, A.; Fromy, B.; Debret, R. Endothelium microenvironment sensing leading to nitric oxide mediated vasodilation: A review of nervous and biomechanical signals. Nitric Oxide 2015, 45, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Vitecek, J.; Lojek, A.; Valacchi, G.; Kubala, L. Arginine-based inhibitors of nitric oxide synthase: therapeutic potential and challenges. Mediators Inflamm. 2012, 2012, 318087. [Google Scholar] [CrossRef] [PubMed]
- Furfine, E.S.; Harmon, M.F.; Paith, J.E.; Knowles, R.G.; Salter, M.; Kiff, R.J.; Duffy, C.; Hazelwood, R.; Oplinger, J.A.; Garvey, E.P. Potent and selective inhibition of human nitric oxide synthases. Selective inhibition of neuronal nitric oxide synthase by S-methyl-l-thiocitrulline and S-ethyl-l-thiocitrulline. J. Biol. Chem. 1994, 269, 26677–26683. [Google Scholar] [PubMed]
- Klatt, P.; Schmidt, K.; Brunner, F.; Mayer, B. Inhibitors of brain nitric oxide synthase. Binding kinetics, metabolism, and enzyme inactivation. J. Biol. Chem. 1994, 269, 1674–1680. [Google Scholar] [PubMed]
- Balligand, J.L.; Kelly, R.A.; Marsden, P.A.; Smith, T.W.; Michel, T. Control of cardiac muscle cell function by an endogenous nitric oxide signaling system. Proc. Natl. Acad. Sci. USA 1993, 90, 347–351. [Google Scholar] [CrossRef] [PubMed]
- Kubes, P.; Suzuki, M.; Granger, D.N. Nitric oxide: An endogenous modulator of leukocyte adhesion. Proc. Natl. Acad. Sci. USA 1991, 88, 4651–4655. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, S.; Leopold, E.; Schmidt, K.; Brunner, F.; Mayer, B. Inhibition of nitric oxide synthesis by NG-nitro-l-arginine methyl ester (l-NAME): Requirement for bioactivation to the free acid, NG-nitro-l-arginine. Br. J. Pharmacol. 1996, 118, 1433–1440. [Google Scholar] [CrossRef] [PubMed]
- Page, C.P.; Curtis, M.J.; Sutter, M. Farmacologia Integrada; Elsevier Espanha: Madrid, Spain, 1998. [Google Scholar]
- Suleyman, H.; Demircan, B.; Karagoz, Y. Anti-inflammatory and side effects of cyclooxygenase inhibitors. Pharmacol. Rep. 2007, 59, 247–258. [Google Scholar] [PubMed]
- Riendeau, D.; Percival, M.D.; Boyce, S.; Brideau, C.; Charleson, S.; Cromlish, W.; Ethier, D.; Evans, J.; Falgueyret, J.P.; Ford-Hutchinson, A.W.; et al. Biochemical and pharmacological profile of a tetrasubstituted furanone as a highly selective COX-2 inhibitor. Br. J. Pharmacol. 1997, 121, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Dannhardt, G.; Kiefer, W. Cyclooxygenase inhibitors—Current status and future prospects. Eur. J. Med. Chem. 2001, 36, 109–126. [Google Scholar] [CrossRef]
- Smith, W.L.; DeWitt, D.L.; Garavito, R.M. Cyclooxygenases: Structural, cellular, and molecular biology. Annu. Rev. Biochem. 2000, 69, 145–182. [Google Scholar] [CrossRef] [PubMed]
- Mayer, B.; Brunner, F.; Schmidt, K. Inhibition of nitric oxide synthesis by methylene blue. Biochem. Pharmacol. 1993, 45, 367–374. [Google Scholar] [CrossRef]
- Kontos, H.A.; Wei, E.P. Hydroxyl radical-dependent inactivation of guanylate cyclase in cerebral arterioles by methylene blue and by LY83583. Stroke 1993, 24, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Olson, L.J.; Knych, E.T., Jr.; Herzig, T.C.; Drewett, J.G. Selective guanylyl cyclase inhibitor reverses nitric oxide-induced vasorelaxation. Hypertension 1997, 29, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Moro, M.A.; Russel, R.J.; Cellek, S.; Lizasoain, I.; Su, Y.; Darley-Usmar, V.M.; Radomski, M.W.; Moncada, S. cGMP mediates the vascular and platelet actions of nitric oxide: confirmation using an inhibitor of the soluble guanylyl cyclase. Proc. Natl. Acad. Sci. USA 1996, 93, 1480–1485. [Google Scholar] [CrossRef] [PubMed]
- Schrammel, A.; Behrends, S.; Schmidt, K.; Koesling, D.; Mayer, B. Characterization of 1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one as a heme-site inhibitor of nitric oxide-sensitive guanylyl cyclase. Mol. Pharmacol. 1996, 50, 1–5. [Google Scholar] [PubMed]
- Valtcheva, N.; Nestorov, P.; Beck, A.; Russwurm, M.; Hillenbrand, M.; Weinmeister, P.; Feil, R. The commonly used cGMP-dependent protein kinase type I (cGKI) inhibitor Rp-8-Br-PET-cGMPS can activate cGKI in vitro and in intact cells. J. Biol. Chem. 2009, 284, 556–562. [Google Scholar] [CrossRef] [PubMed]
- Baltoumas, F.A.; Theodoropoulou, M.C.; Hamodrakas, S.J. Interactions of the α-subunits of heterotrimeric G-proteins with GPCRs, effectors and RGS proteins: A critical review and analysis of interacting surfaces, conformational shifts, structural diversity and electrostatic potentials. J. Struct. Biol. 2013, 182, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Weir, M.R.; Dzau, V.J. The renin-angiotensin-aldosterone system: A specific target for hypertension management. Am. J. Hypertens. 1999, 12, 205S–213S. [Google Scholar] [CrossRef]
- de Gasparo, M.; Catt, K.J.; Inagami, T.; Wright, J.W.; Unger, T. International union of pharmacology. XXIII. The angiotensin II receptors. Pharmacol. Rev. 2000, 52, 415–472. [Google Scholar] [PubMed]
