Cell Suspension Culture of Eriobotrya japonica Regulates the Diabetic and Hyperlipidemic Signs of High-Fat-Fed Mice
Abstract
:Abbreviations
| AMPK | AMP-activated protein kinase |
| aP2 | adipocyte fatty acid binding protein |
| ATGL | adipose triglyceride lipase |
| BA | 6-benzylaminopurine |
| BAT | brown adipose tissue |
| CON | control |
| CPT-1 | carnitine palmitoyl transferase I |
| DGAT | acyl-coenzyme A: diacylglycerol acyltransferase |
| EWAT | epididymal white adipose tissue |
| FAS | fatty acid synthase |
| FFA | free fatty acid |
| HF | high-fat control |
| HOMA-IR | homeostasis model assessment for insulin resistance |
| MS | Murashige and Skoog |
| MWAT | mesenteric white adipose tissue |
| NAA | α-naphthaleneacetic acid |
| OA | oleanolic acid |
| PPARγ | peroxisome proliferator-activated receptor γ |
| PPARs | peroxisomal proliferator-activated receptors |
| Rosi | rosiglitazone |
| RWAT | retroperitoneal white adipose tissue |
| SREBP-1 | sterol regulatory element binding protein 1 |
| TA | cell suspension culture of Eriobotrya japonica |
| TC | total cholesterol |
| TG | triglyceride |
| WAT | white adipose tissue |
1. Introduction
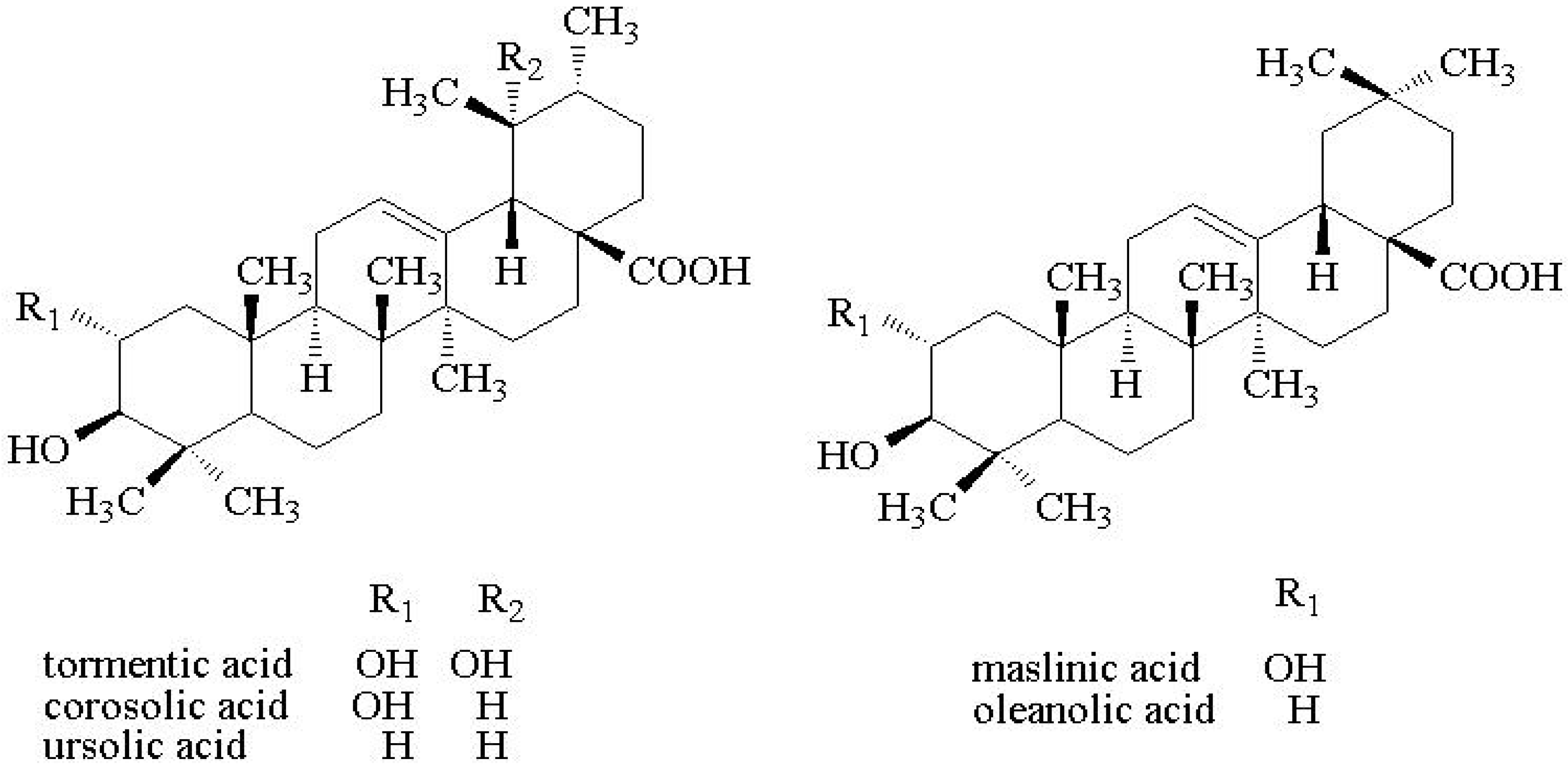
2. Results and Discussion
2.1. Body Weight, Body Weight Gain, Food Intake and Tissue Weight
| Parameter | CON | HF | HF+T1 | HF+T2 | HF+T3 | HF+Rosi |
|---|---|---|---|---|---|---|
| 0.2 a | 0.5 a | 1.0 a | 0.01 a | |||
| Absolute tissue weight (g) | ||||||
| EWAT | 0.513 ± 0.037 | 1.365 ± 0.134 ### | 0.859 ± 0.135 * | 0.838 ± 0.170 * | 0.827 ± 0.138 * | 0.801 ± 0.083 * |
| MWAT | 0.375 ± 0.020 | 0.592 ± 0.041 ### | 0.451 ± 0.041 * | 0.424 ± 0.038 * | 0.438 ± 0.042 * | 0.383 ± 0.031 ** |
| RWAT | 0.142 ± 0.020 | 0.525 ± 0.050 ### | 0.301 ± 0.059 * | 0.304 ± 0.076 * | 0.315 ± 0.043 * | 0.255 ± 0.045 ** |
| Visceral fat | 0.888 ± 0.053 | 1.957 ± 0.173 ### | 1.310 ± 0.180 * | 1.262 ± 0.203 * | 1.231 ± 0.165 ** | 1.184 ± 0.073 ** |
| BAT | 0.081 ± 0.005 | 0.092 ± 0.006 | 0.082 ± 0.007 | 0.079 ± 0.007 | 0.077 ± 0.007 | 0.126 ± 0.006 *** |
| Liver | 0.890 ± 0.032 | 0.928 ± 0.020 | 0.917 ± 0.042 | 0.899 ± 0.040 | 0.875 ± 0.050 | 0.893 ± 0.033 |
| Spleen | 0.085 ± 0.006 | 0.091 ± 0.003 | 0.087 ± 0.003 | 0.078 ± 0.004 | 0.082 ± 0.005 | 0.079 ± 0.002 |
| final bodyweight | 26.56 ± 0.74 | 30.44 ± 1.02# | 28.80 ± 1.08 | 28.10 ± 1.17 | 27.11 ± 1.42 | 28.27 ± 1.25 |
| 4-week cumulative food intake (kcal/mouse) | 293.71 ± 9.94 | 326.50 ± 13.87 | 307.91 ± 7.56 | 306.30 ± 12.32 | 300.58 ± 7.31 | 304.62 ± 7.41 |
| Liver lipids | ||||||
| total lipid (mg/g) | 56.6 ± 2.3 | 92.1 ± 5.6 ### | 78.4 ± 6.1 | 66.22 ± 5.3 ** | 63.1 ± 4.2 ** | 76.3 ± 5.1 |
| Triacylglycerol (μmol/g) | 30.7 ± 3.9 | 74.3 ± 8.2 ### | 63.8 ± 5.9 | 48.5 ± 5.1 *** | 43.6 ± 5.6 *** | 65.6 ± 7.7 |
| Blood profiles | ||||||
| FFA (meq/L) | 1.563 ± 0.083 | 2.262 ± 0.093 # | 2.182 ± 0.213 | 1.800 ± 0.136 | 1.663 ± 0.171 * | 1.558 ± 0.091 * |
| TG (mg/dL) | 92.5 ± 5.4 | 137.9 ± 7.6 # | 126.0 ± 9.2 | 107.3 ± 4.6 * | 95.3 ± 12.6 * | 102.5 ± 7.3 * |
| TC (mg/dL) | 86.4 ± 4.3 | 150.4 ± 2.6 ### | 134.8 ± 7.2 | 124.5 ± 7.3 * | 121.8 ± 7.8 * | 113.3 ± 5.2 *** |
| Leptin (μg/mL) | 1.49 ± 0.29 | 6.35 ± 0.83 ### | 4.69 ± 1.04* | 4.37 ± 0.78 * | 2.70 ± 0.36 ** | 4.36 ± 0.87 * |
