In Vitro Antioxidant and Cytotoxic Activity of Some Synthetic Riparin-Derived Compounds
Abstract
:1. Introduction
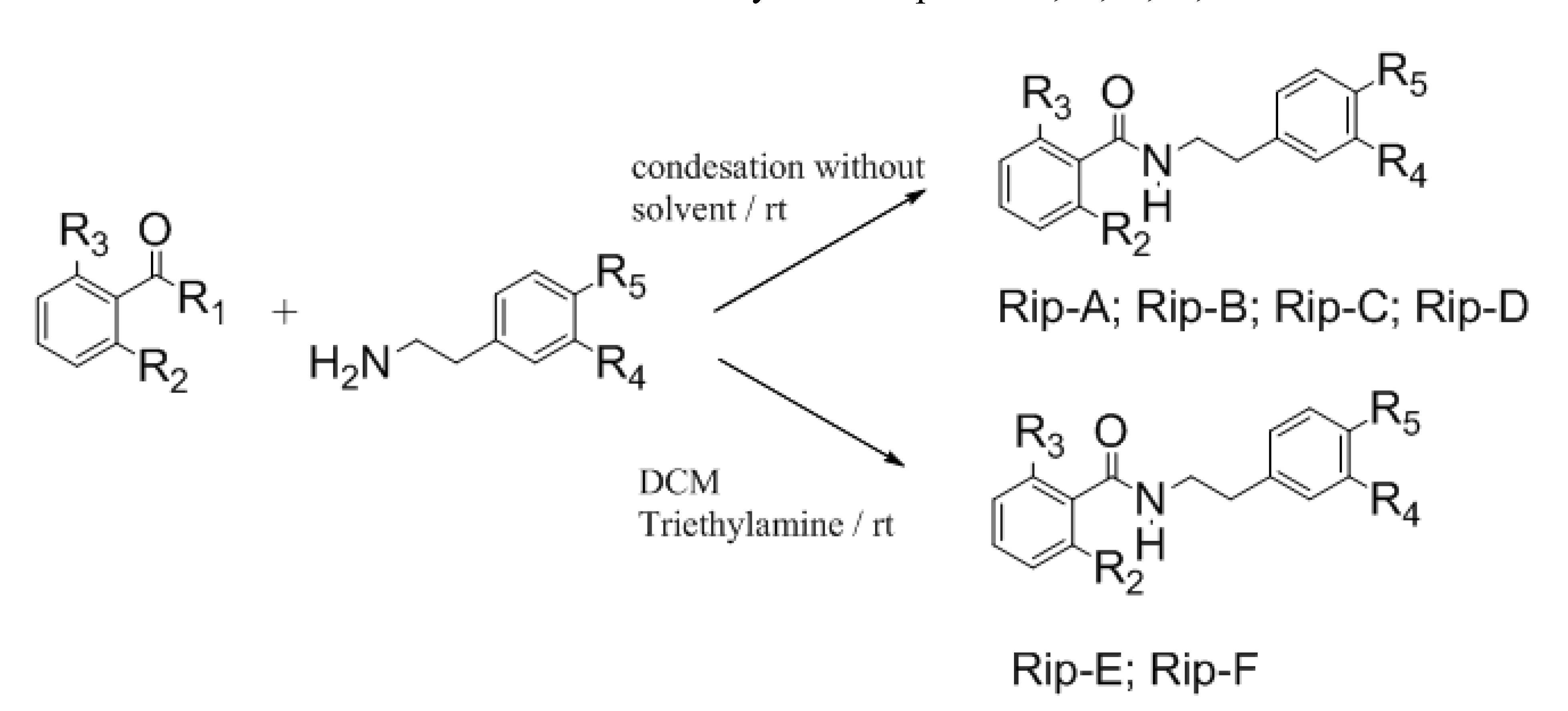
2. Results and Discussion
| H | Rip-A | Rip-B | Rip-C | Rip-D | Rip-E | Rip-F |
|---|---|---|---|---|---|---|
| 1 | - | - | - | - | - | - |
| 2 | 7.81 (dd 2.0;8.0) | 7.82 (dd 1.8;7.8) | - | - | - | - |
| 3 | 7.45(m) | 7.45(m) | 7.28(m) | 6.86(t 7.6) | 7.24(m) | 6.33(d 8.4) |
| 4 | 7.45(m) | 7.45(m) | 7.28(m) | 6.86(t 7.6) | 7.24(m) | 7.13(t 8.2) |
| 5 | 7.45(m) | 7.45(m) | 7.28(m) | 7.38(t 7.0) | 7.24(m) | 6.33(d 8.4) |
| 6 | 7.81 (dd 2.0;8.0) | 7.82 (dd 1.8;7.8) | 7.66 (dd 1.4;8.1) | 7.83 (d 7.6) | - | - |
| 1' | - | - | - | - | - | - |
| 2' | 7.24(m) | 6.81 (s) | 7.28(m) | 6.8(s) | 6.35(d 8.0) | 6.81 (s) |
| 3' | 7.24(m) | - | 7.28(m) | - | 7.24(m) | - |
| 4' | 7.24(m) | - | 6.872 (t7.8) | - | 7.24(m) | - |
| 5' | 7.24(m) | 6.84(d8.4) | 7.28(m) | 6.8(d) | 7.24(m) | 6.75(d 8.0) |
| 6' | 7.24(m) | 6.73(dd2;8.0) | 7.28(m) | 6.73(dd1.8;8.2) | 6.35(d 8.0) | 6.86(d 8.0) |
| 7' | 2.83(t 7.8) | 2.77(t 7.6) | 2.86(t 7.8) | 2.78(t 7.40) | 2.84(t7.4) | 2.77(t 7.2) |
| 8' | 3.47(q 6.8) | 3.46(q 7.6) | 3.52(q 6.8) | 3.50( q 6.8) | 3.58(q 6.8) | 3.56(q 7.4) |
| N-H | 8.57(sl) | 8.552(s) | 8.93(t 5.4) | 8.87(t 5.6) | 8.99(t 4.8) | 8.9(sl) |
| 2-OH | - | - | 12.628 (s) | 12.59(s) | 12.6(s) | 12.58(s) |
| 6-OH | - | - | - | - | 12.6(s) | 12.58(s) |
| 3'-OMe | - | 3.69 (s) | - | 3.69(s) | - | 3.7(s) |
| 4'-OMe | - | 3.69 (s) | - | 3.69(s) | - | 3.7(s) |
| Rip-A | Rip-B | Rip-C | Rip-D | Rip-E | Rip-F | |
|---|---|---|---|---|---|---|
| 1 | 134.64 | 134.67 | 115.26 | 115.33 | 102.50 | 102.50 |
| 2 | 127.11 | 127.06 | 160.03 | 159.99 | 160.23 | 160.23 |
| 3 | 128.65 | 128.19 | 117.37 | 117.37 | 107.17 | 107.18 |
| 4 | 131.04 | 132.02 | 133.60 | 133.60 | 133.32 | 133.34 |
| 5 | 128.65 | 128.19 | 118.54 | 118.55 | 107.17 | 107.18 |
| 6 | 127.11 | 127.06 | 127.67 | 127.71 | 160.23 | 160.23 |
| 1' | 139.54 | 130.99 | 139.24 | 131.70 | 139.14 | 131.52 |
| 2' | 128.24 | 112.61 | 128.63 | 112.55 | 128.44 | 112.48 |
| 3' | 128.32 | 148.62 | 128.36 | 148.64 | 128.66 | 148.70 |