- Jakala, P.; Pere, E.; Lehtinen, R.; Turpeinen, A.; Korpela, R.; Vapaatalo, H. Cardiovascular activity of milk casein-derived tripeptides and plant sterols in spontaneously hypertensive rats. J. Physiol. Pharmacol. 2009, 60, 11–20. [Google Scholar] [PubMed]
- Chen, J.; Yildiz, O.; Purdy, R.E. Phenylephrine precontraction increases the sensitivity of rabbit femoral artery to serotonin by enabling 5-HT1-like receptors. J. Cardiovasc. Pharmacol. 2000, 35, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Bockaert, J.; Claeysen, S.; Bécamel, C.; Dumuis, A.; Marin, P. Neuronal 5-HT metabotropic receptors: fine-tuning of their structure, signaling, and roles in synaptic modulation. Cell Tissue Res. 2006, 326, 553–572. [Google Scholar] [CrossRef] [PubMed]
- Schoeffter, P.; Hoyer, D. 5-Hydroxytryptamine (5-HT)-induced endothelium-dependent relaxation of pig coronary arteries is mediated by 5-HT receptors similar to the 5-HT1D receptor subtype. J. Pharmacol. Exp. Ther. 1990, 252, 387–395. [Google Scholar] [PubMed]
- Yildiz, O.; Cicek, S.; Ay, I.; Demirkilic, U.; Tuncer, M. Hypertension increases the contractions to sumatriptan in the human internal mammary artery. Ann. Thorac. Surg. 1996, 62, 1392–1395; discussion 1396. [Google Scholar] [CrossRef]
- Kuhr, F.; Lowry, J.; Zhang, Y.; Brovkovych, V.; Skidgel, R.A. Differential regulation of inducible and endothelial nitric oxide synthase by kinin B1 and B2 receptors. Neuropeptides 2010, 44, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Leeb-Lundberg, L.M.; Marceau, F.; Muller-Esterl, W.; Pettibone, D.J.; Zuraw, B.L. International union of pharmacology. XLV. Classification of the kinin receptor family: From molecular mechanisms to pathophysiological consequences. Pharmacol. Rev. 2005, 57, 27–77. [Google Scholar] [CrossRef] [PubMed]
- Davenport, A.P.; O′Reilly, G.; Molenaar, P.; Maguire, J.J.; Kuc, R.E.; Sharkey, A.; Bacon, C.R.; Ferro, A. Human endothelin receptors characterized using reverse transcriptase-polymerase chain reaction, in situ hybridization, and subtype-selective ligands BQ123 and BQ3020: Evidence for expression of ETB receptors in human vascular smooth muscle. J. Cardiovasc. Pharmacol. 1993, 22, S22–S25. [Google Scholar] [CrossRef] [PubMed]
- Levin, E.R. Endothelins. N. Engl. J. Med. 1995, 333, 356–363. [Google Scholar] [PubMed]
- Mazzuca, M.Q.; Khalil, R.A. Vascular endothelin receptor type B: Structure, function and dysregulation in vascular disease. Biochem. Pharmacol. 2012, 84, 147–162. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, Y.; Nakao, K.; Arai, H.; Nakagawa, O.; Hosoda, K.; Suga, S.; Nakanishi, S.; Imura, H. Molecular cloning of a non-isopeptide-selective human endothelin receptor. Biochem. Biophys. Res. Commun. 1991, 178, 248–255. [Google Scholar] [CrossRef]
- Seo, B.; Oemar, B.S.; Siebenmann, R.; Von Segesser, L.; Lüscher, T. Both ETA and ETB receptors mediate contraction to endothelin-1 in human blood vessels. Circulation 1994, 89, 1203–1208. [Google Scholar] [CrossRef] [PubMed]
- Caulfield, M.P.; Birdsall, N.J. International Union of Pharmacology. XVII. Classification of muscarinic acetylcholine receptors. Pharmacol. Rev. 1998, 50, 279–290. [Google Scholar] [PubMed]
- Chabner, B.; Brunton, L.; Knollman, B. Goodman and Gilman's The Pharmacological Basis of Therapeutics, 20th ed.; McGraw-Hill Education: New York, NY, USA, 2011. [Google Scholar]
- Ishii, M.; Kurachi, Y. Muscarinic acetylcholine receptors. Curr. Pharm. Des. 2006, 12, 3573–3581. [Google Scholar] [CrossRef] [PubMed]
- Benoist, C.C.; Wright, J.W.; Zhu, M.; Appleyard, S.M.; Wayman, G.A.; Harding, J.W. Facilitation of hippocampal synaptogenesis and spatial memory by C-terminal truncated Nle1-angiotensin IV analogs. J. Pharmacol. Exp. Ther. 2011, 339, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Catt, K.J.; Mendelsohn, F.A.; Millan, M.A.; Aguilera, G. The role of angiotensin II receptors in vascular regulation. J. Cardiovasc. Pharmacol. 1984, 6 (Suppl. 4), S575–S586. [Google Scholar] [CrossRef] [PubMed]
- D’Amore, A.; Black, M.J.; Thomas, W.G. The angiotensin II type 2 receptor causes constitutive growth of cardiomyocytes and does not antagonize angiotensin II type 1 receptor-mediated hypertrophy. Hypertension 2005, 46, 1347–1354. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.W.; Harding, J.W. Brain renin-angiotensin—A new look at an old system. Prog. Neurobiol. 2011, 95, 49–67. [Google Scholar] [CrossRef] [PubMed]
- Wagner, O.F.; Christ, G.; Wojta, J.; Vierhapper, H.; Parzer, S.; Nowotny, P.J.; Schneider, B.; Waldhausl, W.; Binder, B.R. Polar secretion of endothelin-1 by cultured endothelial cells. J. Biol. Chem. 1992, 267, 16066–16068. [Google Scholar] [PubMed]
- Cocks, T.M.; Angus, J.A. Endothelium-dependent relaxation of coronary arteries by noradrenaline and serotonin. Nature 1983, 305, 627–630. [Google Scholar] [CrossRef] [PubMed]
- He, G.W.; Yang, C.Q. Effect of thromboxane A2 antagonist GR32191B on prostanoid and nonprostanoid receptors in the human internal mammary artery. J. Cardiovasc. Pharmacol. 1995, 26, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Leemhuis, J.; Boutillier, S.; Schmidt, G.; Meyer, D.K. The protein kinase A inhibitor H89 acts on cell morphology by inhibiting Rho kinase. J. Pharmacol. Exp. Ther. 2002, 300, 1000–1007. [Google Scholar] [CrossRef] [PubMed]