| Insulin (μg/L) | 0.579 ± 0.029 | 1.117 ± 0.040 ### | 0.836 ± 0.052 * | 0.821 ± 0.056 ** | 0.706 ± 0.090 *** | 0.660 ± 0.034 *** |
| Adiponectin (ng/mL) | 2.56 ± 0.23 | 1.76 ± 0.35 # | 1.96 ± 0.31 | 2.31 ± 0.49 * | 2.79 ± 0.35 ** | 3.05 ± 0.57 ** |
| HOMA-IR index | 0.81 ± 0.09 | 2.64 ± 0.39 ### | 1.58 ± 0.31 ** | 1.34 ± 0.21 *** | 1.14 ± 0.15 *** | 1.03 ± 0.18 *** |
2.2. Plasma Glucose Levels and Homeostasis Model Assessment for Insulin Resistance (HOMA-IR)

2.3. Plasma Lipids
2.4. Leptin, Adiponectin and Insulin Concentration
2.5. Liver Lipids
2.6. Epididymal WAT Histology

2.7. Expressions of Apo CIII, DGAT1, DGAT2, PEPCK, 11β-HSD1, ATGL, SREBP1c and Carnitine Palmitoyl Transferase I (CPT-1) in Liver Tissue
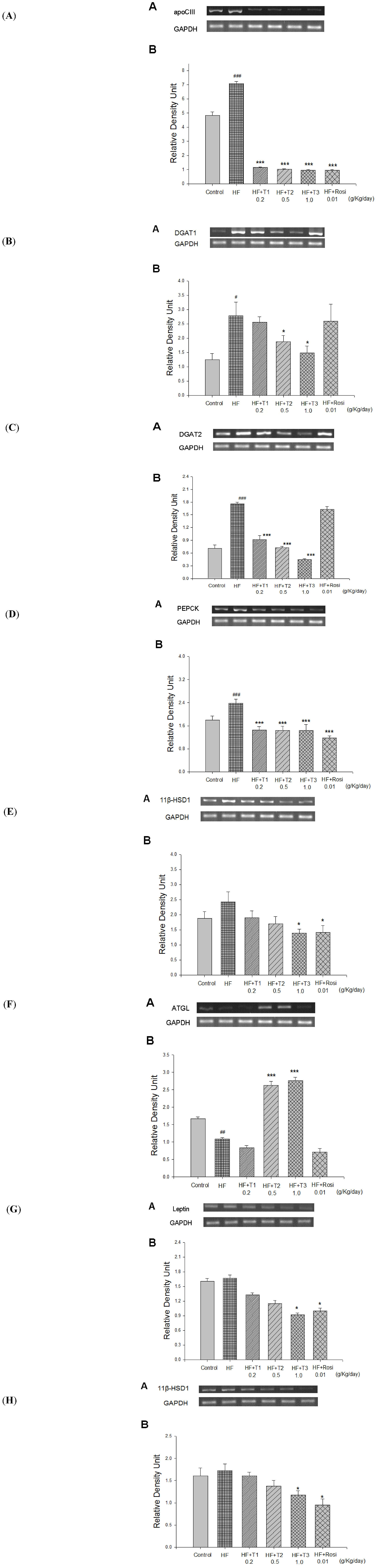
2.8. Expressions of Leptin, 11β-HSD1 and aP2 in White Adipose tissue
2.9. GLUT4 Gene Expression in Skeletal Muscle
| Parameter | CON | HF | HF+T1 | HF+T2 | HF+T3 | HF+Rosi |
|---|---|---|---|---|---|---|
| 0.2 a | 0.5 a | 1.0 a | 0.01 a | |||
| Liver | ||||||
| SREBP-1c | 1.117 ± 0.057 | 1.257 ± 0.096 | 1.186 ± 0.130 | 1.046 ± 0.057 | 1.125 ± 0.062 | 0.945 ± 0.065 |
| CPT1a | 1.438 ± 0.136 | 1.317 ± 0.165 | 1.946 ± 0.169 | 2.052 ± 0.209 * | 2.057 ± 0.152 * | 1.980 ± 0.432 |
| White Adipose tissue | ||||||
| PPARγ | 1.179 ± 0.093 | 1.056 ± 0.052 | 1.078 ± 0.027 | 1.067 ± 0.113 | 1.151 ± 0.142 | 1.410 ± 0.127 * |
| aP2 | 1.117 ± 0.050 | 1.256 ± 0.147 | 0.958 ± 0.073 * | 0.952 ± 0.046 * | 0.798 ± 0.045 *** | 0.984 ± 0.049 |
| Skeletal muscle | ||||||
| Glut4 | 1.441 ± 0.083 | 1.140 ± 0.064 # | 1.188 ± 0.075 | 1.412 ± 0.099 | 1.542 ± 0.173 * | 1.538 ± 0.053 * |
2.10. The Phospho-AMPK (Thr172) Protein Contents in White Adipose and Liver Tissue

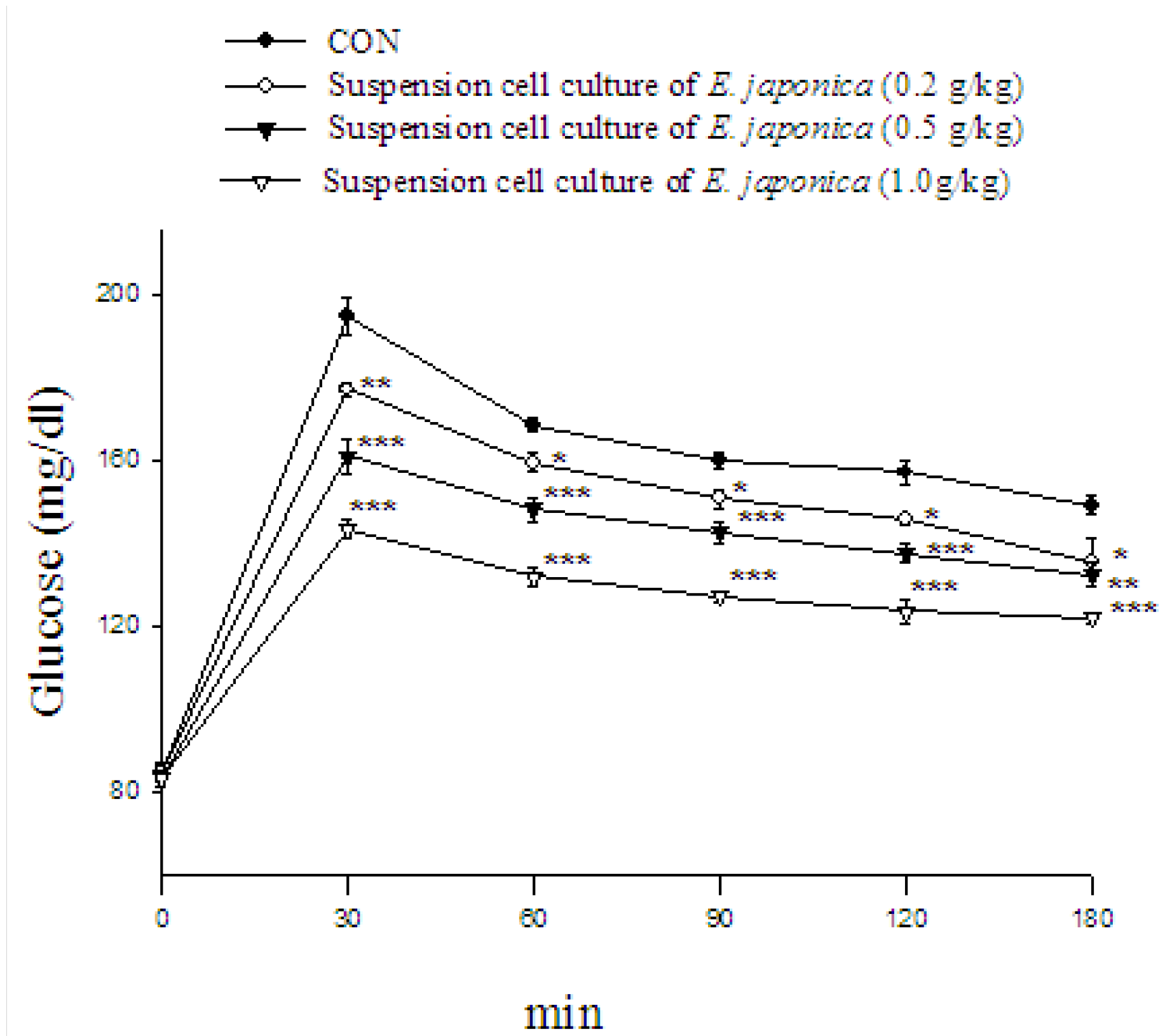
2.11. Oral Glucose Tolerance Test
2.12. Discussion
3. Experimental
3.1. Callus Culture Establishment
3.2. Callus Induction from Leaves of E. japonica Lindl and Initiation of Suspension Cultures
3.3. Bioreactor Method
3.4. Determination of Triterpene Content
3.5. Animals and Experimental Design
| Ingredient | Low-fat | High-fat |
|---|---|---|
| Casein | 800 | 800 |
| L-Cystine | 12 | 12 |
| Corn starch | 1,260 | 291 |
| Maltodextrin 10 | 140 | 400 |
| Sucrose | 1,400 | 691 |
| Cellulose, BW200 | 0 | 0 |
| Soybean Oil | 225 | 225 |
| Lard | 180 | 1,598 |
| Mineral Mix S10026 | 0 | 0 |
| Dicalcium carbonate | 0 | 0 |
| Calcium carbonate | 0 | 0 |
| Potassium citrate, 1H2O | 0 | 0 |
| Vitamin Mix V10001 | 40 | 40 |
| Choline bitartrate | 0 | 0 |
| FD&C Yellow Dye #5 | 0 | |
| FD&C Red Dye #40 | 0 | |
| FD&C Blue Dye #1 | ||
| Total | 4,057 | 4,057 |
3.6. Food Intake and Body Weight Assay
3.7. Blood Parameters Assay
3.8. Adipocytokine Levels Assay
3.9. Histology
3.10. Measurement of Hepatic Lipids
3.11. Isolation of RNA and Relative Quantization of mRNA Indicating Gene Expression
| Gene | Accession numbers | Forward primer and reverse primer | PCR product (bp) | Annealing temperature (°C) |
|---|---|---|---|---|
| White adipose tissue | ||||
| PPARγ | NM_013124 | F: CATGCTTGTGAAGGATGCAAG | 190 | 55 |
| R: TTCTGAAACCGACAGTACTGACAT | ||||
| Leptin | NM_008493 | F: GGCATTTTCTTACCTCTGTG | 303 | 55 |
| R: ACTTTGGATGAACCAATCAG | ||||
| aP2 | NM_024406 | F: TCACCTGGAAGACAGCTCCT | 143 | 50 |
