Natural Products as Leads in Schistosome Drug Discovery
Abstract
:1. Introduction
2. Natural Products as Lead Compounds against Schistosomiasis
2.1. Artemisinins

2.2. Further Terpenoids

Tanshinones
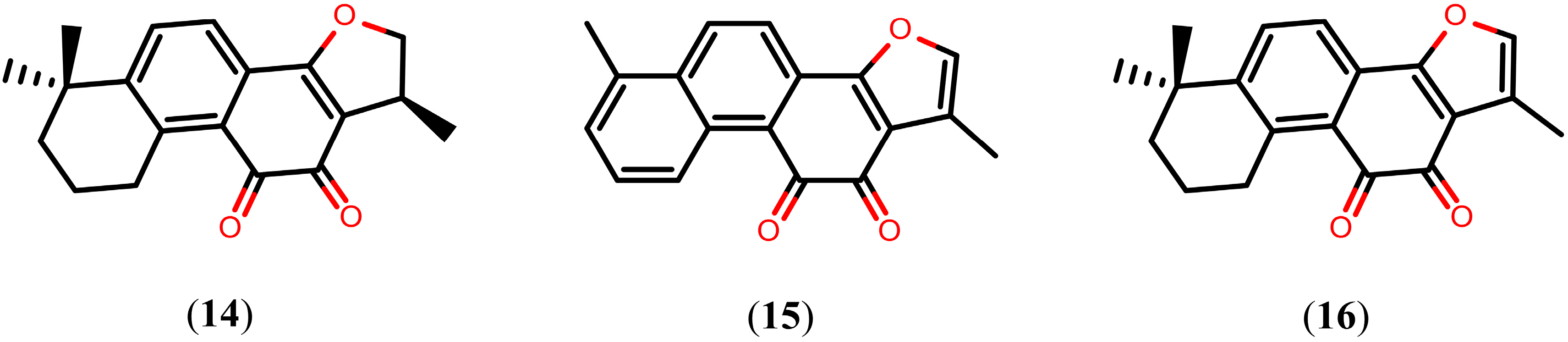
2.3. Alkaloids

Quinoline Methanols

2.4. Flavonoids

2.5. Arachidonic Acid

2.6. Quinones

2.7. Other Natural Products

3. Drawbacks in NP Lead Discovery
4. Integration of Natural Products and Virtual Screening
| Database Name | Nº of Entries | Website |
|---|---|---|
| NuBBE Database | 643 | [144] |
| Traditional Chinese Medicine Database | >32,300 | [150,151] |
| Dictionary of Natural Products | >260,000 | [152] |
| Specs Natural Products | 400 | [27] |
| Herbal Ingredients’ Targets Database | 586 | [153] |
| Super Natural Database | >325,500 | [145] |
| NPACT Database | 1574 | [154] |
| Database of Indonesian Medicinal Plants | 6776 | [155] |
| Greenpharma Natural Compound Library | >150,000 | [156] |
| Tea Metabolome Database | 1450 | [157] |
| TimTec Natural Products Library | 720 | [25] |
| Seaweed Metabolite Database | 1055 | [147] |
| InterBioScreen Natural Products Database | >17,500 | [26] |
| MAPS Database | >1200 | [158] |
| Database of Traditional Chinese Medicine | >12,000 | [146] |
| TIP Database | 8856 | [159,160] |
| AMRI’s Natural Product Library | >290,000 | [161] |
4.1. Ligand-Based Virtual Screening

4.2. Structure-Based Virtual Screening
4.3. Inverse Virtual Screening
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Colley, D.G.; Bustinduy, A.L.; Secor, W.E.; King, C.H. Human schistosomiasis. Lancet 2014, 383, 2253–2264. [Google Scholar] [CrossRef] [PubMed]
- WHO Schistosomiasis. 2014. Fact Sheet. Available online: http://www.who.int/mediacentre/factsheets/fs115/en (accessed on 10 December 2014).
- Gryseels, B.; Polman, K.; Clerinx, J.; Kestens, L. Human schistosomiasis. Lancet 2006, 368, 1106–1118. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.G.P.; Bartley, P.B.; Sleigh, A.C.; Olds, G.R.; Li, Y.; Williams, G.M.; McManus, D.P. Schistosomiasis. N. Engl. J. Med. 2002, 346, 1212–1120. [Google Scholar] [CrossRef] [PubMed]
- Gönnert, R.; Andrews, P. Praziquantel, a new board-spectrum antischistosomal agent. Z. Parasitenkd. 1977, 52, 129–150. [Google Scholar] [CrossRef]
- Angelucci, F.; Miele, A.E.; Boumis, G.; Brunori, M.; Dimastrogiovanni, D.; Bellelli, A. Macromolecular bases of antischistosomal therapy. Curr. Top. Med. Chem. 2011, 11, 2012–2028. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Xu, Y.; Yu, C.; Li, H.; Yin, X.; Wang, T.; Wang, W.; Liang, Y. Effect of praziquantel prolonged administration on granuloma formation around Schistosoma japonicum eggs in lung of sensitized mice. Parasitol. Res. 2011, 109, 1453–1459. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hafeez, E.H.; Ahmad, A.K.; Abdulla, A.M.; Aabdel-Wahab, S.; Mosalem, F.A. Therapeutic effect of alpha lipoic acid combined with praziquantel on liver fibrosis induced by Schistosoma mansoni challenged mice. Parasitol. Res. 2012, 111, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Ismail, M.; Metwally, A.; Farghaly, A.; Bruce, J.; Tao, L.F.; Bennett, J.L. Characterization of isolates of Schistosoma mansoni from Egyptian villagers that tolerate high doses of praziquantel. Am. J. Trop. Med. Hyg. 1996, 55, 214–218. [Google Scholar] [PubMed]
- Melman, S.D.; Steinauer, M.L.; Cunningham, C.; Kubatko, L.S.; Mwangi, I.N.; Wynn, N.B.; Mutuku, M.W.; Karanja, D.M.S.; Colley, D.G.; Black, C.L.; et al. Reduced susceptibility to praziquantel among naturally occurring Kenyan isolates of Schistosoma mansoni. PLoS Negl. Trop. Dis. 2009, 3, e504. [Google Scholar] [CrossRef] [PubMed]
- Fallon, P.G.; Sturrock, R.F.; Niang, A.C.; Doenhoff, M.J. Short report: Diminished susceptibility to praziquantel in a Senegal isolate of Schistosoma mansoni. Am. J. Trop. Med. Hyg. 1995, 53, 61–62. [Google Scholar] [PubMed]
- Couto, F.F.B.; Coelho, P.M.Z.; Araújo, N.; Kusel, J.R.; Katz, N.; Jannotti-Passos, L.K.; Mattos, A.C.A. Schistosoma mansoni: A method for inducing resistance to praziquantel using infected Biomphalaria glabrata snails. Mem. Inst. Oswaldo Cruz 2011, 106, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Fallon, P.G.; Doenhoff, M.J. Drug-resistant schistosomiasis: Resistance to praziquantel and oxamniquine induced in Schistosoma mansoni in mice is drug specific. Am. J. Trop. Med. Hyg. 1994, 51, 83–88. [Google Scholar] [PubMed]
- Ismail, M.M.; Taha, S.A.; Farghaly, A.M.; El-Azony, A.S. Laboratory induced resistance to praziquantel in experimental schistosomiasis. J. Egypt. Soc. Parasitol. 1994, 24, 685–695. [Google Scholar] [PubMed]
- Schuffenhauer, A.; Varin, T. Rule-Based Classification of Chemical Structures by Scaffold. Mol. Inf. 2011, 47, 646–664. [Google Scholar]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J. Nat. Prod. 2012, 75, 311–335. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.L. Natural products in drug discovery. Drug Discov. Today 2008, 13, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Bohlin, L.; Göransson, U.; Alsmark, C.; Wedén, C.; Backlund, A. Natural products in modern life science. Phytochem. Rev. 2010, 9, 279–301. [Google Scholar] [CrossRef]
- Harvey, A.L.; Clark, R.L.; Mackay, S.P.; Johnston, B.F. Current strategies for drug discovery through natural products. Expert Opin. Drug Discov. 2010, 5, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Datry, A.; Thellier, M.; Alfa-Cissé, O.; Danis, M.; Caumes, E. Ivermectin, a broad spectrum antiparasitic drug. Press. Méd. 2002, 31, 607–611. [Google Scholar]
- Butler, M.S. The role of natural product chemistry in drug discovery. J. Nat. Prod. 2004, 67, 2141–2153. [Google Scholar] [CrossRef] [PubMed]
- Guido, R.V.C.; Oliva, G.; Andricopulo, A.D. Modern drug discovery technologies: Opportunities and challenges in lead discovery. Comb. Chem. High Throughput Screen. 2011, 14, 830–839. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.D.; Chu, M.; Oza, U.; Rajgarhia, V. The value of natural products to future pharmaceutical discovery. Nat. Prod. Rep. 2007, 24, 1225–1244. [Google Scholar] [CrossRef] [PubMed]
- Ojima, I. Modern natural products chemistry and drug discovery. J. Med. Chem. 2008, 51, 2587–2588. [Google Scholar] [CrossRef] [PubMed]
- TimTec Natural Products Library. Available online: http://www.timtec.net/natural–compound–library.htm (accessed on 12 December 2014).
