2.4.2. Identification of Warfarin and Ibuprofen Binding Sites
Crystallographic analyses show that HSA, a 585 amino acid residue monomer protein, contains three homologous α-helical domains (domains I–III), each of which includes subdomain A and B, to comprise six subdomains, IA, IB, IIA, IIB, IIIA, and IIIB. The two subdomains in each domain constitute a hydrophobic cavity [
25]. HSA has a limited number of binding sites for a variety of ligands that are typically bound reversibly [
1]. Crystal structure analyses also indicate that the two principal regions of ligand binding sites in albumin, site I and site II, are positioned in hydrophobic cavities. Site I, located within subdomain IIA, preferentially integrates with bulky heterocyclic ligands, such as warfarin and, bilirubin [
9,
26]. Site II, located within subdomain IIIA, preferentially recognizes aromatic compounds, and is more active in the accommodation of ligands, for example, digitoxin, ibuprofen, and tryptophan [
27].
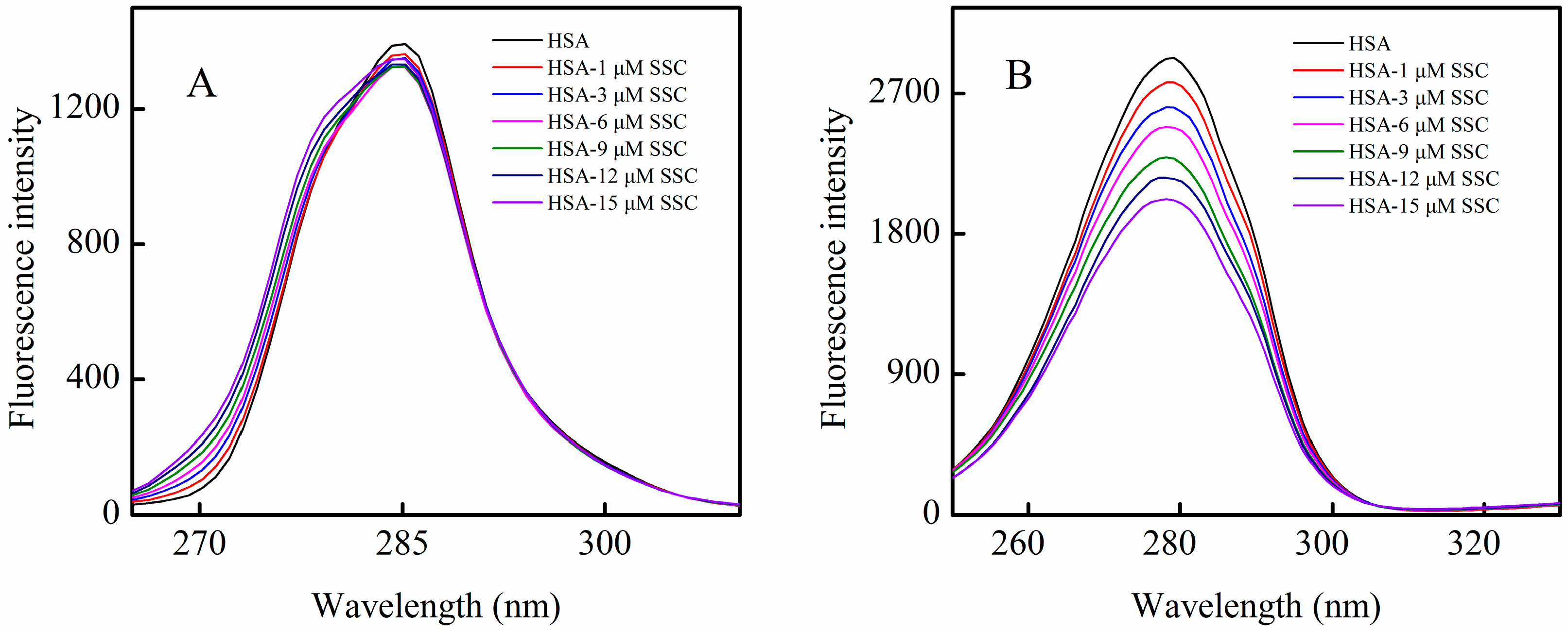
In order to identify SSC binding sites on the region of HSA molecule, site competitive binding experiments have been commonly carried out. Warfarin (an anticoagulant drug, a marker of site I) and ibuprofen (a nonsteroidal anti-inflammatory agent, a marker of site II) were utilized in the present experiment. Following the addition of SSC into the HSA-warfarin solution, the fluorescence intensity of HSA became significantly reduced (
Figure 6A). A shift in fluorescence spectra at the maximum emission wavelength was not found in the warfarin mixture system, while the minor blue shift was observed at ∆λ = 15 nm or ∆λ = 60 nm in
Figure 4, suggesting that the binding of warfarin to HSA inhibits the formation of hydrophobic structures surrounding tyrosine or tryptophan residues in HSA-SSC complex. As shown in
Figure 6B, following the addition of SSC into the HSA-ibuprofen solution, HSA fluorescence intensity weakened dose-dependently. In addition, ibuprofen induced a blue shit of 11 nm (from 336 nm to 325 nm) in the ibuprofen mixture system, indicating that the hydrophobicity of amino acid residues arises around ibuprofen. The competitive effect of ibuprofen on HSA binding reveals that SSC mainly binds site II (subdomain IIIA) in HSA.