- Petersen, R.K.; Madsen, L.; Pedersen, L.M.; Hallenborg, P.; Hagland, H.; Viste, K.; Doskeland, S.O.; Kristiansen, K. Cyclic AMP (cAMP)-mediated stimulation of adipocyte differentiation requires the synergistic action of Epac- and cAMP-dependent protein kinase-dependent processes. Mol. Cell. Biol. 2008, 28, 3804–3816. [Google Scholar] [CrossRef] [PubMed]
- Emery, A.C.; Eiden, M.V.; Eiden, L.E. A new site and mechanism of action for the widely used adenylate cyclase inhibitor SQ22,536. Mol. Pharmacol. 2013, 83, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Bouschet, T.; Perez, V.; Fernandez, C.; Bockaert, J.; Eychene, A.; Journot, L. Stimulation of the ERK pathway by GTP-loaded Rap1 requires the concomitant activation of Ras, protein kinase C, and protein kinase A in neuronal cells. J. Biol. Chem. 2003, 278, 4778–4785. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.; Bers, D.M. Protein kinase inhibitor H-89 reverses forskolin stimulation of cardiac l-type calcium current. Am. J. Physiol. 1995, 268, C651–C659. [Google Scholar] [PubMed]
- Satake, N.; Fujimoto, S.; Shibata, S. The potentiation of nitroglycerin-induced relaxation by PKG inhibition in rat aortic rings. Gen. Pharmacol. 1996, 27, 701–705. [Google Scholar] [CrossRef]
- Buch, J.G. Clinically Oriented Pharmacology; Version 2; PDU Medical College: Rajkot, Gujarat, India, 2010. [Google Scholar]
- Walch, L.; Gascard, J.P.; Dulmet, E.; Brink, C.; Norel, X. Evidence for a M1 muscarinic receptor on the endothelium of human pulmonary veins. Br. J. Pharmacol. 2000, 130, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, A.K.; Pomponio, J.W.; Evans, S.A.; Pervitsky, D.; Gordon, R.K. Discovery of subtype selective muscarinic receptor antagonists as alternatives to atropine using in silico pharmacophore modeling and virtual screening methods. Bioorg. Med. Chem. 2013, 21, 2651–2662. [Google Scholar] [CrossRef] [PubMed]
- Zholos, A.V.; Bolton, T.B. Muscarinic receptor subtypes controlling the cationic current in guinea-pig ileal smooth muscle. Br. J. Pharmacol. 1997, 122, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Archer, S.L.; Rusch, N.J. Potassium Channels in Cardiovascular Biology; Springer: New York, NY, USA, 2001. [Google Scholar]
- Burnham, M.P.; Bychkov, R.; Feletou, M.; Richards, G.R.; Vanhoutte, P.M.; Weston, A.H.; Edwards, G. Characterization of an apamin-sensitive small-conductance Ca2+-activated K+ channel in porcine coronary artery endothelium: Relevance to EDHF. Br. J. Pharmacol. 2002, 135, 1133–1143. [Google Scholar] [CrossRef] [PubMed]
- Bychkov, R.; Burnham, M.P.; Richards, G.R.; Edwards, G.; Weston, A.H.; Feletou, M.; Vanhoutte, P.M. Characterization of a charybdotoxin-sensitive intermediate conductance Ca2+-activated K+ channel in porcine coronary endothelium: relevance to EDHF. Br. J. Pharmacol. 2002, 137, 1346–1354. [Google Scholar] [CrossRef] [PubMed]
- Feletou, M.; Vanhoutte, P. EDHF: The Complete Story; CRC Press: Hoboken, NJ, USA, 2005. [Google Scholar]
- Frieden, M.; Sollini, M.; Beny, J. Substance P and bradykinin activate different types of KCa currents to hyperpolarize cultured porcine coronary artery endothelial cells. J. Physiol. 1999, 519, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Marchenko, S.M.; Sage, S.O. Calcium-activated potassium channels in the endothelium of intact rat aorta. J. Physiol. 1996, 492, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.S.; Bonev, A.D.; Gross, T.P.; Eckman, D.M.; Brayden, J.E.; Bond, C.T.; Adelman, J.P.; Nelson, M.T. Altered expression of small-conductance Ca2+-activated K+ (SK3) channels modulates arterial tone and blood pressure. Circ. Res. 2003, 93, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Hille, B. The selective inhibition of delayed potassium currents in nerve by tetraethylammonium ion. J. Gen. Physiol. 1967, 50, 1287–1302. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.A.; Rivard, A.F.; Bachinger, H.P.; Adelman, J.P. Structure of the gating domain of a Ca2+-activated K+ channel complexed with Ca2+/calmodulin. Nature 2001, 410, 1120–1124. [Google Scholar] [CrossRef] [PubMed]
- Boyd, A.E., 3rd; Aguilar-Bryan, L.; Nelson, D.A. Molecular mechanisms of action of glyburide on the β cell. Am. J. Med. 1990, 89, 3S–10S. [Google Scholar] [CrossRef]
- Standen, N.B.; Quayle, J.M.; Davies, N.W.; Brayden, J.E.; Huang, Y.; Nelson, M.T. Hyperpolarizing vasodilators activate ATP-sensitive K+ channels in arterial smooth muscle. Science 1989, 245, 177–180. [Google Scholar] [CrossRef] [PubMed]
