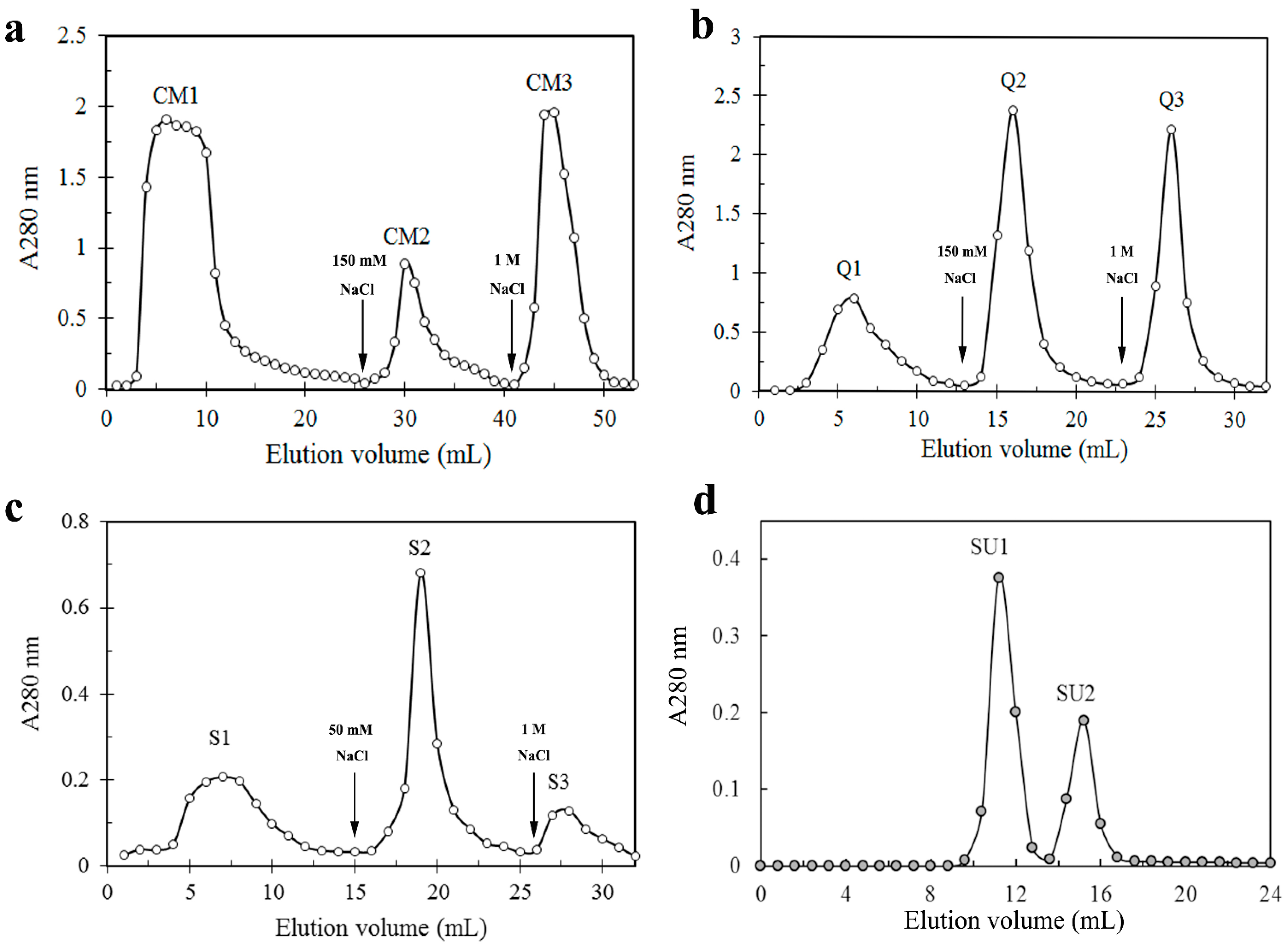
2.1. Results
The extract of the
Stropharia rugosoannulata fruiting bodies was pretreated by precipitation by 70% ammonium sulfate. We obtained three fractions (CM 1–3) after loading the sample, which had been dialyzed against sodium acetate buffer (pH5.2), on a CM-cellulose chromatography column, and the lectin activity was detected in fraction CM2 (
Figure 1a). Fraction CM2 was then subjected to chromatography on Q-Sepharose and lectin activities was enriched in the second (Q2) of the three resulting fractions Q1-3 (
Figure 1b). Subsequently, fraction Q2 was then separated into three fractions S1–S3 by SP-sepharose chromatography. The lectin activity resided in fraction S2 (
Figure 1c). Upon gel filtration on Superdex 75, S2 was resolved into a large peak SU1 and a small peak SU2 (
Figure 1d). Hemagglutinating activity was confined to SU1, which possessed a molecular mass of 36 kDa.