- IBScreen Natural Products. Available online: http://www.ibscreen.com/index.htm (accessed on 10 December 2014).
- Specs Natural Products. Available online: http://www.specs.net/ (accessed on 10 December 2014).
- Schuster, D.; Wolber, G. Identification of bioactive natural products by pharmacophore-based virtual screening. Curr. Pharm. Des. 2010, 16, 1666–1681. [Google Scholar] [CrossRef] [PubMed]
- Geldenhuys, W.J.; Bishayee, A.; Darvesh, A.S.; Carroll, R.T. Natural products of dietary origin as lead compounds in virtual screening and drug design. Curr. Pharm. Biotechnol. 2012, 13, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Ndjonka, D.; Rapado, L.N.; Silber, A.M.; Liebau, E.; Wrenger, C. Natural products as a source for treating neglected parasitic diseases. Int. J. Mol. Sci. 2013, 14, 3395–3439. [Google Scholar] [CrossRef] [PubMed]
- Erasmus, D.A. A comparative study of the reproductive system of mature, immature and “unisexual” female Schistosoma mansoni. Parasitology 1973, 67, 165–183. [Google Scholar] [CrossRef]
- Patocka, N.; Sharma, N.; Rashid, M.; Ribeiro, P. Serotonin signaling in Schistosoma mansoni: A serotonin-activated G protein-coupled receptor controls parasite movement. PLoS Pathog. 2014, 10, e1003878. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, P.; Patocka, N. Neurotransmitter transporters in schistosomes: Structure, function and prospects for drug discovery. Parasitol. Int. 2013, 62, 629–638. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.-G.; Brindley, P.J.; Wang, S.-Y.; Chen, Z. Schistosoma genomics: New perspectives on schistosome biology and host-parasite interaction. Annu. Rev. Genomics Hum. Genet. 2009, 10, 211–240. [Google Scholar] [CrossRef]
- Van Agtmael, M.A.; Eggelte, T.A.; van Boxtel, C.J. Artemisinin drugs in the treatment of malaria: From medicinal herb to registered medication. Trends Pharmacol. Sci. 1999, 20, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Ho, W.E.; Peh, H.Y.; Chan, T.K.; Wong, W.S.F. Artemisinins: Pharmacological actions beyond anti-malarial. Pharmacol. Ther. 2014, 142, 126–139. [Google Scholar] [CrossRef] [PubMed]
- Li, Y. Qinghaosu (artemisinin): Chemistry and pharmacology. Acta Pharmacol. Sin. 2012, 33, 1141–1146. [Google Scholar] [CrossRef] [PubMed]
- Frezza, T.F.; Fernandes de Oliveira, C.N.; Banin, T.M.; Garcia Rehder, V.L.; Boaventura, S., Jr.; Allegretti, S.M. Tegumentary changes in two different strains of Schistosoma mansoni treated with artemisinin and artesunic acid. Rev. Patol. Trop. 2013, 42, 309–321. [Google Scholar] [CrossRef]
- Li, H.-J.; Wang, W.; Qu, G.-L.; Tao, Y.-H.; Xing, Y.-T.; Li, Y.-Z.; Wei, J.-Y.; Dai, J.-R.; Liang, Y.-S. In-vivo activity of dihydroartemisinin against Schistosoma japonicum. Ann. Trop. Med. Parasitol. 2011, 105, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Wang, W.; Qu, G.; Li, Y.; Tao, Y.; Xing, Y.; Wang, X.; Dai, Y.; Wei, J.; Dai, J.; et al. Effect of the in vivo activity of dihydroartemisinin against Schistosoma mansoni infection in mice. Parasitol. Res. 2012, 110, 1727–1732. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.J.; Fu, L.F.; Shao, P.P.; Wu, F.Z.; Fan, C.Z.; Shu, H.; Ren, C.X.; Sheng, X.L. Experimental studies on antischistosomal activity of qinghaosu. Chin. Med. J. (Engl.) 1980, 60, 4222–4425. [Google Scholar]
- Shaohong, L.; Kumagai, T.; Qinghua, A.; Xiaolan, Y.; Ohmae, H.; Yabu, Y.; Siwen, L.; Liyong, W.; Maruyama, H.; Ohta, N. Evaluation of the anthelmintic effects of artesunate against experimental Schistosoma mansoni infection in mice using different treatment protocols. Parasitol. Int. 2006, 55, 63–68. [Google Scholar] [CrossRef]
- Wang, W.; Li, T.-Y.; Ji, Y.; Qu, G.-L.; Qian, Y.-L.; Li, H.-J.; Dai, J.-R.; Liang, Y.-S. Efficacy of artemether and artesunate in mice infected with praziquantel non-susceptible isolate of Schistosoma japonicum. Parasitol. Res. 2014, 113, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Jiraungkoorskul, W.; Sahaphong, S.; Sobhon, P.; Riengrojpitak, S.; Kangwanrangsan, N. Effects of praziquantel and artesunate on the tegument of adult Schistosoma mekongi harboured in mice. Parasitol. Int. 2005, 54, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Shuhua, X.; Chollet, J.; Weiss, N.; Bergquist, R.N.; Tanner, M. Preventive effect of artemether in experimental animals infected with Schistosoma mansoni. Parasitol. Int. 2000, 49, 19–24. [Google Scholar] [CrossRef] [PubMed]
- El-Beshbishi, S.N.; Taman, A.; El-Malky, M.; Azab, M.S.; El-Hawary, A.K.; El-Tantawy, D. In vivo effect of single oral dose of artemether against early juvenile stages of Schistosoma mansoni Egyptian strain. Exp. Parasitol. 2013, 135, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Shuhua, X.; Utzinger, J.; Chollet, J.; Endriss, Y.; N’Goran, E.K.; Tanner, M. Effect of artemether against Schistosoma haematobium in experimentally infected hamsters. Int. J. Parasitol. 2000, 30, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.-H.; Tanner, M.; Shen, B.-G.; Utzinger, J.; Chollet, J. Effect of artemether on the tegument of adult Schistosoma haematobium recovered from mice. Chin. J. Parasitol. Parasit. Dis. 2006, 24, 425–432. [Google Scholar]
- Li, H.-J.; Wang, W.; Li, Y.-Z.; Qu, G.-L.; Xing, Y.-T.; Tao, Y.-H.; Wei, J.-Y.; Dai, J.-R.; Liang, Y.-S. Effects of artemether, artesunate and dihydroartemisinin administered orally at multiple doses or combination in treatment of mice infected with Schistosoma japonicum. Parasitol. Res. 2011, 109, 515–519. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.-H.; Wu, Y.-L.; Tanner, M.; Wu, W.-M.; Utzinger, J.; Mei, J.-Y.; Scorneaux, B.; Chollet, J.; Zhai, Z. Schistosoma japonicum: In vitro effects of artemether combined with haemin depend on cultivation media and appraisal of artemether products appearing in the media. Parasitol. Res. 2003, 89, 459–466. [Google Scholar] [PubMed]
- Xiao, S.; Chollet, J.; Utzinger, J.; Matile, H.; Mei, J.; Tanner, M. Artemether administered together with haemin damages schistosomes in vitro. Trans. R. Soc. Trop. Med. Hyg. 2001, 95, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Mei, H.; LoVerde, P.T. Schistosoma mansoni: The developmental regulation and immunolocalization of antioxidant enzymes. Exp. Parasitol. 1997, 86, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Panic, G.; Duthaler, U.; Speich, B.; Keiser, J. Repurposing drugs for the treatment and control of helminth infections. Int. J. Parasitol. 2014, 4, 185–200. [Google Scholar]
