Hybrid Curcumin Compounds: A New Strategy for Cancer Treatment
Abstract
:1. Introduction
2. Curcumin
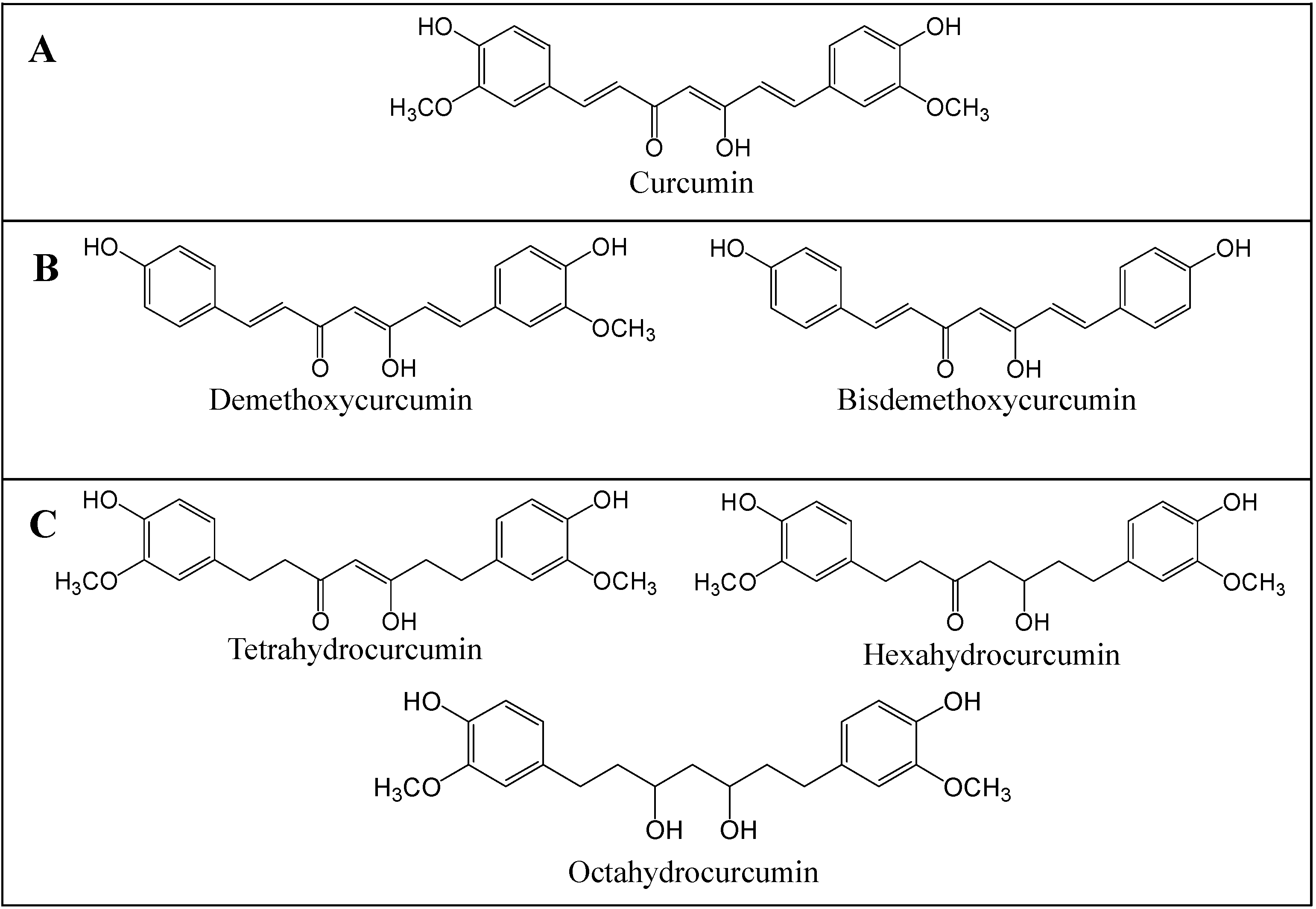
3. Strategies to Enhance Curcumin Bioavailability
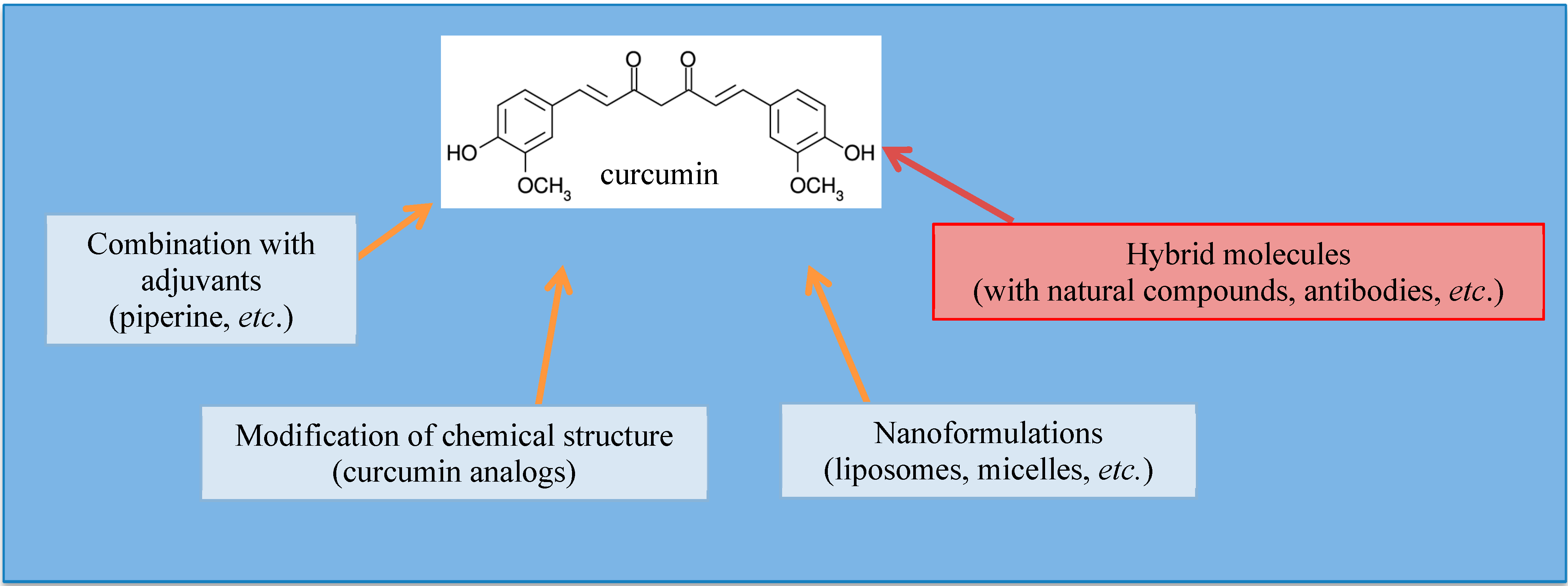
3.1. Curcumin Analogs
3.2. Combination with Specific Adjuvants
3.3. Curcumin Nano-Formulations
3.4. Concept of “Hybrid Molecules”

4. Multifunctional Curcumin Molecules in the War against Cancer
4.1. Curcumin Conjugated with Piperine and Amino Acids

4.2. [DLys6]-LHRH-Curcumin Conjugate
4.3. Curcumin Conjugates with Antiandrogens
4.4. Curcumin Immunoconjugates
4.5. Curcumin and Thalidomide
4.6. Curcumin-Diaminothiazole Hybrids
4.7. Oxovanadium (IV) Curcumin Complexes
4.8. Binding of Curcumin to β-Lactoglobulin
4.9. Limitations of the Hybrid Approach
5. Curcumin Hybrids Used for the Treatment of Other Multipotent Diseases

6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- DeVita, V.T., Jr.; Chu, E. A history of cancer chemotherapy. Cancer Res. 2008, 68, 8643–8653. [Google Scholar] [PubMed]
- Kong, D.X.; Li, X.J.; Zhang, H.Y. Where is the hope for drug discovery? Let history tell the future. Drug Discov. Today 2009, 14, 115–119. [Google Scholar] [PubMed]
- Patwardhan, B.; Mashelkar, R.A. Traditional medicine-inspired approaches to drug discovery: Can ayurveda show the way forward? Drug Discov. Today 2009, 14, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Rather, M.A.; Bhat, B.A.; Qurishi, M.A. Multicomponent phytotherapeutic approach gaining momentum: Is the “one drug to fit all” model breaking down? Phytomedicine 2013, 21, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Wahile, A. Integrated approaches towards drug development from ayurveda and other indian system of medicines. J. Ethnopharmacol. 2006, 103, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Keith, C.T.; Borisy, A.A.; Stockwell, B.R. Multicomponent therapeutics for networked systems. Nat. Rev. Drug Discov. 2005, 4, 71–78. [Google Scholar] [CrossRef]
- Dancey, J.E.; Chen, H.X. Strategies for optimizing combinations of molecularly targeted anticancer agents. Nat. Rev. Drug Discov. 2006, 5, 649–659. [Google Scholar] [CrossRef]
- Zimmermann, G.R.; Lehar, J.; Keith, C.T. Multi-target therapeutics: When the whole is greater than the sum of the parts. Drug Discov. Today 2007, 12, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Morphy, R.; Rankovic, Z. Designed multiple ligands. An emerging drug discovery paradigm. J. Med. Chem. 2005, 48, 6523–6543. [Google Scholar] [CrossRef] [PubMed]
- Medina-Franco, J.L.; Giulianotti, M.A.; Welmaker, G.S.; Houghten, R.A. Shifting from the single to the multi-target paradigm in drug discovery. Drug Discov. Today 2013, 18, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Paterson, I.; Anderson, E.A. Chemistry. The renaissance of natural products as drug candidates. Science 2005, 310, 451–453. [Google Scholar] [CrossRef] [PubMed]
- Bhanot, A.; Sharma, R.; Noolvi, M.N. Natural sources as potential anti-cancer agents: A review. Int. J. Phytomed. 2011, 3, 9–26. [Google Scholar]
- Kelkel, M.; Jacob, C.; Dicato, M.; Diederich, M. Potential of the dietary antioxidants resveratrol and curcumin in prevention and treatment of hematologic malignancies. Molecules 2010, 15, 7035–7074. [Google Scholar] [CrossRef] [PubMed]
- Orlikova, B.; Diederich, M. Power from the garden: Plant compounds as inhibitors of the hallmarks of cancer. Curr. Med. Chem. 2012, 19, 2061–2087. [Google Scholar] [CrossRef] [PubMed]
- Sawadogo, W.R.; Schumacher, M.; Teiten, M.H.; Cerella, C.; Dicato, M.; Diederich, M. A survey of marine natural compounds and their derivatives with anti-cancer activity reported in 2011. Molecules 2013, 18, 3641–3673. [Google Scholar] [CrossRef] [PubMed]
- Folmer, F.; Jaspars, M.; Dicato, M.; Diederich, M. Marine natural products as targeted modulators of the transcription factor nf-kappab. Biochem. Pharmacol. 2008, 75, 603–617. [Google Scholar] [CrossRef] [PubMed]
- Koeberle, A.; Werz, O. Multi-target approach for natural products in inflammation. Drug Discov. Today 2014. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Bhatt, I.D.; Ichikawa, H.; Ahn, K.S.; Sethi, G.; Sandur, S.K.; Sundaram, C.; Seeram, N.; Shishodia, S. Curcumin: Biological and medicinal properties. In Turmeric: The Genus Curcuma; CRC Press: New york, NY, USA, 2007; Volume 45, pp. 297–368. [Google Scholar]
- Hatcher, H.; Planalp, R.; Cho, J.; Torti, F.M.; Torti, S.V. Curcumin: From ancient medicine to current clinical trials. Cell. Mol. Life Sci. 2008, 65, 1631–1652. [Google Scholar] [CrossRef] [PubMed]