| 4' | 126.07 | 147.25 | 126.17 | 147.31 | 126.28 | 147.34 |
| 5' | 128.32 | 111.97 | 128.36 | 111.94 | 128.66 | 111.92 |
| 6' | 128.24 | 120.48 | 128.63 | 120.50 | 128.44 | 120.49 |
| 7' | 35.13 | 34.60 | 34.85 | 34.37 | 34.77 | 34.33 |
| 8' | 40.90 | 41.01 | 40.54 | 40.69 | 40.19 | 40.29 |
| N-H | - | - | - | - | - | - |
| 2-OH | - | - | - | - | - | - |
| 6-OH | - | - | - | - | - | - |
| 3'-OMe | - | 55.51 | - | 55.49 | - | 55.48 |
| 4'-OMe | - | 55.33 | - | 55.32 | - | 55.33 |
| C=O | 166.18 | 166.15 | 168.87 | 168.79 | 170.00 | 169.95 |
| Antioxidant tests | μg/mL | Rip-A | Rip-B | Rip-C | Rip-D | Rip-E | Rip-F | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Nitrite Production (% induced by SNP) | 1 | 75.1 ± 1.31 a | 75.8 ± 1.02 a | 69.3 ± 1.07 a | 72.6 ± 1.81 a | 65.9 ± 1.30 a | 74.9 ± 2.27 a | |||
| 5 | 29.1 ± 0.61 a | 34.1 ± 1.85 a | 34.9 ± 0.23 a | 32.2 ± 1.45 a | 37.7 ± 0.95 a | 35.3 ± 0.96 a | ||||
| Vehicle 1 | System | Trolox (140 μg/mL) | 10 | 29.2 ± 2.05 a | 29.1 ± 2.00 a | 22.9 ± 1.31 a | 28.9 ± 1.74 a | 25.1 ± 1.37 a | 21.5 ± 0.98 a | |
| 34.88 ± 0.96 | 100.4 ± 2.01 * | 40.2 ± 2.80 a | 25 | 24.3 ± 0.62 a | 23.1 ± 0.97 a | 19.8 ± 0.83 a | 24.7 ± 1.77 a | 20.1 ± 0.71 a | 19.9 ± 0.71 a | |
| 100 | 20.4 ± 1.20 a | 20.6 ± 0.98 a | 16.5 ± 1.44 a | 17.3 ± 2.38 a | 15.4 ± 1.11 a | 17.3 ± 0.22 a | ||||
| 2-Deoxyribose degradation (%) | 1 | 97.1 ± 0.78 a | 98.4 ± 0.39 a | 95.5 ± 1.30 a | 93.5 ± 1.17 a | 79.9 ± 0.64 a | 83.3 ± 2.13 a | |||
| 5 | 76.9 ± 1.10 a | 79.8 ± 1.34 a | 80.9 ± 1.29 a | 67.9 ± 1.74 a | 62.2 ± 2.14 a | 61.5 ± 0.98 a | ||||
| Vehicle1 | System | Trolox (140 μg/mL) | 10 | 54.5 ± 0.85 a | 58.5 ± 0.85 a | 58.7 ± 1.37 a | 56.9 ± 0.82 a | 53.4 ± 1.79 a | 57.6 ± 1.47 a | |
| 13.96 ± 0.96 | 100.2 ± 1.35 * | 21.4 ± 0.95 a | 25 | 49.6 ± 0.89 a | 47.7 ± 0.44 a | 52.1 ± 0.13 a | 51.9 ± 1.27 a | 51.4 ± 1.47 a | 55.1 ± 1.78 a | |
| 100 | 44.8 ± 0.30 a | 47.2 ± 0.43 a | 46.7 ± 0.35 a | 47.3 ± 0.88 a | 49.4 ± 0.85 a | 50.9 ± 2.33 a | ||||
| TBARS levels (% AAPH) | 1 | 40.9 ± 1.77 a | 44.5 ± 1.31 a | 39.3 ± 0.60 a | 41.2 ± 0.96 a | 41.4 ± 0.69 a | 38.8 ± 0.99 a | |||