- Katsuda, Y.; Egashira, K.; Ueno, H.; Akatsuka, Y.; Narishige, T.; Arai, Y.; Takayanagi, T.; Shimokawa, H.; Takeshita, A. Glibenclamide, a selective inhibitor of ATP-sensitive K+ channels, attenuates metabolic coronary vasodilatation induced by pacing tachycardia in dogs. Circulation 1995, 92, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Chandy, K.G.; Wulff, H.; Beeton, C.; Pennington, M.; Gutman, G.A.; Cahalan, M.D. K+ channels as targets for specific immunomodulation. Trends Pharmacol. Sci. 2004, 25, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Feske, S.; Gwack, Y.; Prakriya, M.; Srikanth, S.; Puppel, S.H.; Tanasa, B.; Hogan, P.G.; Lewis, R.S.; Daly, M.; Rao, A. A mutation in Orai1 causes immune deficiency by abrogating CRAC channel function. Nature 2006, 441, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Wellman, G.C.; Bevan, J.A. Barium inhibits the endothelium-dependent component of flow but not acetylcholine-induced relaxation in isolated rabbit cerebral arteries. J. Pharmacol. Exp. Ther. 1995, 274, 47–53. [Google Scholar] [PubMed]
- Golan, D.E.; Tashjian, A.H.; Armstrong, E.J. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy; Wolters Kluwer Health: Philadelphia, PA, USA, 2011. [Google Scholar]
- Furberg, C.D.; Psaty, B.M.; Meyer, J.V. Nifedipine. Dose-related increase in mortality in patients with coronary heart disease. Circulation 1995, 92, 1326–1331. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.T.; Venkatachalam, K.; Parys, J.B.; Gill, D.L. Modification of store-operated channel coupling and inositol trisphosphate receptor function by 2-aminoethoxydiphenyl borate in DT40 lymphocytes. J. Biol. Chem. 2002, 277, 6915–6922. [Google Scholar] [CrossRef] [PubMed]
- Prakriya, M.; Lewis, R.S. Potentiation and inhibition of Ca2+ release-activated Ca2+ channels by 2-aminoethyldiphenyl borate (2-APB) occurs independently of IP3 receptors. J. Physiol. 2001, 536, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Bootman, M.D.; Collins, T.J.; MacKenzie, L.; Roderick, H.L.; Berridge, M.J.; Peppiatt, C.M. 2-aminoethoxydiphenyl borate (2-APB) is a reliable blocker of store-operated Ca2+ entry but an inconsistent inhibitor of InsP3-induced Ca2+ release. FASEB J. 2002, 16, 1145–1150. [Google Scholar] [CrossRef] [PubMed]
- Adding, L.C.; Bannenberg, G.L.; Gustafsson, L.E. Basic experimental studies and clinical aspects of gadolinium salts and chelates. Cardiovasc. Drug Rev. 2001, 19, 41–56. [Google Scholar] [CrossRef] [PubMed]
- Fellner, S.K.; Arendshorst, W.J. Store-operated Ca2+ entry is exaggerated in fresh preglomerular vascular smooth muscle cells of SHR. Kidney Int. 2002, 61, 2132–2141. [Google Scholar] [CrossRef] [PubMed]
- Rogers, T.B.; Inesi, G.; Wade, R.; Lederer, W. Use of thapsigargin to study Ca2+ homeostasis in cardiac cells. Biosci. Rep. 1995, 15, 341–349. [Google Scholar] [CrossRef] [PubMed]
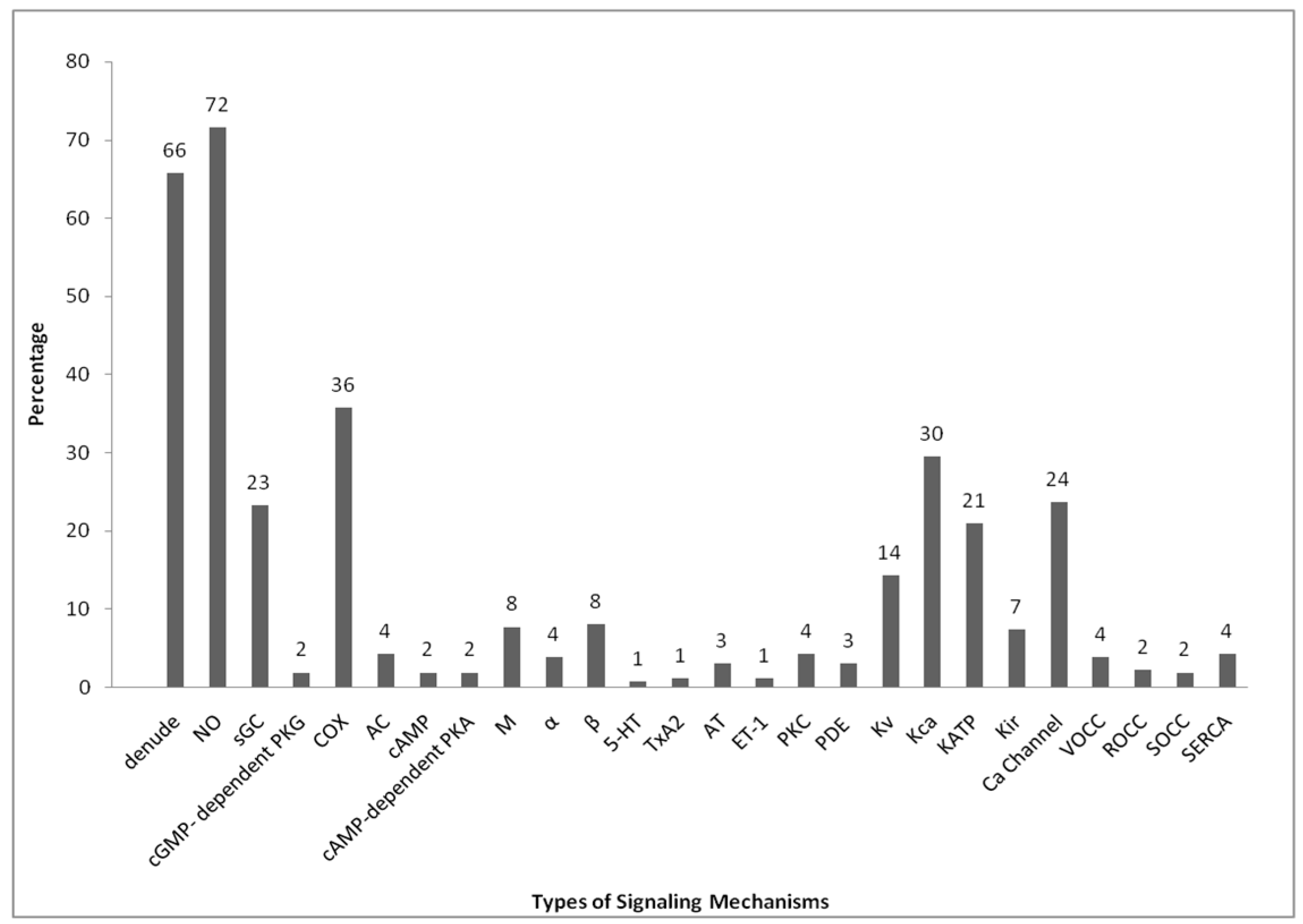
| TOV | D | NO | GC | PKG | COX | AC | cAMP | PKA | M | α | β | Sr | T | AT | ET | PKC | PDE | Kv | Kca | KATP | Kir | Ca | V | R | S | SE | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| A | + | + | + | - | + | - | - | - | + | + | + | - | - | - | - | - | - | - | - | + | - | + | - | - | - | - | [6] |
| MA | + | + | + | - | + | + | - | - | - | - | - | - | - | - | - | - | - | + | + | + | + | + | - | - | - | - | [7] |
| A | + | + | + | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | + | - | - | + | - | - | - | - | [8] |
| A & SA | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [9] |
| A | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [10] |
| A | + | + | + | - | + | - | - | - | + | - | + | + | - | - | - | + | - | + | + | + | + | + | - | - | - | - | [11] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [12] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [13] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | [14] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [15] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | + | + | - | - | - | - | [16] |
| A | + | + | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [17] |
| MA | + | + | + | - | + | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [18] |
| A | + | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | [19] | ||
| A | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [20] |
| A & MA | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [21] |
| A | + | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | + | - | - | - | - | [22] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | - | - | - | - | - | - | [23] |
| RAS | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | - | - | - | - | - | - | [24] |
| A | + | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [25] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [26] |
| A | + | + | + | + | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | [27] |
| RAS | - | + | - | - | + | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | [28] |
| A | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | + | - | - | - | - | [29] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | + | - | - | - | - | - | [30] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [31] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | + | - | - | - | - | - | [32] |
| A | + | + | + | - | + | - | - | - | + | - | + | - | - | - | - | - | - | - | + | + | - | + | - | - | + | + | [33] |