Figure 1.
Purification of S. rugosoannulata lectin (SRL) by chromatography on (a) CM-celluose (2.5 cm × 30 cm); (b) Q-Sepharose (1 cm × 10 cm); (c) SP-Sepharose (1 cm × 10 cm) and (d) gel-filtration on Superdex G-75 HR10/30 column. Arrows indicate the point at which buffer was changed.
Figure 1.
Purification of S. rugosoannulata lectin (SRL) by chromatography on (a) CM-celluose (2.5 cm × 30 cm); (b) Q-Sepharose (1 cm × 10 cm); (c) SP-Sepharose (1 cm × 10 cm) and (d) gel-filtration on Superdex G-75 HR10/30 column. Arrows indicate the point at which buffer was changed.
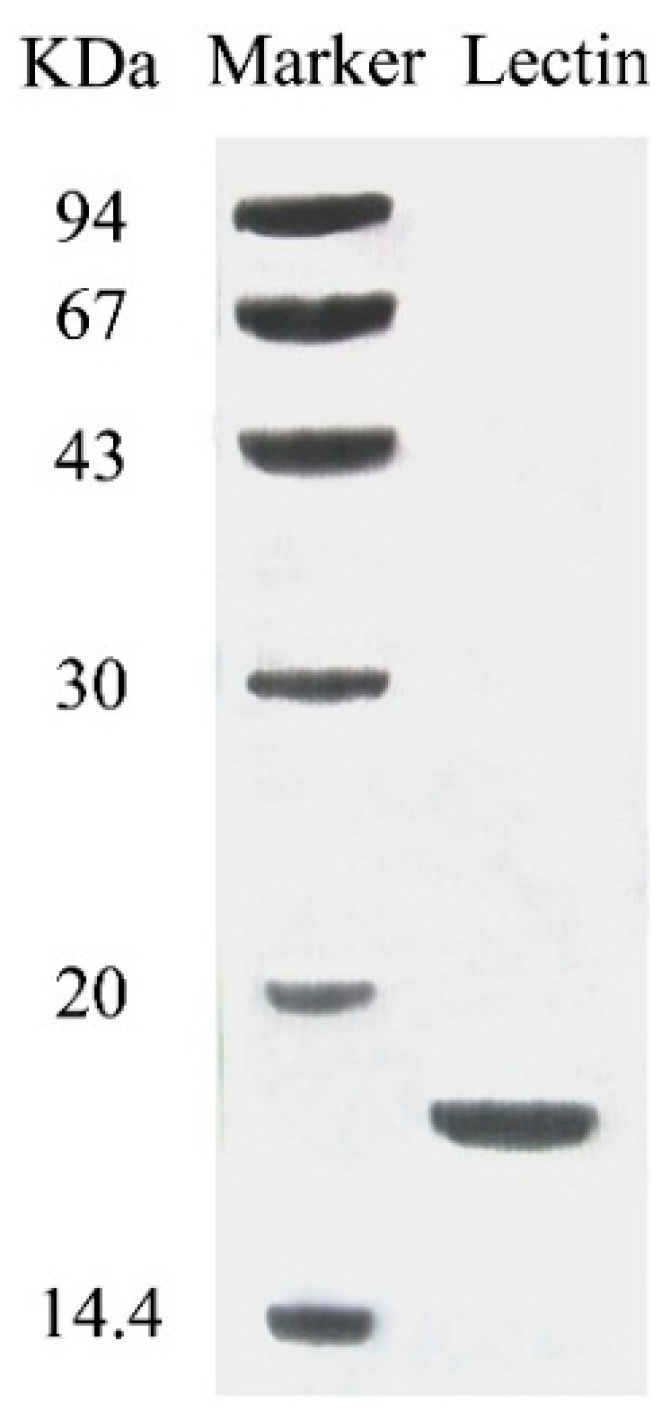
The purified lectin appeared as a single band with a molecular mass of 18 kDa in SDS-PAGE (
Figure 2). This suggests that the lectin was composed of two subunits, each with a molecular mass of 18 kDa. The N-terminal sequence of this lectin was IKSGVYRIVSWQGALGPEAR. A BLAST search did not reveal sequence resemblance to any previously published mushroom lectin or other lectins.