- Teiten, M.H.; Eifes, S.; Dicato, M.; Diederich, M. Curcumin-the paradigm of a multi-target natural compound with applications in cancer prevention and treatment. Toxins (Basel) 2010, 2, 128–162. [Google Scholar] [CrossRef]
- Teiten, M.H.; Gaigneaux, A.; Chateauvieux, S.; Billing, A.M.; Planchon, S.; Fack, F.; Renaut, J.; Mack, F.; Muller, C.P.; Dicato, M.; et al. Identification of differentially expressed proteins in curcumin-treated prostate cancer cell lines. Omics 2012, 16, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Goel, A.; Kunnumakkara, A.B.; Aggarwal, B.B. Curcumin as “curecumin”: From kitchen to clinic. Biochem. Pharmacol. 2008, 75, 787–809. [Google Scholar] [CrossRef] [PubMed]
- Bar-Sela, G.; Epelbaum, R.; Schaffer, M. Curcumin as an anti-cancer agent: Review of the gap between basic and clinical applications. Curr. Med. Chem. 2010, 17, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, B.B.; Kumar, A.; Bharti, A.C. Anticancer potential of curcumin: Preclinical and clinical studies. Anticancer Res. 2003, 23, 363–398. [Google Scholar] [PubMed]
- Jurenka, J.S. Anti-inflammatory properties of curcumin, a major constituent of curcuma longa: A review of preclinical and clinical research. Altern. Med. Rev. 2009, 14, 141–153. [Google Scholar] [PubMed]
- Epstein, J.; Sanderson, I.R.; Macdonald, T.T. Curcumin as a therapeutic agent: The evidence from in vitro, animal and human studies. Br. J. Nutr. 2010, 103, 1545–1557. [Google Scholar] [CrossRef] [PubMed]
- Teiten, M.H.; Dicato, M.; Diederich, M. Curcumin as a regulator of epigenetic events. Mol. Nutr. Food Res. 2013, 57, 1619–1629. [Google Scholar] [CrossRef] [PubMed]
- Reuter, S.; Gupta, S.C.; Park, B.; Goel, A.; Aggarwal, B.B. Epigenetic changes induced by curcumin and other natural compounds. Genes Nutr. 2011, 6, 93–108. [Google Scholar] [CrossRef] [PubMed]
- Balogun, E.; Hoque, M.; Gong, P.; Killeen, E.; Green, C.J.; Foresti, R.; Alam, J.; Motterlini, R. Curcumin activates the haem oxygenase-1 gene via regulation of nrf2 and the antioxidant-responsive element. Biochem. J. 2003, 371, 887–895. [Google Scholar] [CrossRef] [PubMed]
- Sandur, S.K.; Pandey, M.K.; Sung, B.; Ahn, K.S.; Murakami, A.; Sethi, G.; Limtrakul, P.; Badmaev, V.; Aggarwal, B.B. Curcumin, demethoxycurcumin, bisdemethoxycurcumin, tetrahydrocurcumin and turmerones differentially regulate anti-inflammatory and anti-proliferative responses through a ros-independent mechanism. Carcinogenesis 2007, 28, 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- Menon, V.P.; Sudheer, A.R. Antioxidant and anti-inflammatory properties of curcumin. Adv. Exp. Med. Biol. 2007, 595, 105–125. [Google Scholar] [PubMed]
- Reuter, S.; Charlet, J.; Juncker, T.; Teiten, M.H.; Dicato, M.; Diederich, M. Effect of curcumin on nuclear factor kappab signaling pathways in human chronic myelogenous k562 leukemia cells. Ann. N. Y. Acad. Sci. 2009, 1171, 436–447. [Google Scholar] [CrossRef] [PubMed]
- Teiten, M.H.; Eifes, S.; Reuter, S.; Duvoix, A.; Dicato, M.; Diederich, M. Gene expression profiling related to anti-inflammatory properties of curcumin in k562 leukemia cells. Ann. N. Y. Acad. Sci. 2009, 1171, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Duvoix, A.; Blasius, R.; Delhalle, S.; Schnekenburger, M.; Morceau, F.; Henry, E.; Dicato, M.; Diederich, M. Chemopreventive and therapeutic effects of curcumin. Cancer Lett. 2005, 223, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Reuter, S.; Eifes, S.; Dicato, M.; Aggarwal, B.B.; Diederich, M. Modulation of anti-apoptotic and survival pathways by curcumin as a strategy to induce apoptosis in cancer cells. Biochem. Pharmacol. 2008, 76, 1340–1351. [Google Scholar] [CrossRef] [PubMed]
- Bhandarkar, S.S.; Arbiser, J.L. Curcumin as an inhibitor of angiogenesis. Adv. Exp. Med. Biol. 2007, 595, 185–195. [Google Scholar] [PubMed]
- Kunnumakkara, A.B.; Anand, P.; Aggarwal, B.B. Curcumin inhibits proliferation, invasion, angiogenesis and metastasis of different cancers through interaction with multiple cell signaling proteins. Cancer Lett. 2008, 269, 199–225. [Google Scholar] [CrossRef] [PubMed]
- Reddy, A.R.; Dinesh, P.; Prabhakar, A.S.; Umasankar, K.; Shireesha, B.; Raju, M.B. A comprehensive review on sar of curcumin. Mini Rev. Med. Chem. 2013, 13, 1769–1777. [Google Scholar] [CrossRef] [PubMed]
- Priyadarsini, K.I. Chemical and structural features influencing the biological activity of curcumin. Curr. Pharm. Des. 2013, 19, 2093–2100. [Google Scholar] [PubMed]
- Jitoe-Masuda, A.; Fujimoto, A.; Masuda, T. Curcumin: From chemistry to chemistry-based functions. Curr. Pharm. Des. 2013, 19, 2084–2092. [Google Scholar] [PubMed]