- Liu, Y.-X.; Wu, W.; Liang, Y.-J.; Jie, Z.-L.; Wang, H.; Wang, W.; Huang, Y.-X. New uses for old drugs: The tale of artemisinin derivatives in the elimination of schistosomiasis japonica in China. Molecules 2014, 19, 15058–15074. [Google Scholar] [CrossRef] [PubMed]
- N’Goran, E.K.; Utzinger, J.; Gnaka, H.N.; Yapi, A.; N’Guessan, N.A.; Kigbafori, S.D.; Lengeler, C.; Chollet, J.; Shuhua, X.; Tanner, M. Randomized, double-blind, placebo-controlled trial of oral artemether for the prevention of patent Schistosoma haematobium infections. Am. J. Trop. Med. Hyg. 2003, 68, 24–32. [Google Scholar] [PubMed]
- Utzinger, J.; N’Goran, E.K.; N’Dri, A.; Lengeler, C.; Xiao, S.; Tanner, M. Oral artemether for prevention of Schistosoma mansoni infection: Randomised controlled trial. Lancet 2000, 355, 1320–1325. [Google Scholar] [CrossRef] [PubMed]
- Keiser, J.; N’Guessan, N.A.; Adoubryn, K.D.; Silué, K.D.; Vounatsou, P.; Hatz, C.; Utzinger, J.; N’Goran, E.K. Efficacy and safety of mefloquine, artesunate, mefloquine-artesunate, and praziquantel against Schistosoma haematobium: Randomized, exploratory open-label trial. Clin. Infect. Dis. 2010, 50, 1205–1213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Obonyo, C.O.; Muok, E.M.O.; Mwinzi, P.N.M. Efficacy of artesunate with sulfalene plus pyrimethamine versus praziquantel for treatment of Schistosoma mansoni in Kenyan children: An open-label randomised controlled trial. Lancet Infect. Dis. 2010, 10, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.A.; Mahgoub, H.M.; Magzoub, M.; Gasim, G.I.; Eldein, W.N.; Ahmed, A.A.A.; Adam, I. Artesunate plus sulfadoxine/pyrimethamine versus praziquantel in the treatment of Schistosoma mansoni in eastern Sudan. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 1062–1064. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Dong, H.-F.; Guo, Y.; Zhao, Q.-P.; Jiang, M.-S. Efficacy of praziquantel and artemisinin derivatives for the treatment and prevention of human schistosomiasis: A systematic review and meta-analysis. Parasit. Vectors 2011, 4, 201. [Google Scholar] [CrossRef] [PubMed]
- Pérez del Villar, L.; Burguillo, F.J.; López-Abán, J.; Muro, A. Systematic review and meta-analysis of artemisinin based therapies for the treatment and prevention of schistosomiasis. PLoS One 2012, 7, e45867. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.T.; Saify, Z.S.; Sultana, N.; Ahmad, I.; Saeed-Ul-Hassan, S.; Tariq, I.; Khanum, M. Malaria and artemisinin derivatives: An updated review. Mini Rev. Med. Chem. 2013, 13, 1879–1902. [Google Scholar] [CrossRef] [PubMed]
- Ledwitch, K.; Ogburn, R.; Cox, J.; Graham, R.; Fritzsche, A.; Gosnell, D.; Manning, T. Taxol: Efficacy against oral squamous cell carcinoma. Mini Rev. Med. Chem. 2013, 13, 509–521. [Google Scholar] [CrossRef] [PubMed]
- Huang, M.; Lu, J.-J.; Huang, M.-Q.; Bao, J.-L.; Chen, X.-P.; Wang, Y.-T. Terpenoids: Natural products for cancer therapy. Expert Opin. Investig. Drugs 2012, 21, 1801–1818. [Google Scholar] [CrossRef] [PubMed]
- De Moraes, J.; Almeida, A.A.C.; Brito, M.R.M.; Marques, T.H.C.; Lima, T.C.; de Sousa, D.P.; Nakano, E.; Mendonça, R.Z.; Freitas, R.M. Anthelmintic activity of the natural compound (+)-limonene epoxide against Schistosoma mansoni. Planta Med. 2013, 79, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Porto, T.S.; da Silva Filho, A.A.; Magalhães, L.G.; dos Santos, R.A.; Furtado, N.A.J.C.; Arakawa, N.S.; Said, S.; de Oliveira, D.C.R.; Gregório, L.E.; Rodrigues, V.; et al. Fungal transformation and schistosomicidal effects of pimaradienoic acid. Chem. Biodivers. 2012, 9, 1465–1474. [Google Scholar] [CrossRef] [PubMed]
- Bandeira, S.O.; Gaspar, F.; Pagula, F.P. African ethnobotany and healthcare: Emphasis on mozambique. Pharm. Biol. 2001, 39 (Suppl. S1), 70–73. [Google Scholar] [CrossRef]
- Ramalhete, C.; Magalhães, L.G.; Rodrigues, V.; Mulhovo, S.; Da Silva Filho, A.A.; Ferreira, M.-J.U. In vitro schistosomicidal activity of balsaminol F and karavilagenin C. Planta Med. 2012, 78, 1912–1917. [Google Scholar] [CrossRef] [PubMed]
- Ramalhete, C.; Lopes, D.; Molnár, J.; Mulhovo, S.; Rosário, V.E.; Ferreira, M.-J.U. Karavilagenin C derivatives as antimalarials. Bioorg. Med. Chem. 2011, 19, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Ramalhete, C.; da Cruz, F.P.; Lopes, D.; Mulhovo, S.; Rosário, V.E.; Prudêncio, M.; Ferreira, M.-J.U. Triterpenoids as inhibitors of erythrocytic and liver stages of Plasmodium infections. Bioorg. Med. Chem. 2011, 19, 7474–7481. [Google Scholar] [CrossRef] [PubMed]
- Johann, S.; Oliveira, F.B.; Siqueira, E.P.; Cisalpino, P.S.; Rosa, C.A.; Alves, T.M.A.; Zani, C.L.; Cota, B.B. Activity of compounds isolated from Baccharis dracunculifolia D.C. (Asteraceae) against Paracoccidioides brasiliensis. Med. Mycol. 2012, 50, 843–851. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.P.N.; Oliveira, G.L.S.; de Carvalho, R.B.F.; de Sousa, D.P.; Freitas, R.M.; Pinto, P.L.S.; de Moraes, J. Antischistosomal activity of the terpene nerolidol. Molecules 2014, 19, 3793–3803. [Google Scholar] [CrossRef] [PubMed]
- Barth, L.R.; Fernandes, A.P.M.; Ribeiro-Paes, J.T.; Rodrigues, V. Effects of Goyazensolide during in Vitro Cultivation of Schistosoma mansoni. Mem. Inst. Oswaldo Cruz 1997, 92, 427–429. [Google Scholar] [CrossRef] [PubMed]
- Jisaka, M.; Kawanaka, M.; Sugiyama, H.; Takegawa, K.; Huffman, M.A.; Ohigashi, H.; Koshimizu, K. Antischistosomal activities of sesquiterpene lactones and steroid glucosides from Vernonia amygdalina, possibly used by wild chimpanzees against parasite-related diseases. Biosci. Biotechnol. Biochem. 1992, 56, 845–846. [Google Scholar] [CrossRef] [PubMed]
- Berriman, M.; Haas, B.J.; LoVerde, P.T.; Wilson, R.A.; Dillon, G.P.; Cerqueira, G.C.; Mashiyama, S.T.; Al-Lazikani, B.; Andrade, L.F.; Ashton, P.D.; et al. The genome of the blood fluke Schistosoma mansoni. Nature 2009, 460, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Consortium, F.A. The Schistosoma japonicum genome reveals features of host-parasite interplay. Nature 2009, 460, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Ross, F.; Hernández, P.; Porcal, W.; López, G.V.; Cerecetto, H.; González, M.; Basika, T.; Carmona, C.; Fló, M.; Maggioli, G.; et al. Identification of thioredoxin glutathione reductase inhibitors that kill cestode and trematode parasites. PLoS One 2012, 7, e35033. [Google Scholar] [CrossRef] [PubMed]
