Coumarins as Potential Antioxidant Agents Complemented with Suggested Mechanisms and Approved by Molecular Modeling Studies
Abstract
:1. Introduction
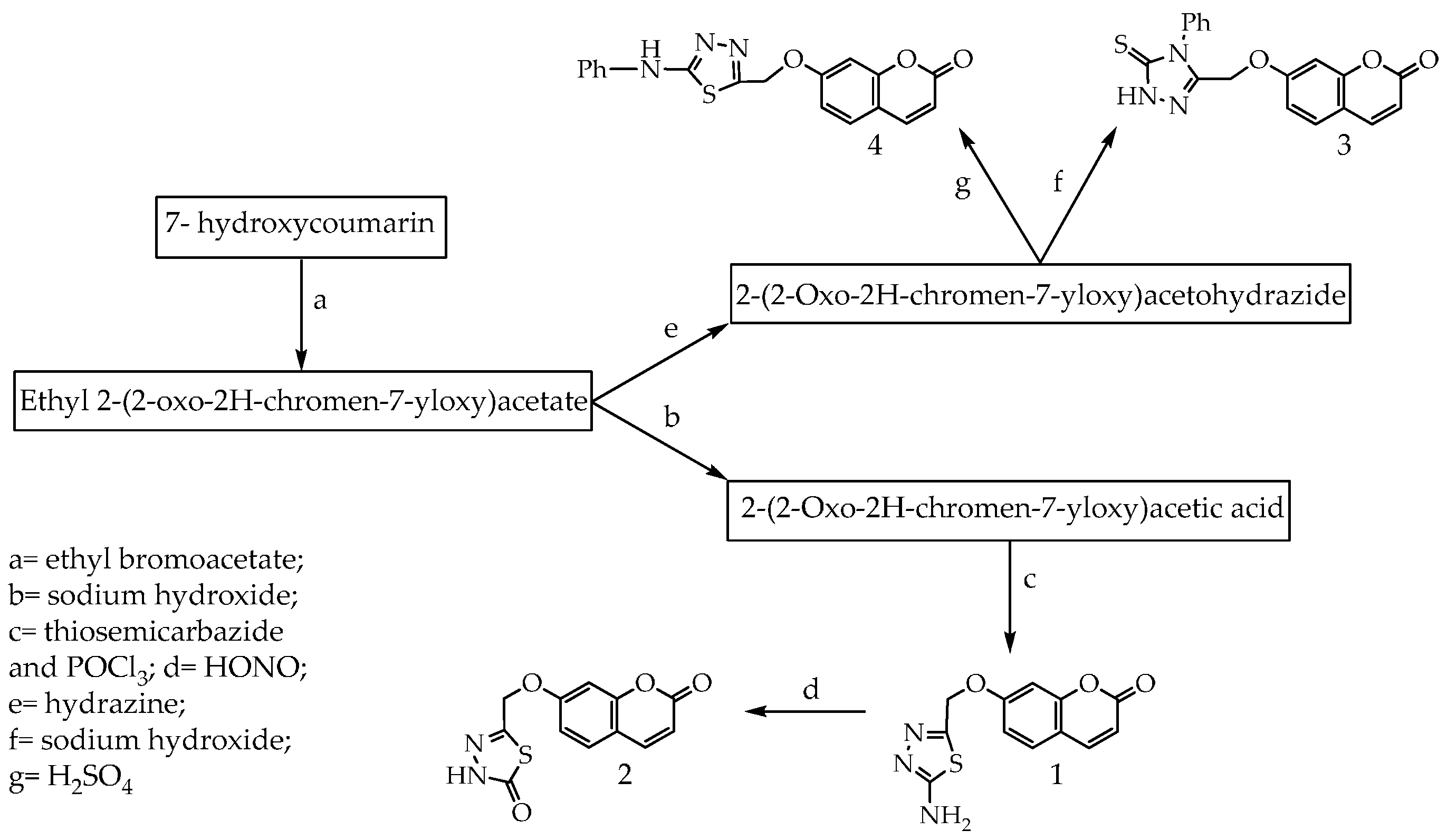
2. Results and Discussion
2.1. Antioxidant Activity
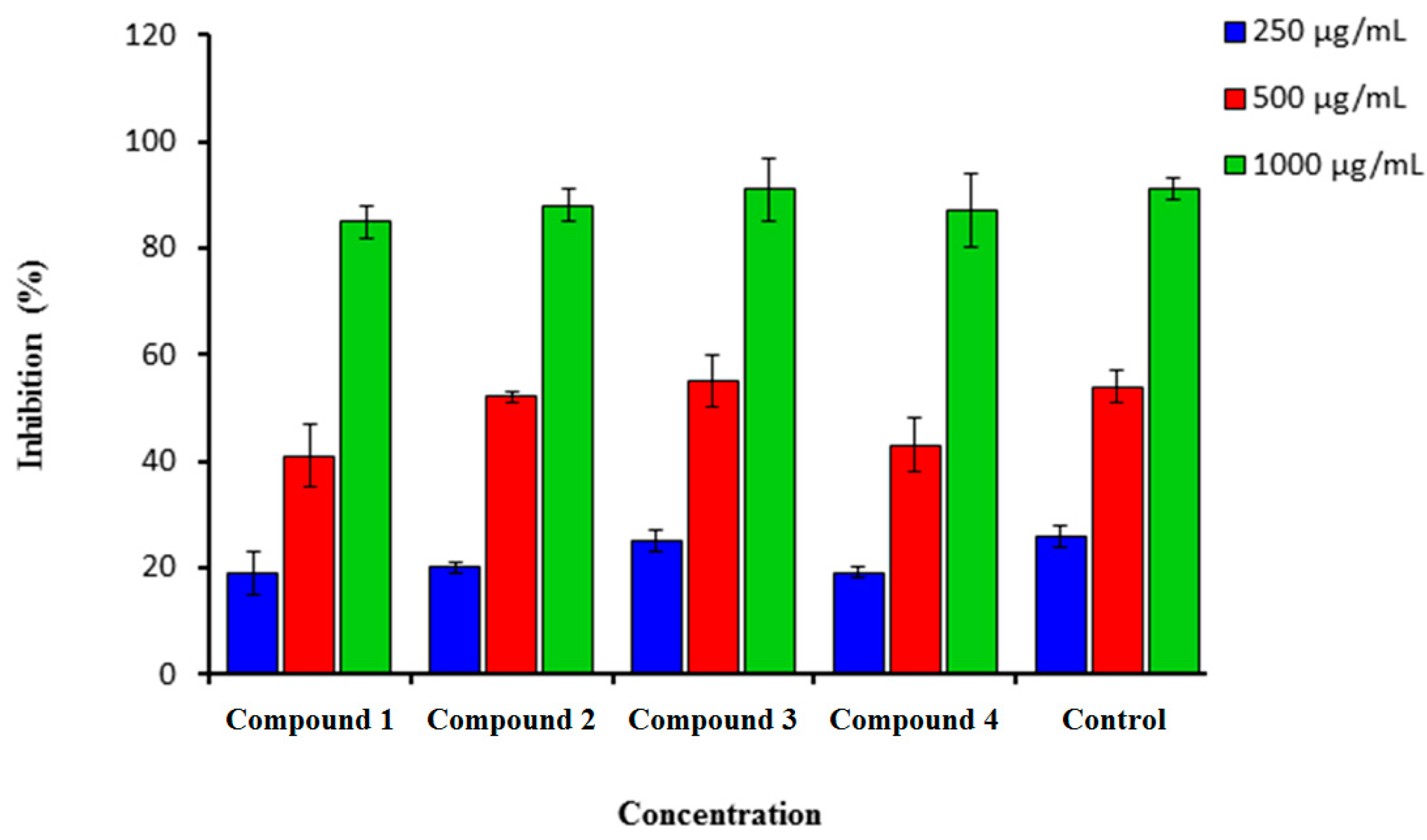
2.1.1. DPPH Scavenging Assay
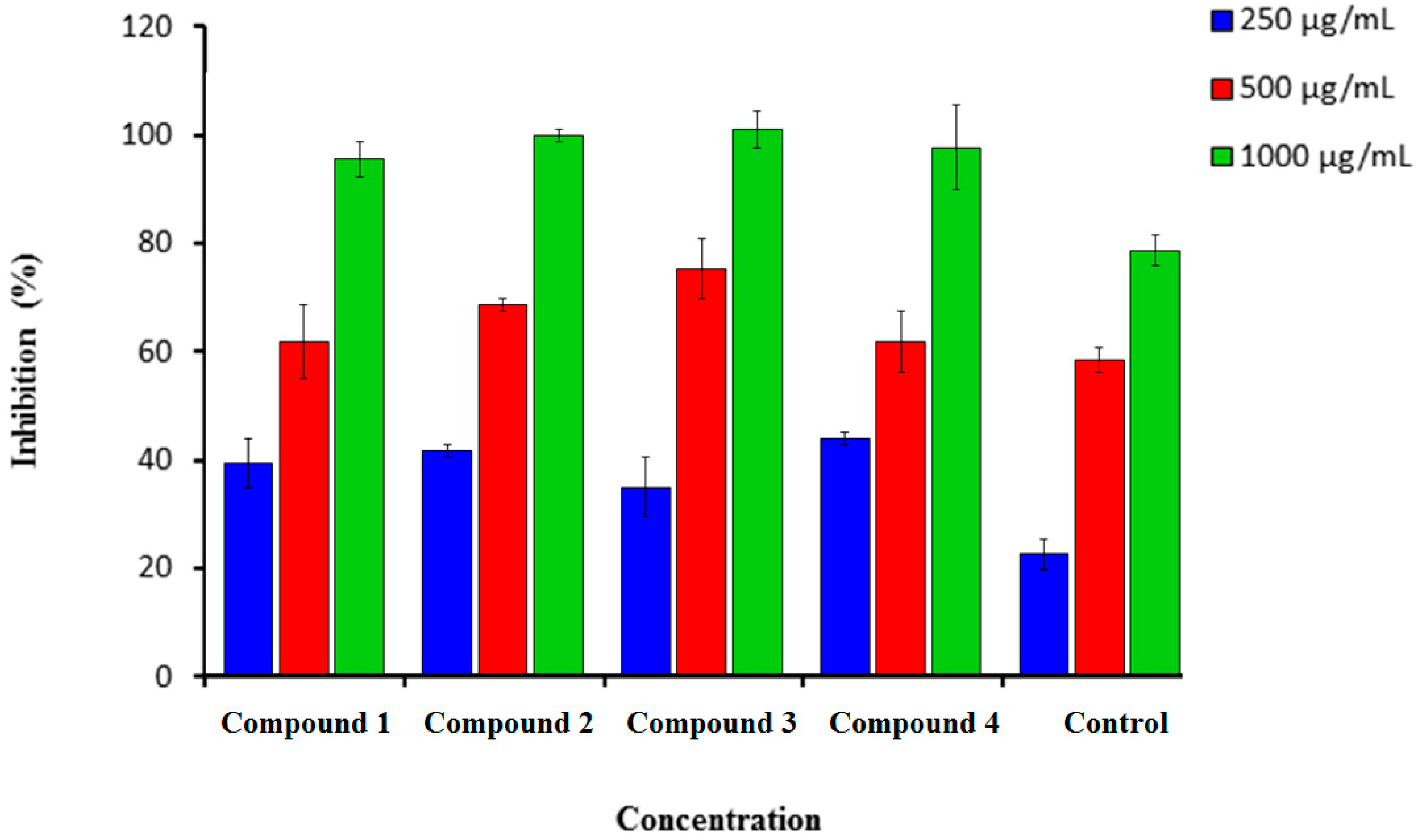
2.1.2. H2O2 Scavenging Assay
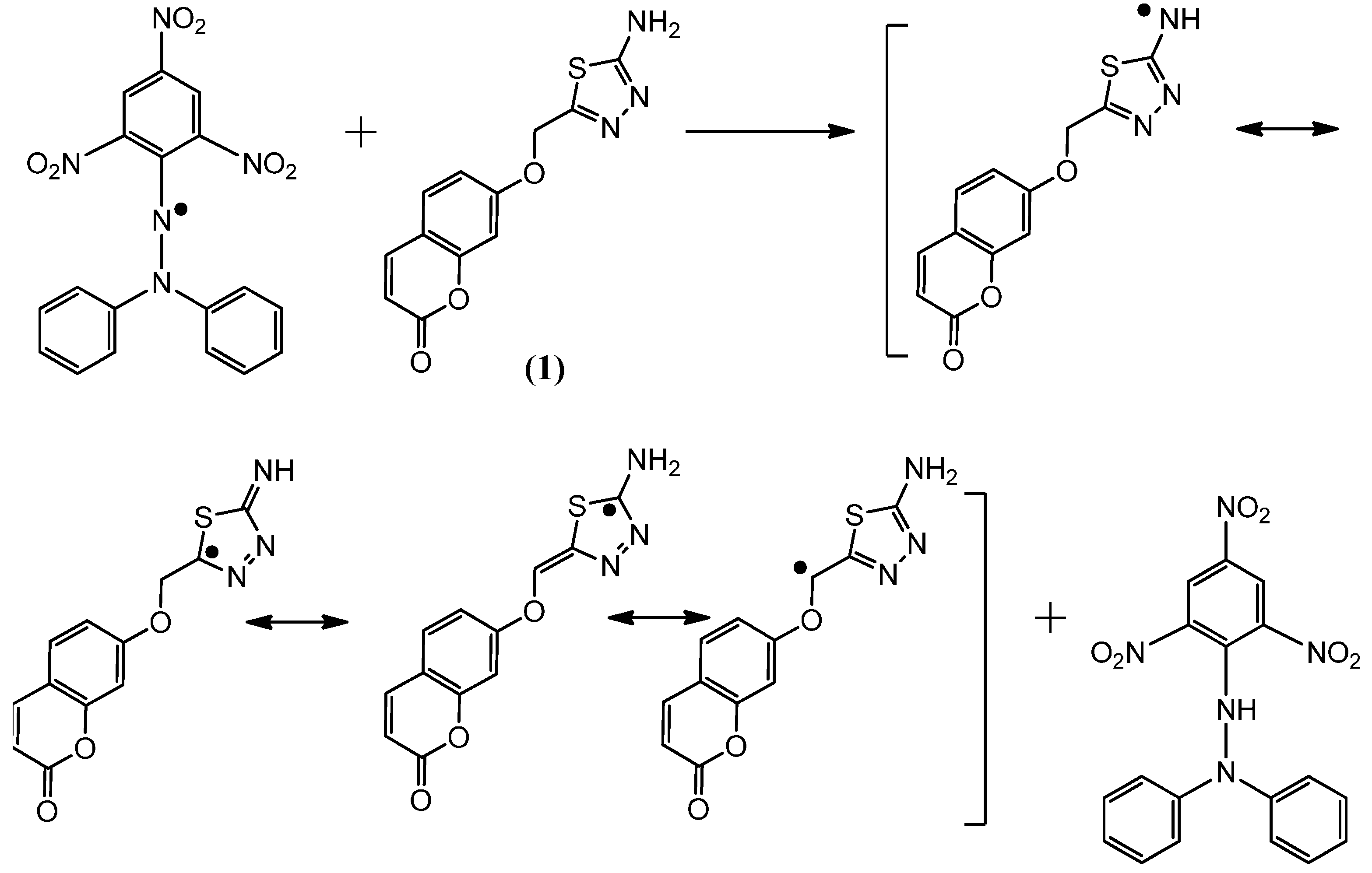
2.2. Postulated Mechanisms for Coumarins 1, 2, 3 and 4 as Antioxidants

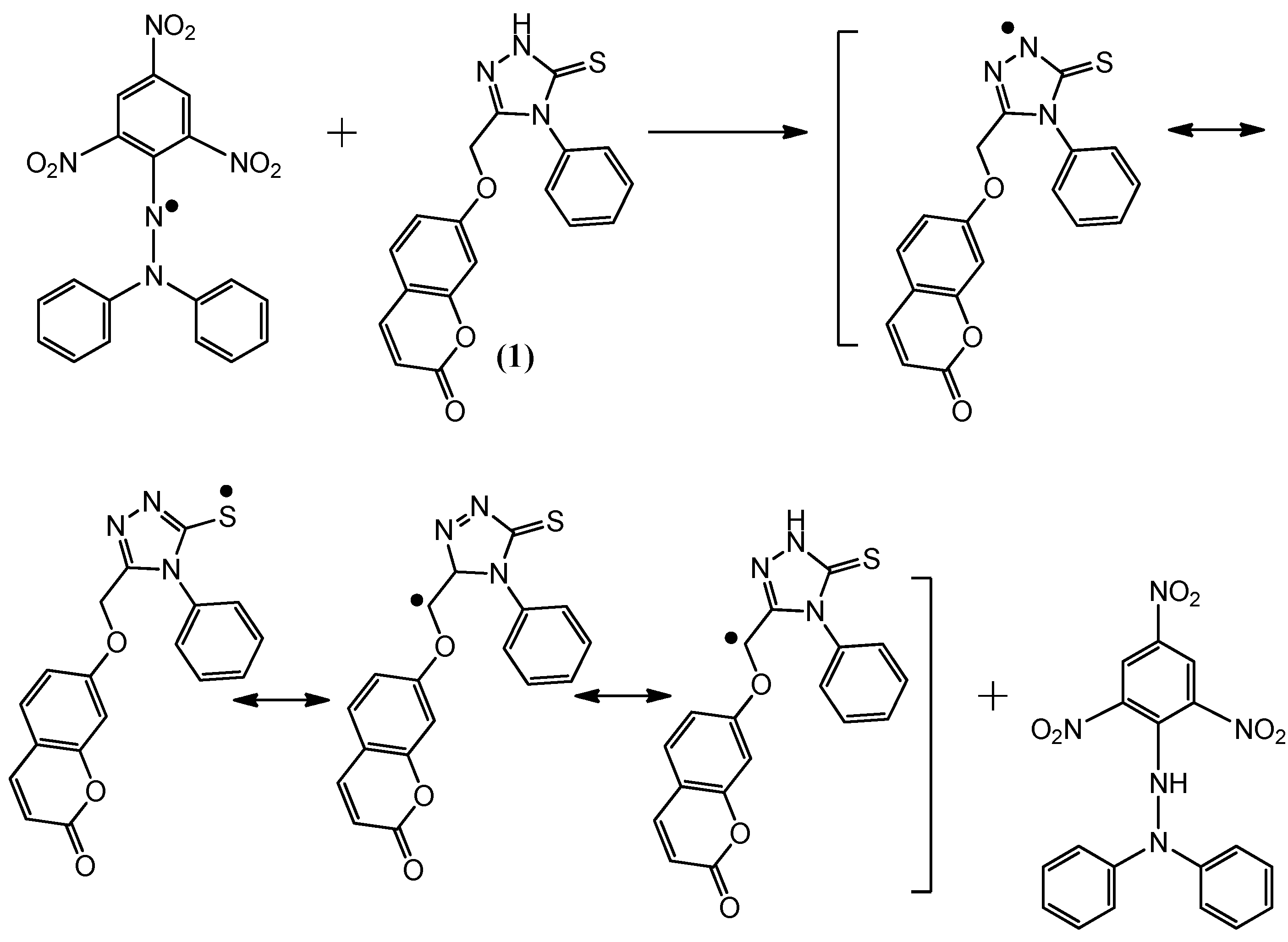
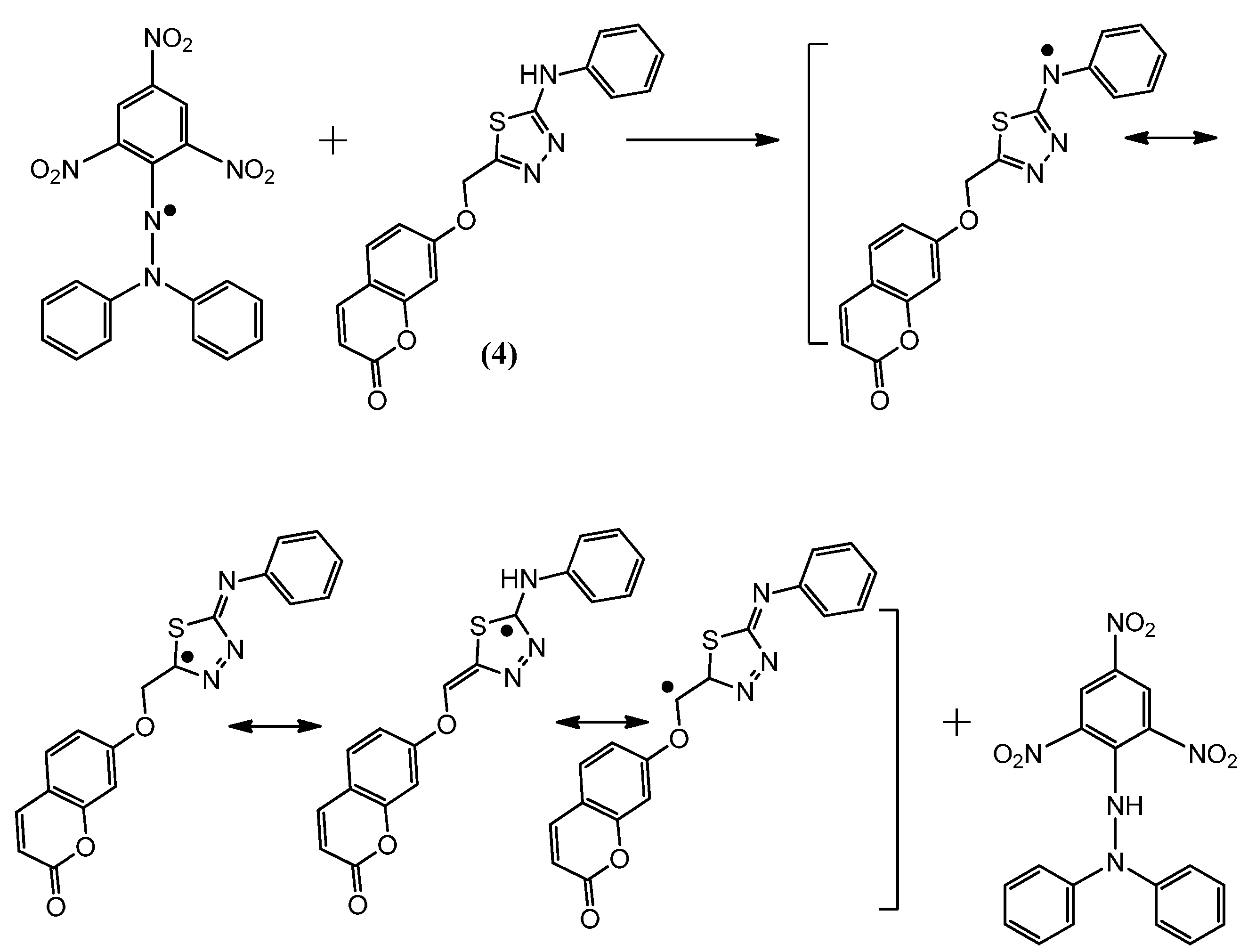
2.3. Molecular Modeling Studies

| Parameters | Compound 1 | Compound 2 | Compound 4 | Compound 3 | Ascorbic Acid |
|---|---|---|---|---|---|
| Dipole moment Depy | 4.665 | 7.117 | 6.613 | 5.229 | 9.549 |
| Ionization potential (IP) eV | 8.504 | 10.102 | 8.753 | 8.532 | 10.772 |
| Electron affinity (EA) eV | 4.332 | 4.132 | 4.532 | 4.776 | 1.115 |
| Hardness (η) | 2.86 | 2.895 | 2.110 | 1.378 | 4.827 |
| Softness (S) | 0.239 | 0.172 | 0.236 | 0.266 | 0.207 |
| Electro negativity(µ) | 6.418 | 7.117 | 6.142 | 6.518 | 5.9435 |
| EHOMO | −8.504 | −10.102 | −8.753 | −8.532 | −10.772 |
| ELUMO | −4.332 | −4.132 | −4.532 | −4.776 | −1.115 |
| Band gap = EHOMO − ELUMO | 4.172 | 5.790 | 4.221 | 3.756 | 9.655 |
3. Materials and Methods
3.1. Synthesis of Compounds 1–4
3.2. Antioxidant Activity
3.2.1. DPPH Free Radical Scavenging Activity
