Synthesis and Preliminary Biological Evaluations of 5′-Substituted Derivatives of Uridine as Glycosyltransferase Inhibitors
Abstract
:1. Introduction
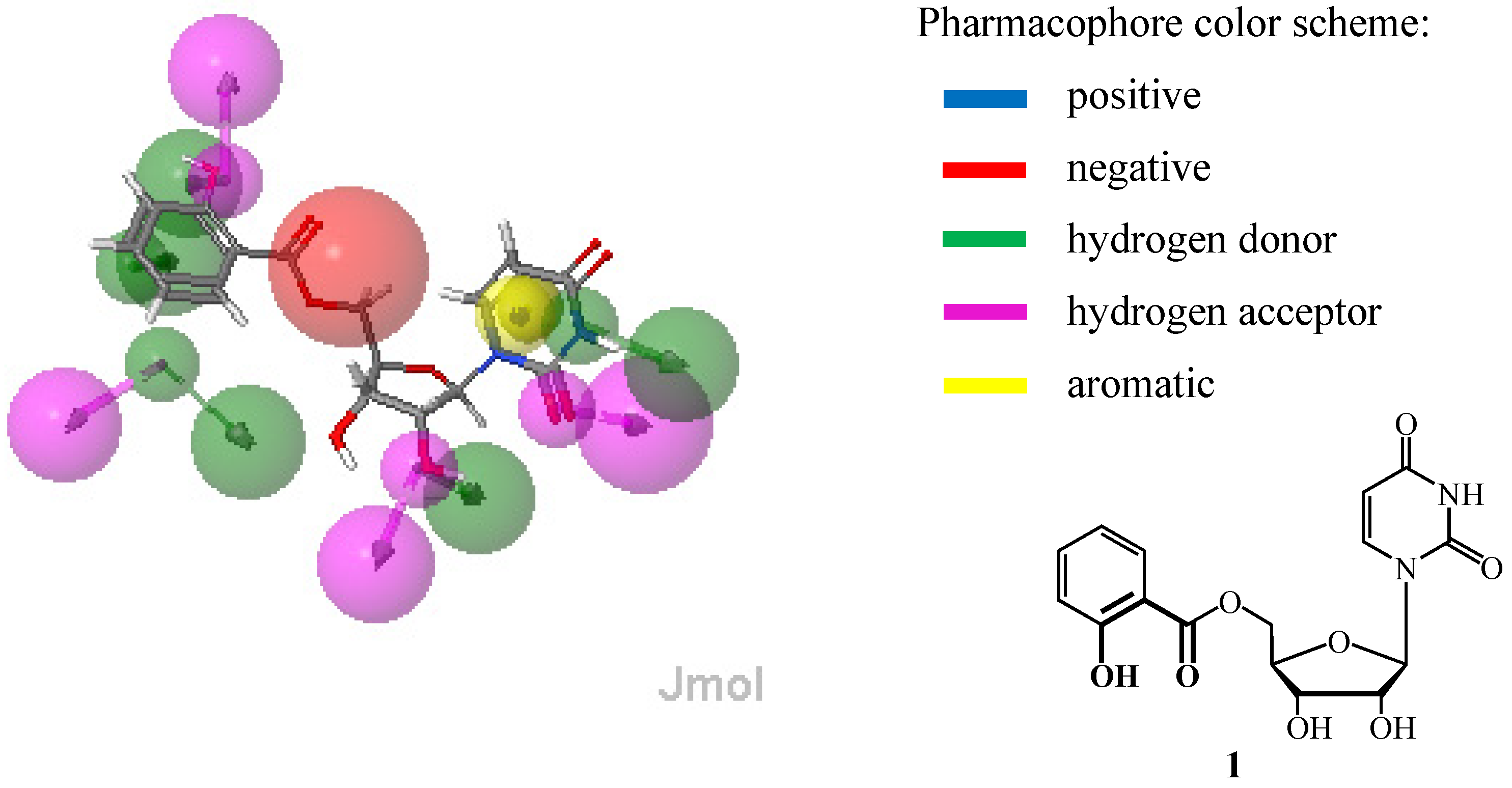
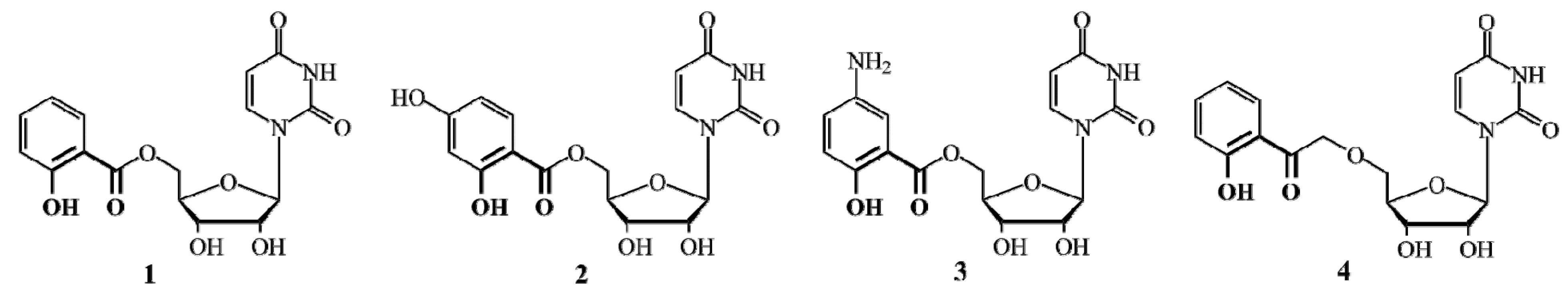
2. Results and Discussion
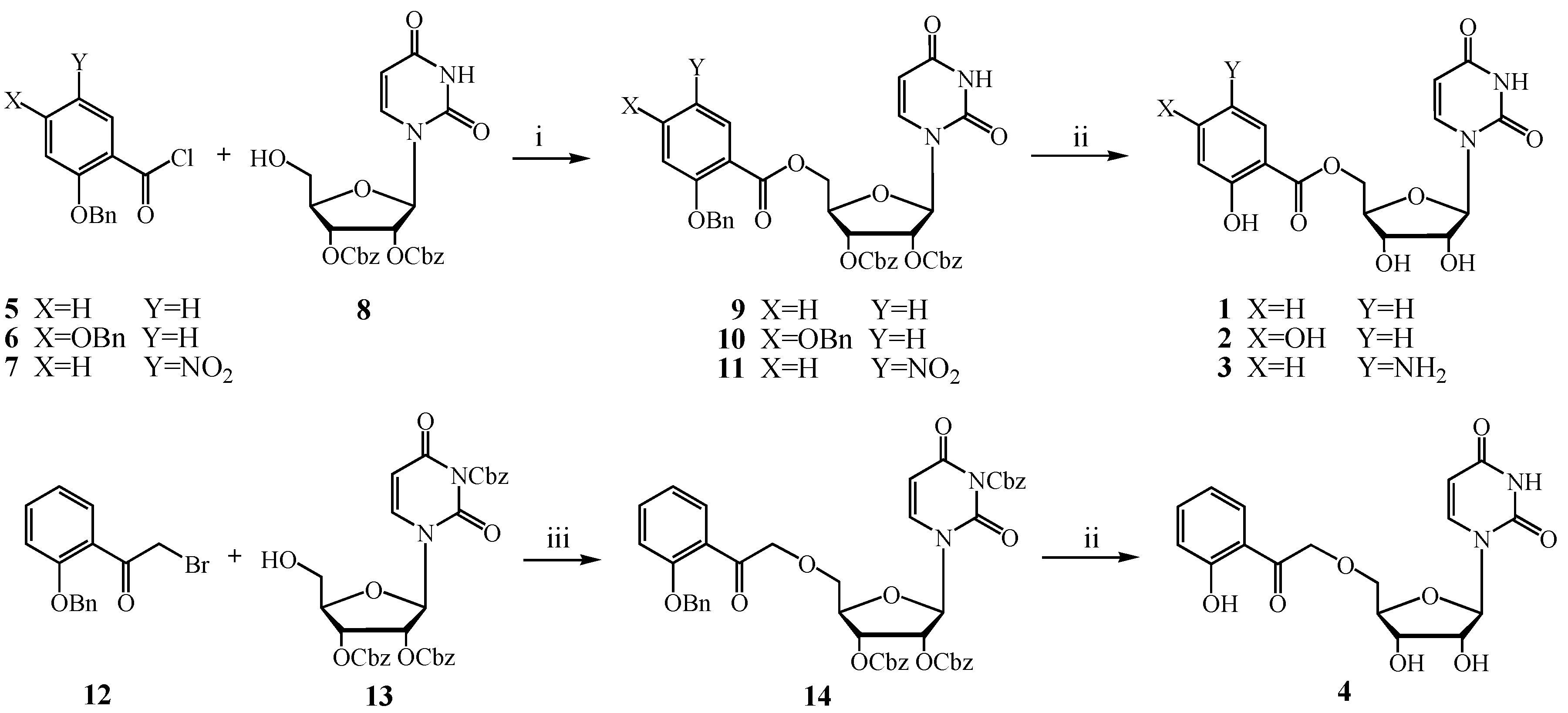
3. Experimental
3.1. General
3.2. Chemistry
3.2.1. General Procedure for the Synthesis of Ester C-5′-Substituted Derivatives 1–3
3.2.2. Procedure for the Synthesis of Ether C-5′-Substituted Derivative 4
3.3. Biological Evaluation
3.3.1. Bovine Milk β-1,4-Galactosyltransferase I Assay
3.3.2. Antifungal Assays
3.4. In Silico Target Identification
4. Conclusion
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Varki, A.; Cummings, R.D.; Esco, J.D.; Freeze, H.H.; Stanley, P.; Bertozzi, C.R.; Hart, G.W.; Etzler, M.E. Essentials of Glycobiology, 2nd ed.; CSHL Press: New York, NY, USA, 2009. [Google Scholar]
- Breton, C.; Šnajdrowa, L.; Jeanneau, C.; Koča, J.; Imberty, A. Structures and mechanisms of glycosyltransferases. Glycobiology 2006, 16, 29R–37R. [Google Scholar]
- Jung, K.-H.; Schmidt, R.R. Glycosylotransferase Inhibitors. In Carbohydrate-Based Drug Discovery, 2nd ed.; Wong, C.-H., Ed.; Wiley-VCh Verlag: Weinheim, Germany, 2003; pp. 609–659. [Google Scholar]
- Compain, P.; Martin, O.R. Carbohydrate mimetics-based glycosyltransferase inhibitors. Bioorg. Med. Chem. 2001, 9, 3077–3092. [Google Scholar] [CrossRef]
- Zou, W. C-Glycosides and Aza-C-Glycosides as potential glycosidase and glycosyltransferase inhibitors. Curr. Top. Med. Chem. 2005, 5, 1363–1391. [Google Scholar] [CrossRef]
- Kajimoto, T.; Node, M. Synthesis of inhibitors against glycosyltransferases. J. Synth. Org. Chem. Jpn. 2006, 64, 894–912. [Google Scholar] [CrossRef]