| R: TGCCTGCCACTTTCCTTGT | ||||
| SREBP1c | NM_011480 | F: GGCTGTTGTCTACCATAAG | 219 | 55 |
| R: AGGAAGAAACGTGTCAAGAA | ||||
| FAS | NM_007988 | F: TGGAAAGATAACTGGGTGAC | 240 | 55 |
| R: TGCTGTCGTCTGTAGTCTTG | ||||
| Liver | ||||
| apo C-III | NM_023114.3 | F: CAGTTTTATCCCTAGAAGCA | 349 | 47 |
| R: TCTCACGACTCAATAGCTG | ||||
| CPT-1 | NM_153679 | F: GCAGGAAATTTACCTCTGTG | 288 | 55 |
| R: ACATGAAGGGTGAAGATGAG | ||||
| DGAT1 | NM_010046.2 | F: ATCTTTGCTCCTACTTTGTGTT | 333 | 50 |
| R: ATTCCACCAATCTCTGTAGAAC | ||||
| DGAT2 | NM_026384.3 | F: AGTGGCAATGCTATCATCATCGT | 149 | 50 |
| R: AAGGAATAAGTGGGAACCAGATCA | ||||
| 11β-HSD1 | NM_008288.2 | F:AAGCAGAGCAATGGCAGCAT | 300 | 50 |
| R: GAGCAATCATAGGCTGGGTCA | ||||
| PPARα | NM_011144 | F: ACCTCTGTTCATGTCAGACC | 352 | 55 |
| R: ATAACCACAGACCAACCAAG | ||||
| FAS | NM_007988 | F: TGGAAAGATAACTGGGTGAC | 240 | 50 |
| R: TGCTGTCGTCTGTAGTCTTG | ||||
| SREBP1c | NM_011480 | F: GGCTGTTGTCTACCATAAGC | 219 | 50 |
| R: AGGAAGAAACGTGTCAAGAA | ||||
| PEPCK | NM_011044.2 | F: CTACAACTTCGGCAAATACC | 330 | 52 |
| R: TCCAGATACCTGTCGATCTC | ||||
| Skeletal muscle | ||||
| Glut4 | M25482 | F: ACTGGCGCTTTCACTGAACT | 106 | 55 |
| R: CGAGGCAAGGCTAGATTTTG | ||||
| GAPDH | NM_031144 | F: TGTGTCCGTCGTGGATCTGA | 99 | 55 |
| R: CCTGCTTCACCACCTTCTTGA | ||||
3.12. Western Immunoblotting Analysis of Phospho-AMPK (Thr172) Proteins
3.13. Oral Glucose Tolerance test (OGTT)
3.14. Statistical Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- O’Rahilly, S.; Turner, R.C.; Matthews, D.R. Impaired pulsatile secretion of insulin in relatives of patients with non-insulin-dependent diabetes. N. Engl. J. Med. 1988, 318, 1225–1230. [Google Scholar] [CrossRef]
- Samuel, V.T.; Liu, Z.X.; Qu, X.; Elder, B.D.; Bilz, S.; Befroy, D.; Romanelli, A.J.; Shulman, G.I. Mechanism of hepatic insulin resistance in non-alcoholic fatty liver disease. J. Biol. Chem. 2004, 279, 32345–32353. [Google Scholar]
- Noreen, W.; Wadood, A.; Hidayat, H.K.; Wahid, S.A. Effect of Eriobotrya japonica on blood glucose levels of normal and alloxan-diabetic rabbits. Planta Med. 1988, 54, 196–199. [Google Scholar] [CrossRef]
- Tommasi, N.D.; Shimone, F.D.; Pizza, C. Constituents of Eriobotrya japonica: A study of their antiviral properties. J. Nat. Prod. 1992, 55, 1067–1073. [Google Scholar] [CrossRef]
- Louati, S.; Simmonds, M.S.J.; Grayer, R.J.; Kite, G.C.; Damak, M. Flavonoids from Eriobotrya japonica (Rosaceae) growing in Tunisia. Biochem. Syst. Ecol. 2003, 31, 99–101. [Google Scholar] [CrossRef]
- Ding, C.K.; Chachin, K.; Ueda, Y.; Imahori, Y.; Wang, C.Y. Metabolism of phenolic compounds during loquat fruit development. J. Agric. Food Chem. 2001, 49, 2883–2888. [Google Scholar]
- Zhuang, Y.F. Determination of amygdalin content in Eriobotrya japonica leaves by high performance liquid chromatography (HPLC). Strait Pharm. J. 2002, 14, 64–65. [Google Scholar]
- Liang, Z.Z.; Aquino, R.; De Feo, V.; De Simone, F.; Pizza, C. Polyhydroxylated triterpenes from Eriobotrya japonica. Planta Med. 1990, 56, 330–332. [Google Scholar] [CrossRef]
- Godoy, H.T.; Amaya, D.B. Carotenoid composition and vitamin A value of Brazilian loquat (Eriobotrya japonica Lindl.). Arch. Latinoam. Nutr. 1995, 45, 336–339. [Google Scholar]