| A | + | + | - | - | + | - | - | - | + | - | - | - | - | - | - | - | - | - | + | + | + | + | - | - | - | - | [34] |
| CA | + | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | + | + | + | + | + | - | - | - | - | [35] |
| A | + | + | + | - | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [36] |
| A | + | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [37] |
| GA, BLA & A | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [38] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [39] |
| CA | - | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [40] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | + | - | - | - | [41] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | [42] |
| MA | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | - | [43] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [44] |
| A | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | + | - | - | - | - | - | - | + | [45] |
| A | - | + | - | - | + | - | - | - | + | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | - | [46] |
| A | + | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | [47] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [48] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | [49] |
| MA | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [50] |
| A | + | + | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [51] |
| A | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [52] |
| A | + | - | - | - | - | - | - | - | - | + | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | [53] |
| IMA | - | + | + | - | + | - | - | - | + | - | - | - | - | - | + | - | - | + | + | + | + | - | - | - | - | - | [54] |
| A | + | + | - | - | - | - | - | - | - | + | + | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | [55] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | [56] |
| IMA | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | + | - | - | - | + | [57] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | + | + | - | - | - | - | [58] |
| A | + | + | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [59] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [60] |
| A | + | + | + | - | + | - | - | - | + | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | [61] |
| CBA | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [62] |
| A | + | + | + | - | + | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [63] |
| A | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [64] |
| A | + | + | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | + | + | - | - | - | - | - | - | [65] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [66] |
| MA | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | [67] |
| A | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | + | + | - | - | - | - | [68] |
| A | + | + | + | - | + | - | - | - | - | - | + | - | - | - | - | - | - | - | + | + | + | + | - | - | - | - | [69] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | [70] |
| CA | - | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [71] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [72] |
| A | + | + | - | - | + | - | - | - | + | - | + | - | - | - | - | - | - | - | + | + | - | - | - | - | - | - | [73] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [74] |
| A | + | + | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [75] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [76] |
| RA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | [77] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [78] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | [79] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [80] |
| A | - | + | - | - | - | - | - | - | - | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [81] |
| A | + | + | + | - | + | - | - | - | + | - | + | - | - | - | - | - | - | - | + | + | - | - | - | - | - | - | [82] |
| MA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | [83] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | + | + | - | - | - | - | - | - | - | - | - | - | - | - | [84] |
| A | - | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | + | - | - | - | - | [85] |
| A | + | - | - | - | + | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | [86] |
| A | + | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [87] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [88] |
| A | + | + | + | - | + | - | - | + | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | - | [89] |
| A | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | - | [90] |
| A | + | + | + | - | + | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [91] |
| A | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [92] |
| A | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [93] |
| A | + | + | + | - | + | - | - | - | + | - | + | - | - | - | - | - | - | - | + | + | - | + | - | - | + | + | [94] |
| A | + | + | + | - | + | - | - | - | + | - | + | - | - | - | - | - | - | - | + | + | - | + | - | - | + | + | [95] |
| CRA | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | + | - | - | + | + | [96] |
| A | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [97] |