- Fuchs, J.R.; Pandit, B.; Bhasin, D.; Etter, J.P.; Regan, N.; Abdelhamid, D.; Li, C.; Lin, J.; Li, P.K. Structure-activity relationship studies of curcumin analogues. Bioorg. Med. Chem. Lett. 2009, 19, 2065–2069. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, R.; Mishra, B.; Tyagi, E.; Nath, C.; Shukla, R. Effect of curcumin on brain insulin receptors and memory functions in stz (icv) induced dementia model of rat. Pharmacol. Res. 2010, 61, 247–252. [Google Scholar] [CrossRef]
- Ishida, J.; Ohtsu, H.; Tachibana, Y.; Nakanishi, Y.; Bastow, K.F.; Nagai, M.; Wang, H.K.; Itokawa, H.; Lee, K.H. Antitumor agents. Part 214: Synthesis and evaluation of curcumin analogues as cytotoxic agents. Bioorg. Med. Chem. 2002, 10, 3481–3487. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, Y.; Kawakishi, S.; Osawa, T. Involvement of the beta-diketone moiety in the antioxidative mechanism of tetrahydrocurcumin. Biochem. Pharmacol. 1996, 52, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Somparn, P.; Phisalaphong, C.; Nakornchai, S.; Unchern, S.; Morales, N.P. Comparative antioxidant activities of curcumin and its demethoxy and hydrogenated derivatives. Biol. Pharm. Bull. 2007, 30, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, E.; Lazzari, S.; Marverti, G.; Pignedoli, F.; Spagnolo, F.; Saladini, M. Synthesis, cytotoxic and combined cddp activity of new stable curcumin derivatives. Bioorg. Med. Chem. 2009, 17, 3043–3052. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Lazaro, M. Anticancer and carcinogenic properties of curcumin: Considerations for its clinical development as a cancer chemopreventive and chemotherapeutic agent. Mol. Nutr. Food Res. 2008, 52 (Suppl. 1), S103–S127. [Google Scholar]
- Burgos-Moron, E.; Calderon-Montano, J.M.; Salvador, J.; Robles, A.; Lopez-Lazaro, M. The dark side of curcumin. Int. J. Cancer 2010, 126, 1771–1775. [Google Scholar] [PubMed]
- Anand, P.; Kunnumakkara, A.B.; Newman, R.A.; Aggarwal, B.B. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Bisht, S.; Maitra, A. Systemic delivery of curcumin: 21st century solutions for an ancient conundrum. Curr. Drug Discov. Technol. 2009, 6, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Shehzad, A.; Khan, S.; Shehzad, O.; Lee, Y.S. Curcumin therapeutic promises and bioavailability in colorectal cancer. Drugs Today (Barc.) 2010, 46, 523–532. [Google Scholar] [CrossRef]
- Liu, Z.; Tang, L.; Zou, P.; Zhang, Y.; Wang, Z.; Fang, Q.; Jiang, L.; Chen, G.; Xu, Z.; Zhang, H.; et al. Synthesis and biological evaluation of allylated and prenylated mono-carbonyl analogs of curcumin as anti-inflammatory agents. Eur. J. Med. Chem. 2014, 74, 671–682. [Google Scholar] [CrossRef] [PubMed]
- Tamvakopoulos, C.; Dimas, K.; Sofianos, Z.D.; Hatziantoniou, S.; Han, Z.; Liu, Z.L.; Wyche, J.H.; Pantazis, P. Metabolism and anticancer activity of the curcumin analogue, dimethoxycurcumin. Clin. Cancer Res. 2007, 13, 1269–1277. [Google Scholar] [CrossRef] [PubMed]
- Steward, W.P.; Gescher, A.J. Curcumin in cancer management: Recent results of analogue design and clinical studies and desirable future research. Mol. Nutr. Food Res. 2008, 52, 1005–1009. [Google Scholar] [CrossRef] [PubMed]
- Patwardhan, R.S.; Checker, R.; Sharma, D.; Kohli, V.; Priyadarsini, K.I.; Sandur, S.K. Dimethoxycurcumin, a metabolically stable analogue of curcumin, exhibits anti-inflammatory activities in murine and human lymphocytes. Biochem. Pharmacol. 2011, 82, 642–657. [Google Scholar] [CrossRef] [PubMed]
- Ohori, H.; Yamakoshi, H.; Tomizawa, M.; Shibuya, M.; Kakudo, Y.; Takahashi, A.; Takahashi, S.; Kato, S.; Suzuki, T.; Ishioka, C.; et al. Synthesis and biological analysis of new curcumin analogues bearing an enhanced potential for the medicinal treatment of cancer. Mol. Cancer Ther. 2006, 5, 2563–2571. [Google Scholar] [CrossRef] [PubMed]
- Yadav, B.; Taurin, S.; Rosengren, R.J.; Schumacher, M.; Diederich, M.; Somers-Edgar, T.J.; Larsen, L. Synthesis and cytotoxic potential of heterocyclic cyclohexanone analogues of curcumin. Bioorg. Med. Chem. 2010, 18, 6701–6707. [Google Scholar] [CrossRef] [PubMed]
- Kasinski, A.L.; Du, Y.; Thomas, S.L.; Zhao, J.; Sun, S.Y.; Khuri, F.R.; Wang, C.Y.; Shoji, M.; Sun, A.; Snyder, J.P.; et al. Inhibition of ikappab kinase-nuclear factor-kappab signaling pathway by 3,5-bis(2-flurobenzylidene)piperidin-4-one (ef24), a novel monoketone analog of curcumin. Mol. Pharmacol. 2008, 74, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Zambre, A.P.; Kulkarni, V.M.; Padhye, S.; Sandur, S.K.; Aggarwal, B.B. Novel curcumin analogs targeting tnf-induced nf-kappab activation and proliferation in human leukemic kbm-5 cells. Bioorg. Med. Chem. 2006, 14, 7196–7204. [Google Scholar] [CrossRef] [PubMed]
