Bioprospecting of Marine Invertebrates for New Natural Products — A Chemical and Zoogeographical Perspective
Abstract
:1. Introduction
2. Results
2.1. Chemical Trends
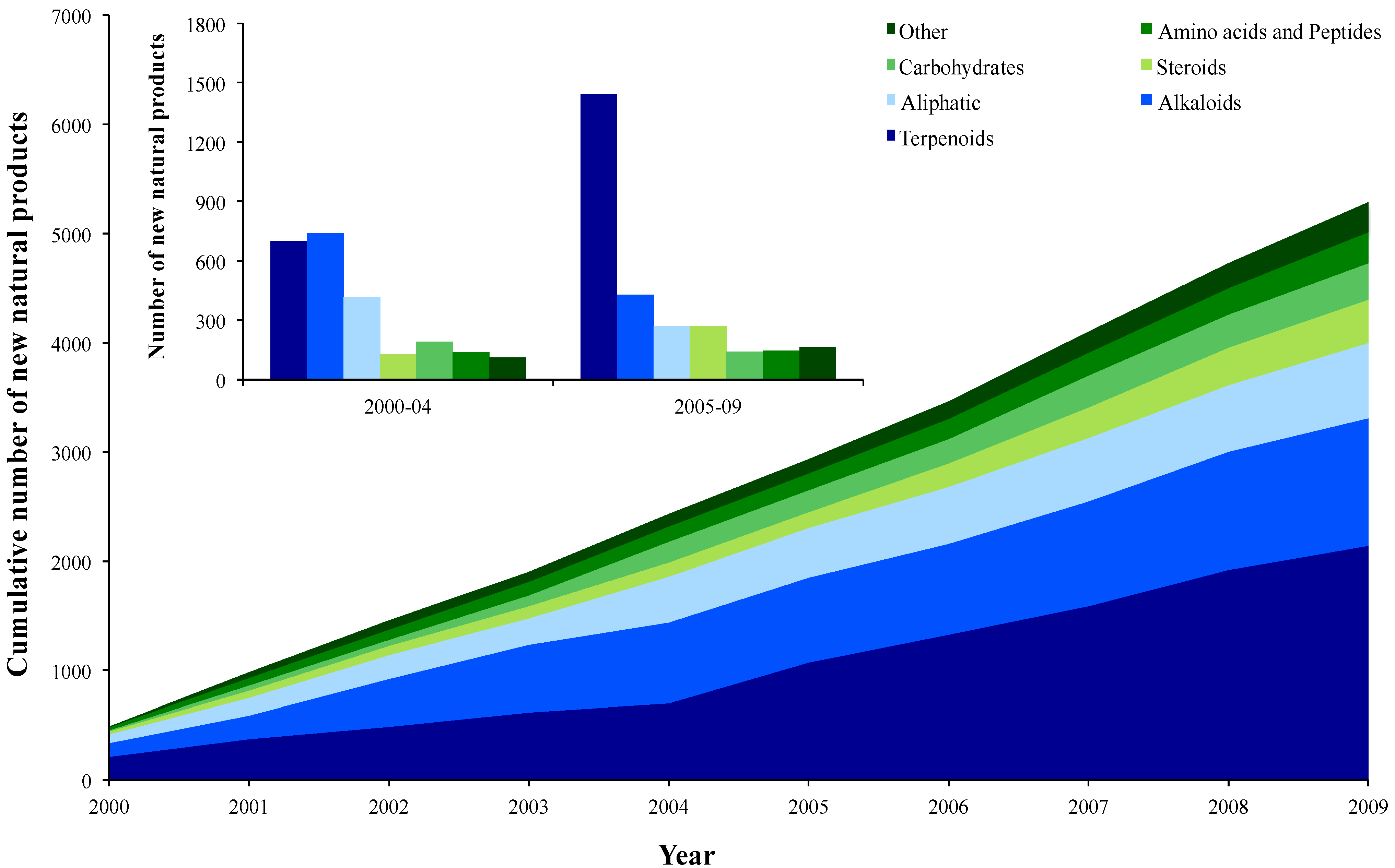
2.2. Taxonomical Trends
| Phyla | Terpenoids | Alkaloids | Aliphatic | Steroids | Carbohydrates | Amino acids and |
|---|---|---|---|---|---|---|
| (%) | (%) | (%) | (%) | (%) | peptides (%) | |
| Porifera | 30.0 | 30.9 | 12.0 | 6.5 | 4.9 | 8.1 |
| Cnidaria | 65.9 | 10.1 | 7.7 | 8.9 | 6.1 | 0.4 |
| Echinodermata | 10.5 | 10.7 | 35.8 | 14.3 | 20.7 | 0.8 |
| Chordata | 23.0 | 39.4 | 16.1 | 2.8 | 4.4 | 7.9 |
| Mollusca | 42.6 | 12.5 | 19.0 | 1.1 | 3.0 | 17.1 |
2.3. Zoogeographical Trends
| Hemisphere | Region | Terpenoids | Alkaloids | Aliphatic | Steroids | Carbohydrates | Amino acids and peptides | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2000–2004 | 2005–2009 | 2000–2004 | 2005–2009 | 2000–2004 | 2005–2009 | 2000–2004 | 2005–2009 | 2000–2004 | 2005–2009 | 2000–2004 | 2005–2009 | ||
| North | Polar | 0 | 0 | 2 | 3 | 4 | 0 | 0 | 0 | 0 | 0 | 1 | 0 |
| Temperate | 222 | 281 | 251 | 121 | 179 | 136 | 55 | 52 | 55 | 56 | 18 | 42 | |
| Tropical | 176 | 395 | 200 | 111 | 38 | 63 | 32 | 86 | 55 | 67 | 53 | 41 | |
| South | Tropical | 202 | 643 | 216 | 147 | 111 | 65 | 35 | 94 | 49 | 10 | 42 | 50 |
| Temperate | 36 | 42 | 48 | 31 | 35 | 1 | 4 | 14 | 5 | 2 | 10 | 6 | |
| Polar | 25 | 24 | 5 | 8 | 10 | 0 | 0 | 10 | 7 | 5 | 2 | 0 | |
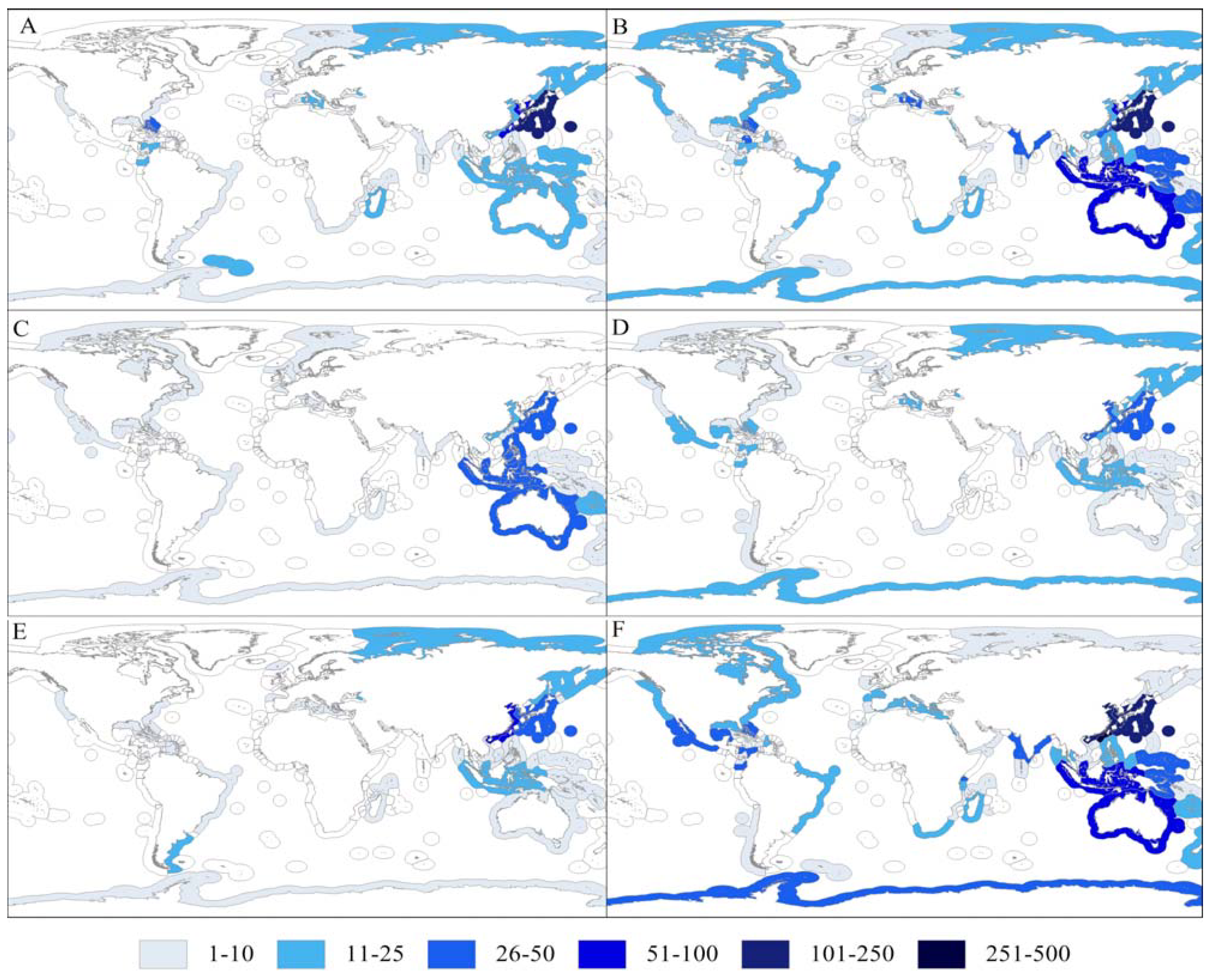
| Exclusive Economic Zone | Terpenoids (%) | Alkaloids (%) | Aliphatic (%) | Steroids (%) | Carbohydrates (%) | Amino acids and peptides (%) | Total |
|---|---|---|---|---|---|---|---|
| Antarctica | 42.5 | 13.8 | 12.5 | 12.5 | 15.0 | 2.5 | 80 |
| Australia | 37.5 | 33.5 | 6.0 | 2.8 | 3.6 | 10.9 | 248 |
| Bahamas | 22.4 | 28.0 | 28.0 | 3.2 | 10.4 | 1.6 | 125 |
| China | 63.3 | 3.8 | 3.3 | 14.9 | 8.7 | 3.3 | 390 |
| Colombia | 46.0 | 18.4 | 19.5 | 0.0 | 16.1 | 0.0 | 87 |
| Fiji | 16.7 | 36.7 | 23.3 | 0.0 | 0.0 | 16.7 | 60 |
| India | 47.7 | 34.9 | 3.5 | 0.0 | 7.0 | 3.5 | 86 |