- Ju, J.H.; Zhou, L.; Lin, G.; Liu, D.; Wang, L.W.; Yang, J.S. Studies on constituents of triterpene acids from Eriobotrya japonica and their anti-inflammatory and antitussive effects. J. Chin. Pharmacol. 2003, 38, 752–757. [Google Scholar]
- Park, H.J.; Yoon, S.H.; Han, L.S.; Zheng, L.T.; Jung, K.H.; Uhm, Y.K.; Lee, J.H.; Jeong, J.S.; Joo, W.S.; Yim, S.Y.; et al. Amygdalin inhibits genes related to cell cycle in SNU-C4 human colon cancer cells. World J. Gastroenterol. 2005, 11, 5156–5161. [Google Scholar]
- Wen, X.; Sun, H.; Liu, J.; Wu, G.; Zhang, L.; Wu, X.; Ni, P. Pentacyclic triterpenes. Part 1: The first examples of naturally occurring pentacyclic triterpenes as a new class of inhibitors of glycogen phosphorylases. Bioorg. Med. Chem. Lett. 2005, 15, 4944–4948. [Google Scholar] [CrossRef]
- Wen, X.; Xia, J.; Cheng, K.; Zhang, L.; Zhang, P.; Liu, J.; Zhang, L.; Ni, P.; Sun, H. Pentacyclic triterpenes. Part 5: Synthesis and SAR study of corosolic acid derivatives as inhibitors of glycogen phosphorylases. Bioorg. Med. Chem. Lett. 2007, 17, 5777–5782. [Google Scholar]
- Liu, J. Oleanolic acid and ursolic acid: Research perspectives. J. Ethnopharmacol. 2005, 100, 92–94. [Google Scholar] [CrossRef]
- Ivorra, M.D.; Paya, M.; Villar, A. Hypoglycemic and insulin release effects of tormentic acid: A new hypoglycemic natural product. Planta Med. 1988, 54, 282–285. [Google Scholar] [CrossRef]
- Zhang, Q.; Chang, Z.; Wang, Q. Ursane triterpenoids inhibit atherosclerosis and xanthoma in LDL receptor knockout mice. Cardiovasc. Drugs Ther. 2006, 20, 349–357. [Google Scholar] [CrossRef]
- Miura, T.; Itoh, Y.; Kaneko, T.; Ueda, N.; Ishida, T.; Fukushima, M.; Matsuyama, F.; Seino, Y. Corosolic acid induces GLUT4 translocation in genetically type 2 diabetic mice. Biol. Pharm. Bull. 2004, 27, 1103–1105. [Google Scholar] [CrossRef]
- Yamada, K.; Hosokawa, M.; Yamada, C.; Watanabe, R.; Fujimoto, S.; Fujiwara, H.; Kunitomo, M.; Miura, T.; Kaneko, T.; Tsuda, K.; et al. Dietary corosolic acid ameliorates obesity and hepatic steatosis in KK-Ay mice. Biol. Pharm. Bull. 2008, 31, 651–655. [Google Scholar] [CrossRef]
- Wen, X.A.; Zhang, P.; Liu, J. Pentacyclic triterpenes. Part 2: Synthesis and biological evaluation of maslinic acid derivatives as glycogen phosphorylase inhibitors. Bioorg. Med. Chem. Lett. 2006, 16, 722–726. [Google Scholar] [CrossRef]
- Jang, S.M.; Yee, S.T.; Choi, J. Ursolic acid enhances the cellular immune system and pancreatic β-cell function in streptozotocin-induced diabetic mice fed a high-fat diet. Int. Pharmacol. 2009, 9, 113–119. [Google Scholar]
- Somova, L.O.; Ndar, A.; Rammanan, P.; Shode, F.O. Cardiovascular, antihyperlipidemic and oxidant effects of oleanolic and ursolic acids in experimental hypertension. Phytomedicine 2003, 10, 115–121. [Google Scholar] [CrossRef]
- Jayaprakasam, B.; Olson, L.K.; Schutzki, R.E.; Tai, M.H.; Nair, M.G. Amelioration of obesity and glucose intolerance in high-fat-fed C57BL/6 mice by anthocyanins and ursolic acid in Cornelian cherry (Cornus mas). J. Agric. Food Chem. 2006, 54, 243–248. [Google Scholar] [CrossRef]
