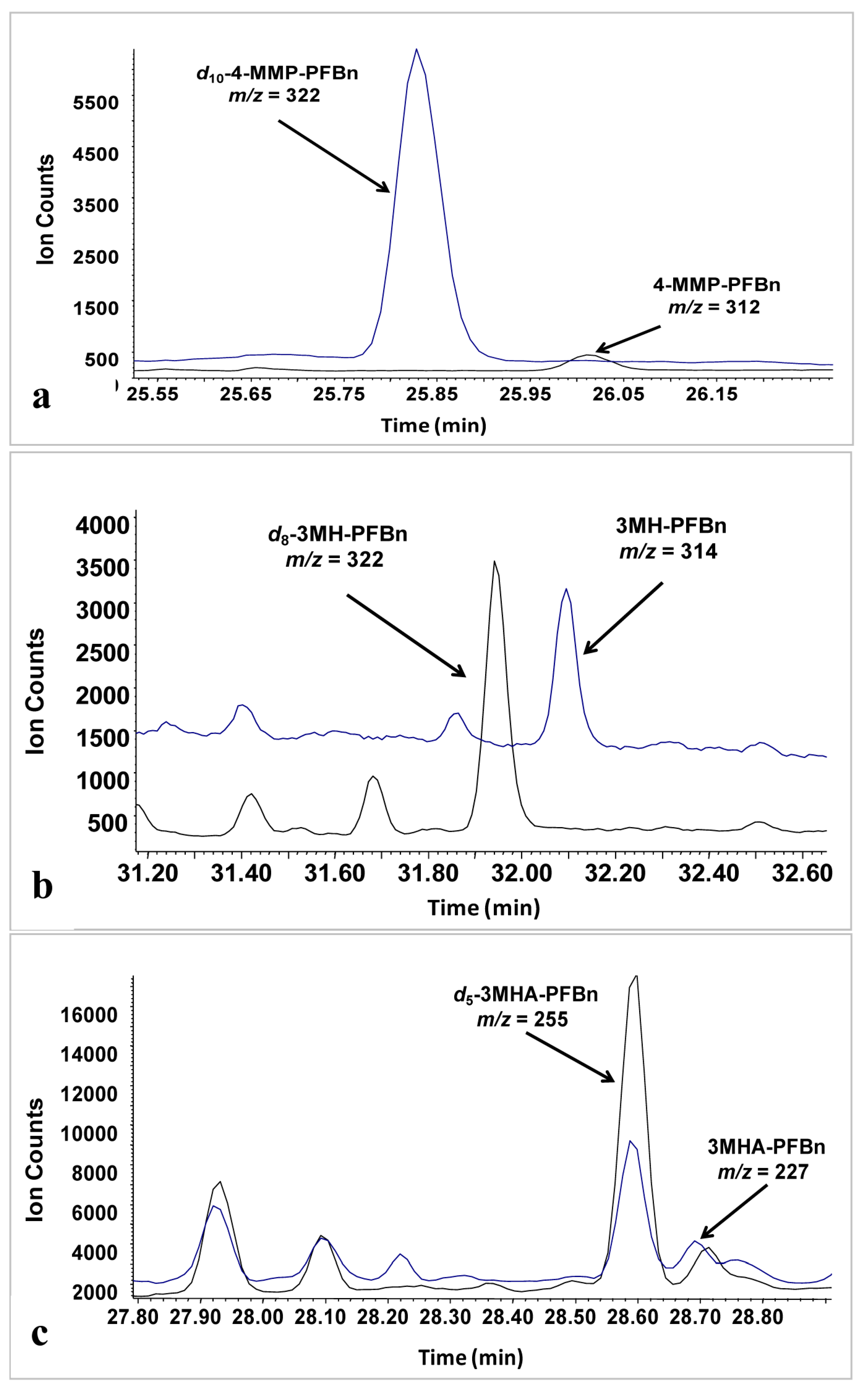
Analysis of key wine polyfunctional thiols (3-MHA, 3-MH, 4-MMP) in underivatized form is challenging, and reports that have achieved detection limits below sensory threshold have required the use of organomercuric reagents to achieve a high degree of selective pre-concentration. Alternate quantification schemes for thiols involving derivatization prior to analysis exist [
18]. We chose to develop an optimized method based on pentafluorobenzylbromide (PFBBr) derivatization, as this approach has previously been demonstrated to be effective for the key wine polyfunctional thiols [
9,
10,
11,
19]. Representative chromatograms for wine samples analyzed by the optimized method are shown in
Figure 1.