3.2.2. Hydrogen Peroxide Scavenging Activity
3.3. Quantum Studies
3.4. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hoult, J.R.; Payá, M. Pharmacological and biochemical actions of simple coumarins: Natural products with therapeutic potential. Gen. Pharmacol. 1996, 27, 713–722. [Google Scholar] [CrossRef]
- Al-Amiery, A.A.; Al-Majedy, Y.K.; Kadhum, A.A.H.; Mohamad, A.B. Hydrogen. Peroxide Scavenging Activity of Novel Coumarins Synthesized Using Different Approaches. PLoS ONE 2015, 10, e0132175. [Google Scholar] [CrossRef]
- Lin, M.H.; Chou, Y.S.; Tsai, Y.J.; Chou, D.S. Antioxidantroperties of 5,7-dihydroxy coumarin derivatives in vitro cell free and cell-containing system. J. Exp. Clin. Med. 2011, 3, 126–131. [Google Scholar] [CrossRef]
- Al-Amiery, A.A.; Al-Majedy, Y.K.; Kadhum, A.A.H.; Mohamad, A.B. Novel macromolecules derived from coumarin: Synthesis and antioxidant activity. Sci. Rep. 2015, 5, 11825. [Google Scholar] [CrossRef] [PubMed]
- Egan, D.; O’Kennedy, R.; Moran, E.; Cox, D.; Prosser, E.; Thornes, R.D. The pharmacology, metabolism, analysis, and applications of coumarin and coumarin-related compounds. Drug Metab. Rev. 1990, 22, 503–529. [Google Scholar] [CrossRef] [PubMed]
- Završnik, D.; Muratović, S.; Makuc, D.; Plavec, J.; Cetina, M.; Nagl, A.; Clercq, E.D.; Balzarini, J.; Mintas, M. Benzylidene-bis-(4-hydroxycoumarin) and benzopyrano-coumarin derivatives: Synthesis, ¹H/¹³C-NMR conformational and X-ray crystal structure studies and in vitro antiviral activity evaluations. Molecules 2011, 19, 6023–6040. [Google Scholar] [CrossRef] [PubMed]
