RETRACTED: Dynamic Action of Carotenoids in Cardioprotection and Maintenance of Cardiac Health
Abstract
:1. Introduction
2. Mechanisms of Redox Signaling
3. Antioxidants in Redox Status
4. Carotenoids as Redox Agents
4.1. Structure of Carotenoids
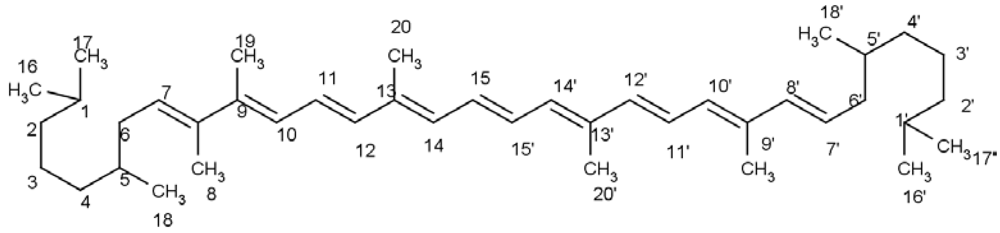
4.2. Bioavailability and Metabolism of Carotenoids
5. Cardioprotective Mechanisms of Carotenoids
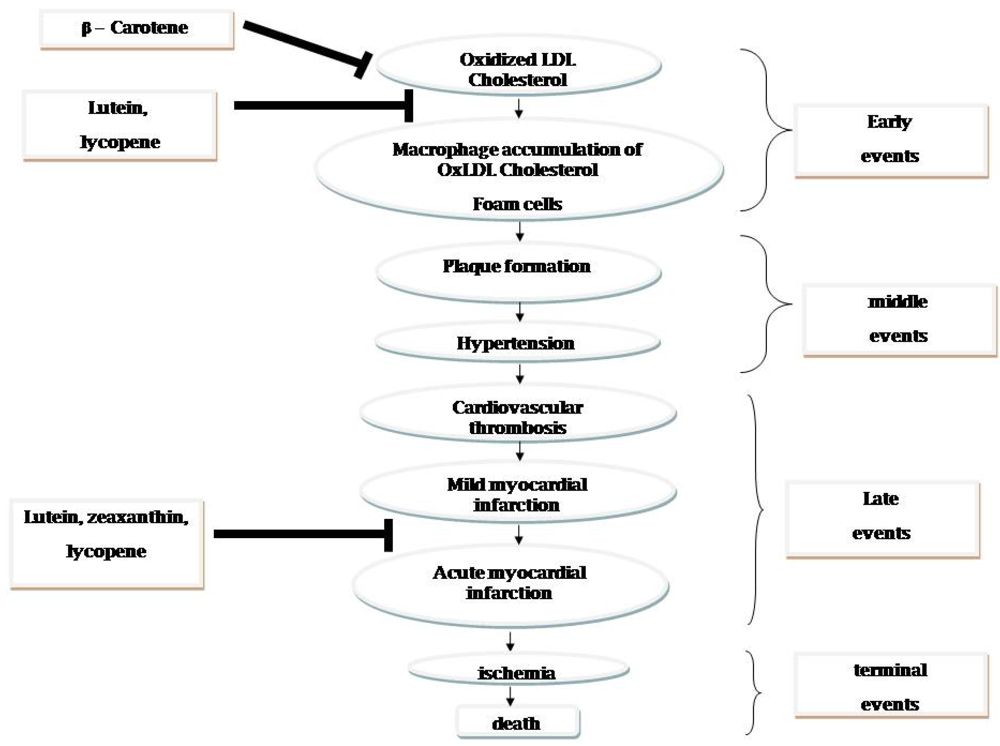
5.1. Antioxidant Activity
5.2. Hypocholesterolemic Activity
5.3. Antiinflammatory Activity
6. Role on Endothelial Function
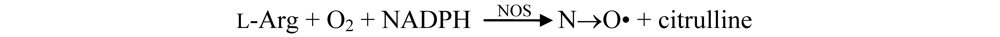
7. Influence on Gap Junction Communication
8. Attenuation of Ischemia-Reperfusion Injury
9. Conclusions
Acknowledgment
References and Notes
- Trachootham, D.; Lu, W.; Ogasawara, M.A.; Nilsa Rivera, D.V.; Huang, P. Redox regulation of cell survival. Antioxid. Redox Sign. 2008, 10, 1343–1357. [Google Scholar]
- Turan, B. Role of antioxidants in redox regulation of diabetic cardiovascular complications. Curr. Pharm. Biotechnol. 2010, 11, 819–836. [Google Scholar]
- Grassi, D.; Desideri, G.; Tiberti, S.; Ferri, C. Oxidative stress, endothelial dysfunction and prevention of cardiovascular diseases. Agro Food Industry Hi-Tech. 2009, 20, 76–79. [Google Scholar]
- Stocker, R.; Keaney, J.F., Jr. Role of oxidative modifications in atherosclerosis. Physiol. Rev. 2004, 84, 1381–1478. [Google Scholar]
- Madamanchi, N.R.; Vendrov, A.; Runge, M.S. Oxidative stress and vascular disease. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 29–38. [Google Scholar]
- Joshipura, K.J.; Hu, F.B.; Manson, J.E.; Stampfer, M.J.; Rimm, E.B.; Speizer, F.E.; Colditz, G.; Ascherio, A.; Rosner, B.; Spiegelman, D.; et al. The effect of fruit and vegetable intake on risk for coronary heart disease. Ann. Intern. Med. 2001, 134, 1106–1114. [Google Scholar]
- Finkel, T. Signal transduction by reactive oxygen species in non-phagocytic cells. J. Leukoc. Biol. 1999, 65, 337–340. [Google Scholar]
- Gao, W.D.; Liu, Y.; Marban, E. Selective effects of oxygen free radicals on excitation-contraction coupling in ventricular muscle. Implications for the mechanism of stunned myocardium. Circ. Res. 1996, 94, 2597–2604. [Google Scholar]
- Xie, Y.W.; Kaminski, P.M.; Wolin, M.S. Inhibition of rat cardiac muscle contraction and mitochondrial respiration by endogenous peroxynitrite formation during post hypoxic reoxygenation. Circ. Res. 1998, 82, 891–897. [Google Scholar]
- Sies, H. Antioxidants in Disease, Mechanisms and Therapy; Academic Press: New York, NY, USA, 1996. [Google Scholar]
- Palozza, P.; Krinsky, N.I. Antioxidant effects of carotenoids in vitro and in vivo: An overview. Methods Enzymol. 1992, 213, 403–420. [Google Scholar] [CrossRef]
- Krinsky, N.I. Actions of carotenoids in biological systems. Ann. Rev. Nutr. 1993, 13, 561–588. [Google Scholar]