The yields and specific hemagglutinating activities of chromatographic fractions are given in
Table 1 which shows that the lectin was purified more than 450-fold. The hemagglutinating activity of SRL remained stable between 10 °C and 70 °C, while 50% activity remained at 80 °C and 25% activity was left at 90 °C. No activity was detectable at 100 °C (
Table 2). The activity was retained in the presence of 6 mM and 12.5 mM HCl (pH 1.9) and 6 mM NaOH (pH 11.7), but it was only 50% when the HCl concentration was increased to 25 mM (pH 1.6) and that of NaOH to 12.5 mM (pH 12.1). There was no activity detectable at 100 mM NaOH or HCl concentration (pH below 1 and above 13, respectively) (
Table 3).
Figure 2.
SDS-PAGE results of fraction SU1 which represents purified SRL.
Figure 2.
SDS-PAGE results of fraction SU1 which represents purified SRL.
Table 1.
Summary of purification of S. rugosoannulata lectin (from 400 g fresh fruiting bodies).
Table 1.
Summary of purification of S. rugosoannulata lectin (from 400 g fresh fruiting bodies).
| Fraction | Yield (mg) | Specific Activity (U/mg) | Total Activity (U) | Recovery of Activity (%) | Folds of Purification |
|---|
| CM | 11,203.8 | 0.7 | 8105.0 | 100.0 | 1.0 |
| CM1 | 2125.7 | - | - | - | - |
| CM2 | 280.5 | 11.3 | 3170.6 | 39.1 | 16.1 |
| CM3 | 1286.3 | - | - | - | - |
| Q1 | 51. | - | - | - | - |
| Q2 | 44.6 | 44.4 | 1978.2 | 24.4 | 63.4 |
| Q3 | 46.9 | - | - | - | - |
| S1 | 11.9 | - | - | - | - |
| S2 | 12.3 | 117.8 | 1453.6 | 17.9 | 168.3 |
| S3 | 5.6 | - | - | - | - |
| SU1 | 2.0 | 320.0 | 640.0 | 7.9 | 457.1 |
| SU2 | 1.2 | - | - | - | - |
Table 2.
Effects of temperature on hemagglutinating activity of S. rugosoannulata lectin. (initial hemagglutinating activity: 64U). S. rugosoannulata lectin was incubated at different temperatures for 30 min before determination of hemagglutinating activity.
Table 2.
Effects of temperature on hemagglutinating activity of S. rugosoannulata lectin. (initial hemagglutinating activity: 64U). S. rugosoannulata lectin was incubated at different temperatures for 30 min before determination of hemagglutinating activity.
| Temperature (°C) | 10 | 20 | 30 | 40 | 50 | 60 | 70 | 80 | 90 | 100 |
|---|
| Hemagglutinating activity (U) | 64 | 64 | 64 | 64 | 64 | 64 | 32 | 16 | 16 | 0 |
Table 3.
Effects of NaOH and HCl solutions on hemagglutinating activity of S. rugosoannulata lectin. (initial hemagglutinating activity: 64U). S. rugosoannulata lectin was incubated at different pH values for 60 min before determination of hemagglutinating activity.
Table 3.
Effects of NaOH and HCl solutions on hemagglutinating activity of S. rugosoannulata lectin. (initial hemagglutinating activity: 64U). S. rugosoannulata lectin was incubated at different pH values for 60 min before determination of hemagglutinating activity.