- Cravotto, G.; Tagliapietra, S.; Capello, R.; Palmisano, G.; Curini, M.; Boccalini, M. Long-Chain 3-Acyl-4-hydroxycoumarins: Structure and Antibacterial Activity. Arch. Pharm. 2006, 339, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Naser, T.; Bondock, S.; Youns, M. Anticancer activity of new coumarin substituted hydrazide-hydrazone derivative. Eur. J. Med. Chem. 2014, 76, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Skalicka-Woźniak, K.; Orhan, E.; Cordell, G.; Nabavi, S.; Budzyńska, B. Implication of coumarins towards central nervous system disorders. Pharmacol. Res. 2016, 103, 188–203. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chen, Y.; Wang, S.; Xu, X.; Liu, X.; Yu, M.; Zhao, S.; Liu, S.; Qiu, Y.; Zhang, T.; et al. Synthesis and Biological Investigation of Coumarin Piperazine (Piperidine) Derivatives as Potential Multireceptor Atypical Antipsychotics. J. Med. Chem. 2013, 56, 4671–4690. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.; Jiang, H.; Zhou, Z.; Lei, D.; Xue, Y.; Yao, Q. Ultrasound-promoted greener synthesis of novel trifurcate 3-substituted-chroman-2,4-dione derivatives and their drug-likeness evaluation. Molecules 2012, 17, 14146–14158. [Google Scholar] [CrossRef] [PubMed]