- Jia, Y.; Bhuiyan, M.J.H.; Jun, H.; Lee, J.H.; Hoang, M.H.; Lee, H.-J.; Kim, N.; Lee, D.; Hwang, K.Y.; Hwang, B.Y.; et al. Ursolic acid is a PPAR-α agonist that regulates hepatic lipid metabolism. Bioorg. Med. Chem. Lett. 2011, 21, 5876–5880. [Google Scholar]
- Taniguchi, S.; Imayoshi, Y.; Kobayashi, E.; Takamatsu, Y.; Ito, H.; Hatano, T.; Sakagami, H.; Tokuda, H.; Nishino, H.; Sugita, D.; Shimura, S.; Yoshida, T. Production of bioactive triterpenes by Eriobotrya japonica calli. Photochemistry 2002, 59, 315–323. [Google Scholar]
- Liu, C.Z.; Wang, Y.C.; Ouyang, F.; Ye, H.C.; Li, G.F.; Guo, Z.C. Research advance in plant tissue culture using bioreactor technology. Eng. Chem. Metall. 1999, 20, 329–336. [Google Scholar]
- Shakti, M.; Manoj, K.G.; Arun, K.K.; Bhartendu, N.M. Efficiency of liquid culture systems over conventional micropropagation: A progress towards commercialization. Afr. J. Biotechnol. 2007, 6, 1484–1492. [Google Scholar]
- Kim, H.J.; Kim, S.K.; Shim, W.S.; Lee, J.H.; Hur, K.Y.; Kang, E.S.; Ahn, C.W.; Lim, S.K.; Lee, H.C.; Cha, B.S. Rosiglitazone improves insulin sensitivity with increased serum leptin levels in patients with type 2 diabetes mellitus. Diabetes Res. Clin. Pract. 2008, 81, 42–49. [Google Scholar] [CrossRef]
- Yuen, V.G.; Bhanot, S.; Battell, M.L.; Orvig, C.; McNeill, J.H. Chronic glucose-lowering effects of rosiglitazone and bis (ethylmaltolato) oxovanadium (IV) in ZDF rats. Can. J. Physiol. Pharmacol. 2003, 81, 1049–1055. [Google Scholar] [CrossRef]
- Harrity, T.; Farrelly, D.; Tieman, A.; Chu, C.; Kunselman, L.; Hariharan, N. Muraglitazar, a novel dual (α/γ) peroxisome proliferator activated receptor activator, improves diabetes and other metabolic abnormalities and preserves β-cell function in db / db mice. Diabetes 2006, 55, 240–248. [Google Scholar] [CrossRef]
- Rangwala, S.M.; Lazar, M.A. Peroxisome proliferator-activated receptor gamma in diabetes and metabolism. PPAR gamma, Glucose homeostasis. Trends Pharmacol. Sci. 2004, 25, 331–336. [Google Scholar] [CrossRef]
- Yu, S.; Matsusue, K.; Kashireddy, P.; Cao, W.Q.; Yeldandi, V.; Reddy, J.K. Adipocyte-specific gene expression and adipogenic steatosis in the mice liver due to peroxisome proliferator-activated receptor gamma1 (PPARgamma1) overexpression. J. Biol. Chem. 2003, 278, N498–N505. [Google Scholar]
- Karalliedde, J.; Buckingham, R.E. Thiazolidinediones and their fluid-related adverse effects: Facts, Fiction and putative management strategies. Drug Safety 2007, 30, 741–53. [Google Scholar] [CrossRef]
- Cases, S.; Smith, S.J.; Zheng, Y.W.; Myers, H.M.; Lear, S.R.; Sande, E.; Novak, S.; Colin, C.; Welch, C.B.; Lusis, A.J.; et al. Identification of a gene encoding an acyl CoA: Diacylglycerol acyltransferase, a key enzyme in triacylglycerol synthesis. Proc. Natl. Acad. Sci. USA 1998, 95, 13018–13023. [Google Scholar]
- Oelkers, P.; Behari, A.; Cromley, D.; Billheimer, J.T.; Sturley, S.L. Characterization of two human genes encoding acyl coenzyme A: Cholesterol acyltransferase-related enzymes. J. Biol. Chem. 1998, 273, 26765–26771. [Google Scholar]
- Cases, S.; Stone, S.J.; Zhou, P.; Yen, E.; Tow, B.; Lardizabal, K.D.; Voelker, T.; Farese Jr, R.V. Cloning of DGAT2, a second mammalian diacylglycerol acyltransferase, and related family members. J. Biol. Chem. 2001, 276, 38870–38876. [Google Scholar]