- Amengual, J.; Lobo, G.P.; Golczak, M.; Li, H.N.; Klimova, T.; Hoppel, C.L.; Wyss, A.; Palczewski, K.; von Lintig, J. A mitochondrial enzyme degrades carotenoids and protects against oxidative stress. FASEB J. 2011, 25, 948–959. [Google Scholar]
- Sies, H.; Stahl, W. Vitamins E and C, β-carotene, and other carotenoids as antioxidants. Am. J. Clin. Nutr. 1995, 62, 1315S–1321S. [Google Scholar]
- Burton, G.W.; Ingold, K.U. β-Carotene: An unusual type of lipid antioxidant. Science 1984, 224, 569–573. [Google Scholar]
- Palozza, P. Prooxidant actions of carotenoids in biologic systems. Nutr. Rev. 1998, 56, 257–265. [Google Scholar]
- Parker, R.S. Bioavailability of carotenoids. Eur. J. Clin. Nutr. 1997, 51, 586–590. [Google Scholar]
- Parker, R.S. Absorption, metabolism and transport of carotenoids. FASEB J. 1996, 10, 542–551. [Google Scholar]
- Gartner, C.; Stahl, W.; Sies, H. Lycopene is more bioavailable from tomato paste than from fresh tomatoes. Am. J. Clin. Nutr. 1997, 66, 116–122. [Google Scholar]
- Erdman, J.W., Jr.; Bierer, T.L.; Gugger, E.T. Absorption and transport of carotenoids. Ann. NY Acad. Sci. 1993, 691, 76–85. [Google Scholar]
- Parker, R.S. Carotenoid and tocopherol composition in human adipose tissue. Am. J. Clin. Nutr. 1988, 47, 33–36. [Google Scholar]
- Britton, G. Structure and properties of carotenoids in relation to function. FASEB J. 1995, 9, 1551–1558. [Google Scholar]
- Stahl, W.; Nicolai, S.; Briviba, K.; Hanusch, M.; Broszeit, G.; Peters, M.; Martin, H.D.; Sies, H. Biological activities of natural and synthetic carotenoids: Induction of gap junctional communication and singlet oxygen quenching. Carcinogenesis 1997, 18, 89–92. [Google Scholar]
- Ziegler, R.G.; Mayne, S.T.; Swanson, C.A. Nutrition and lung cancer. Cancer Causes Control 1996, 7, 157–177. [Google Scholar]
- Peto, R.; Doll, R.; Buckley, J.D.; Sporn, M.B. Can dietary beta-carotene materially reduce human cancer rates? Nature 1981, 290, 201–208. [Google Scholar]
- Voutilainen, S.; Nurmi, T.; Mursu, J.; Rissanen, T.H. Carotenoids and cardiovascular health. Am. J. Clin. Nutr. 2006, 83, 1265–1271. [Google Scholar]
- Conn, P.F.; Schalch, W.; Truscott, T.G. The singlet oxygen and β-carotene interaction. J. Photochem. Photobiol. B Biol. 1991, 11, 41–47. [Google Scholar]
- di Mascio, P.; Kaiser, S.; Sies, H. Lycopene as the most efficient biological carotenoid singlet oxygen quencher. Arch. Biochem. Biophys. 1989, 274, 532–528. [Google Scholar]
- Burton, G.W.; Ingold, K.U. β-Carotene: An unusual type of lipid anti-oxidant. Science 1984, 224, 569–573. [Google Scholar]
- Hill, T.J.; Land, E.J.; McGarvey, D.J.; Schalch, W.; Tinkler, J.H.; Truscott, T.G. Interactions between carotenoids and the CCl3O2• radical. J. Am. Chem. Soc. 1995, 117, 8322–8326. [Google Scholar]
- Everett, S.A.; Dennis, M.F.; Patel, K.B.; Maddix, S.; Kundu, S.C.; Willson, R.L. Scavenging of nitrogen dioxide, thiyl, and sulfonyl free radicals by the nutritional anti-oxidant β-carotene. J. Biol. Chem. 1996, 271, 3988–3994. [Google Scholar]