- Phadtare, B.; Jarag, K.; Shankarling, G. Greener protocol for one pot synthesis of coumarin styryl dyes. Dyes Pigments 2013, 97, 105–112. [Google Scholar] [CrossRef]
- Elnagdi, M.; Abdallah, S.; Ghoneim, K.; Ebied, E.; Kassab, K. Synthesis of Some Coumarin Derivatives as Potential Laser Dyes. J. Chem. Res. 1997, 44–45. [Google Scholar] [CrossRef]
- Garcia-Belto, O.; Mena, N.; Friedrich, L.C.; Neto-Ferreira, J.C.; Vargas, V.; Quina, F.H.; Nuez, M.T.; Cassels, B.K. Design and synthesis of a new coumarin-based fluorescent probe selective for Cu2+. Tetrahedron Lett. 2012, 53, 5280–5283. [Google Scholar] [CrossRef]
- Garcia-Belto, O.; Mena, N.; Yaez, O.; Caballero, J.; Vargas, V.; Nuez, M.T.; Cassels, B.K. Design, synthesis and cellular dynamics studies in membranes of a new coumarin-based fluorescent probe selective for Fe2+. Eur. J. Med. Chem. 2013, 67, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Belto, O.; Cassels, B.K.; Pérez, C.; Mena, N.; Nuez, M.T.; Martnez, N.P.; Pavez, P.; Aliaga, M.E. Coumarin-based fluorescent probes for dual recognition of copper(II) and iron(III) ions and their application in bio-imaging. Sensors 2014, 14, 1358–1371. [Google Scholar] [CrossRef] [PubMed]
- Moylan, C.R. Molecular hyperpolarize abilities of coumarin dyes. J. Phys. Chem. 1994, 98, 13513–13516. [Google Scholar] [CrossRef]
- Li, N.; Xiang, Y.; Tong, A. Highly sensitive and selective “turn-on” fluorescent chemodosimeter for Cu2+ in water via Cu2+-promoted hydrolysis of lactone moiety incoumarin. Chem. Commun. 2010, 46, 3363–3365. [Google Scholar] [CrossRef] [PubMed]