| A | + | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | + | [98] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | + | - | - | - | - | [99] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [100] |
| A | - | + | - | - | - | - | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [101] |
| BA | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | + | + | - | - | - | - | [102] |
| A | + | + | + | - | + | - | - | - | + | - | + | - | - | - | - | - | - | + | + | + | - | + | + | + | - | - | [103] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [104] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [105] |
| A | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | [106] |
| A | + | + | + | - | + | - | - | - | - | - | + | - | - | - | - | + | - | + | + | + | - | + | - | - | - | - | [107] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | + | + | + | - | + | [108] |
| A | + | + | + | + | + | - | - | - | + | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [109] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | + | - | - | - | - | [110] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [111] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [112] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [113] |
| CA | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [114] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [115] |
| RA & A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [116] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [117] |
| RAS | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | + | + | - | - | - | [118] |
| A | + | + | - | - | + | - | - | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [119] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [120] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [121] |
| A & TA | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [122] |
| PA | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [123] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | [124] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [125] |
| FA | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [126] |
| MA & A | + | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | - | [127] |
| CA | + | + | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | [128] |
| CA & SPA | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | + | - | - | + | - | - | - | - | [129] |
| CRA | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | - | [130] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [131] |
| MA | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [132] |
| A | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [133] |
| A | + | + | + | - | + | - | - | - | + | - | + | - | - | - | - | - | - | - | + | + | - | + | - | - | + | + | [134] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [135] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | - | [136] |
| PA | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [137] |
| PA | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [138] |
| BA | - | + | - | - | + | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [139] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [140] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [141] |
| A | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [142] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [143] |
| IA | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [144] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [145] |
| CA | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [146] |
| A | + | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [147] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [148] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [149] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [150] |
| RA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [151] |
| VA | - | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [152] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | - | - | - | - | - | - | - | [153] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | - | - | - | - | - | - | - | [154] |
| A | + | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | [155] |
| GCA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [156] |
| PA | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [157] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [158] |
| A | + | + | + | - | + | + | - | - | - | - | - | - | - | - | - | + | - | - | + | + | - | + | - | - | - | - | [159] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | - | + | - | - | - | - | - | [160] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [161] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [162] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [163] |
| MA | - | + | - | - | + | + | - | + | - | - | + | - | - | - | - | - | - | - | + | + | - | - | - | - | - | - | [164] |
| CA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [165] |
| A | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [166] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [167] |