| 5 | 38.1 ± 0.49 a | 40.4 ± 0.85 a | 38.8 ± 1.07 a | 39.4 ± 0.71 a | 38.6 ± 0.63 a | 35.9 ± 0.41 a | ||||
| Vehicle1 | System | Trolox (140 μg/mL) | 10 | 36.7 ± 0.66 a | 38.3 ± 1.05 a | 34.3 ± 0.92 a | 37.3 ± 0.68 a | 35.5 ± 0.37 a | 35.2 ± 0.44 a | |
| 18.38 ± 1.84 | 99.90 ± 1.32 * | 55.3 ± 6.48 a | 25 | 35.1 ± 0.88 a | 36.5 ± 1.21 a | 29.5 ± 1.91 a | 34.4 ± 0.76 a | 31.3 ± 0.76 a | 34.6 ± 0.49 a | |
| 100 | 25.8 ± 0.48 a | 29.7 ± 1.31 a | 25.7 ± 2.62 a | 25.6 ± 0.66 a | 28.2 ± 0.39 a | 30.3 ± 0.47 a | ||||
| Riparins | Riparin A | Riparin B | Riparin C | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Parameters | OH radical | Nitrite | TBARS | OH radical | Nitrite | TBARS | OH radical | Nitrite | TBARS |
| IC50 (μg/mL) | 1.501 | 0.8404 | 1.147 | 1.639 | 0.7948 | 1.032 | 0.9516 | 0.7240 | 0.8971 |
| CI | 0.81–2.77 | 0.41–1.69 | 0.59–2.20 | 0.82–3.24 | 0.40–1.57 | 0.57–1.86 | 0.47–1.89 | 0.37–1.40 | 0.49–1.63 |
| r2 | 0.84 | 0.80 | 0.83 | 0.80 | 0.81 | 0.85 | 0.81 | 0.81 | 0.85 |
| Riparins | Riparin D | Riparin E | Riparin F | ||||||
| Parameters | OH radical | Nitrite | TBARS | OH radical | Nitrite | TBARS | OH radical | Nitrite | TBARS |
| IC50 (μg/mL) | 1.358 | 1.270 | 0.9958 | 1.054 | 0.5224 | 0.7821 | 1.016 | 0.5817 | 0.6231 |
| CI | 0.68–2.71 | 0.69–2.31 | 0.55–1.80 | 0.55–1.99 | 0.28–0.94 | 0.45–1.33 | 0.56–1.81 | 0.32–1.03 | 0.37–1.04 |
| r2 | 0.81 | 0.85 | 0.85 | 0.83 | 0.83 | 0.87 | 0.86 | 0.84 | 0.87 |
| Riparins | NCIH-292 | HEP-2 | HL-60 |
|---|---|---|---|
| Rip-A | >25 | >25 | >25 |
| Rip-B | >25 | >25 | >25 |
| Rip-C | >25 | 7.3 (5.2–10.1) | 3.3 (2.0–5.6) |
| Rip-D | >25 | 7.3 (5.4–9.9) | 9.0 (6.8–11.9) |
| Rip-E | >25 | nt | 1.9 (1.5–2.4) |
| Rip-F | 10.3 (7.6–13.8) | 7.8 (4.5–13.5) | 11.4 (9.1–14.3) |
| Riparins | LDH | |
|---|---|---|
| % Release | Deviation | |
| Negative Control | 3.13 | 0.68 |
| Rip-A | 28.13 | 3.02 a |
| Rip-B | 17.31 | 1.71 a |
| Rip-C | 87.35 | 2.37 a |
| Rip-D | 83.12 | 1.04 a |
| Rip-E | 96.91 | 0.15 a |
| Rip-F | 91.01 | 0.19 a |
| Triton X-100 1% | 99.96 | 1.21 a |
3. Experimental
3.1. General Information
3.2. Compound Synthesis
3.2.1. N-Phenethylbenzamide (Rip-A)
3.2.2. N-[2-(3,4-Dimethoxyphenyl)ethyl]benzamide (Rip-B)