Figure 6.
Effect of site marker probes on the SSC-HSA system. The experiment was carried out at CHSA = 2 μM, T = 26 °C, pH = 7.4, and λex = 280 nm. (A) Cwarfarin = 0.025 M; (B) Cibuprofen = 0.25 M; (C) Double-reciprocal plots for log (F0 − F)/F vs. log [Q]; [Q], CSSC = 0, 1, 3, 6, 9, and 12 μM. War, Warfarin; Ibu, Ibuprofen.
Figure 6.
Effect of site marker probes on the SSC-HSA system. The experiment was carried out at CHSA = 2 μM, T = 26 °C, pH = 7.4, and λex = 280 nm. (A) Cwarfarin = 0.025 M; (B) Cibuprofen = 0.25 M; (C) Double-reciprocal plots for log (F0 − F)/F vs. log [Q]; [Q], CSSC = 0, 1, 3, 6, 9, and 12 μM. War, Warfarin; Ibu, Ibuprofen.
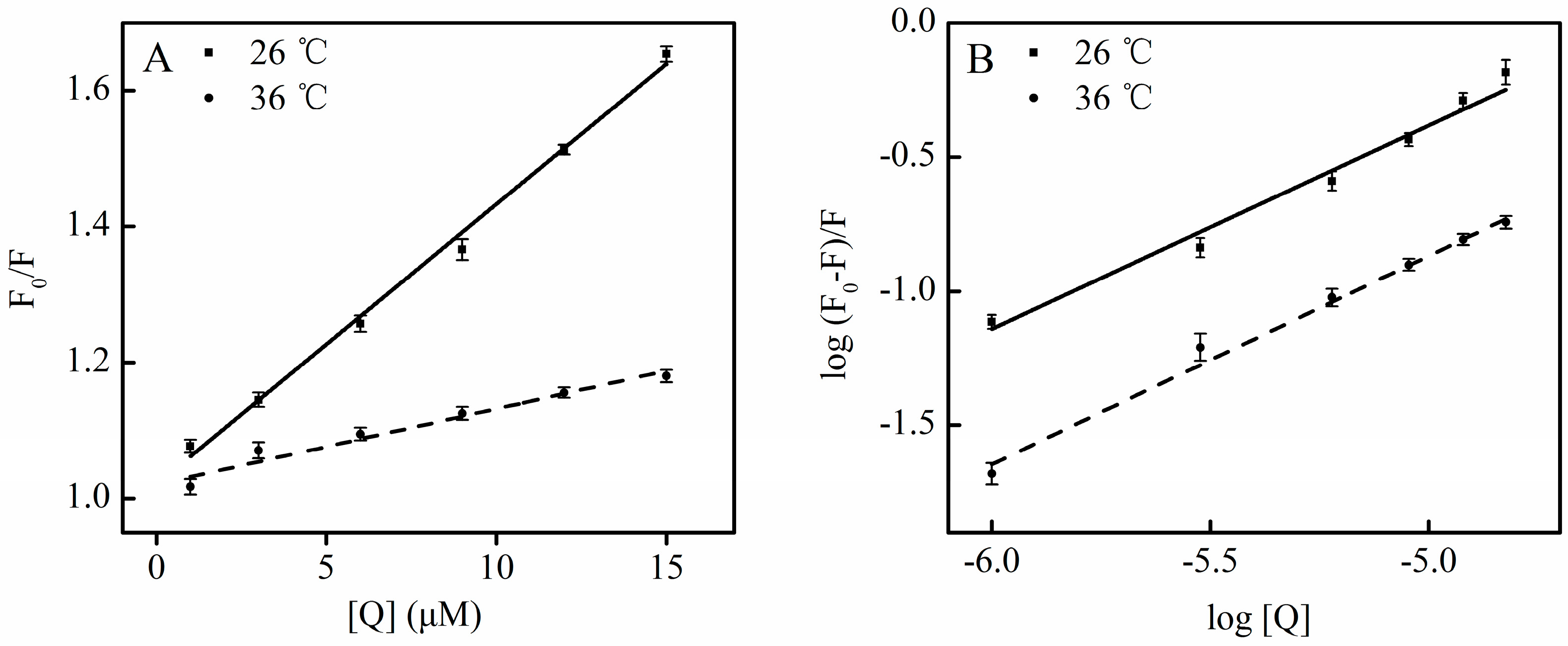
According to the Equation (4), the corresponding plot can be obtained from the fluorescence data (
Figure 6C) [
28]. The
Ka in the warfarin or ibuprofen mixture was 4.38 × 10
4 L·mol
−1 or 2.22 × 10
3 L·mol
−1, respectively (
Table 3). Although the binding constants in the study system are low, lower binding constants (about 10
3 L·mol
−1) have also been reported for protein–ligand interactions [
29]. Commonly, weak binding to the protein results in a shorter lifetime or poor distribution of drugs in plasma, whereas the strong binding reduces the levels of free plasma drugs [
10]. The results showed that the
Ka of the warfarin complex was much higher than that of SSC-HSA binding, whereas the
Ka of the ibuprofen complex is below the binding constant of SSC and HSA (
Table 3). Confusedly, the
Ka in the mixture system is higher in the presence of warfarin. Obviously, the effect requires further elucidation in the future. Albumin-binding events play an essential role in the transportation, distribution and free fraction of drugs. Free drugs are responsible for pharmacological activities [
30]. The calculated results show that the effect of warfarin or ibuprofen is of practical significance in clinical application. If warfarin is given at the same time with SSC, the warfarin-HSA binding will be able to regulate the binding of SSC and HSA, extend the retention time in plasma, decrease free concentration, and inhibit the effect of SSC/Chaihu. If ibuprofen combines with SSC at the same time, the ibuprofen-HSA interaction can influence SSC and HSA binding, elevate the free concentration, and enhance the effect of SSC/Chaihu. Warfarin, a coumarin representative, and other coumarin anticoagulants, such as acenocoumarol and phenprocoumon, widely exist, not only among Chinese herbal medicines, but also among prescribed drugs worldwide [
31]. Ibuprofen, a classical anti-inflammatory drug, and its derivatives are also among the most common prescribed drugs in the world [
32]. Chaihu enriching SSC is often prescribed as a major herbal medicine both in some classical Chinese medicinal formulae, such as “xiao yao powder”, and in any prescription, such as Chaihu and