- Al-Amiery, A.A.; Al-Majedy, Y.K.; Abdulreazak, H.; Abood, H. Synthesis, characterization, theoretical crystal structure, and antibacterial activities of some transition metal complexes of the thiosemicarbazone. Bioinorg. Chem. Appl. 2011, 2011, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Abdalla, A.E.; Roozen, J.P. Effect of plant extracts on the oxidative stability of sunflower oil and emulsion. Food Chem. 1999, 64, 323–329. [Google Scholar] [CrossRef]
- Bast, A.; Haenen, G.R.; Doelman, C.J. Oxidants and antioxidants: State of the art. Am. J. Med. 1999, 30, 2–13. [Google Scholar] [CrossRef]
- Paya, M.; Goodwin, P.A.; de Las Heras, B.; Hoult, J.R. Superoxide scavenging activity in leucocytes and absence of cellular toxicity of a series of coumarins. Biochem. Pharmacol. 1994, 48, 445–451. [Google Scholar] [CrossRef]
- Symeonidis, T.; Chamilos, M.; Hadjipavlou-Litina, D.J.; Kallitsakis, M.; Litinas, K.E. Synthesis of hydroxycoumarins and hydroxybenzo[f]- or [h]coumarins as lipid peroxidation inhibitors. Bioorg. Med. Chem. Lett. 2009, 19, 1139–1142. [Google Scholar] [CrossRef] [PubMed]
- Fylaktakidou, K.C.; Hadjipavlou-Litina, D.J.; Litinas, K.; Nicolaides, D.N. Natural and synthetic coumarin derivatives with anti-inflammatory/antioxidant activities. Curr. Pharm. Des. 2004, 10, 3813–3833. [Google Scholar] [CrossRef] [PubMed]