Figure 1.
Typical chromatogram obtained in selective ion monitoring (SIM) mode: (a) White wine spiked with 10 ng/L 4-MMP and 540 ng/L d10-4-MMP; (b) White wine spiked with 25 ng/L 3-MH and 440 ng/L d8-3-MH; (c) White wine spiked with 60 ng/L 3-MHA and 400 ng/L d5-3-MHA.
2.4. Comparison of Current Method to Existing GC-MS Methods for Wine Polyfunctional Thiol Analysis
Literature methods that rely on a low selectivity extraction step (e.g., SPE, SPME) to pre-concentrate underivatized wine polyfunctional thiols (3-MH, 3-MHA, 4-MMP) prior to GC-EI-MS have not achieved detection limits below sensory thresholds [
6,
7]. Quantification of these thiols below sensory threshold can be achieved using organomercuric reagents and anion exchanges for selective pre-concentration [
4], but the approach has been criticized for needing large sample volumes and hazardous reagents [
10,
11].
More recent reports have achieved detection limits below sensory thresholds by preparing PFBBr derivatives of the thiols prior to GC-NICI-MS detection. The detection limits of these methods are generally better than what we have achieved with our current report: for example Mateo-Vivaracho,
et al. reported LODs of 0.1 ng/L, 2 ng/L, and 0.3 ng/L, for 4-MMP, 3-MH, and 3-MHA, respectively, as compared to 0.9 ng/L, 1 ng/L, and 17 ng/L in our work. Correcting for the fact that this earlier report used a 6 mL wine sample compared to 40 mL in our work, NICI-MS provides about a 1–3 order of magnitude improvement in detection limits compared to EI-MS for PFB derivatives, comparable to values reported for other compound classes [
25].
However, an advantage of our current method is that EI-MS is more widespread than NICI-MS in laboratories. One other group has utilized GC-EI-MS for quantification of the 3-MH-PFB derivative, although they did not report characterization of 3-MHA and 4-MMP [
13]. In that report, 3-MH was initially extracted into pentane and back extracted into aqueous alkaline buffer prior to derivatization. Similar to our approach, the 3-MH-PFB derivative was extracted by SPME prior to GC-EI-MS. We achieved a detection limit of 1 ng/L for a 40 mL wine sample, as compared to 30 ng/L for a 200 mL wine sample in the previous report. The most notable difference between the two reports is that our current work utilized a dry-down of the organic layer step prior to reconstitution and HS-SPME, as opposed to the back-extraction step in the other report. Potentially, this dry-down step removed lower boiling volatiles that could compete with the thiol-PFB derivatives for absorptive sites on the SPME fiber. While we observed a considerable enhancement in S/N when aqueous NaCl solution was added to our dried-down SPME vials, this should have been comparable to the conditions in the other report for alkaline back-extraction, and is thus unlikely to account for the differences. Other potential explanations for the enhanced sensitivity of our current report are more optimal derivatization or HS-SPME conditions, or decreased losses during sample preparation due to fewer work-up steps. GC-EI-MS has also been reported for quantification of ethyl propiolate derivatives of thiols, but detection limits were well above sensory thresholds for 4-MMP and 3-MH [
14].