| Indonesia | 33.9 | 31.8 | 6.4 | 6.4 | 4.2 | 10.2 | 283 |
| Italy | 17.5 | 33.8 | 16.3 | 5.0 | 13.8 | 3.8 | 80 |
| Japan | 33.6 | 25.7 | 20.5 | 3.6 | 4.7 | 5.4 | 717 |
| Madagascar | 21.1 | 36.8 | 21.1 | 5.3 | 0.0 | 7.0 | 57 |
| Mexico | 59.5 | 3.8 | 0.0 | 0.0 | 30.4 | 6.3 | 79 |
| Micronesia | 32.4 | 29.5 | 18.1 | 3.8 | 4.8 | 7.6 | 105 |
| New Zealand | 29.7 | 37.5 | 12.5 | 0.0 | 3.1 | 6.3 | 64 |
| Palau | 21.3 | 31.3 | 0.0 | 10.0 | 3.8 | 10.0 | 80 |
| Papua New Guinea | 39.5 | 31.5 | 12.9 | 1.6 | 3.2 | 6.5 | 124 |
| Philippines | 24.2 | 18.2 | 4.0 | 9.1 | 7.1 | 26.3 | 99 |
| Russia | 7.1 | 14.3 | 21.4 | 20.2 | 26.2 | 0.0 | 84 |
| South Korea | 34.9 | 17.4 | 28.3 | 11.1 | 6.9 | 0.0 | 350 |
| Taiwan | 69.6 | 6.0 | 8.3 | 11.7 | 2.6 | 0.1 | 684 |
| United States | 33.3 | 30.2 | 15.9 | 9.5 | 1.6 | 1.6 | 63 |
| Vanuatu | 18.6 | 48.8 | 4.7 | 8.1 | 1.2 | 12.8 | 86 |
3. Discussion
3.1. Chemical Trends
3.2. Taxonomical Trends
3.3. Geographical Trends
4. Experimental
Methods
5. Conclusions
Acknowledgments
References
- Blunt, J.W.; Copp, B.R.; Munro, M.H.G.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2010, 27, 165–237. [Google Scholar] [CrossRef]
- Hu, G.-P.; Yuan, J.; Sun, L.; She, Z.-G.; Wu, J.-H.; Lan, X.-J.; Zhu, X.; Lin, Y.-C.; Chen, S.-P. Statistical research on marine natural products based on data obtained between 1985 and 2008. Mar. Drugs 2011, 9, 514–525. [Google Scholar] [CrossRef]
- Riguera, R. Isolating bioactive compounds from maine organisms. J. Mar. Biotechnol. 1997, 5, 187–193. [Google Scholar]
- Costantino, V.; Fattorusso, E.; Menna, M. Chemical diversity of bioactive marine natural products: An illustrative case study. Curr. Med. Chem. 2004, 11, 1671–1692. [Google Scholar]
- Munro, M.H.G.; Ludibrand, R.T.; Blunt, J.W. The Research for Antiviral and Anticancer Compounds from Marine Organisms. In Bioorganic Marine Chemistry; Scheuer, P., Ed.; Springer-Verlag: Berlin, Germany, 1987; Volume 1, pp. 93–176. [Google Scholar]