- Smith, S.J.; Cases, S.; Jensen, D.R.; Chen, H.C.; Sande, E.; Tow, B.; Sanan, D.A.; Raber, J.; Eckel, R.H.; Farese, R.V., Jr. Obesity resistance and multiple mechanisms of triglyceride synthesis in mice lacking DGAT. Nat. Genet. 2000, 25, 87–90. [Google Scholar]
- Chen, H.C.; Jensen, D.R.; Myers, H.M.; Eckel, R.H.; Farese, R.V., Jr. Obesity resistance and enhanced glucose metabolism in mice transplanted with white adipose tissue lacking acyl CoA:diacylglycerol acyltransferase 1. J. Clin. Invest. 2003, 111, 1715–1722. [Google Scholar]
- Zhao, G.; Souers, A.J.; Voorbach, M.; Falls, H.D.; Droz, B.; Brodjian, S.; Lau, Y.Y.; Iyengar, RR.; Gao, J.; Judd, A.S. Validation of diacyl glycerolacyltransferase I as a novel target for the treatment of obesity and dyslipidemia using a potent and selective small molecule inhibitor. J. Med. Chem. 2008, 51, 380–383. [Google Scholar] [CrossRef]
- Birch, A.M.; Birtles, S.; Buckett, L.K.; Kemmitt, P.D.; Smith, G.J.; Smith, T.J.D.; Turnbull, A.V.; Wang, S.J.Y. Discovery of a potent, selective, and orally efficacious pyrimidinooxazinyl bicyclo-octaneacetic acid diacylglycerol acyltransferase-1 inhibitor. J. Med. Chem. 2009, 52, 1558–1568. [Google Scholar]
- Yamamoto, T.; Yamaguchi, H.; Miki, H.; Shimada, M.; Nakada, Y.; Ogino, M.; Asano, K.; Aoki, K.; Tamura, N.; Masago, M.; et al. Coenzyme A:diacylglycerol acyltransferase 1 inhibitor ameliorates obesity, liver steatosis, and lipid metabolism abnormality in KKAy mice fed high-fat or high-carbohydrate diets. Eur. J. Pharmacol. 2010, 640, 243–249. [Google Scholar] [CrossRef]
- King, A.J.; Segreti, J.A.; Larson, K.J.; Souers, A.J.; Kym, P.R.; Reilly, R.M.; Zhao, G.; Mittelstadt, S.W.; Cox, B.F. Diacylglycerol acyltransferase inhibition lowers serum triglycerides in the Zucker Fatty rat and the hyperlipidemic hamster. J. Pharmacol. Exp. Ther. 2009, 330, 526–531. [Google Scholar] [CrossRef]
- Foretz, M.; Taleux, N.; Guigas, B.; Horman, S.; Beauloye, C.; Andreelli, F.; Viollet, B. Regulation of energy metabolism by AMPK: a novel therapeutic approach for the treatment of metabolic and cardiovascular diseases. Med. Sci. (Paris) 2006, 22, 381–383. [Google Scholar] [CrossRef]
- Viollet, B.; Lantier, L.; Devin-Leclerc, J.; Hebrard, S.; Amouyal, C.; Mounier, R.; Foretz, M.; Andreelli, F. Targeting the AMPK pathway for the treatment of Type 2 diabetes. Front. Biosci. 2009, 14, 3380–3400. [Google Scholar]
- Zhou, G.; Myers, R.; Li, Y.; Chen, Y.; Shen, X.; Fenyk-Melody, J.; Wu, M.; Ventre, J.; Doebber, T.; Fujii, N.; et al. Role of AMP-activated protein kinase in mechanism of metformin action. J. Clin. Invest. 2001, 108, 1167–1174. [Google Scholar]
- Stein, S.C.; Woods, A.; Jones, N.A.; Davison, M.D.; Carling, D. The regulation of AMP-activated protein kinase by phosphorylation. Biochem. J. 2000, 345, 437–443. [Google Scholar] [CrossRef]
- Kato, M.; Higuchi, N.; Enjoji, M. Reduced hepatic expression of adipose tissue triglyceride lipase and CGI-58 may contribute to the development of non-alcoholic fatty liver disease inpatients with insulin resistance. Scand. J. Gastroenterol. 2008, 43, 1018–1019. [Google Scholar] [CrossRef]