- Meghwal, M.; Goswami, T.K. Piper nigrum and piperine: An update. Phytother. Res. 2013, 27, 1121–1130. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, K. Black pepper and its pungent principle-piperine: A review of diverse physiological effects. Crit Rev. Food Sci. Nutr. 2007, 47, 735–748. [Google Scholar] [CrossRef] [PubMed]
- Shoba, G.; Joy, D.; Joseph, T.; Majeed, M.; Rajendran, R.; Srinivas, P.S. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Kondo, A.; Takeda, T.; Li, B.; Tsuiji, K.; Kitamura, M.; Wong, T.F.; Yaegashi, N. Epigallocatechin-3-gallate potentiates curcumin’s ability to suppress uterine leiomyosarcoma cell growth and induce apoptosis. Int. J. Clin. Oncol. 2013, 18, 380–388. [Google Scholar] [CrossRef] [PubMed]
- Naksuriya, O.; Okonogi, S.; Schiffelers, R.M.; Hennink, W.E. Curcumin nano-formulations: A review of pharmaceutical properties and preclinical studies and clinical data related to cancer treatment. Biomaterials 2014, 35, 3365–3383. [Google Scholar] [CrossRef] [PubMed]
- Shehzad, A.; Ul-Islam, M.; Wahid, F.; Lee, Y.S. Multifunctional polymeric nanocurcumin for cancer therapy. J. Nanosci. Nanotechnol. 2014, 14, 803–814. [Google Scholar] [CrossRef] [PubMed]
- Yallapu, M.M.; Jaggi, M.; Chauhan, S.C. Curcumin nano-formulations: A future nanomedicine for cancer. Drug Discov. Today 2012, 17, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Zhang, S.; Kong, D.; Gao, X.; Zhao, Y.; Wang, Z. Biodegradable polymer-curcumin conjugate micelles enhance the loading and delivery of low-potency curcumin. Pharm. Res. 2012, 29, 3512–3525. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, C.; Acharya, S.; Mohanty, A.K.; Dilnawaz, F.; Sahoo, S.K. Curcumin-encapsulated mepeg/pcl diblock copolymeric micelles: A novel controlled delivery vehicle for cancer therapy. Nanomedicine (Lond.) 2010, 5, 433–449. [Google Scholar] [CrossRef]
- Sahu, A.; Bora, U.; Kasoju, N.; Goswami, P. Synthesis of novel biodegradable and self-assembling methoxy poly(ethylene glycol)-palmitate nanocarrier for curcumin delivery to cancer cells. Acta Biomater. 2008, 4, 1752–1761. [Google Scholar] [CrossRef] [PubMed]
- Gou, M.; Men, K.; Shi, H.; Xiang, M.; Zhang, J.; Song, J.; Long, J.; Wan, Y.; Luo, F.; Zhao, X.; et al. Curcumin-loaded biodegradable polymeric micelles for colon cancer therapy in vitro and in vivo. Nanoscale 2011, 3, 1558–1567. [Google Scholar] [CrossRef] [PubMed]
- Dey, S.; Sreenivasan, K. Conjugation of curcumin onto alginate enhances aqueous solubility and stability of curcumin. Carbohydr. Polym. 2014, 99, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Manju, S.; Sreenivasan, K. Conjugation of curcumin onto hyaluronic acid enhances its aqueous solubility and stability. J. Colloid Interface Sci. 2011, 359, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Goyal, P.; Goyal, K.; Vijaya Kumar, S.G.; Singh, A.; Katare, O.P.; Mishra, D.N. Liposomal drug delivery systems--clinical applications. Acta Pharm. 2005, 55, 1–25. [Google Scholar] [PubMed]
- Li, L.; Braiteh, F.S.; Kurzrock, R. Liposome-encapsulated curcumin: In vitro and in vivo effects on proliferation, apoptosis, signaling, and angiogenesis. Cancer 2005, 104, 1322–1331. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.; Belhaj, N.; Benachour, H.; Barberi-Heyob, M.; Kahn, C.J.; Jabbari, E.; Linder, M.; Arab-Tehrany, E. Liposome encapsulation of curcumin: Physico-chemical characterizations and effects on mcf7 cancer cell proliferation. Int. J. Pharm. 2014, 461, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Saengkrit, N.; Saesoo, S.; Srinuanchai, W.; Phunpee, S.; Ruktanonchai, U.R. Influence of curcumin-loaded cationic liposome on anticancer activity for cervical cancer therapy. Colloids Surf. B Biointerfaces 2014, 114, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Veena, M.S.; Stevenson, K.; Tang, C.; Ho, B.; Suh, J.D.; Duarte, V.M.; Faull, K.F.; Mehta, K.; Srivatsan, E.S.; et al. Liposome-encapsulated curcumin suppresses growth of head and neck squamous cell carcinoma in vitro and in xenografts through the inhibition of nuclear factor kappab by an akt-independent pathway. Clin. Cancer Res. 2008, 14, 6228–6236. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Ahmed, B.; Mehta, K.; Kurzrock, R. Liposomal curcumin with and without oxaliplatin: Effects on cell growth, apoptosis, and angiogenesis in colorectal cancer. Mol. Cancer Ther. 2007, 6, 1276–1282. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, N.K.; Nargi, D.; Randolph, C.; Narayanan, B.A. Liposome encapsulation of curcumin and resveratrol in combination reduces prostate cancer incidence in pten knockout mice. Int. J. Cancer 2009, 125, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Dilnawaz, F.; Mewar, S.; Sharma, U.; Jagannathan, N.R.; Sahoo, S.K. Composite polymeric magnetic nanoparticles for co-delivery of hydrophobic and hydrophilic anticancer drugs and mri imaging for cancer therapy. ACS Appl. Mater. Interfaces 2011, 3, 842–856. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, S.; Girija, A.R.; Nagaoka, Y.; Iwai, S.; Suzuki, M.; Kizhikkilot, V.; Yoshida, Y.; Maekawa, T.; Nair, S.D. Curcumin and 5-fluorouracil-loaded, folate- and transferrin-decorated polymeric magnetic nano-formulation: A synergistic cancer therapeutic approach, accelerated by magnetic hyperthermia. Int. J. Nanomed. 2014, 9, 437–459. [Google Scholar]
- Pramanik, D.; Campbell, N.R.; Das, S.; Gupta, S.; Chenna, V.; Bisht, S.; Sysa-Shah, P.; Bedja, D.; Karikari, C.; Steenbergen, C.; et al. A composite polymer nanoparticle overcomes multidrug resistance and ameliorates doxorubicin-associated cardiomyopathy. Oncotarget 2012, 3, 640–650. [Google Scholar] [PubMed]
- Misra, R.; Sahoo, S.K. Coformulation of doxorubicin and curcumin in poly(d,l-lactide-co-glycolide) nanoparticles suppresses the development of multidrug resistance in k562 cells. Mol. Pharm. 2011, 8, 852–866. [Google Scholar] [CrossRef] [PubMed]
- Frei, E.; Eder, J.P. Combination chemotherapy. In Holland-Frei Cancer Medicine, 6th ed.; Kufe, D.W., Weichselbaum, R.R., Bast, R.C., Jr., Gansler, T.S., Holland, J.F., Frei, E., III, Eds.; BC Decker: Hamilton, ON, USA, 2003. [Google Scholar]
- Meunier, B. Hybrid molecules with a dual mode of action: Dream or reality? Acc. Chem. Res. 2008, 41, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, S.S.; Sharma, M.; Chauhan, P.M. Trioxaquines: Hybrid molecules for the treatment of malaria. Drug News Perspect. 2010, 23, 632–646. [Google Scholar] [CrossRef] [PubMed]
- Bajorath, J. Chemoinformatics methods for systematic comparison of molecules from natural and synthetic sources and design of hybrid libraries. J. Comput. Aided Mol. Des. 2002, 16, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Zhu, F.; Ma, X.; Cao, Z.; Li, Y.; Chen, Y.Z. Mechanisms of drug combinations: Interaction and network perspectives. Nat. Rev. Drug Discov. 2009, 8, 111–128. [Google Scholar] [CrossRef] [PubMed]
- Fortin, S.; Berube, G. Advances in the development of hybrid anticancer drugs. Expert Opin. Drug Discov. 2013, 8, 1029–1047. [Google Scholar] [CrossRef] [PubMed]
- Mehta, G.; Singh, V. Hybrid systems through natural product leads: An approach towards new molecular entities. Chem. Soc. Rev. 2002, 31, 324–334. [Google Scholar] [CrossRef] [PubMed]
- Nepali, K.; Sharma, S.; Kumar, D.; Budhiraja, A.; Dhar, K.L. Anticancer hybrids—A patent survey. Recent Pat. Anticancer Drug Discov. 2014, 9, 303–339. [Google Scholar] [CrossRef] [PubMed]
- Bansal, Y.; Silakari, O. Multifunctional compounds: Smart molecules for multifactorial diseases. Eur. J. Med. Chem. 2014, 76, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Morphy, R.; Kay, C.; Rankovic, Z. From magic bullets to designed multiple ligands. Drug Discov. Today 2004, 9, 641–651. [Google Scholar] [CrossRef] [PubMed]
- Nepali, K.; Sharma, S.; Sharma, M.; Bedi, P.M.; Dhar, K.L. Rational approaches, design strategies, structure activity relationship and mechanistic insights for anticancer hybrids. Eur. J. Med. Chem. 2014, 77, 422–487. [Google Scholar] [CrossRef] [PubMed]
- Muregi, F.W.; Ishih, A. Next-generation antimalarial drugs: Hybrid molecules as a new strategy in drug design. Drug Dev. Res. 2010, 71, 20–32. [Google Scholar] [PubMed]
- Tietze, L.F.; Bell, H.P.; Chandrasekhar, S. Natural product hybrids as new leads for drug discovery. Angew. Chem. Int. Ed. 2003, 42, 3996–4028. [Google Scholar] [CrossRef]
- Gademann, K. Natural produc hybrids. Chimia 2006, 60, 841–845. [Google Scholar] [CrossRef]
- Tsogoeva, S.B. Recent progress in the development of synthetic hybrids of natural or unnatural bioactive compounds for medicinal chemistry. Mini. Rev. Med. Chem. 2010, 10, 773–793. [Google Scholar] [CrossRef] [PubMed]
- Decker, M. Hybrid molecules incorporating natural products: Applications in cancer therapy, neurodegenerative disorders and beyond. Curr. Med. Chem. 2011, 18, 1464–1475. [Google Scholar] [CrossRef] [PubMed]