| The Hemagglutinating Activity of SRL in Different Concentrations of HCl |
|---|
| HCl concentration (M) | 6 (pH 2.2) | 12.5 (pH 1.9) | 25 (pH 1.6) | 50 (pH 1.3) | 100 (pH 1.0) | 200 (pH 0.7) |
| Hemagglutinating Activity (U) | 64 | 64 | 32 | 8 | 0 | 0 |
| The Hemagglutinating Activity of SRL in Different Concentrations of NaOH |
| NaOH concentration (M) | 6 (pH 11.7) | 12.5 (pH 12.1) | 25 (pH 12.4) | 50 (pH 12.7) | 100 (pH 13.0) | 200 (pH 13.3) |
| Hemagglutinating Activity (U) | 64 | 32 | 16 | 8 | 0 | 0 |
The hemagglutinating activity of the lectin was not affected by the majority of divalent and trivalent metallic ions tested (Fe
2+, Cu
2+, Al
3+, Mg
2+, and K
+). Zn
2+, Ca
2+, and Mg
2+ ions at a higher concentration (10 mM) slightly affected the hemagglutinating activity. The activity was drastically reduced by Cd
2+ and Hg
2+ ions at a concentration range of 1.25 mM to 10 mM, and dose-dependently reduced by Pb
2+ and Fe
3+ ions (
Table 4).
Table 4.
Effects of cations on hemagglutinating activity of S. rugosoannulata lectin. (initial hemagglutinating activity: 64U). S. rugosoannulata lectin was incubated in presence of different metal ions for 60 min before determination of hemagglutinating activity.
Table 4.
Effects of cations on hemagglutinating activity of S. rugosoannulata lectin. (initial hemagglutinating activity: 64U). S. rugosoannulata lectin was incubated in presence of different metal ions for 60 min before determination of hemagglutinating activity.
| Cations | 10 mM | 5 mM | 2.5 mM | 1.25 mM |
|---|
| Cd2+ | 0 | 0 | 2 | 4 |
| Fe3+ | 64 | 64 | 64 | 64 |
| Cu2+ | 64 | 64 | 64 | 64 |
| Hg2+ | 0 | 0 | 2 | 4 |
| Al3+ | 64 | 64 | 64 | 64 |
| Pb2+ | 4 | 16 | 32 | 64 |
| Zn2+ | 32 | 32 | 64 | 64 |
| Ca2+ | 32 | 64 | 64 | 64 |
| Mn2+ | 32 | 64 | 64 | 64 |
| Mg2+ | 64 | 64 | 64 | 64 |
| K+ | 64 | 64 | 64 | 64 |
| Fe2+ | 0 | 4 | 16 | 64 |
All sugars tested excepted inulin had no effect on the hemagglutinating activity of the lectin when tested at various concentrations from 0.87 mM to 200 mM. The minimum concentration which inhibited the hemagglutinating of inulin was 50 mM (
Table 5).