| GCA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [168] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | [169] |
| A | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [170] |
| PA | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [171] |
| A | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [172] |
| CA | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [173] |
| MA | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [174] |
| A | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [175] |
| A | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [176] |
| CA | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [177] |
| A | + | - | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [178] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | + | + | + | - | - | [179] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | + | + | + | - | - | [180] |
| A | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [181] |
| FA | + | + | - | - | + | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | [182] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | [183] |
| CA | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | + | - | - | - | - | [184] |
| A | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [185] |
| A | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [186] |
| A | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | [187] |
| PA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [188] |
| A | + | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [189] |
| MA | + | + | + | - | + | - | - | - | + | - | - | - | - | - | - | - | - | - | + | - | - | + | - | - | - | - | [190] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [191] |
| A | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [192] |
| PA | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | + | + | - | - | - | - | [193] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [194] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [195] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [196] |
| PA | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [197] |
| BA | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [198] |
| PA | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [199] |
| BA | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | + | - | - | - | - | - | [200] |
| PSA | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | + | - | - | - | - | [201] |
| A | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | - | [202] |
| MA | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | - | [203] |
| CA & FA | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [204] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | + | + | + | - | - | [205] |
| RA | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | - | [206] |
| AA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [207] |
| A | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [208] |
| A | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [209] |
| TA | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [210] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [211] |
| A | + | + | - | - | - | - | - | + | - | - | - | - | - | - | - | + | - | - | + | - | - | - | - | - | - | [212] | |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | + | - | - | - | - | [213] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | + | - | - | + | + | + | - | - | [214] |
| PA | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | [215] |
| RAS | + | + | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | + | + | + | - | - | - | - | - | - | [216] |
| MA | - | - | - | - | + | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [217] |
| CA | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [218] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | + | [219] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [220] |
| A | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [221] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [222] |
| MA | - | + | + | + | + | + | + | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | + | - | - | - | [223] |
| CRA | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | + | - | - | - | - | [224] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [225] |
| A, CA, MA & RA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [226] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [227] |
| A & FA | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [228] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [229] |
| A | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [230] |
| RA | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | + | - | - | - | - | [231] |
| MA | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [232] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [233] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [234] |
| MA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [235] |
| RSA | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [236] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [237] |
| A | + | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [238] |