- Li, S.Y.; Wang, X.B.; Xie, S.S.; Jiang, N.; Wang, K.D.; Yao, H.Q.; Sun, H.B.; Kong, L.Y. Multifunctional tacrine-flavonoid hybrids with cholinergic, beta-amyloid-reducing, and metal chelating properties for the treatment of alzheimer’s disease. Eur. J. Med. Chem. 2013, 69, 632–646. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, C.S.; Paul, S.; Suh, N.; Rimando, A.M. Synthesis and biological evaluation of retinoid-chalcones as inhibitors of colon cancer cell growth. Bioorg. Med. Chem. Lett. 2010, 20, 7385–7387. [Google Scholar] [CrossRef] [PubMed]
- Mao, F.; Yan, J.; Li, J.; Jia, X.; Miao, H.; Sun, Y.; Huang, L.; Li, X. New multi-target-directed small molecules against alzheimer’s disease: A combination of resveratrol and clioquinol. Org. Biomol. Chem. 2014, 12, 5936–5944. [Google Scholar] [CrossRef] [PubMed]
- Belluti, F.; Fontana, G.; dal Bo, L.; Carenini, N.; Giommarelli, C.; Zunino, F. Design, synthesis and anticancer activities of stilbene-coumarin hybrid compounds: Identification of novel proapoptotic agents. Bioorg. Med. Chem. 2010, 18, 3543–3550. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Sarkar, J.; Sashidhara, K.V.; Ali, S.; Sinha, S. Anti-tumour activity of a novel coumarin-chalcone hybrid is mediated through intrinsic apoptotic pathway by inducing puma and altering bax/bcl-2 ratio. Apoptosis 2014, 19, 1017–1028. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Aziz, M.; Park, S.E.; Abuo-Rahma Gel, D.; Sayed, M.A.; Kwon, Y. Novel n-4-piperazinyl-ciprofloxacin-chalcone hybrids: Synthesis, physicochemical properties, anticancer and topoisomerase i and ii inhibitory activity. Eur. J. Med. Chem. 2013, 69, 427–438. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.H.; Batist, G.; Zhou, J.; Geng, G.; Lin, R.; Li, Y. Hybrid-Ionone and Curcumin Molecules as Anticancer Agents. U.S. Patent 8470889 B2, 25 June 2013. [Google Scholar]
- Dimauro, T.M. Methylated Curcumin-Resveratrol Hybrid Molecules for Treating Cancer. U.S. Patent 8350093 B2, 8 January 2013. [Google Scholar]
- Dimauro, T.M. Curcumin-Resveratrol Hybrids. European Patent 2749552 A1, 2 July 2014. [Google Scholar]
- Mishra, S.; Kapoor, N.; Mubarak Ali, A.; Pardhasaradhi, B.V.; Kumari, A.L.; Khar, A.; Misra, K. Differential apoptotic and redox regulatory activities of curcumin and its derivatives. Free Radic. Biol. Med. 2005, 38, 1353–1360. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, F.; Banks, L. The human papillomavirus e6 protein and its contribution to malignant progression. Oncogene 2001, 20, 7874–7887. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Misra, K. Human papilloma virus 16 e6 protein as a target for curcuminoids, curcumin conjugates and congeners for chemoprevention of oral and cervical cancers. Interdiscip. Sci. 2013, 5, 112–118. [Google Scholar] [CrossRef] [PubMed]
- Dubey, S.K.; Sharma, A.K.; Narain, U.; Misra, K.; Pati, U. Design, synthesis and characterization of some bioactive conjugates of curcumin with glycine, glutamic acid, valine and demethylenated piperic acid and study of their antimicrobial and antiproliferative properties. Eur. J. Med. Chem. 2008, 43, 1837–1846. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.V.; Agarwal, S.; Singh, P.; Godbole, M.M.; Misra, K. Curcumin conjugates induce apoptosis via a mitochondrion dependent pathway in mcf-7 and mda-mb-231 cell lines. Asian Pac. J. Cancer Prev. 2013, 14, 5797–5804. [Google Scholar] [CrossRef] [PubMed]
- Trecul, A.; Morceau, F.; Dicato, M.; Diederich, M. Dietary compounds as potent inhibitors of the signal transducers and activators of transcription (stat) 3 regulatory network. Genes Nutr. 2012, 7, 111–125. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Bora, U. Molecular docking studies on inhibition of stat3 dimerization by curcumin natural derivatives and its conjugates with amino acids. Bioinformation 2012, 8, 988–993. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Rojas, A.; Huerta-Reyes, M. Human gonadotropin-releasing hormone receptor-activated cellular functions and signaling pathways in extra-pituitary tissues and cancer cells (review). Oncol. Rep. 2009, 22, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Limonta, P.; Montagnani Marelli, M.; Mai, S.; Motta, M.; Martini, L.; Moretti, R.M. Gnrh receptors in cancer: From cell biology to novel targeted therapeutic strategies. Endocr. Rev. 2012, 33, 784–811. [Google Scholar] [CrossRef] [PubMed]