3.2.3. 2-Hydroxy-N-phenethylbenzamide (Rip-C)
3.2.4. 2-Hydroxy-N-[2-(3,4-methoxyphenyl)ethyl]benzamide (Rip-D)
3.2.5. 2,6-Dihydroxy-N-phenethylbenzamide (Rip-E)
3.2.6. N-[2-(3,4-Dimethoxyphenyl)ethyl]-2,6-dihydroxybenzamide (Rip-F)
3.3. In Vitro Antioxidant Potential Tests
3.3.1. Hydroxyl Radical Scavenging Activity
3.3.2. Evaluation of the Antioxidant Potential of Riparins against the Formation of TBARS
3.3.3. Scavenging Activity of Nitric Oxide (NO)
3.4. Cytotoxicity against Tumor Cell Lines
3.5. Cytotoxicity Assay on RAW 264.7
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ferreira, P.M.P.; Costa-Lotufo, L.V.; Moraes, M.O.; Barros, F.W.A.; Martins, A.M.A.; Cavalheiro, A.J.; Bolzani, V.S.; Santos, A.G.; Pessoa, C. Folk uses and pharmacological properties of Casearia sylvestris: A medicinal review. An. Acad. Bras. Cienc. 2011, 83, 1373–1384. [Google Scholar] [CrossRef]
- Marques, C.A. Importância econômica da família Lauraceae Lindl. Floresta Ambiente 2001, 8, 195–206. [Google Scholar]
- Sousa, F.C.F.; Melo, C.T.V.; Citó, C.O.M.; Félix, F.H.C.; Vasconcelos, S.M.M.; Fonteles, M.M.F.; Barbosa Filho, J.M.; Viana, G.S.B. Plantas medicinais e seus constituintes bioativos: Uma revisão da bioatividade e potenciais benefícios nos distúrbios da ansiedade em modelos animais. Rev. Bras. Farmacogn. 2008, 18, 642–654. [Google Scholar] [CrossRef]
- Ferreira, J.R.O.; Cavalcanti, B.C.; Costa, P.M.; Arantes, F.F.P.; Alvarenga, E.S.; Maltha, C.R.A.; Barbosa, L.C.A.; Militão, G.C.G.; Pessoa, C.; Ferreira, P.M.P. Induction of G2/M arrest, caspase activation and apoptosis by α-santonin derivatives in HL-60 cells. Toxicol. In Vitro 2013, 27, 1458–1466. [Google Scholar] [CrossRef]
- Brasil. Ministério da Saúde. ANVISA. In Portaria 116, de 08 de Agosto de 1996; DOU: Brasília, Brazil, 1996; Seção I; pp. 15198–151989. [Google Scholar]
- Barbosa-Filho, J.M.; Yoshida, M.; Gottlieb, O.R.; Barbosa, R.C.S.B.C.; Giesbrecht, A.M.; Yong, M.C.M. Benzoyl esters and amides, stryrylpyrones and neolignans from the fruits of Aniba riparia. Phytochemistry 1987, 26, 2615–2617. [Google Scholar] [CrossRef]