- Kostova, I. Synthetic and natural coumarins as antioxidants. Mini Rev. Med. Chem. 2006, 6, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Tabart, J.; Kevers, C.; Pincelmail, J.; Defraigne, J.O.; Dommes, J. Comparative antioxidant capacities of phenolic compounds measured by various tests. Food Chem. 2009, 113, 1226–1233. [Google Scholar] [CrossRef]
- Kadhum, A.A.H.; Mohamad, A.B.; Al-Amiery, A.A.; Takriff, M.S. Antimicrobial and antioxidant activities of new metal complexes derived from 3-aminocoumarin. Molecules 2011, 16, 6969–6984. [Google Scholar] [CrossRef] [PubMed]
- Al-Amiery, A.A.; Al-Bayati, R.I.H.; Saour, K.Y.; Radi, M.F. Cytotoxicity, antioxidant and antimicrobial activities of novel 2-quinolone derivatives derived from coumarins. Res. Chem. Intermed. 2011, 38, 559–569. [Google Scholar] [CrossRef]
- Al-Amiery, A.; Al-Majedy, Y.; Ibrahim, H.; Al-Tamimi, A. Antioxidant, antimicrobial, and theoretical studies of the thiosemicarbazone derivative Schiff base 2-(2-imino-1-methylimidazolidin-4-ylidene)hydrazinecarbothioamide (IMHC). Org. Med. Chem. Lett. 2012, 2, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kadhum, A.A.H.; Al-Amiery, A.A.; Musa, A.Y.; Mohamad, A. The antioxidant activity of new coumarin derivatives. Int. J. Mol. Sci. 2012, 12, 5747–5761. [Google Scholar] [CrossRef] [PubMed]