- Beckmann, S.; Leutner, S.; Gouignard, N.; Dissous, C.; Grevelding, C.G. Protein kinases as potential targets for novel anti-schistosomal strategies. Curr. Pharm. Des. 2012, 18, 3579–3594. [Google Scholar] [PubMed]
- Ray, D.; Williams, D.L. Characterization of the phytochelatin synthase of Schistosoma mansoni. PLoS Negl. Trop. Dis. 2011, 5, e1168. [Google Scholar] [CrossRef] [PubMed]
- Ziniel, P.D.; Desai, J.; Cass, C.L.; Gatto, C.; Oldfield, E.; Williams, D.L. Characterization of potential drug targets farnesyl diphosphate synthase and geranylgeranyl diphosphate synthase in Schistosoma mansoni. Antimicrob. Agents Chemother. 2013, 57, 5969–5976. [Google Scholar] [PubMed]
- Lancelot, J.; Caby, S.; Dubois-Abdesselem, F.; Vanderstraete, M.; Trolet, J.; Oliveira, G.; Bracher, F.; Jung, M.; Pierce, R.J. Schistosoma mansoni Sirtuins: Characterization and potential as chemotherapeutic targets. PLoS Negl. Trop. Dis. 2013, 7, e2428. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Wang, Y. Pharmacological actions and therapeutic applications of Salvia miltiorrhiza depside salt and its active components. Acta Pharmacol. Sin. 2012, 33, 1119–1130. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Zuo, Z.; Chow, M.S.S. Danshen: An overview of its chemistry, pharmacology, pharmacokinetics, and clinical use. J. Clin. Pharmacol. 2005, 45, 1345–1359. [Google Scholar] [CrossRef] [PubMed]
- Lea, W.A.; Jadhav, A.; Rai, G.; Sayed, A.A.; Cass, C.L.; Inglese, J.; Williams, D.L.; Austin, C.P.; Simeonov, A. A 1,536-well-based kinetic HTS assay for inhibitors of Schistosoma mansoni thioredoxin glutathione reductase. Assay Drug Dev. Technol. 2008, 6, 551–555. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.L.; Bonilla, M.; Gladyshev, V.N.; Salinas, G. Thioredoxin glutathione reductase-dependent redox networks in platyhelminth parasites. Antioxid. Redox Signal. 2013, 19, 735–745. [Google Scholar] [CrossRef] [PubMed]
- Kuntz, A.N.; Davioud-Charvet, E.; Sayed, A.A.; Califf, L.L.; Dessolin, J.; Arnér, E.S.J.; Williams, D.L. Thioredoxin glutathione reductase from Schistosoma mansoni: An essential parasite enzyme and a key drug target. PLoS Med. 2007, 4, e206. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, M.; Denicola, A.; Marino, S.M.; Gladyshev, V.N.; Salinas, G. Linked thioredoxin-glutathione systems in platyhelminth parasites: Alternative pathways for glutathione reduction and deglutathionylation. J. Biol. Chem. 2011, 286, 4959–4967. [Google Scholar] [CrossRef] [PubMed]
- Veras, L.M.; Guimaraes, M.A.; Campelo, Y.D.; Vieira, M.M.; Nascimento, C.; Lima, D.F.; Vasconcelos, L.; Nakano, E.; Kuckelhaus, S.S.; Batista, M.C.; et al. Activity of epiisopiloturine against Schistosoma mansoni. Curr. Med. Chem. 2012, 19, 2051–2058. [Google Scholar] [CrossRef] [PubMed]
- De Moraes, J.; Nascimento, C.; Lopes, P.O.M.V.; Nakano, E.; Yamaguchi, L.F.; Kato, M.J.; Kawano, T. Schistosoma mansoni: In vitro schistosomicidal activity of piplartine. Exp. Parasitol. 2011, 127, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, A.F.; Fonseca, S.A.; César, F.A.; de Azevedo Albuquerque, M.C.P.; Santana, J.V.; Santana, A.E.G. A penta-substituted pyridine alkaloid from the rhizome of Jatropha elliptica (Pohl) Muell. Arg. is active against Schistosoma mansoni and Biomphalaria glabrata. Parasitol. Res. 2014, 113, 1077–1084. [Google Scholar] [CrossRef] [PubMed]
- Silva-Moraes, V.; Couto, F.F.B.; Vasconcelos, M.M.; Araújo, N.; Coelho, P.M.Z.; Katz, N.; Grenfell, R.F.Q.; Fernanda, F.; Couto, B.; Marcos, P.; et al. Antischistosomal activity of a calcium channel antagonist on schistosomula and adult Schistosoma mansoni worms. Mem. Inst. Oswaldo Cruz 2013, 108, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.A.; Magalhães, L.G.; Tiossi, R.F.J.; Kuehn, C.C.; Oliveira, L.G.R.; Rodrigues, V.; McChesney, J.D.; Bastos, J.K. Evaluation of the schistosomicidal activity of the steroidal alkaloids from Solanum lycocarpum fruits. Parasitol. Res. 2012, 111, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-M.; Coultas, K. Identification of plumbagin and sanguinarine as effective chemotherapeutic agents for treatment of schistosomiasis. Int. J. Parasitol. Drugs Drug Resist. 2013, 3, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Corrêa Soares, J.B.R.; Menezes, D.; Vannier-Santos, M.A.; Ferreira-Pereira, A.; Almeida, G.T.; Venancio, T.M.; Verjovski-Almeida, S.; Zishiri, V.K.; Kuter, D.; Hunter, R.; et al. Interference with hemozoin formation represents an important mechanism of schistosomicidal action of antimalarial quinoline methanols. PLoS Negl. Trop. Dis. 2009, 3, e477. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oliveira, M.F.; D’Avila, J.C.P.; Torres, C.R.; Oliveira, P.L.; Tempone, A.J.; Rumjanek, F.D.; Braga, C.M.S.; Silva, J.R.; Dansa-Petretski, M.; Oliveira, M.A.; et al. Haemozoin in Schistosoma mansoni. Mol. Biochem. Parasitol. 2000, 111, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, M.F.; Timm, B.L.; Machado, E.A.; Miranda, K.; Attias, M.; Silva, J.R.; Dansa-Petretski, M.; de Oliveira, M.A.; de Souza, W.; Pinhal, N.M.; et al. On the pro-oxidant effects of haemozoin. FEBS Lett. 2002, 512, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Keiser, J.; Chollet, J.; Xiao, S.-H.; Mei, J.-Y.; Jiao, P.-Y.; Utzinger, J.; Tanner, M. Mefloquine—An aminoalcohol with promising antischistosomal properties in mice. PLoS Negl. Trop. Dis. 2009, 3, e350. [Google Scholar] [CrossRef] [PubMed]
- Manneck, T.; Haggenmüller, Y.; Keiser, J. Morphological effects and tegumental alterations induced by mefloquine on schistosomula and adult flukes of Schistosoma mansoni. Parasitology 2010, 137, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Manneck, T.; Braissant, O.; Ellis, W.; Keiser, J. Schistosoma mansoni: Antischistosomal activity of the four optical isomers and the two racemates of mefloquine on schistosomula and adult worms in vitro and in vivo. Exp. Parasitol. 2011, 127, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Ingram, K.; Ellis, W.; Keiser, J. Antischistosomal activities of mefloquine-related arylmethanols. Antimicrob. Agents Chemother. 2012, 56, 3207–3215. [Google Scholar] [CrossRef] [PubMed]