- Zimmermann, R.; Strauss, J.G.; Haemmerle, G.; Schoiswohl, G.; Birner-Gruenberger, R.; Riederer, M.; Lass, A.; Neuberger, G.; Eisenhaber, F.; Hermetter, A.; et al. Fat mobilization in adipose tissue is promoted by adipose triglyceride lipase. Science 2004, 306, 1383–1386. [Google Scholar]
- Shih, C.C.; Lin, C.H.; Wu, J.B. Eriobotrya japonica improves hyperlipidaemia and reverses insulin resistance in high-fat-fed mice. Phytother. Res. 2010, 24, 1769–1780. [Google Scholar] [CrossRef]
- Surwit, R.S.; Kuhn, C.M.; Cochrane, C.; McCubbin, J.A.; Feinglos, M.N. Diet-induced type II diabetes in C57BL/6J mice. Diabetes 1988, 37, 1163–1167. [Google Scholar]
- Ahren, B.; Pacini, G. Importance of quantifying insulin secretion in relation to insulin sensitivity to accurately assess beta cell function in clinical studies. Eur. J. Endocrinol. 2005, 15, 97–104. [Google Scholar]
- Gil-Campos, M.; Canete, R.; Gil, A. Adiponectin, the missing link in insulin resistance and obesity. Clin. Nutr. 2004, 23, 963–974. [Google Scholar] [CrossRef]
- Giorgino, F.; Laviola, L.; Eriksson, J.W. Regional differences of insulin action in adipose tissue: Insights from in vivo and in vitro studies. Acta Physiol. Scand. 2005, 183, 13–30. [Google Scholar] [CrossRef]
- Pan, Y.; Zheng, J.M.; Zhao, H.Y.; Li, Y.J.; Xu, H.; Wei, G. Relationship between drug effects and particle size of insulin-loaded bioadhesive microsperes. Acta Pharmacol. Sin. 2002, 23, 1051–1056. [Google Scholar]
- Koeilevtsev, Y.; Holmes, M.C.; Burchell, A.; Houston, P.M.; Schmoll, D.; Jamieson, P.; Best, R.; Brown, R.; Edwards, C.R.W.; Seckl, J.R.; et al. 11β-hydroxysteroid dehydrogenase type 1 knockout mice show attenuated glucocorticoid-inducible responses and resist hyperglycemia on obesity or stress. Proc. Natl. Acad. Sci. USA 1997, 94, 14924–14929. [Google Scholar]
- Alberts, P.; Nilsson, C.; Selen, G.; Engblom, L.O.M.; Edling, N.M.H; Norling, S.;Klingström, G.; Larsson, C.; Forsgren, M.; Ashkzari, M.; et al. Selective inhibition of 11β-hydroxysteroid dehydrogenase type 1 improves hepatic insulin sensitivity in hyperglycemic mice strains. Endocrinology 2003, 144, 4755–4762. [Google Scholar] [CrossRef]
- Alberti, L.; Girola, A.; Gilardini, L.; Conti, A.; Cattaldo, S.; Micheletto, G.; Invitti, C. Type 2 diabetes and metabolic syndrome are associated with increased expression of 11beta hydroxysteroid dehydrogenase 1 in obese subjects. Int. J. Obes. (Lond) 2007, 31, 1826–1831. [Google Scholar] [CrossRef]
- Kissebah, A.H. Central obesity: Measurement and metabolic effects. Diabetes Rev. 1997, 5, 8–20. [Google Scholar]
- Gaidhu, M.P.; Fediuc, S.; Anthony, N.M.; So, M.; Mirpourian, M.; Perry, R.L.; Ceddia, R.B. Prolonged AICAR-induced AMP-kinase activation promotes energy dissipation in white adipocytes: novel mechanisms integrating HSL and ATGL. J. Lipid Res. 2009, 50, 704–715. [Google Scholar]
- McGarry, J.D.; Brown, N.F. The mitochondrial carnitine palmitoyl-transferase system from concept to molecular analysis. Eur. J. Biochem. 1997, 244, 1–14. [Google Scholar]
- Kallen, C.B.; Lazar, M.A. Antidiabetic thiazolidinediones inhibit leptin (ob) gene expression in 3T3-L1 adipocytes. Proc. Natl. Acad. Sci. USA 1996, 93, 5793–5796. [Google Scholar] [CrossRef]
- Considine, R.V.; Sinha, M.K.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Nyce, M.R. Serum immunoreactive-leptin concentrations in normal-weight and obese humans. N. Engl. J. Med. 1996, 334, 292–295. [Google Scholar] [CrossRef]