- Barbosa-Filho, J.M.; Silva, E.C.; Bhattacharyya, J. Synthesis of severall new phenylethylamides of substituited benzoic acids. Quim. Nova 1990, 13, 332–334. [Google Scholar]
- Barbosa-Filho, J.M. Quimiodiversidade e potencialidade farmacológica da flora paraibana. Cad. Farm. 1997, 13, 85–102. [Google Scholar]
- Gutierrez, S.J.C. Síntese do Bowdenol, um Dihidrobenzofuranoide Isolado de Bowdichia Virgilioides e Preparação de Derivados da Riparina Isolada de Aniba Riparia com Potencial Atividade Biológica . Ph.D. Thesis, UFPB/CCS, João Pessoa, Brazil, 2007. [Google Scholar]
- Xavier, S.M.; Barbosa, C.O.; Barros, D.O.; Silva, R.F.; Oliveira, A.A.; Freitas, R.M. Vitamin C antioxidant effects in hippocampus of adult Wistar rats after seizures and status epilepticus induced by pilocarpine. Neurosci. Lett. 2007, 420, 76–79. [Google Scholar] [CrossRef]
- Fleschin, S.; Fleschin, M.; Nita, S.; Pavel, E.; Magearu, V. Free radicals mediated protein oxidation in biochemistry. Roum. Biotechnol. Lett. 2000, 5, 479–495. [Google Scholar]
- Hirata, L.L.; Sato, M.E.O.; Santos, C.A.M. Radicais livres e o envelhecimento cutâneo. Acta Farmac. Bonaerense 2004, 23, 418–424. [Google Scholar]
- Halliwell, B.; Whiteman, M. Measuring reactive species and oxidative damage in vivo and in cell culture: How should you do it and what do the results mean? Br. J. Clin. Pharmacol. 2004, 142, 231–255. [Google Scholar] [CrossRef]
- Stadtman, E.R. Protein oxidation and aging. Science 1992, 257, 1220–1224. [Google Scholar]
- Finkel, T.; Holbrook, N.J. Oxidants, oxidative stress and the biology of ageing. Nature 2000, 408, 239–247. [Google Scholar] [CrossRef]
- Maldonado, O.S.; Lucas, R.; Comelles, F.; Gonzalez, M.J.; Parra, J.L.; Medina, I.; Morales, J.C. Synthesis and characterization of phenolic antioxidants with surfactant properties: Glucosyl- and glucuronosyl alkyl gallates. Tetrahedron 2011, 67, 7268–7279. [Google Scholar] [CrossRef]
- Reed, T.T. Lipid peroxidation and neurodegenerative disease. Free Radic. Biol. Med. 2011, 51, 1302–1319. [Google Scholar] [CrossRef]