- Keiser, J.; Manneck, T.; Vargas, M. Interactions of mefloquine with praziquantel in the Schistosoma mansoni mouse model and in vitro. J. Antimicrob. Chemother. 2011, 66, 1791–1797. [Google Scholar] [CrossRef] [PubMed]
- Mierziak, J.; Kostyn, K.; Kulma, A. Flavonoids as Important Molecules of Plant Interactions with the Environment. Molecules 2014, 19, 16240–16265. [Google Scholar] [CrossRef] [PubMed]
- Buer, C.S.; Imin, N.; Djordjevic, M.A. Flavonoids: New roles for old molecules. J. Integr. Plant Biol. 2010, 52, 98–111. [Google Scholar] [CrossRef] [PubMed]
- Ravishankar, D.; Rajora, A.K.; Greco, F.; Osborn, H.M.I. Flavonoids as prospective compounds for anti-cancer therapy. Int. J. Biochem. Cell Biol. 2013, 45, 2821–2831. [Google Scholar] [CrossRef] [PubMed]
- Bhullar, K.S.; Rupasinghe, H.P.V. Polyphenols: Multipotent therapeutic agents in neurodegenerative diseases. Oxid. Med. Cell. Longev. 2013, 2013. [Google Scholar] [CrossRef]
- Van Dam, R.M.; Naidoo, N.; Landberg, R. Dietary flavonoids and the development of type 2 diabetes and cardiovascular diseases: Review of recent findings. Curr. Opin. Lipidol. 2013, 24, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Lyddiard, J.R.A.; Whitfield, P.J.; Bartlett, A. Antischistosomal bioactivity of isoflavonoids from Millettia thonningii (Leguminosae). J. Parasitol. 2002, 88, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Cunha, N.L.; Uchôa, C.J.D.M.; Cintra, L.S.; de Souza, H.C.; Peixoto, J.A.; Silva, C.P.; Magalhães, L.G.; Gimenez, V.M.M.; Groppo, M.; Rodrigues, V.; et al. In vitro schistosomicidal activity of some brazilian cerrado species and their isolated compounds. Evid. Based. Complement. Altern. Med. 2012, 2012, 173614. [Google Scholar] [CrossRef]
- Braguine, C.G.; Bertanha, C.S.; Gonçalves, U.O.; Magalhães, L.G.; Rodrigues, V.; Melleiro Gimenez, V.M.; Groppo, M.; Silva, M.L.A.E.; Cunha, W.R.; Januário, A.H.; et al. Schistosomicidal evaluation of flavonoids from two species of Styrax against Schistosoma mansoni adult worms. Pharm. Biol. 2012, 50, 925–929. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, I.; Kellenberger, E.; Said-Hassane, F.; Villa, P.; Rognan, D.; Lobstein, A.; Haiech, J.; Hibert, M.; Schuber, F.; Muller-Steffner, H. Identification by high-throughput screening of inhibitors of Schistosoma mansoni NAD(+) catabolizing enzyme. Bioorg. Med. Chem. 2010, 18, 7900–7910. [Google Scholar] [CrossRef] [PubMed]
- You, J.-Y.; Peng, C.; Liu, X.; Ji, X.-J.; Lu, J.; Tong, Q.; Wei, P.; Cong, L.; Li, Z.; Huang, H. Enzymatic hydrolysis and extraction of arachidonic acid rich lipids from Mortierella alpina. Bioresour. Technol. 2011, 102, 6088–6094. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Ji, X.; Zhang, H.; Fu, N.; Yan, L.; Deng, Z.; Huang, H. Development of a defined medium for arachidonic acid production by Mortierella alpina using a visualization method. Appl. Biochem. Biotechnol. 2012, 168, 1516–527. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.-J.; Ren, L.-J.; Nie, Z.-K.; Huang, H.; Ouyang, P.-K. Fungal arachidonic acid-rich oil: Research, development and industrialization. Crit. Rev. Biotechnol. 2014, 34, 197–214. [Google Scholar] [CrossRef] [PubMed]
- El Ridi, R.; Aboueldahab, M.; Tallima, H.; Salah, M.; Mahana, N.; Fawzi, S.; Mohamed, S.H.; Fahmy, O.M. In vitro and in vivo activities of arachidonic acid against Schistosoma mansoni and Schistosoma haematobium. Antimicrob. Agents Chemother. 2010, 54, 3383–3389. [Google Scholar] [CrossRef] [PubMed]
- Tallima, H.; Salah, M.; El-Ridi, R. In vitro and in vivo effects of unsaturated fatty acids on Schistosoma mansoni and S. haematobium lung-stage larvae. J. Parasitol. 2005, 91, 1094–1102. [Google Scholar] [CrossRef] [PubMed]
- El Ridi, R.; Tallima, H. Equilibrium in lung schistosomula sphingomyelin breakdown and biosynthesis allows very small molecules, but not antibody, to access proteins at the host-parasite interface. J. Parasitol. 2006, 92, 730–737. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials. Arachidonic Acid Treatment Against Schistosomiasis Infection in Children. 2014; Fact Sheet. Available online: https://clinicaltrials.gov/ct2/show/NCT02144389 (accessed on 10 December 2014). [Google Scholar]
- Schmidt, T.J.; Khalid, S.A.; Romanha, A.J.; Alves, T.M.; Biavatti, M.W.; Brun, R.; da Costa, F.B.; de Castro, S.L.; Ferreira, V.F.; de Lacerda, M.V.G.; et al. The potential of secondary metabolites from plants as drugs or leads against protozoan neglected diseases—Part II. Curr. Med. Chem. 2012, 19, 2176–2228. [Google Scholar] [PubMed]
- De Lima, O.G.; d’ Albuquerque, I.L.; Maciel, G.M.; Maciel, M.C. Antimicrobial substances of superior plants. XXVII. Isolation of plumbagin from Plumbago scandens L. Rev. Inst. Antibiot. (Recife) 1968, 8, 95–97. [Google Scholar]
- Cichewicz, R.H.; Lim, K.-C.; McKerrow, J.H.; Nair, M.G. Kwanzoquinones A–G and other constituents of Hemerocallis fulva “Kwanzo” roots and their activity against the human pathogenic trematode Schistosoma mansoni. Tetrahedron 2002, 58, 8597–8606. [Google Scholar] [CrossRef]
- Magalhães, L.G.; Kapadia, G.J.; da Silva Tonuci, L.R.; Caixeta, S.C.; Parreira, N.A.; Rodrigues, V.; Da Silva Filho, A.A. In vitro schistosomicidal effects of some phloroglucinol derivatives from Dryopteris species against Schistosoma mansoni adult worms. Parasitol. Res. 2010, 106, 395–401. [Google Scholar] [CrossRef] [PubMed]
- Abdulla, M.-H.; Ruelas, D.S.; Wolff, B.; Snedecor, J.; Lim, K.-C.; Xu, F.; Renslo, A.R.; Williams, J.; McKerrow, J.H.; Caffrey, C.R. Drug discovery for schistosomiasis: Hit and lead compounds identified in a library of known drugs by medium-throughput phenotypic screening. PLoS Negl. Trop. Dis. 2009, 3, e478. [Google Scholar] [CrossRef] [PubMed]
- Ramos, H.P.; Simão, M.R.; de Souza, J.M.; Magalhães, L.G.; Rodrigues, V.; Ambrósio, S.R.; Said, S. Evaluation of dihydroisocoumarins produced by the endophytic fungus Arthrinium state of Apiospora montagnei against Schistosoma mansoni. Nat. Prod. Res. 2013, 27, 2240–2243. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, L.G.; Machado, C.B.; Morais, E.R.; Moreira, E.B.D.C.; Soares, C.S.; da Silva, S.H.; Da Silva Filho, A.A.; Rodrigues, V. In vitro schistosomicidal activity of curcumin against Schistosoma mansoni adult worms. Parasitol. Res. 2009, 104, 1197–1201. [Google Scholar] [CrossRef] [PubMed]