- Munro, M.H.G.; Blunt, J.W.; Dumdei, E.; Hickford, S.; Lill, R.; Li, S.; Battershill, C.; Duckworth, A. The discovery and development of marine compounds with pharmaceutical potential. J. Biotechnol. 1999, 70, 15–25. [Google Scholar]
- Blunt, J.W.; Munro, M.H.G.; Laatsch, H. AntiMarin Database; University of Canterbury: Christchurch, New Zealand, 2006. [Google Scholar]
- Department of Chemistry, University of Canterbury. MarinLit database. Available online: http://www.chem.canterbury.ac.nz/marinlit/marinlit.shtml (accessed on 1 May 2012).
- Jha, R.K.; Xu, Z. Biomedical compounds from marine organisms. Mar. Drugs 2004, 2, 123–146. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Hu, W.-P.; Munro, M.H.G.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2008, 25, 35–94. [Google Scholar] [CrossRef]
- Avila, C.; Taboada, S.; Núñez-Pons, L. Antarctic marine chemical ecology: What is next? Mar. Ecol. 2008, 29, 1–71. [Google Scholar]
- McClintock, J.; Baker, B.J. Marine Chemical Ecology; Taylor & Francis Group: Boca Raton, FL, USA, 2001. [Google Scholar]
- Paul, V.J.; Arthur, K.E.; Ritson-Williams, R.; Ross, C.; Sharp, K. Chemical defenses: From compounds to communities. Biol. Bull. 2007, 213, 226–251. [Google Scholar] [CrossRef]
- Paul, V.J. Ecological Roles of Marine Natural Products; Comstock Publications Association: Ithaca, NY, USA, 1992. [Google Scholar]
- Leal, M.C.; Puga, J.; Serôdio, J.; Gomes, N.C.M.; Calado, R. Trends in the discovery of new marine natural products from invertebrates over the last two decades—Where and what are we bioprospecting? PLoS One 2012, 7, e30580. [Google Scholar]
- Molinski, T.F.; Dalisay, D.S.; Lievens, S.L.; Saludes, J.P. Drug development from marine natural products. Nat. Rev. Drug Discov. 2009, 8, 69–85. [Google Scholar]
- Shirsath, S.; Sonawane, S.; Gogate, P. Intensification of extraction of natural products using ultrasonic irradiations—A review of current status. Chem. Eng. Process. 2012, 53, 10–23. [Google Scholar] [CrossRef]
- Rocha, J.; Peixe, L.; Gomes, N.C.M.; Calado, R. Cnidarians as a source of new marine bioactive compounds—An overview of the last decade and future steps for bioprospecting. Mar. Drugs 2011, 9, 1860–1886. [Google Scholar] [CrossRef]