- Sousa, C.M.M.; Silva, H.R.; Vieira, G.M., Jr.; Ayres, M.C.C.; Costa, C.L.S.; Araújo, D.S.; Cavalcante, L.C.D.; Barros, E.D.S.; Araújo, P.B.M.; Brandão, M.S.; et al. Total phenolics and antioxidant activity of five medicinal plants. Quim. Nova 2007, 30, 351–355. [Google Scholar] [CrossRef]
- Rizo, W.F.; Ferreira, L.E.; Colnaghi, V.; Martins, J.S.; Franchi, L.P.; Takahashi, C.S.; Beleboni, R.O.; Marins, M.; Pereira, P.S.; Fachin, A.L. Cytotoxicity and genotoxicity of coronaridine from Tabernaemontana catharinensis A.DC in a human laryngeal epithelial carcinoma cell line (Hep-2). Genet. Mol. Biol. 2013, 36, 105–110. [Google Scholar] [CrossRef]
- Morrison, R.T.; Boyd, R.N. Organic Chemistry, 6th ed.; Prentice Hall: Englewood Cliffs, NJ, USA, 1992. [Google Scholar]
- Trost, B.M.; Pearson, W.H. Sulfur activation of azides toward addition of organometallics. Amination of aliphatic carbanions. J. Am. Chem. Soc. 1983, 105, 1054–1056. [Google Scholar] [CrossRef]
- Sugasawa, S. Synthesis of papaverine derivatives. II. Synthesis of 1-(3',4',5'-trimethylphenyl)-6,7-diethoxyisoquinoline. Yakugaku Zasshi 1935, 55, 224–233. [Google Scholar]
- Shapiro, S.L.; Rose, I.M.; Freedman, L. Pyridylethylated benzoxazinediones. J. Am. Chem. Soc. 1957, 79, 2811–2814. [Google Scholar] [CrossRef]
- Fritzson, I.; Bedingfield, P.T.P.; Sundin, A.P.; McConkey, G.; Nilsson, U.J. N-Substituted salicylamides as selective malaria parasite dihydroorotate dehydrogenase inhibitors. Med. Chem. Comm. 2011, 2, 895–898. [Google Scholar] [CrossRef]
- Lopes, G.K.; Schulman, H.M.; Hermes-Lima, M. Polyphenol tannic acid inhibits hydroxyl radical formation from Fenton reaction by complexing ferrous ions. Biochim. Biophys. Acta 1999, 1472, 142–152. [Google Scholar]
- Esterbauer, H.; Cheeseman, K.H. Determination of aldehydic lipid peroxidation products: Malonaldehyde and 4-hydroxynonenal. Methods Enzymol. 1990, 186, 407–421. [Google Scholar] [CrossRef]
- Basu, S.; Hazra, B. Evaluation of nitric oxide scavenging activity, in vitro and ex vivo, of selected medicinal plants traditionally used in inflammatory diseases. Phytother. Res. 2006, 20, 896–900. [Google Scholar] [CrossRef]