- Mortensen, A.; Skibsted, L.H. Kinetics of parallel electron transfer from β-carotene to phenoxyyl radical and adduct formation between phenoxyl radical and β-carotene. Free Radic. Res. 1996, 25, 515–523. [Google Scholar]
- Christensen, R.L. The Electronic States of Carotenoids. In The Photochemistry of Carotenoids; Frank, H.A., Young, A.J., Britton, G., Cogdell, R.J., Eds.; Kluwer Academic: Dordrecht, The Netherlands, 1999; pp. 137–157. [Google Scholar]
- El-Agamey, A.; Lowe, G.M.; McGarvey, D.J.; Mortensen, A.; Phillip, D.M.; Truscott, T.G.; Young, A.J. Carotenoid radical chemistry and antioxidant/pro-oxidant properties. Arch. Biochem. Biophys. 2004, 430, 37–48. [Google Scholar]
- di Mascio, P.; Murphy, M.E.; Seis, H. Antioxidant defense systems: The role of carotenoids, tocopherols, and thiols. Am. J. Clin. Nutr. 1994, 53, 194–200. [Google Scholar]
- Arab, L.; Steck, S. Lycopene and cardiovascular disease. Am. J. Clin. Nutr. 2000, 71, 1691S–1695S. [Google Scholar]
- Fuhrman, B.; Elis, A.; Aviram, M. Hypocholesterolemic effect of lycopene and betacarotene is related to suppression of cholesterol synthesis and augmentation of LDL receptor activity in macrophages. BiochemBiophys Res. Commun. 1997, 233, 658–662. [Google Scholar]
- Agarwal, S.; Rao, A.V. Tomato lycopene and low density lipoprotein oxidation: a human dietary intervention study. Lipids 1998, 33, 981–984. [Google Scholar]
- Palooza, P.; Krinsky, N.I. Astaxanthin and canthaxanthin are potent antioxidants in a membrane model. Arch. Biochem. Biophys. 1992, 297, 291–295. [Google Scholar]
- Shimidzu, N. Carotenoids as singlet oxygen quenchers in marine organisms. Fish. Sci. 1996, 62, 134–137. [Google Scholar]
- Miki, W. Biological functions and activities of animal carotenoids. Pure Appl. Chem. 1991, 63, 141–146. [Google Scholar]
- Krinsky, N.I. Antioxidant functions of carotenoids. Free Radic. Biol. Med. 1989, 7, 617–635. [Google Scholar]
- Jackson, H.; Braun, C.L.; Ernst, H. The chemistry of novel xanthophyll carotenoids. Am. J. Cardiol. 2008, 101, 50D–57D. [Google Scholar]
- Beutner, S.; Bloedorn, B.; Frixel, S.; Blanco, I.H.; Hoffman, T.; Martin, H.D.; Mayer, B.; Noach, P.; Rack, C.; Schmidt, M.; et al. Quantitative assessment of antioxidant properties of natural colorants and phytochemicals: Carotenoids, flavonoids, phenols and indigoids. The role of β-carotene in antioxidant functions. J. Sci. Food Agric. 2001, 81, 559–568. [Google Scholar] [CrossRef]
- Pashkow, F.J.; Watumull, D.G.; Campbell, C.L. Astaxanthin: A novel potential treatment for oxidative stress and inflammation in cardiovascular disease. Am. J. Cardiol. 2008, 101, 58D–68D. [Google Scholar]