- Paul, V.J.; Ritson-Williams, R.; Sharp, K. Marine chemical ecology in benthic environments. Nat. Prod. Rep. 2011, 28, 345–388. [Google Scholar] [CrossRef]
- Ianora, A.; Boersma, M.; Casotti, R.; Fontana, A.; Harder, J.; Hoffmann, F.; Pavia, H.; Potin, P.; Poulet, S.A.; Toth, G. New trends in marine chemical ecology. Estuar. Coast. 2006, 29, 531–551. [Google Scholar]
- Skropeta, D. Deep-sea natural products. Nat. Prod. Rep. 2008, 25, 1131–1166. [Google Scholar] [CrossRef]
- Zwenger, S.; Basu, C. Plant terpenoids: Applications and future potentials. Biotechnol. Mol. Biol. Rev. 2008, 3, 001–007. [Google Scholar]
- Gavagnin, M.; Fontana, A. Diterpenes from marine opisthobranch molluscs. Curr. Org. Chem. 2000, 4, 1201–1248. [Google Scholar] [CrossRef]
- Maimone, T.; Baran, P. Modern synthetic efforts toward biologically active terpenes. Nat. Chem. Biol. 2007, 3, 396–407. [Google Scholar] [CrossRef]
- Blunt, J.W.; Munro, M.H.G. Dictionary of Marine Natural Products; Chapman & Hall/CRC: Boca Raton, FL, USA, 2008; p. 2108. [Google Scholar]
- Erwin, P.; López-Legentil, S.; Schuhmann, P. The pharmaceutical value of marine biodiversity for anti-cancer drug discovery. Ecol. Econ. 2010, 70, 445–451. [Google Scholar] [CrossRef]
- Lewis, R.; Garcia, M. Therapeutic potential of venom peptides. Nat. Rev. Drug Discov. 2003, 2, 790–802. [Google Scholar] [CrossRef]
- Taylor, M.W.; Radaz, R.; Steger, D.; Wagner, M. Sponge-associated microorganisms: Evolution, ecology, and biotechnological potential. Microbiol. Mol. Biol. Rev. 2007, 71, 295–347. [Google Scholar] [CrossRef]
- Kobayashi, J.; Ishibashi, M. Bioactive metabolites of symbiotic marine microorganisms. Chem. Rev. 1993, 93, 1753–1769. [Google Scholar]
- Kelecom, A. Secondary metabolites from marine microorganisms. An. Acad. Bras. Ciênc. 2002, 74, 151–170. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Munro, M.H.G.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2011, 28, 165–238. [Google Scholar]