- Vaghefi, M.M.; Bernacki, R.J.; Dalley, N.K.; Wilson, B.E.; Robins, R.K. Synthesis of glycopyranosylphosphonate analogs of certain natural nucleoside diphosphate sugars as potential inhibitors of glycosyltransferases. J. Med. Chem. 1987, 30, 1383–1391. [Google Scholar] [CrossRef]
- Vaghefi, M.M.; Bernacki, R.J.; Hennen, W.J.; Robins, R.K. Synthesis of certain nucleoside methylenediphosphonate sugars as potential inhibitors of glycosyltransferases. J. Med. Chem. 1987, 30, 1391–1399. [Google Scholar] [CrossRef]
- Vidal, S.; Bruyère, I.; Malleron, A.; Augé, C.; Praly, J.-P. Non-isosteric C-glycosyl analogues of natural nucleotide diphosphate sugars as glycosyltransferase inhibitors. Bioorg. Med. Chem. 2006, 14, 7293–7301. [Google Scholar] [CrossRef]
- Wang, R.; Steensma, D.H.; Takaoka, Y.; Yun, J.W.; Kajimoto, T.; Wong, C.-H. A Search for pyrophosphate mimics for the development of substrates and inhibitors of glycosyltransferases. Bioorg. Med. Chem. 1997, 5, 661–672. [Google Scholar] [CrossRef]
- Behr, J.-B.; Gourlain, T.; Helimi, A.; Guillerm, G. Design, synthesis and biological evaluation of hetaryl-nucleoside derivatives as inhibitors of chitin synthase. Bioorg. Med. Chem. Lett. 2003, 13, 1713–1716. [Google Scholar] [CrossRef]
- Ballell, L.; Young, R.J.; Field, R.A. Synthesis and evaluation of mimetics of UDP and UDP-alpha-D-galactose, dTDP and dTDP-alpha-D-glucose with monosaccharides replacing the key pyrophosphate unit. Org. Biomol. Chem. 2005, 3, 1109–1115. [Google Scholar] [CrossRef]
- Vembaiyan, K.; Pearcey, J.A.; Bhasin, M.; Lowary, T.L.; Zou, W. Synthesis of sugar–amino acid–nucleosides as potential glycosyltransferase inhibitors. Bioorg. Med. Chem. 2011, 19, 58–66. [Google Scholar] [CrossRef]