- Wu, X.; Motoshima, H.; Mahadev, K.; Stalker, T.J.; Scalia, R.; Goldstein, B.J. Involvement of AMP-activated protein kinase in glucose uptake stimulated by the globular domain of adiponectin in primary rat adipocytes. Diabetes 2003, 52, 1355–1363. [Google Scholar] [CrossRef]
- Yamauchi, T.; Kamon, J.; Minokoshi, Y.; Ito, Y.; Waki, H.; Uchida, S.; Yamashita, S.; Noda, M.; Kita, S.; Ueki, K.; et al. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat. Med. 2002, 8, 1288–1295. [Google Scholar] [CrossRef]
- Minokoshi, Y.; Kim, Y.-B.; Peroni, O.D.; Fryer, L.G.D.; Müller, C.; Carling, D.; Kahn, B.B. Leptin stimulates fatty-acid oxidation by activating AMP-activated protein. Nature 2002, 415, 339–343. [Google Scholar]
- Hotamisligil, G.S.; Johnson, R.S,.; Distel, R.J.; Ellis, R.; Papaioannou, V.E.; Spiegelman, B.M. Uncoupling of obesity from insulin resistance through a targeted mutation in aP2, the adipocyte fatty acid binding protein. Science 1996, 274, 1377–1379. [Google Scholar] [CrossRef]
- Uysal, K.T.; Scheja, L.; Wiesbrock, S.M.; Bonner-Weir, S.; Hotamisligil, G.S. Improved glucose and lipid metabolism in genetically obese mice lacking aP2. Endocrinology 2000, 141, 3388–3396. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassaya with tobacco tissue cultures. Physiol. Plant 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Ho, H.A.; Lin, W.C.; Kitanaka, S.; Chang, C.T.; Wu, J.B. Analysis of bioactive triterpenes in Eriobotrya japonica Lindl. by high-performance liquid chromatography. J. Food Drug Anal. 2008, 16, 41–45. [Google Scholar]
- Ho, H.Y.; Ling, K.Y.; Lin, W.C.; Kitanaka, S.; Wu, J.B. Regulation and improvement of triterpene formation in plant cultured cells of Eriobotrya japonica Lindl. J. Biosci. Bioeng. 2010, 11, 588–592. [Google Scholar]
- Folch, J.; Lees, M.; Sloan-Stanley, G.H. A sample method for isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- Shen, Q.W.; Jones, C.S.; Kalchayanand, N.; Zhu, M.J.; Du, M. Effect of dietary alpha-lipoic acid on growth, Body composition, Muscle pH, and AMP-activated protein kinase phosphorylation in mice. J. Anim. Sci. 2005, 83, 2611–2617. [Google Scholar]
- Laemmli, U.K. Cleavage of structure proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds Cell Suspension Culture of Eriobotrya japonica are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shih, C.-C.; Ciou, J.-L.; Lin, C.-H.; Wu, J.-B.; Ho, H.-Y. Cell Suspension Culture of Eriobotrya japonica Regulates the Diabetic and Hyperlipidemic Signs of High-Fat-Fed Mice. Molecules 2013, 18, 2726-2753. https://doi.org/10.3390/molecules18032726
Shih C-C, Ciou J-L, Lin C-H, Wu J-B, Ho H-Y. Cell Suspension Culture of Eriobotrya japonica Regulates the Diabetic and Hyperlipidemic Signs of High-Fat-Fed Mice. Molecules. 2013; 18(3):2726-2753. https://doi.org/10.3390/molecules18032726
Chicago/Turabian StyleShih, Chun-Ching, Jiun-Lin Ciou, Cheng-Hsiu Lin, Jin-Bin Wu, and Hui-Ya Ho. 2013. "Cell Suspension Culture of Eriobotrya japonica Regulates the Diabetic and Hyperlipidemic Signs of High-Fat-Fed Mice" Molecules 18, no. 3: 2726-2753. https://doi.org/10.3390/molecules18032726