| A | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [239] |
| MA | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [240] |
| GS | - | + | - | - | - | - | - | + | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | [241] |
| OPA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [242] |
| A | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [243] |
| CRA | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [244] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [245] |
| MA | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [246] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [247] |
| CA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | [248] |
| MA | - | + | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [249] |
| CBA, BA & MA | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | [250] |
| CA | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [251] |
| A | + | + | + | - | - | - | - | - | + | - | + | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | [252] |
| A | + | + | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [253] |
| A & MA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [254] |
| OPA | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [255] |
| MA & OMA | + | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | [256] |
| A | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [257] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | [258] |
| A | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [259] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [260] |
| A | + | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | [261] |
| A | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | - | - | - | - | - | [262] |
| Mechanism Pathways | Antagonists | Ref. | |
|---|---|---|---|
| Selective | Non-Selective | ||
| Endothelial nitric oxide synthase (eNOS) | l-NG-NitroArginine-Methyl Ester, l-NG-NitroArginine | [268,269,270,271,272,273] | |
| Cyclooxygenase (COX) | Ibuprofen, mefenamic acid, piroxicam, diclofenac, flubiprofen, indomethacin, aspirin | [276,277,278] | |
| Soluble guanylyl cycles (sGC) | 1H-[1,2,4] oxadiazolo [4,3-a]-quinoxalin-1-one | Methylene blue | [279,281,282,283] |
| Protein kinase G (PKG) | KT 5823 | Rp-8-Br-PET-cGMPs | [284] |
| Adenylyl cyclase (AC) | SQ22536, MDL12330A, 2′5′-dideoxyadenosine | [312] | |
| Protein kinase A (PKA) | Rp-cAMPs, KT 5720, H-89 | [313,314,315] | |
| β-adrenergic receptor | Atenolol | Propanolol, nadolol, pindolol | [316] |
| α-adrenergic receptor | Prazosin (α1), RX 821002 (α2) | [288,292] | |
| Endothelin receptor | BQ 123 (ETAR), BQ 788 (ETBR) | Bosentan | [295,296,297,298,299] |
| Angiotensin II receptor (AT1) | Candesartan cilexetil (ARB & AT1), losartan (AT1), valsartan (ARB & AT1) | [286,287,288,303,304,305,306] | |
| Muscarinic receptor | N-methylatropine, tiotropium | Atropine | [300,301,302,318,319] |
| Thromboxane (TxA2) | Ridogrel, ozagrel, furegrelate | [288,292,309] | |
| Serotonin receptor (Sr) | Katanserin (5-HT2), WAY 100635 (5-HT1A) | [289,290,291,292] | |
| Protein kinase C (PKC) | GF 109203x, staurosporine, Go 6983, chelerythrine, BIM | [286,287,288] | |
| Phosphodiesterase (PDE) | Slidenafil (PDE 5), dipyridamole (PDE 5), zaprinast (PDE 5), T 1032 (PDE 5), rolipram (PDE 4), milrinone (PDE 3) | Papaverine | [152] |
| Calcium-activated potassium channel (Kca) | Iberiotoxin (BKca), UCL 1684 (BKca), paxilline (BKca), apamin (SKca), TRAM-34 (IKca), clotrimazole (IKca) | Charybdotoxin (BKca & IKca), TEA | [323,327,328] |
| ATP-sensitive potassium channel (KATP) | Glibenclamide | [330,331] | |
| Voltage-activated potassium channel (Kv) | XE 991 (KV 7) | 4-aminopyridine | [323,332,333] |
| Inwardly-rectifier potassium channel (Kir) | Barium chloride | [334] | |
| Voltage-operated calcium channel (VOCC) | Nifedipine, nicardipine, diltiazem | Verapamil | [335,336] |
| Store-operated calcium channel (SOCC) | Gandolinium | [340,341] | |
| Inositol triphosphate receptor (IP3R) | 2-Aminoethoxydiphenyl borate | [337,338,339,340,341] | |
| SERCA | Thapsigargin, cyclopiazonic acid | [342] | |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loh, Y.C.; Tan, C.S.; Ch’ng, Y.S.; Ahmad, M.; Asmawi, M.Z.; Yam, M.F. Overview of Antagonists Used for Determining the Mechanisms of Action Employed by Potential Vasodilators with Their Suggested Signaling Pathways. Molecules 2016, 21, 495. https://doi.org/10.3390/molecules21040495
Loh YC, Tan CS, Ch’ng YS, Ahmad M, Asmawi MZ, Yam MF. Overview of Antagonists Used for Determining the Mechanisms of Action Employed by Potential Vasodilators with Their Suggested Signaling Pathways. Molecules. 2016; 21(4):495. https://doi.org/10.3390/molecules21040495
Chicago/Turabian StyleLoh, Yean Chun, Chu Shan Tan, Yung Sing Ch’ng, Mariam Ahmad, Mohd Zaini Asmawi, and Mun Fei Yam. 2016. "Overview of Antagonists Used for Determining the Mechanisms of Action Employed by Potential Vasodilators with Their Suggested Signaling Pathways" Molecules 21, no. 4: 495. https://doi.org/10.3390/molecules21040495