- Blunt, J.W.; Copp, B.R.; Hu, W.-P.; Munro, M.H.G.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2009, 26, 170–244. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Hu, W.-P.; Munro, M.H.G.; Northcote, P.T.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2007, 24, 31–86. [Google Scholar] [CrossRef]
- Blunt, J.; Copp, B.; Munro, M.; Northcote, P.; Prinsep, M. Marine natural products. Nat. Prod. Rep. 2006, 23, 26–78. [Google Scholar] [CrossRef]
- Blunt, J.; Copp, B.; Munro, M.; Northcote, P.; Prinsep, M. Marine natural products. Nat. Prod. Rep. 2005, 22, 15–61. [Google Scholar] [CrossRef]
- Blunt, J.; Copp, B.; Munro, M.; Northcote, P.; Prinsep, M. Marine natural products. Nat. Prod. Rep. 2004, 21, 1–49. [Google Scholar] [CrossRef]
- Blunt, J.; Copp, B.; Munro, M.; Northcote, P.; Prinsep, M. Marine natural products. Nat. Prod. Rep. 2003, 20, 1–48. [Google Scholar] [CrossRef]
- Faulkner, D.J. Marine natural products. Nat. Prod. Rep. 2002, 19, 1–48. [Google Scholar]
- Paul, V.J.; Ritson-Williams, R. Marine chemical ecology. Nat. Prod. Rep. 2008, 25, 662–695. [Google Scholar] [CrossRef]
- Paul, V.J.; Puglisi, M.P. Chemical mediation of interactions among marine organisms. Nat. Prod. Rep. 2004, 21, 189–209. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Leal, M.C.; Madeira, C.; Brandão, C.A.; Puga, J.; Calado, R. Bioprospecting of Marine Invertebrates for New Natural Products — A Chemical and Zoogeographical Perspective. Molecules 2012, 17, 9842-9854. https://doi.org/10.3390/molecules17089842
Leal MC, Madeira C, Brandão CA, Puga J, Calado R. Bioprospecting of Marine Invertebrates for New Natural Products — A Chemical and Zoogeographical Perspective. Molecules. 2012; 17(8):9842-9854. https://doi.org/10.3390/molecules17089842
Chicago/Turabian StyleLeal, Miguel Costa, Carolina Madeira, Cláudio Alexandre Brandão, João Puga, and Ricardo Calado. 2012. "Bioprospecting of Marine Invertebrates for New Natural Products — A Chemical and Zoogeographical Perspective" Molecules 17, no. 8: 9842-9854. https://doi.org/10.3390/molecules17089842