- Tchertanov, L.; Mouscadet, J.-F. Target recognition by catechols and β-ketoenols: Potential contribution of hydrogen bonding and Mn/Mg chelation to HIV-1 integrase inhibition. J. Med. Chem. 2007, 50, 1133–1145. [Google Scholar] [CrossRef]
- Liu, X.; Ouyang, S.; Yu, B.; Huang, K.; Liu, Y.; Gong, J.; Zheng, S.; Li, Z.; Li, H.; Jiang, H. PharmMapper server: A web server for potential drug target identification via pharmacophore mapping approach. Nucleic Acids Res. 2010, 38, W609–W614. [Google Scholar] [CrossRef]
- Huang, S.-T.; Hsei, I.-J.; Chen, C. Synthesis and anticancer evaluation of bis(benzimidazoles), bis(benzoxazoles), and benzothiazoles. Bioorg. Med. Chem. 2006, 14, 6106–6119. [Google Scholar] [CrossRef]
- Fujimura, O.; Fu, G.C.; Grubbs, R.H. The synthesis of cyclic enol ethers via molybdenum alkylidene-catalyzed ring-closing metathesis. J. Org. Chem. 1994, 59, 4029–4031. [Google Scholar] [CrossRef]
- Khasanova, T.V.; Khakshoor, O.; Nowick, J.S. Functionalized analogues of an unnatural amino acid that mimics a tripeptide β-strand. Org. Lett. 2008, 10, 5293–5296. [Google Scholar] [CrossRef]
- Hanessian, S.; Liak, T.J.; Vanasse, B. Facile cleavage of benzyl ethers by catalytic transfer hydrogenation. Synthesis 1981, 396–398. [Google Scholar] [CrossRef]
- Pearlman, W.M. Noble metal hydroxides on carbon nonpyrophoric dry catalysts. Tetrahedron Lett. 1967, 17, 1663–1664. [Google Scholar] [CrossRef]
- Dueno, E.E.; Chu, F.; Kim, S.-I.; Jung, K.W. Cesium promoted O-alkylation of alcohols for the efficient ether synthesis. Tetrahedron Lett. 1999, 40, 1843–1846. [Google Scholar] [CrossRef]
- Wandzik, I.; Bieg, T.; Czaplicka, M. Synthesis of 2-deoxy-hexopyranosyl derivatives of uridine as donor substrate analogues for glycosyltransferases. Bioorg. Chem. 2009, 37, 211–216. [Google Scholar] [CrossRef]