Beyond using EI-MS, another advantage of our extractive alkylation method is that it requires fewer clean-up steps than many other methods that have analyzed PFB derivatives. For example, one report describes an initial loading of the sample onto an SPE cartridge followed by multiple wash steps, introduction of a catalyst and derivatizing reagents, addition of mercaptoglycerol to remove excess PFBBr, and, finally, elution of the derivatized thiol with organic solvent [
12]. The organic phase is then evaporated in a SPME vial prior to HS-SPME analysis, similar to our current work. Other reports also use multiple LLE or SPE clean-up steps, often prior to derivatization [
9,
10,
13]. By comparison, the extractive alkylation described here can be performed in a single step.
2.5. Quantification of Polyfunctional Thiols in Commercial Wines
Using the optimized method, the three polyfunctional thiols were measured in 31 California wines and 30 commercial New York State wines representing seven wine types: four
V. vinifera (Gewurztraminer, Riesling, Sauvignon blanc, Cabernet Sauvignon), one non-varietal
V. vinifera (rosé blends), one
V. labruscana (Niagara), and one
Vitis spp. (Cayuga White). Results are summarized in
Table 3. The only thiol detected in all wine samples was 3-MH, and concentrations were significantly higher in the
vinifera-based wines than the interspecific hybrid-based wines (Cayuga White and Niagara). However, 3-MH concentrations were not significantly higher in Sauvignon blanc wines than the other white varietal wines, unlike results reported elsewhere [
3].
Table 3.
Concentrations of volatile polyfunctional thiols in California and NY wines.
Table 3.
Concentrations of volatile polyfunctional thiols in California and NY wines.
| Variety | Region | Concentration (ng/L) |
|---|
| 4-MMP | 3-MH | 3-MHA |
|---|
| Cayuga White | Finger Lakes, NY (n = 5) | <LOD | 195 ± 38 | <LOD |
| Niagara | Finger Lakes, NY (n = 5) | 18 ± 13 | 230 ± 159 | <LOD |
| Riesling | Finger Lakes, NY (n = 5) | 2.3 ± 2.5 | 569 ± 334 | <LOD |
| Gewürztraminer | Finger Lakes, NY (n = 5) | <LOD | 373 ± 134 | <LOD |
| Rosé | Finger Lakes, NY (n = 5) | <LOD | 296 ± 116 | a |
| Sauvignon blanc | Finger Lakes, NY (n = 5) | 27 ± 8 | 446 ± 154 | a |
| Napa Valley, CA (n = 4) | 44 ± 22 | 438 ± 87 | b |
| Sonoma County, CA (n = 4) | 43 ± 18 | 712 ± 342 | b |
| Central Coast, CA (n = 3) | 50 ± 25 | 835 ± 137 | b |
| Cabernet Sauvignon | Napa Valley, CA (n = 8) | <LOD | 765 ± 396 | 57 ± 16 |
| Sonoma County, CA (n = 5) | <LOD. | 405 ± 106 | 60 ± 21 |
| Central coast, CA (n = 6) | <LOD | 498 ± 113 | 67 ± 24 |
| Lodi (n = 1) | <LOD | 396 | 46 |
Mean 3-MH concentrations were lower in New York and California Sauvignon blanc (less than 1000 ng/L) than reports from other regions. For example, a survey of commercial Sauvignon blanc wines from Australia, New Zealand, France, and South Africa reported mean concentrations ranging from 1700 to 7100 ng/L [
24]. Mean concentrations of 3-MH in Sauvignon blanc in our survey were also lower than what was reported in an earlier survey of US Sauvignon blanc, ~2000 ng/L for eight wines over 3 vintages [
24], although the lack of specific information regarding wine region and large standard deviations (~100%) in the previous report makes direct comparison challenging. In contrast, mean 3-MH in New York Gewürztraminer and Riesling (373 and 579 ng/L, respectively) were in the middle of ranges reported for Australian sources of these varietal wines [
26]. 3-MH in the California Cabernet Sauvignon was within the broad range reported for Bordeaux red wines previously (10–5000 ng/L) [
27]. The contribution of volatile thiols like 3-MH to red wines has not been well studied, but the mean values reported in this study (396–765 ng/L) are comparable to other non-Sauvignon blanc white wines, both in the current study and elsewhere [
26].