Table 5.
Effects of various carbohydrates on hemagglutinating induced by S. rugosoannulata lectin.
Table 5.
Effects of various carbohydrates on hemagglutinating induced by S. rugosoannulata lectin.
| Sugar | Minimum Inhibitory Concentration of Sugar (mM) |
|---|
| l-Sorbose | No |
| Raffinose | No |
| l-Rhamnose | No |
| d-Fructose | No |
| d-Mannose | No |
| Cellobiose | No |
| l-Arabinose | No |
| d-Xylose | No |
| d-Melibiose | No |
| Lactose | No |
| Inulin | 0.1 M |
| Maltose | No |
| d-Galactose | No |
| d-Glucose | No |
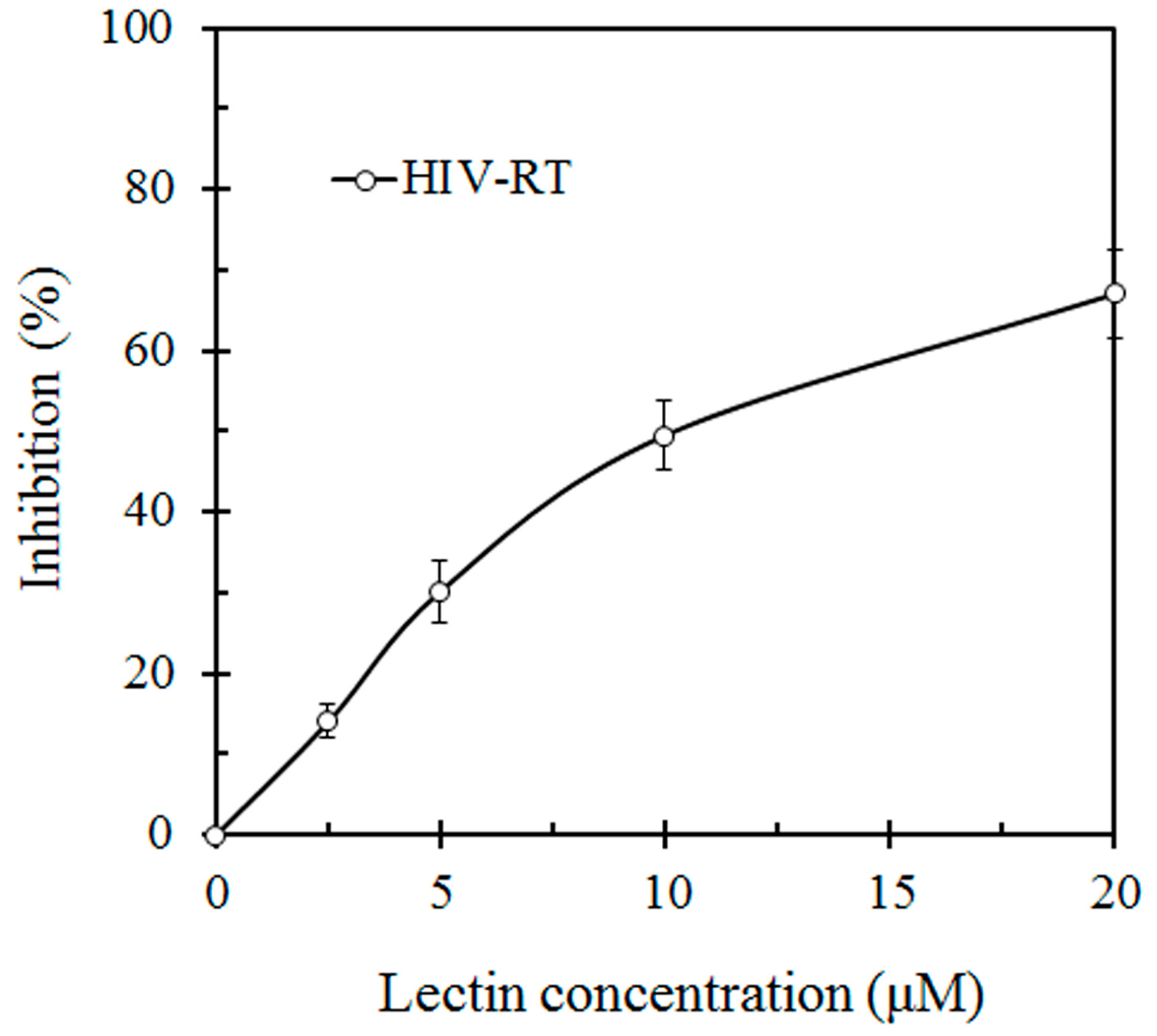
This lectin inhibited the proliferation of HepG2 and L1210 tumor cells with an IC
50 value of 7 μM and 19 μM, respectively (
Figure 3). SRL also had anti HIV-1 reverse transcriptase activity with an IC
50 value of 10 μM (
Figure 4).
Figure 3.
In vitro inhibitory effect of S. rugosoannulata lectin (SRL) on proliferation of hepatoma Hep G2 cells and murine leukemia L1210 cells. Proliferation of HepG2 cells and L1210 cells was inhibited with an IC50 of 7 μM and 19 μM, respectively.
Figure 3.
In vitro inhibitory effect of S. rugosoannulata lectin (SRL) on proliferation of hepatoma Hep G2 cells and murine leukemia L1210 cells. Proliferation of HepG2 cells and L1210 cells was inhibited with an IC50 of 7 μM and 19 μM, respectively.
Figure 4.
Inhibitory effect of S. rugosoannulata lectin (SRL) on HIV-1 reverse transcriptase (HIV-RT). HIV-RT activity was inhibited with an IC50 of 10 μM.
Figure 4.
Inhibitory effect of S. rugosoannulata lectin (SRL) on HIV-1 reverse transcriptase (HIV-RT). HIV-RT activity was inhibited with an IC50 of 10 μM.
2.2. Discussion
Stropharia rugosoannulata is an edible mushroom which can be cultivated for food. In the genus Stropharia, S. rugosoannulata and Stropharia aeruginosa are the two species which are relatively better known than others. However, few studies about either of them have been reported. In this study, we isolated a lectin (SRL) from S. rugosoannulata which represents the first protein purified from this species. The finding of SRL may promote the development of S. rugosoannulata to some degree.