- Behr, J.-B. Chitin synthase, a fungal glycosyltransferase that is a valuable antifungal target. Chimia 2011, 65, 1–5. [Google Scholar]
- Liu, J.; Balasubramanian, M.K. 1,3-beta-Glucan synthase: A useful target for antifungal drugs. Curr. Drug Targets Infect. Disord. 2001, 1, 159–169. [Google Scholar] [CrossRef]
- Obi, K.; Uda, J.; Iwase, K.; Sugimoto, O.; Ebisu, H.; Matsuda, A. Novel nikkomycin analogues: Inhibitors of the fungal cell wall biosynthesis enzyme chitin synthase. Bioorg. Med. Chem. Lett. 2000, 10, 1451–1454. [Google Scholar] [CrossRef]
- Xie, J.; Thellend, A.; Becker, H.; Vidal-Cros, A. Synthesis and evaluation of a C-glycosyl nucleoside as an inhibitor of chitin synthase. Carbohydr. Res. 2001, 334, 177–182. [Google Scholar] [CrossRef]
- Suda, A.; Ohta, A.; Sudoh, M.; Tsukuda, T.; Shimma, N. Combinatorial synthesis of nikkomycin analogues on solid support. Heterocycles 2001, 55, 1023–1028. [Google Scholar] [CrossRef]
- Chaudhary, P.M.; Chavan, S.R.; Shirazi, F.; Razdan, M.; Nimkar, P.; Maybhate, S.P.; Likhite, A.P.; Gonnade, R.; Hazara, B.G.; Deshpande, M.V.; et al. Exploration of click reaction for the synthesis of modified nucleosides as chitin synthase inhibitors. Bioorg. Med. Chem. 2009, 17, 2433–2440. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Cuenca-Estrella, M.; Lass-Flörl, C.; Hope, W. EUCAST technical note on the EUCAST definitive document EDef 7.2: Method for the determination of broth dilution minimum inhibitory concentrations of antifungal agents for yeasts EDef 7.2 (EUCAST-AFST). Clin. Microbiol. Infect. 2012, 18, E246–E247. [Google Scholar] [CrossRef]
- Subcommittee on Antifungal Susceptibility Testing (AFST) of the ESCMID European Committee for Antimicrobial Susceptibility Testing (EUCAST). Clin. Microbiol. Infect. 2008, 14, 982–984. [CrossRef]