- Montagnani Marelli, M.; Moretti, R.M.; Januszkiewicz-Caulier, J.; Motta, M.; Limonta, P. Gonadotropin-releasing hormone (gnrh) receptors in tumors: A new rationale for the therapeutical application of gnrh analogs in cancer patients? Curr. Cancer Drug Targets 2006, 6, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Grundker, C.; Gunthert, A.R.; Westphalen, S.; Emons, G. Biology of the gonadotropin-releasing hormone system in gynecological cancers. Eur. J. Endocrinol. 2002, 146, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.; Ndinguri, M.W.; Solipuram, R.; Wakamatsu, N.; Hammer, R.P.; Ingram, D.; Hansel, W. [dlys(6)]-luteinizing hormone releasing hormone-curcumin conjugate inhibits pancreatic cancer cell growth in vitro and in vivo. Int. J. Cancer 2011, 129, 1611–1623. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Wada, K.; Ohkoshi, E.; Lin, L.; Huang, R.; Morris-Natschke, S.L.; Goto, M.; Lee, K.H. Antitumor agents 290. Design, synthesis, and biological evaluation of new lncap and pc-3 cytotoxic curcumin analogs conjugated with anti-androgens. Bioorg. Med. Chem. 2012, 20, 4020–4031. [Google Scholar] [CrossRef] [PubMed]
- Souriau, C.; Hudson, P.J. Recombinant antibodies for cancer diagnosis and therapy. Expert Opin. Biol. Ther. 2003, 3, 305–318. [Google Scholar] [CrossRef] [PubMed]
- List, T.; Neri, D. Immunocytokines: A review of molecules in clinical development for cancer therapy. Clin. Pharmacol. 2013, 5, 29–45. [Google Scholar] [PubMed]
- Xin, L.; Cao, J.; Cheng, H.; Zeng, F.; Hu, X.; Shao, J. Human monoclonal antibodies in cancer therapy: A review of recent developments. Front Biosci. (Landmark Ed.) 2013, 18, 765–772. [Google Scholar] [CrossRef]
- Cole, L.A. Hcg variants, the growth factors which drive human malignancies. Am. J. Cancer Res. 2012, 2, 22–35. [Google Scholar] [PubMed]
- Vyas, H.K.; Pal, R.; Vishwakarma, R.; Lohiya, N.K.; Talwar, G.P. Selective killing of leukemia and lymphoma cells ectopically expressing hcgbeta by a conjugate of curcumin with an antibody against hcgbeta subunit. Oncology 2009, 76, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Sung, B.; Kunnumakkara, A.B.; Sethi, G.; Anand, P.; Guha, S.; Aggarwal, B.B. Curcumin circumvents chemoresistance in vitro and potentiates the effect of thalidomide and bortezomib against human multiple myeloma in nude mice model. Mol. Cancer Ther. 2009, 8, 959–970. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Zhang, D.; Chojnacki, J.; Du, Y.; Fu, H.; Grant, S.; Zhang, S. Design and biological characterization of hybrid compounds of curcumin and thalidomide for multiple myeloma. Org. Biomol. Chem. 2013, 11, 4757–4763. [Google Scholar] [CrossRef] [PubMed]
- Juneja, M.; Vanam, U.; Paranthaman, S.; Bharathan, A.; Keerthi, V.S.; Reena, J.K.; Rajaram, R.; Rajasekharan, K.N.; Karunagaran, D. 4-amino-2-arylamino-5-indoloyl/cinnamoythiazoles, analogs of topsentin-class of marine alkaloids, induce apoptosis in hela cells. Eur. J. Med. Chem. 2013, 63, 474–483. [Google Scholar] [CrossRef] [PubMed]
- Koon, H.; Leung, A.W.; Yue, K.K.; Mak, N.K. Photodynamic effect of curcumin on npc/cne2 cells. J. Environ. Pathol. Toxicol. Oncol. 2006, 25, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Prasad, P.; Hussain, A.; Khan, I.; Kondaiah, P.; Chakravarty, A.R. Remarkable photocytotoxicity of curcumin in hela cells in visible light and arresting its degradation on oxovanadium(iv) complex formation. Chem. Commun. 2012, 48, 7702–7704. [Google Scholar] [CrossRef]
- Prasad, P.; Khan, I.; Kondaiah, P.; Chakravarty, A.R. Mitochondria-targeting oxidovanadium(iv) complex as a near-ir light photocytotoxic agent. Chemistry 2013, 19, 17445–17455. [Google Scholar] [CrossRef] [PubMed]
- Banik, B.; Somyajit, K.; Nagaraju, G.; Chakravarty, A.R. Oxovanadium(iv) complexes of curcumin for cellular imaging and mitochondria targeted photocytotoxicity. Dalton Trans. 2014, 43, 13358–13369. [Google Scholar] [CrossRef] [PubMed]
- Balaji, B.; Somyajit, K.; Banik, B.; Nagaraju, G.; Chakravarty, A.R. Photoactivated DNA cleavage and anticancer activity of oxovanadium(iv) complexes of curcumin. Inorg. Chim. Acta 2013, 400, 142–150. [Google Scholar] [CrossRef]
- Kanakis, C.D.; Tarantilis, P.A.; Polissiou, M.G.; Tajmir-Riahi, H.A. Probing the binding sites of resveratrol, genistein, and curcumin with milk beta-lactoglobulin. J. Biomol. Struct. Dyn. 2013, 31, 1455–1466. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Ma, Y.; Ngadi, M.O. Binding of curcumin to beta-lactoglobulin and its effect on antioxidant characteristics of curcumin. Food Chem. 2013, 141, 1504–1511. [Google Scholar] [CrossRef] [PubMed]