- Jana, N.R.; Dikshit, P.; Goswami, A.; Nukina, N. Inhibition of proteasomal function by curcumin induces apoptosis through mitochondrial pathway. J. Biol. Chem. 2004, 279, 11680–11685. [Google Scholar] [CrossRef] [PubMed]
- Milacic, V.; Banerjee, S.; Landis-Piwowar, K.R.; Sarkar, F.H.; Majumdar, A.P.N.; Dou, Q.P. Curcumin inhibits the proteasome activity in human colon cancer cells in vitro and in vivo. Cancer Res. 2008, 68, 7283–7292. [Google Scholar] [CrossRef] [PubMed]
- Sticher, O. Natural product isolation. Nat. Prod. Rep. 2008, 25, 517–554. [Google Scholar] [CrossRef] [PubMed]
- Bucar, F.; Wube, A.; Schmid, M. Natural product isolation--how to get from biological material to pure compounds. Nat. Prod. Rep. 2013, 30, 525–545. [Google Scholar] [CrossRef] [PubMed]
- Keiser, J. In vitro and in vivo trematode models for chemotherapeutic studies. Parasitology 2010, 137, 589–603. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, B.; Bickle, Q.; Yousif, F.; Fakorede, F.; Mouries, M.-A.; Nwaka, S. Schistosomes: Challenges in compound screening. Expert Opin. Drug Discov. 2007, 2, S53–S61. [Google Scholar] [CrossRef] [PubMed]
- Mansour, N.R.; Bickle, Q.D. Comparison of microscopy and Alamar blue reduction in a larval based assay for schistosome drug screening. PLoS Negl. Trop. Dis. 2010, 4, e795. [Google Scholar] [CrossRef] [PubMed]
- Paveley, R.A.; Mansour, N.R.; Hallyburton, I.; Bleicher, L.S.; Benn, A.E.; Mikic, I.; Guidi, A.; Gilbert, I.H.; Hopkins, A.L.; Bickle, Q.D. Whole organism high-content screening by label-free, image-based Bayesian classification for parasitic diseases. PLoS Negl. Trop. Dis. 2012, 6, e1762. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Moody-Davis, A.; Saha, U.; Suzuki, B.M.; Asarnow, D.; Chen, S.; Arkin, M.; Caffrey, C.R.; Singh, R. Quantification and clustering of phenotypic screening data using time-series analysis for chemotherapy of schistosomiasis. BMC Genomics 2012, 13 (Suppl. 1), S4. [Google Scholar] [PubMed]
- Paveley, R.A.; Bickle, Q.D. Automated imaging and other developments in whole-organism anthelmintic screening. Parasite Immunol. 2007, 35, 302–313. [Google Scholar] [CrossRef]
- Singh, S.; Carpenter, A.E.; Genovesio, A. Increasing the Content of High-Content Screening: An Overview. J. Biomol. Screen. 2014, 19, 640–650. [Google Scholar] [CrossRef] [PubMed]
- Asarnow, D.E.; Singh, R. Segmenting the etiological agent of schistosomiasis for high-content screening. IEEE Trans. Med. Imaging 2013, 32, 1007–1018. [Google Scholar] [CrossRef] [PubMed]
- Peak, E.; Chalmers, I.W.; Hoffmann, K.F. Development and validation of a quantitative, high-throughput, fluorescent-based bioassay to detect schistosoma viability. PLoS Negl. Trop. Dis. 2010, 4, e759. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.-L.; Chan, D.S.-H.; Leung, C.-H. Molecular docking for virtual screening of natural product databases. Chem. Sci. 2011, 2, 1656–1665. [Google Scholar] [CrossRef]
- Kannan, S.; Melesina, J.; Hauser, A.-T.; Chakrabarti, A.; Heimburg, T.; Schmidtkunz, K.; Walter, A.; Marek, M.; Pierce, R.J.; Romier, C.; et al. Discovery of inhibitors of Schistosoma mansoni HDAC8 by combining homology modeling, virtual screening, and in vitro validation. J. Chem. Inf. Model. 2014, 54, 3005–3019. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Dyer, D.; Wang, J.; Wang, S.; Du, X.; Xu, B.; Zhang, H.; Wang, X.; Hu, W. 3-Oxoacyl-ACP reductase from Schistosoma japonicum: Integrated in silico-in vitro strategy for discovering antischistosomal lead compounds. PLoS One 2013, 8, e64984. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Dyer, D.H.; Cheng, J.; Wang, J.; Wang, S.; Yang, Z.; Wang, X.; Hu, W. Aldose reductase from Schistosoma japonicum: Crystallization and structure-based inhibitor screening for discovering antischistosomal lead compounds. Parasit. Vectors 2013, 6, 162. [Google Scholar] [CrossRef] [PubMed]
- Postigo, M.P.; Guido, R.V.C.; Oliva, G.; Castilho, M.S.; da R Pitta, I.; de Albuquerque, J.F.C.; Andricopulo, A.D. Discovery of new inhibitors of Schistosoma mansoni PNP by pharmacophore-based virtual screening. J. Chem. Inf. Model. 2010, 50, 1693–1705. [Google Scholar] [CrossRef] [PubMed]
- Braga, R.C.; Andrade, C.H. Assessing the performance of 3D pharmacophore models in virtual screening: How good are they? Curr. Top. Med. Chem. 2013, 13, 1127–1138. [Google Scholar] [CrossRef] [PubMed]
- Valli, M.; dos Santos, R.N.; Figueira, L.D.; Nakajima, C.H.; Castro-Gamboa, I.; Andricopulo, A.D.; Bolzani, V.S. Development of a natural products database from the biodiversity of Brazil. J. Nat. Prod. 2013, 76, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Dunkel, M.; Fullbeck, M.; Neumann, S.; Preissner, R. SuperNatural: A searchable database of available natural compounds. Nucleic Acids Res. 2006, 34, D678–D683. [Google Scholar] [CrossRef] [PubMed]
- Ehrman, T.M.; Barlow, D.J.; Hylands, P.J. Phytochemical informatics of traditional Chinese medicine and therapeutic relevance. J. Chem. Inf. Model. 2006, 47, 2316–2334. [Google Scholar] [CrossRef]
- Davis, G.D.J.; Vasanthi, A.H.R. Seaweed metabolite database (SWMD): A database of natural compounds from marine algae. Bioinformation 2011, 5, 361–364. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.H.; Zhu, F.; Liu, X.; Shi, Z.; Zhang, J.X.; Yang, S.Y.; Wei, Y.Q.; Chen, Y.Z. Virtual screening methods as tools for drug lead discovery from large chemical libraries. Curr. Med. Chem. 2012, 19, 5562–5571. [Google Scholar] [CrossRef] [PubMed]
- Braga, R.C.; Alves, V.M.; Silva, A.C.; Nascimento, M.N.; Silva, F.C.; Liao, L.M.; Andrade, C.H. Virtual screening strategies in medicinal chemistry: The state of the art and current challenges. Curr. Top. Med. Chem. 2014, 14, 1899–1912. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, K. Databases aim to bridge the East-West divide of drug discovery. Nat. Med. 2011, 17, 1531. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Y.-C. TCM Database@Taiwan: The world’s largest traditional Chinese medicine database for drug screening in silico. PLoS One 2011, 6, e15939. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dictionary of Natural Products. Available online: http://www.chemnetbase.com/ (accessed on 12 December 2014).