- Berridge, M.V.; Tan, A.S.; Mccoy, K.D.; Wang, R. The biochemical and cellular basisof cell proliferation assays that use tetrazolium salts. Biochemica 1996, 4, 14–19. [Google Scholar]
- Muzitano, M.F.; Tinoco, L.W.; Guette, C.; Kaiser, C.R.; Rossi-Bergmann, B.; Costa, S.S. Assessment of antileishmanial activity of new and unusual flavonoids from Kalanchoepinnata. Phytochemistry 2006, 67, 2071–2077. [Google Scholar] [CrossRef]
- Lantto, T.A.; Dorman, H.J.D.; Shikov, A.N.; Pozharitskaya, O.N.; Makarov, V.G.; Tikhonov, V.P.; Hiltunen, R.; Raasmaja, A. Chemical composition, antioxidative activity andcell viability effects of a Siberian pine (Pinussibirica Du Tour) extract. Food Chem. 2009, 112, 936–943. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds riparins A–F are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nunes, G.B.L.; Policarpo, P.R.; Costa, L.M.; Da Silva, T.G.; Militão, G.C.G.; Câmara, C.A.; Barbosa Filho, J.M.; Gutierrez, S.J.C.; Islam, M.T.; De Freitas, R.M. In Vitro Antioxidant and Cytotoxic Activity of Some Synthetic Riparin-Derived Compounds. Molecules 2014, 19, 4595-4607. https://doi.org/10.3390/molecules19044595
Nunes GBL, Policarpo PR, Costa LM, Da Silva TG, Militão GCG, Câmara CA, Barbosa Filho JM, Gutierrez SJC, Islam MT, De Freitas RM. In Vitro Antioxidant and Cytotoxic Activity of Some Synthetic Riparin-Derived Compounds. Molecules. 2014; 19(4):4595-4607. https://doi.org/10.3390/molecules19044595
Chicago/Turabian StyleNunes, Geandra B. L., Paola R. Policarpo, Luciana M. Costa, Teresinha G. Da Silva, Gardenia Carmen G. Militão, Celso A. Câmara, José Maria Barbosa Filho, Stanley Juan C. Gutierrez, Mohammed T. Islam, and Rivelilson M. De Freitas. 2014. "In Vitro Antioxidant and Cytotoxic Activity of Some Synthetic Riparin-Derived Compounds" Molecules 19, no. 4: 4595-4607. https://doi.org/10.3390/molecules19044595
APA StyleNunes, G. B. L., Policarpo, P. R., Costa, L. M., Da Silva, T. G., Militão, G. C. G., Câmara, C. A., Barbosa Filho, J. M., Gutierrez, S. J. C., Islam, M. T., & De Freitas, R. M. (2014). In Vitro Antioxidant and Cytotoxic Activity of Some Synthetic Riparin-Derived Compounds. Molecules, 19(4), 4595-4607. https://doi.org/10.3390/molecules19044595