The reasons for lower concentrations of 3-MH in the California and New York State Sauvignon blanc in this study as compared to other reports is unclear, as several factors are known to affect final 3-MH concentrations. For example, wine grapes are typically hand-harvested in the Finger Lakes and Napa, and it is reported that hand harvesting grapes results in lower 3-MH precursor concentrations and eventually lower 3-MH in wines as compared to machine harvesting [
28]. However, this argument would not be appropriate for California Central Coast where machine harvesting is common. Other factors reported to affect final 3-MH concentrations include vineyard nitrogen availability, yeast assimilable nitrogen, abiotic stresses, disease pressure, oxygen status during maceration, and post-fermentation oxidation [
1], and a recent study has reported modest correlations of over 20 grape metabolites (amino acids, fatty acids,
etc.) with wine 3-MH concentrations [
29]. Discerning which of these factors would lead to low 3-MH concentrations in Sauvignon blanc and Gewürztraminer samples in this study is not possible.
It was found that 3-MHA was only detectable in two out of the 30 New York wines evaluated; one rosé wine, and one Sauvignon Blanc wine. Formed by partial enzymatic acetylation of 3-MH during fermentation [
30], 3-MHA will decrease during storage due to acid-catalyzed hydrolysis [
31]. As a result, average 3-MHA values are reported to be 10%–20% the concentration of average 3-MH values in Sauvignon blanc [
29], and can be much lower [
24]. Since the average 3-MH concentration across New York State wines in this study was 195 to 569 ng/L, and many of the wines were 2–3 years old at the time of analysis, it is unsurprising that 3-MHA was generally below the 17 ng/L detection limit of the method. Additionally, New York State grapes tend to be low in α-amino acids as compared to other regions [
32], and several of these amino acids have been correlated with lower 3-MHA/3-MH ratios [
29]. Furthermore, 3-MHA could not be quantified in the California Sauvignon blanc samples due to an unknown interference on the quantifier ion, which could not be identified by library search. However, 3-MHA could be detected in California Cabernet Sauvignon, and mean values across sub-regions (46–67 ng/L) were approximately 10% of 3-MH concentrations. Similar to 3-MH, there is a lack of analytical data regarding 3-MHA in red wines, but values are within the range estimated for Bordeaux red wines, 1–200 ng/L [
27].
In contrast to 3-MH and 3-MHA, mean 4-MMP concentrations in both New York (27 ng/L) and California (44–50 ng/L) Sauvignon blanc wines were greater than the mean values (5–10 ng/L) reported for Sauvignon blanc from most other wine regions [
24]. The highest reported mean value for 4-MMP in this previous survey was for the Wairarapa region of New Zealand (19 ng/L), but this region also had order of magnitude higher concentrations of 3-MH. In addition, 4-MMP was not detectable in other
V. vinifera wines, except for trace concentrations (mean = 2.5 ng/L) in NY State Riesling wines.
To our knowledge, this is the first report of thiol concentrations in any American wines from specific wine regions, rather than the general category of US wine [
24]. The study also included the first analysis of thiols in wines produced from interspecific hybrids-Niagara and Cayuga White. The former contains
V. labrusca parentage, and the latter is a complex interspecific hybrid with mixed parentage. The 3-MH concentrations in Cayuga White were significantly lower than in
vinifera wines, and the other two thiols were not detectable. However, concentrations of 4-MMP in New York Niagara (mean = 18 ng/L) did not differ significantly from New York Sauvignon blanc (
p > 0.05), and were significantly higher than all other varietal wines (
p < 0.05). The “foxy” smelling
o-aminoacetophenone and methyl anthranilate have previously been implicated in contributing to Niagara juice and wines [
33,
34]. Because 4-MMP exceeded sensory threshold by an order of magnitude in the majority of Niagara wines, 4-MMP may also be important to the varietal character of Niagara wines, but confirmation by sensory studies was not part of this study.