- Tsujii, H.; Yamada, T.; Kajimoto, T.; Tanaka, R.; Tokuda, H.; Hasegawa, J.; Hamashima, Y.; Node, M. Hybrids of 3alpha-methoxyserrat-14-en-21beta-ol (pj-1) and 3beta-methoxyserrat-14-en-21beta-ol (pj-2) and various anti-oxidants as cancer chemopreventive agents. Eur. J. Med. Chem. 2010, 45, 2191–2197. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa-Goto, K.; Yamada, K.; Nakamura, S.; Chen, T.H.; Chiang, P.C.; Bastow, K.F.; Wang, S.C.; Spohn, B.; Hung, M.C.; Lee, F.Y.; et al. Antitumor agents. 258. Syntheses and evaluation of dietary antioxidant—Taxoid conjugates as novel cytotoxic agents. Bioorg. Med. Chem. Lett. 2007, 17, 5204–5209. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa-Goto, K.; Nakamura, S.; Bastow, K.F.; Nyarko, A.; Peng, C.Y.; Lee, F.Y.; Lee, F.C.; Lee, K.H. Antitumor agents. 256. Conjugation of paclitaxel with other antitumor agents: Evaluation of novel conjugates as cytotoxic agents. Bioorg. Med. Chem. Lett. 2007, 17, 2894–2898. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, R.; Tsujii, H.; Yamada, T.; Kajimoto, T.; Amano, F.; Hasegawa, J.; Hamashima, Y.; Node, M.; Katoh, K.; Takebe, Y. Novel 3alpha-methoxyserrat-14-en-21beta-ol (pj-1) and 3beta-methoxyserrat-14-en-21beta-ol (pj-2)-curcumin, kojic acid, quercetin, and baicalein conjugates as hiv agents. Bioorg. Med. Chem. 2009, 17, 5238–5246. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.H.; Loo, C.Y.; Bebawy, M.; Luk, F.; Mason, R.S.; Rohanizadeh, R. Curcumin and its derivatives: Their application in neuropharmacology and neuroscience in the 21st century. Curr. Neuropharmacol. 2013, 11, 338–378. [Google Scholar] [CrossRef] [PubMed]
- Chin, D.; Huebbe, P.; Pallauf, K.; Rimbach, G. Neuroprotective properties of curcumin in Alzheimer’s disease--merits and limitations. Curr. Med. Chem. 2013, 20, 3955–3985. [Google Scholar] [CrossRef] [PubMed]
- Reinke, A.A.; Gestwicki, J.E. Structure-activity relationships of amyloid beta-aggregation inhibitors based on curcumin: Influence of linker length and flexibility. Chem. Biol. Drug Des. 2007, 70, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Peng, P.; Tang, L.; Hu, Y.; Sheng, R. Design, synthesis and evaluation of rivastigmine and curcumin hybrids as site-activated multi-target-directed ligands for Alzheimer’s disease therapy. Bioorg. Med. Chem. 2014, 22, 4717–4725. [Google Scholar] [CrossRef] [PubMed]
- Lenhart, J.A.; Ling, X.; Gandhi, R.; Guo, T.L.; Gerk, P.M.; Brunzell, D.H.; Zhang, S. “Clicked” bivalent ligands containing curcumin and cholesterol as multifunctional abeta oligomerization inhibitors: Design, synthesis, and biological characterization. J. Med. Chem. 2010, 53, 6198–6209. [Google Scholar] [CrossRef] [PubMed]
- Chojnacki, J.E.; Liu, K.; Yan, X.; Toldo, S.; Selden, T.; Estrada, M.; Rodriguez-Franco, M.I.; Halquist, M.S.; Ye, D.; Zhang, S. Discovery of 5-(4-hydroxyphenyl)-3-oxo-pentanoic acid [2-(5-methoxy-1h-indol-3-yl)-ethyl]-amide as a neuroprotectant for alzheimer’s disease by hybridization of curcumin and melatonin. ACS Chem. Neurosci. 2014, 5, 690–699. [Google Scholar] [CrossRef] [PubMed]
- Lapchak, P.A.; McKim, J.M., Jr. Ceetox analysis of cnb-001 a novel curcumin-based neurotrophic/neuroprotective lead compound to treat stroke: Comparison with nxy-059 and radicut. Transl. Stroke Res. 2011, 2, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Abdel Aziz, M.T.; Motawi, T.; Rezq, A.; Mostafa, T.; Fouad, H.H.; Ahmed, H.H.; Rashed, L.; Sabry, D.; Senbel, A.; Al-Malki, A.; et al. Effects of a water-soluble curcumin protein conjugate vs. Pure curcumin in a diabetic model of erectile dysfunction. J. Sex. Med. 2012, 9, 1815–1833. [Google Scholar] [CrossRef] [PubMed]
- Zaahkouk, A.M.; Abdel Aziz, M.T.; Rezq, A.M.; Atta, H.M.; Fouad, H.H.; Ahmed, H.H.; Sabry, D.; Yehia, M.H. Efficacy of a novel water-soluble curcumin derivative versus sildenafil citrate in mediating erectile function. Int. J. Impot. Res. 2014. [Google Scholar] [CrossRef]
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teiten, M.-H.; Dicato, M.; Diederich, M. Hybrid Curcumin Compounds: A New Strategy for Cancer Treatment. Molecules 2014, 19, 20839-20863. https://doi.org/10.3390/molecules191220839
Teiten M-H, Dicato M, Diederich M. Hybrid Curcumin Compounds: A New Strategy for Cancer Treatment. Molecules. 2014; 19(12):20839-20863. https://doi.org/10.3390/molecules191220839
Chicago/Turabian StyleTeiten, Marie-Hélène, Mario Dicato, and Marc Diederich. 2014. "Hybrid Curcumin Compounds: A New Strategy for Cancer Treatment" Molecules 19, no. 12: 20839-20863. https://doi.org/10.3390/molecules191220839
APA StyleTeiten, M.-H., Dicato, M., & Diederich, M. (2014). Hybrid Curcumin Compounds: A New Strategy for Cancer Treatment. Molecules, 19(12), 20839-20863. https://doi.org/10.3390/molecules191220839