Scutellaria Baicalensis, Chaihu and peony, and Chaihu and
Liquorice [
33]. Inevitably, with the extensive application of Chaihu in clinical practice, SSC commonly interacts not only with co-administered medicinal herbs, but also likely with co-treated modern drugs. In short, the effect of SSC/Chaihu is likely weakened by warfarin-class drugs, whereas the effect of SSC/Chaihu is likely strengthened when ibuprofen-class drugs and SSC/Chaihu are co-administered.
Table 3.
Binding reactive parameters in site competitive experiments.
Table 3.
Binding reactive parameters in site competitive experiments.
| System | Equation | Ka (L·mol−1) ± SD | R |
|---|
| Warfarin | Y = 0.78X + 3.64 | 4.38 × 104 ± 11.65 * | 0.991 |
| Ibuprofen | Y = 0.87X + 3.35 | 2.22 × 103 ± 3.75 * | 0.985 |
| HSA-SSC | Y = 0.79X + 3.57 | 3.72 × 103 ± 4.76 | 0.983 |
2.4.3. Binding Forces between SSC and HSA
The binding forces between a small molecule and a macromolecule can include several types of interactions, such as hydrogen bonds, van der Waals forces, electrostatic forces, and hydrophobic interactions [
34,
35]. The thermodynamic parameters, enthalpy change (∆
H) and entropy change (∆
S), are the major indicators to confirm binding modes. Based on a number of experimental results, Ross summarized the thermodynamic relationship between proteins and small molecules as follows: ∆
H < 0 and ∆
S < 0 indicates van der Waals force or hydrogen bond formation; ∆
H < 0 and ∆
S > 0 implies an electrostatic force; and ∆
H > 0 and ∆
S > 0 reflects hydrophobic interaction [
36].
The enthalpy change can be considered as a constant when the effect of tested temperatures on binding constants is not statistically significant (
Table 2). From the temperature dependence of the binding constants, the parameters involved in the process were calculated using the following equations (
Table 4) [
37,
38,
39]:
where
Ka is the binding constant calculated by Equation (4), and
R is the gas constant. As shown in
Table 4, the negative value of the free energy change (∆
G) means a spontaneous binding process. Both negative enthalpy and entropy values reveal that van der Waals interactions and hydrogen bonds play a major role in the binding process between SSC and HSA (
Table 4). Therefore, the interaction between SSC and HSA is a forward reaction involving the reduction of free energy change, and enthalpy change and entropy change at a low temperature, which is mainly driven by enthalpy because the entropy value was negative and unfavorable for the binding process.
Table 4.
Thermodynamic parameters of SSC-HSA interaction.
Table 4.
Thermodynamic parameters of SSC-HSA interaction.
| Fluorescence | T (°C) | λex | ∆G (kJ·mol−1) | ∆H (kJ·mol−1) | ∆S (J·mol−1·K−1) |
|---|
| Conventional | 26 | 280 nm | −20.34 | −47.34 | −90.31 |
| | 36 | 280 nm | −19.52 | −90.04 |