- McNulty, H.P.; Byun, J.; Lockwood, S.F.; Jacob, R.F.; Mason, R.P. Differential effects of carotenoids on lipid peroxidation due to membrane interactions: X-ray diffraction analysis. Biochim. Biophys. Acta 2007, 1768, 167–174. [Google Scholar] [CrossRef]
- Stahl, W.; Nicolai, S.; Briviba, K.; Hanusch, M.; Broszeit, G.; Peters, M.; Martin, H.D.; Sies, H. Biological activities of natural and synthetic carotenoids: Induction of gap junctional communication and singlet oxygen quenching. Carcinogenesis 1997, 18, 89–92. [Google Scholar]
- Esterbauer, H.; Striegl, G.; Puhl, J.; Rotheneder, M. Continuous monitoring of in vitro oxidation of human low density lipoprotein. Free Radic. Res. Commun. 1989, 6, 67–75. [Google Scholar] [CrossRef]
- Romanchik, J.E.; Harrison, E.H.; Morel, D.W. Addition of lutein, lycopene or beta-carotene to LDL or serum in vitro: Effects on carotenoid distribution, LDL composition, and LDL oxidation. J. Nutr. Biochem. 1997, 8, 681–688. [Google Scholar] [CrossRef]
- Polyakov, N.E.; Leshina, T.V.; Konovalova, T.A.; Kispert, L.D. Carotenoids as scavengers of free radicals in a fenton reaction: Antioxidants or pro-oxidants? Free Radic. Biol. Med. 2001, 31, 398–404. [Google Scholar] [CrossRef]
- Gruszecki, W. Carotenoids in Membranes. Adv. Photosynth. Resp. 2004, 8, 363–379. [Google Scholar]
- Dwyer, J.H.; Navab, M.; Dwyer, K.M.; Hassan, K.; Sun, P.; Shircore, A.; Hama-Levy, S.; Hough, G.; Wang, X.; Drake, T.; et al. Oxygenated carotenoid lutein and progression of early atherosclerosis: The Los Angeles atherosclerosis study. Circulation 2001, 103, 2922–2927. [Google Scholar]
- Panasenko, O.M.; Sharov, V.S.; Briviba, K.; Sies, H. Interaction of peroxynitrite with carotenoids in human low density lipoproteins. Arch. Biochem. Biophys. 2000, 73, 302–305. [Google Scholar]
- Shaish, A.; Daugherty, A.; O’Sullivan, F.; Schonfeld, G.; Heinecke, J.W. Beta-carotene inhibits atherosclerosis in hypercholesterolemic rabbits. J. Clin. Invest. 1995, 96, 2075–2082. [Google Scholar]
- Steinberg, D. Atherogenesis in perspective: Hypercholesterolemia and inflammation as partners in crime. Nat. Med. 2002, 8, 1211–1217. [Google Scholar]
- Feng, D.; Ling, W.H.; Duan, R.D. Lycopene suppresses LPS-induced NO and IL-6 production by inhibiting the activation of ERK, p38MAPK, and NF-κB in macrophages. Inflamm. Res. 2009, 59, 115–121. [Google Scholar]
- Riso, P.; Visioli, F.; Grande, S.; Guarnieri, S.; Gardana, C.; Simonetti, P.; Porrini, M. Effect of a tomato-based drink on markers of inflammation, immunomodulation and oxidative stress. J. Agric. Food Chem. 2006, 54, 2563–2566. [Google Scholar]
- Napolitano, M.; De Pascale, C.; Wheeler-Jones, C.; Botham, K.M.; Bravo, E. Effects of lycopene on the induction of foam cell formation by modified LDL. Am. J. Physiol. Endocrinol. Metab. 2007, 293, 1820–1827. [Google Scholar]
- Jung, E.M.; Jose, O.L.; Ryan, D.; Joan, A.S.; Richard, M.C.; Maria, L.F. A lutein-enriched diet prevents cholesterol accumulation and decreases oxidized LDL and inflammatory cytokines in the aorta of guinea pigs. J. Nutr. 2011, 141, 1458–1463. [Google Scholar]