SRL has been purified more than 450-fold in this study, which is much higher than the values for other lectins [
7,
18]. During the purification procedure, four chromatographic steps including three ion exchange columns and one gel-filtration step were involved. SRL is not adsorbed on DEAE-cellulose, but is adsorbed on CM-cellulose, Q-sepharose, and SP-sepharose.
The molecular weight determined by SDS-PAGE is half of the amount determined by gel-filtration chromatography. This suggests that SRL is dimeric, like lectins from other mushrooms including
Pleurotus citrinopileatus [
8] and
Agrocybe cylindracea [
19]. The molecular mass of SRL is within the range shown by mushroom lectins [
20].
SRL is the first protein isolated from
Stropharia rugosoannulata with a unique
N-terminal sequence. Little resemblance of this
N-teminal sequence can be found between SRL and other mushroom lectins, such as those from
Agaricus bisporus [
21],
Agrocybe aegerita [
22],
Coprinopsis cinerea [
23],
Flammulina velutipes [
24],
Laccaria bicolor [
25], and
Pleurotus cornucopiae [
26] (shown in
Table 6).
Table 6.
Comparison of N-terminal sequence of S. rugosoannulata lectin with sequences of known mushroom lectins.
Table 6.
Comparison of N-terminal sequence of S. rugosoannulata lectin with sequences of known mushroom lectins.
| Species | N-Terminal Sequence |
|---|
| Stropharia rugosoannulata | IKSGVYRIVSWQGALGPEAR |
| Agaricus bisporus [21] | MGGSGTSGSL |
| Agrocybe aegerita [22] | NISAGTSVDL |
| Coprinopsis cinerea [23] | IPLEGTFGDR |
| Flammulina velutipes [24] | TSLTFQLAYL |
| Laccaria bicolor [25] | SHLYGDGVAL |
| Pleurotus cornucopiae [26] | SDSTWTFAML |
Compared with other lectins isolated from mushrooms, SRL has advantages in its possession of anti-proliferative activity toward cancer cells and anti-HIV-RT activity. SRL is shown to have an ability to inhibit proliferation of two tumor cell lines, Hep G2 and L1210. The potent antiproliferative activity of SRL could facilitate its development into a cancer therapy agent. As a key enzyme of the HIV-1 life cycle, HIV-1 RT is a target of anti-HIV-1 drugs. It is worth mentioning that SRL manifests potent inhibitory activity towards HIV-1 RT with an IC50 of 10 μM.
SRL is moderately thermostable. Its hemagglutinating activitity is not affected below 70 °C. At 80 °C, 50% of the activity remains. It is thus more thermostable than lectins from
Boletus edulis [
27,
28] and some other mushrooms, but similar to lectins from
Russula lepida [
9] and
Tricholoma mongolicum [
29]. SRL is stable in the presence of 0.05 M NaOH and 0.1 M HCl solution. Hemagglutinating activity is detectable even in 0.2 M NaOH and HCl. In this aspect, SRL is more stable than some other mushroom lectins [
18,
28,
30].
The lectin activity of SRL remains unaltered after metal ion chelation with EDTA or in the presence of various cations , like Fe2+, Cu2+, Al3+, Mn2+ and K+ ions, and is slightly inhibited in the presence of high concentrations of Zn2+ and Ca2+ ions. The hemagglutinating activity of SRL is strongly inhibited by Cd2+ and Hg2+ ions. The fact that no increase in activity be detected in the presence of the metal ions suggests that the active region of SRL may not contain any metal ions. The inhibitory effect of Cd2+ and Hg2+ ions indicates that SRL hemagglutinating activity may be reduced by heavy metals, probably due to protein denaturation.
Lectin is the marker for blood grouping. However, SRL cannot agglutinate human erythrocytes of any of the groups of the ABO system. Besides, SRL can agglutinate rabbit red blood cells. This probably suggests SRL cannot bind the SRL receptor of human erythrocytes but has a specific binding with rabbit erythrocytes. However, among the sugars involved in the test of carbohydrate specificity, only inulin was found to have inhibition effect to hemagglutinating activity of SRL. Inulin is a plant polysaccharide, which is a heterogeneous collection of fructose polymers. Lectins from plants and some mushroom specifically recognize simple sugars and disaccharides. However, there are a few lectins isolated from mushrooms whose hemagglutinating activity could only be inhibited by inulin [
31,
32]. To date, only few isolated lectins are found to be inulin-specific [
18,
30,
31]. Hence SRL may have a potential to be used in carbohydrate binding research and affinity chromatography.