- Ye, H.; Ye, L.; Kang, H.; Zhang, D.; Tao, L.; Tang, K.; Liu, X.; Zhu, R.; Liu, Q.; Chen, Y.Z.; et al. HIT: linking herbal active ingredients to targets. Nucleic Acids Res. 2011, 39, D1055–D1059. [Google Scholar] [CrossRef] [PubMed]
- Mangal, M.; Sagar, P.; Singh, H.; Raghava, G.P.S.; Agarwal, S.M. NPACT: Naturally Occurring Plant-based Anti-cancer Compound-Activity-Target database. Nucleic Acids Res. 2013, 41, D1124–D1129. [Google Scholar] [CrossRef] [PubMed]
- Database of Indonesian Medicinal Plants. Available online: http://herbaldb.farmasi.ui.ac.id/ (accessed on 12 December 2014).
- Greenpharma Natural Compound Library. Available online: http://www.greenpharma.com/products/ (accessed on 12 December 2014).
- Yue, Y.; Chu, G.-X.; Liu, X.-S.; Tang, X.; Wang, W.; Liu, G.-J.; Yang, T.; Ling, T.-J.; Wang, X.-G.; Zhang, Z.-Z.; et al. TMDB: A literature-curated database for small molecular compounds found from tea. BMC Plant Biol. 2014, 14, 243. [Google Scholar] [CrossRef] [PubMed]
- Ashfaq, U.A.; Mumtaz, A.; Qamar, T.U.; Fatima, T. MAPS Database: Medicinal plant Activities, Phytochemical and Structural Database. Bioinformation 2013, 9, 993–995. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-C.; Wang, C.-C.; Chen, I.-S.; Jheng, J.-L.; Li, J.-H.; Tung, C.-W. TIPdb: A database of anticancer, antiplatelet, and antituberculosis phytochemicals from indigenous plants in Taiwan. Sci. World J. 2013, 2013. [Google Scholar] [CrossRef]
- Tung, C.-W.; Lin, Y.-C.; Chang, H.-S.; Wang, C.-C.; Chen, I.-S.; Jheng, J.-L.; Li, J.-H. TIPdb-3D: the three-dimensional structure database of phytochemicals from Taiwan indigenous plants. Database (Oxford) 2014, 2014, bau055. [Google Scholar] [CrossRef]
- AMRI’s Natural Product Library. Available online: http://www.amriglobal.com/ (accessed on 12 December 2014).
- Wermuth, C.; Ganellin, C.; Lindberg, P.; Mitscher, L. Glossary of terms used in medicinal chemistry (IUPAC Recommendations 1998). Pure Appl. Chem. 1998, 70, 1129–1143. [Google Scholar] [CrossRef]
- Güner, O.; Clement, O.; Kurogi, Y. Pharmacophore modeling and three dimensional database searching for drug design using catalyst: Recent advances. Curr. Med. Chem. 2004, 11, 2991–3005. [Google Scholar] [CrossRef] [PubMed]
- Richmond, N.J.; Abrams, C.A.; Wolohan, P.R.N.; Abrahamian, E.; Willett, P.; Clark, R.D. GALAHAD: 1. pharmacophore identification by hypermolecular alignment of ligands in 3D. J. Comput. Aided Mol. Des. 2006, 20, 567–587. [Google Scholar] [CrossRef] [PubMed]
- Barnum, D.; Greene, J.; Smellie, A.; Sprague, P. Identification of common functional configurations among molecules. J. Chem. Inf. Comput. Sci. 1996, 36, 563–571. [Google Scholar] [CrossRef] [PubMed]
- Dixon, S.L.; Smondyrev, A.M.; Knoll, E.H.; Rao, S.N.; Shaw, D.E.; Friesner, R.A. PHASE: A new engine for pharmacophore perception, 3D QSAR model development, and 3D database screening: 1. Methodology and preliminary results. J. Comput. Aided. Mol. Des. 2006, 20, 647–671. [Google Scholar] [CrossRef] [PubMed]
- Cherkasov, A.; Muratov, E.N.; Fourches, D.; Varnek, A.; Baskin, I.I.; Cronin, M.; Dearden, J.; Gramatica, P.; Martin, Y.C.; Todeschini, R.; et al. QSAR modeling: Where have you been? Where are you going to? J. Med. Chem. 2014, 57, 4977–5010. [Google Scholar] [CrossRef] [PubMed]
- Melo-Filho, C.C.; Braga, R.C.; Andrade, C.H. 3D-QSAR Approaches in Drug Design: Perspectives to Generate Reliable CoMFA Models. Curr. Comput. Aided Drug Des. 2014, 10, 148–159. [Google Scholar] [CrossRef] [PubMed]
- Neves, B.J.; Bueno, R.V.; Braga, R.C.; Andrade, C.H. Discovery of new potential hits of Plasmodium falciparum enoyl-ACP reductase through ligand- and structure-based drug design approaches. Bioorg. Med. Chem. Lett. 2013, 23, 2436–2441. [Google Scholar] [CrossRef] [PubMed]
- Bueno, R.V.; Toledo, N.R.; Neves, B.J.; Braga, R.C.; Andrade, C.H. Structural and chemical basis for enhanced affinity to a series of mycobacterial thymidine monophosphate kinase inhibitors: Fragment-based QSAR and QM/MM docking studies. J. Mol. Model. 2013, 19, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Tropsha, A. Best Practices for QSAR Model Development, Validation, and Exploitation. Mol. Inf. 2010, 29, 476–488. [Google Scholar] [CrossRef]
- Zhang, L.; Fourches, D.; Sedykh, A.; Zhu, H.; Golbraikh, A.; Ekins, S.; Clark, J.; Connelly, M.C.; Sigal, M.; Hodges, D.; et al. Discovery of novel antimalarial compounds enabled by QSAR-based virtual screening. J. Chem. Inf. Model. 2013, 53, 475–492. [Google Scholar] [CrossRef] [PubMed]
- Roy, K.; Mitra, I.; Kar, S.; Ojha, P.K.; Das, R.N.; Kabir, H. Comparative studies on some metrics for external validation of QSPR models. J. Chem. Inf. Model. 2012, 52, 396–408. [Google Scholar] [CrossRef] [PubMed]
- Roy, K.; Mitra, I. On the use of the metric rm2 as an effective tool for validation of QSAR models in computational drug design and predictive toxicology. Mini Rev. Med. Chem. 2012, 12, 491–504. [Google Scholar] [CrossRef] [PubMed]
- Roy, K.; Mitra, I.; Ojha, P.K.; Kar, S.; Das, R.N.; Kabir, H. Introduction of rm2 (rank) metric incorporating rank-order predictions as an additional tool for validation of QSAR/QSPR models. Chemom. Intell. Lab. Syst. 2012, 118, 200–210. [Google Scholar] [CrossRef]
- Gramatica, P. On the development and validation of QSAR models. Methods Mol. Biol. 2013, 930, 499–526. [Google Scholar] [PubMed]