- Selvaraj, R.K.; Klasing, K.C. Lutein and eicosapentaenoic acid interact to modify iNOS mRNA levels through the PPARg/RXR pathway in chickens and HD11 cell lines. J. Nutr. 2006, 136, 1610–1616. [Google Scholar]
- Zhao, Y.; Yu, W.; Hu, W.; Yuan, Y. Anti-inflammatory and anticoagulant activities of lycopene in mice. Nutr. Res. 2003, 23, 1591–1595. [Google Scholar]
- Ohgami, K.; Shiratori, K.; Kotake, S.; Nishida, T.; Mizuki, N.; Yazawa, K.; Ohno, S. Efects of astaxanthin on lipopolysaccharide-induced inflammation in vitro and in vivo. Invest. Ophthalmol. Visual Sci. 2006, 44, 2694–2701. [Google Scholar]
- Lee, S.J.; Bai, S.K.; Lee, K.S.; Namkoong, S.; Na, H.J.; Ha, K.S.; Han, J.A.; Yim, S.V.; Chang, K.; Kwon, Y.G.; et al. Astaxanthin inhibits nitric oxide production and inflammatory gene expression by suppressing I(kappa)B kinase-dependent NF-kappaB activation. Mol. Cells 2003, 16, 97–105. [Google Scholar]
- Devasagayam, T.P.A.; Tilak, J.C.; Boloor, K.K.; Ketaki, S.S.; Ghaskadbi, S.S.; Lele, R.D. Free radicals and antioxidants in human health: Current status and future prospects. JAPI 2004, 52, 794–804. [Google Scholar]
- Wendy, M.R.; Ineke, A.A.; Bots, M.L.; Kluft, C.; Princen, H.; Henk, F.J.; Lilian, B.M.; Poppel, G.V.; Alwine, F.M. Serum carotenoids and vitamins in relation to markers of endothelial function and inflammation. Eur. J. Epidemiol. 2004, 19, 915–921. [Google Scholar]
- Martin, K.R.; Wu, D.; Meydani, M. The effect of carotenoids on the expression of cell surface adhesion molecules and binding of monocytes to human aortic endothelial cells. Atherosclerosis 2000, 105, 265–274. [Google Scholar]
- Lee, D.K.; Grantham, R.N.; Mannion, J.D.; Trachte, A.L. Carotenoids enhance phosphorylation of Akt and suppress tissue factor activity in human endothelial cells. J. Nutr. Biochem. 2006, 17, 780–786. [Google Scholar]
- Pashkow, F.J.; Watumull, G.D.; Campbell, C.L. Astaxanthin: A novel potential treatment for oxidative stress and inflammation in cardiovascular disease. Am. J. Cardiol. 2008, 101, 58D–68D. [Google Scholar]
- Stahl, W.; von Laar, J.; Martin, H.D.; Emmerich, T.; Sies, H. Stimulation of gap junctional communication: Comparison of acyclo-retinoic acid and lycopene. Arch. Biochem. Biophys. 2000, 373, 271–274. [Google Scholar]
- Bertram, J.S. Carotenoids and gene regulation. Nutr. Rev. 1999, 57, 182–191. [Google Scholar]
- Hix, L.M.; Lockwood, S.F.; Bertram, J.S. Upregulation of connexin 43 protein expression and increased gap junctional communication by water soluble disodium disuccinate astaxanthin derivatives. Cancer Lett. 2004, 211, 25–37. [Google Scholar]
- Maulik, N.; Das, D.K. Apoptosis, heart failure, ischemic heart disease. Heart Failure Rev. 1999, 4, 165–175. [Google Scholar]