- Al-Amiery, A.; Kadhum, A.; Obayes, H.; Mohamad, A. Synthesis and Antioxidant Activities of Novel 5-Chlorocurcumin, Complemented by Semiempirical Calculations. Bioinorg. Chem. Appl. 2013, 2013, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Gocer, H.; Gulcin, I. Caffeic acid phenethyl ester (CAPE): Correlation of structure and antioxidant properties. Int. J. Food Sci. Nutr. 2011, 62, 821–825. [Google Scholar] [CrossRef] [PubMed]
- Widjaja, A.; Yeh, H.; Ju, H. Enzymatic synthesis of caffeic acid phenethyl ester. J. Chin. Inst. Chem. Eng. 2008, 39, 413–418. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, M.; Rosen, T.; Ho, C. 2,2-Diphenyl-1-picrylhydrazyl radical-scavenging active components from Polygonum multiflorum Thunb. J. Agric. Food Chem. 1999, 47, 2226–2228. [Google Scholar] [CrossRef] [PubMed]
- Chang, R. Chemistry, 7th ed.; McGraw-Hill: New York, NY, USA, 2001; Volume 253. [Google Scholar]
- Al-Amiery, A.; Musa, A.; Kadhum, A.; Mohamad, A. The Use of Umbelliferone in the Synthesis of New Heterocyclic Compounds. Molecules 2011, 16, 6833–6843. [Google Scholar] [CrossRef] [PubMed]
- Braca, A.; Sortino, C.; Politi, M.; Morelli, I.; Mendez, J. Anti-oxidant activity of flavonoids from Licania licaniaeflora. J. Ethnopharmacol. 2002, 79, 379–381. [Google Scholar] [CrossRef]
- Bors, W.; Saran, M.; Elstner, E.F. Screening for plant anti-oxidants. In Modern Methods of Plant Analysis-Plant Toxin Analysis-New Series; Linskens, H.F., Jackson, J.F., Eds.; Springer: Berlin, Germany, 1997; Volume 13, pp. 277–295. [Google Scholar]