- Chirico, N.; Gramatica, P. Real external predictivity of QSAR models: How to evaluate it? Comparison of different validation criteria and proposal of using the concordance correlation coefficient. J. Chem. Inf. Model. 2011, 51, 2320–2335. [Google Scholar] [CrossRef] [PubMed]
- Chirico, N.; Gramatica, P. Real external predictivity of QSAR models. Part 2; New intercomparable thresholds for different validation criteria and the need for scatter plot inspection. J. Chem. Inf. Model. 2012, 52, 2044–2058. [Google Scholar] [CrossRef] [PubMed]
- Ten Brink, T.; Exner, T.E. pKa based protonation states and microspecies for protein-ligand docking. J. Comput. Aided Mol. Des. 2010, 24, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Klebe, G. Virtual ligand screening: Strategies, perspectives and limitations. Drug Discov. Today 2006, 11, 580–594. [Google Scholar] [CrossRef] [PubMed]
- Lionta, E.; Spyrou, G.; Vassilatis, D.K.; Cournia, Z. Structure-based virtual screening for drug discovery: Principles, applications and recent advances. Curr. Top. Med. Chem. 2014, 14, 1923–1938. [Google Scholar] [CrossRef] [PubMed]
- McGann, M. FRED pose prediction and virtual screening accuracy. J. Chem. Inf. Model. 2011, 51, 578–596. [Google Scholar] [CrossRef] [PubMed]
- McGann, M. FRED and HYBRID docking performance on standardized datasets. J. Comput. Aided Mol. Des. 2012, 26, 897–906. [Google Scholar] [CrossRef] [PubMed]
- OEDocking. version 3.0.1. OpenEye Scientific Software, Inc.: Santa Fe, NM, USA, 2014. Available online: http://www.eyesopen.com/oedocking (accessed on 10 December 2014).
- Toledo Warshaviak, D.; Golan, G.; Borrelli, K.W.; Zhu, K.; Kalid, O. Structure-based virtual screening approach for discovery of covalently bound ligands. J. Chem. Inf. Model. 2014, 54, 1941–1950. [Google Scholar] [CrossRef] [PubMed]
- Schellhammer, I.; Rarey, M. FlexX-Scan: Fast, structure-based virtual screening. Proteins 2004, 57, 504–517. [Google Scholar] [CrossRef] [PubMed]
- Verdonk, M.L.; Cole, J.C.; Hartshorn, M.J.; Murray, C.W.; Taylor, R.D. Improved protein-ligand docking using GOLD. Proteins 2003, 52, 609–623. [Google Scholar] [CrossRef] [PubMed]
- Waszkowycz, B.; Clark, D.E.; Gancia, E. Outstanding challenges in protein-ligand docking and structure-based virtual screening. Wiley Interdiscip. Rev. Comput. Mol. Sci. 2011, 1, 229–259. [Google Scholar] [CrossRef]
- Spyrakis, F.; BidonChanal, A.; Barril, X.; Luque, F.J. Protein flexibility and ligand recognition: Challenges for molecular modeling. Curr. Top. Med. Chem. 2011, 11, 192–210. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Bleylevens, I.W.M.; Bitorina, A.V.; Wichapong, K.; Nicolaes, G.A.F. Optimization of compound ranking for structure-based virtual ligand screening using an established FRED-Surflex consensus approach. Chem. Biol. Drug Des. 2014, 83, 37–51. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Eom, J.-W.; Kim, Y.-H. Consensus scoring approach to identify the inhibitors of AMP-activated protein kinase α2 with virtual screening. J. Chem. Inf. Model. 2014, 54, 2139–2146. [Google Scholar] [CrossRef] [PubMed]
- Feher, M. Consensus scoring for protein-ligand interactions. Drug Discov. Today 2006, 11, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Wolber, G.; Langer, T. LigandScout: 3-D pharmacophores derived from protein-bound ligands and their use as virtual screening filters. J. Chem. Inf. Model. 2005, 45, 160–169. [Google Scholar] [CrossRef] [PubMed]
- Steindl, T.M.; Schuster, D.; Wolber, G.; Laggner, C.; Langer, T. High-throughput structure-based pharmacophore modelling as a basis for successful parallel virtual screening. J. Comput. Aided Mol. Des. 2006, 20, 703–715. [Google Scholar] [CrossRef] [PubMed]
- Caporuscio, F.; Tafi, A. Pharmacophore modelling: A forty year old approach and its modern synergies. Curr. Med. Chem. 2011, 18, 2543–2553. [Google Scholar] [CrossRef] [PubMed]
- Lauro, G.; Masullo, M.; Piacente, S.; Riccio, R.; Bifulco, G. Inverse Virtual Screening allows the discovery of the biological activity of natural compounds. Bioorg. Med. Chem. 2012, 20, 3596–3602. [Google Scholar] [CrossRef] [PubMed]
- Lauro, G.; Romano, A.; Riccio, R.; Bifulco, G. Inverse virtual screening of antitumor targets: Pilot study on a small database of natural bioactive compounds. J. Nat. Prod. 2011, 74, 1401–1407. [Google Scholar] [CrossRef] [PubMed]
- Schomburg, K.T.; Bietz, S.; Briem, H.; Henzler, A.M.; Urbaczek, S.; Rarey, M. Facing the challenges of structure-based target prediction by inverse virtual screening. J. Chem. Inf. Model. 2014, 54, 1676–1686. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Samples of the compounds described in this review are not available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neves, B.J.; Andrade, C.H.; Cravo, P.V.L. Natural Products as Leads in Schistosome Drug Discovery. Molecules 2015, 20, 1872-1903. https://doi.org/10.3390/molecules20021872
Neves BJ, Andrade CH, Cravo PVL. Natural Products as Leads in Schistosome Drug Discovery. Molecules. 2015; 20(2):1872-1903. https://doi.org/10.3390/molecules20021872
Chicago/Turabian StyleNeves, Bruno J., Carolina H. Andrade, and Pedro V. L. Cravo. 2015. "Natural Products as Leads in Schistosome Drug Discovery" Molecules 20, no. 2: 1872-1903. https://doi.org/10.3390/molecules20021872