- Murata, T.; Shimada, M.; Kadono, H.; Sakakibara, S.; Yoshino, T.; Masuda, T.; Shimazaki, M.; Shintani, T.; Fuschikami, K.; Bacon, K.B.; et al. Synthesis and structure-activity relationships of novel IKK-β inhibitors. Part 2: Improvement of in vitro activity. Bioorg. Med. Chem. Lett. 2004, 14, 4013–4017. [Google Scholar] [CrossRef]
- Johnson, D.C., II; Widlanski, T.S. Facile deprotection of O-Cbz-protected nucleosides by hydrogenolysis: An alternative to O-benzyl ether-protected nucleosides. Org. Lett. 2004, 6, 4643–4646. [Google Scholar] [CrossRef]
- Wandzik, I.; Bieg, T.; Kadela, M. Simultaneous removal of benzyl and benzyloxycarbonyl protective groups in 5′-O-(2-deoxy-α-D-glucopyranosyl)uridine by catalytic transfer hydrogenolysis. Nucleos. Nucleot. Nucl. 2008, 27, 1250–1256. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1-4 are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Paszkowska, J.; Kral, K.; Bieg, T.; Nawrot, U.; Szeja, W.; Wandzik, I. Synthesis and Preliminary Biological Evaluations of 5′-Substituted Derivatives of Uridine as Glycosyltransferase Inhibitors. Molecules 2013, 18, 8018-8027. https://doi.org/10.3390/molecules18078018
Paszkowska J, Kral K, Bieg T, Nawrot U, Szeja W, Wandzik I. Synthesis and Preliminary Biological Evaluations of 5′-Substituted Derivatives of Uridine as Glycosyltransferase Inhibitors. Molecules. 2013; 18(7):8018-8027. https://doi.org/10.3390/molecules18078018
Chicago/Turabian StylePaszkowska, Jadwiga, Katarzyna Kral, Tadeusz Bieg, Urszula Nawrot, Wiesław Szeja, and Ilona Wandzik. 2013. "Synthesis and Preliminary Biological Evaluations of 5′-Substituted Derivatives of Uridine as Glycosyltransferase Inhibitors" Molecules 18, no. 7: 8018-8027. https://doi.org/10.3390/molecules18078018