- Duh, P.; Tu, Y.; Yen, G. Antioxidant Activity of Water Extract of Harng Jyur (Chrysanthemum morifolium Ramat). LWT Food Sci. Technol. 1999, 32, 269–277. [Google Scholar] [CrossRef]
- Ilhami, G.I.; Haci, A.A.; Mehmet, C. Determination of in Vitro Antioxidant and Radical Scavenging Activities of Propofol. Chem. Pharmacol. Bull. 2005, 53, 281–285. [Google Scholar]
- Sample Availability: Samples of the compounds 1–4 are available from the authors.
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Majedy, Y.K.; Al-Duhaidahawi, D.L.; Al-Azawi, K.F.; Al-Amiery, A.A.; Kadhum, A.A.H.; Mohamad, A.B. Coumarins as Potential Antioxidant Agents Complemented with Suggested Mechanisms and Approved by Molecular Modeling Studies. Molecules 2016, 21, 135. https://doi.org/10.3390/molecules21020135
Al-Majedy YK, Al-Duhaidahawi DL, Al-Azawi KF, Al-Amiery AA, Kadhum AAH, Mohamad AB. Coumarins as Potential Antioxidant Agents Complemented with Suggested Mechanisms and Approved by Molecular Modeling Studies. Molecules. 2016; 21(2):135. https://doi.org/10.3390/molecules21020135
Chicago/Turabian StyleAl-Majedy, Yasameen K., Dunya L. Al-Duhaidahawi, Khalida F. Al-Azawi, Ahmed A. Al-Amiery, Abdul Amir H. Kadhum, and Abu Bakar Mohamad. 2016. "Coumarins as Potential Antioxidant Agents Complemented with Suggested Mechanisms and Approved by Molecular Modeling Studies" Molecules 21, no. 2: 135. https://doi.org/10.3390/molecules21020135