- Esterhuyse, A.J.; Van, R.J.; Strijdom, H.; Bester, D.; Du Toit, E.F. Proposed mechanisms for red palm oil induced cardioprotection in a model of hyperlipidaemia in the rat. PLEFA 2006, 75, 375–380. [Google Scholar]
- Engelbrecht, A.M.; Esterhuyse, A.J.; Du Toit, E.F.; Lochner, A.; Van Rooyen, J. p38-MAPK and PKB/Akt, possible role players in red palm oil-induced protection of the isolated perfused rat heart. J. Nutr. Biochem. 2006, 17, 265–271. [Google Scholar]
- Grossa, G.J.; Lockwood, S.F. Cardioprotection and myocardial salvage by a disodium disuccinate astaxanthin derivative (Cardax). Life Sci. 2004, 75, 215–224. [Google Scholar]
- Aoi, W.; Naito, Y.; Sakuma, K.; Kuchide, M.; Tokuda, H.; Maoka, T.; Toyokuni, S.; Oka, S.; Yasuhara, M.; Yoshikawa, T. Astaxanthin limits exercise-induced skeletal and cardiac muscle damage in mice. Antioxid. Redox Sign. 2003, 5, 139–144. [Google Scholar]
- Guerin, M.; Huntley, M.E.; Olaizola, M. Haematococcus astaxanthin: Applications for human health and nutrition. Trends Biotechnol. 2003, 21, 210–216. [Google Scholar]
- Kima, J.H.; Naa, J.H.; Kima, C.K.; Kima, J.Y.; Haa, K.S.; Leeb, H.; Chungc, H.T.; Kwond, H.J.; Kwone, Y.G.; Kim, Y.M. The non-provitamin A carotenoid, lutein, inhibits NF-κB-dependent gene expression through redox-based regulation of the phosphatidylinositol 3-kinase/PTEN/Akt and NF-κB-inducing kinase pathways: Role of H2O2 in NF-κB activation. Free Radic. Biol. Med. 2008, 45, 885–896. [Google Scholar] [CrossRef]
- Simone, R.E.; Russo, M.; Catalano, A.; Monego, G.; Froehlich, K.; Boehm, V.; Palozza, P. Lycopene inhibits NF-кB mediated IL-8 expression and changes redox and PPAR γ signaling in cigarette smoke-stimulated macrophages. PLoS One 2011, 6. [Google Scholar]
- Das, D.K.; Maulik, N. Conversion of death signal into survival signal by redox signaling. Biochemistry 2004, 69, 10–17. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Agarwal, M.; Parameswari, R.P.; Vasanthi, H.R.; Das, D.K. RETRACTED: Dynamic Action of Carotenoids in Cardioprotection and Maintenance of Cardiac Health. Molecules 2012, 17, 4755-4769. https://doi.org/10.3390/molecules17044755
Agarwal M, Parameswari RP, Vasanthi HR, Das DK. RETRACTED: Dynamic Action of Carotenoids in Cardioprotection and Maintenance of Cardiac Health. Molecules. 2012; 17(4):4755-4769. https://doi.org/10.3390/molecules17044755
Chicago/Turabian StyleAgarwal, Mahesh, Royapuram P. Parameswari, Hannah R. Vasanthi, and Dipak K. Das. 2012. "RETRACTED: Dynamic Action of Carotenoids in Cardioprotection and Maintenance of Cardiac Health" Molecules 17, no. 4: 4755-4769. https://doi.org/10.3390/molecules17044755
APA StyleAgarwal, M., Parameswari, R. P., Vasanthi, H. R., & Das, D. K. (2012). RETRACTED: Dynamic Action of Carotenoids in Cardioprotection and Maintenance of Cardiac Health. Molecules, 17(4), 4755-4769. https://doi.org/10.3390/molecules17044755




