Synthesis and Antibacterial Evaluation of New N-acylhydrazone Derivatives from Dehydroabietic Acid
Abstract
:1. Introduction
2. Results and Discussion
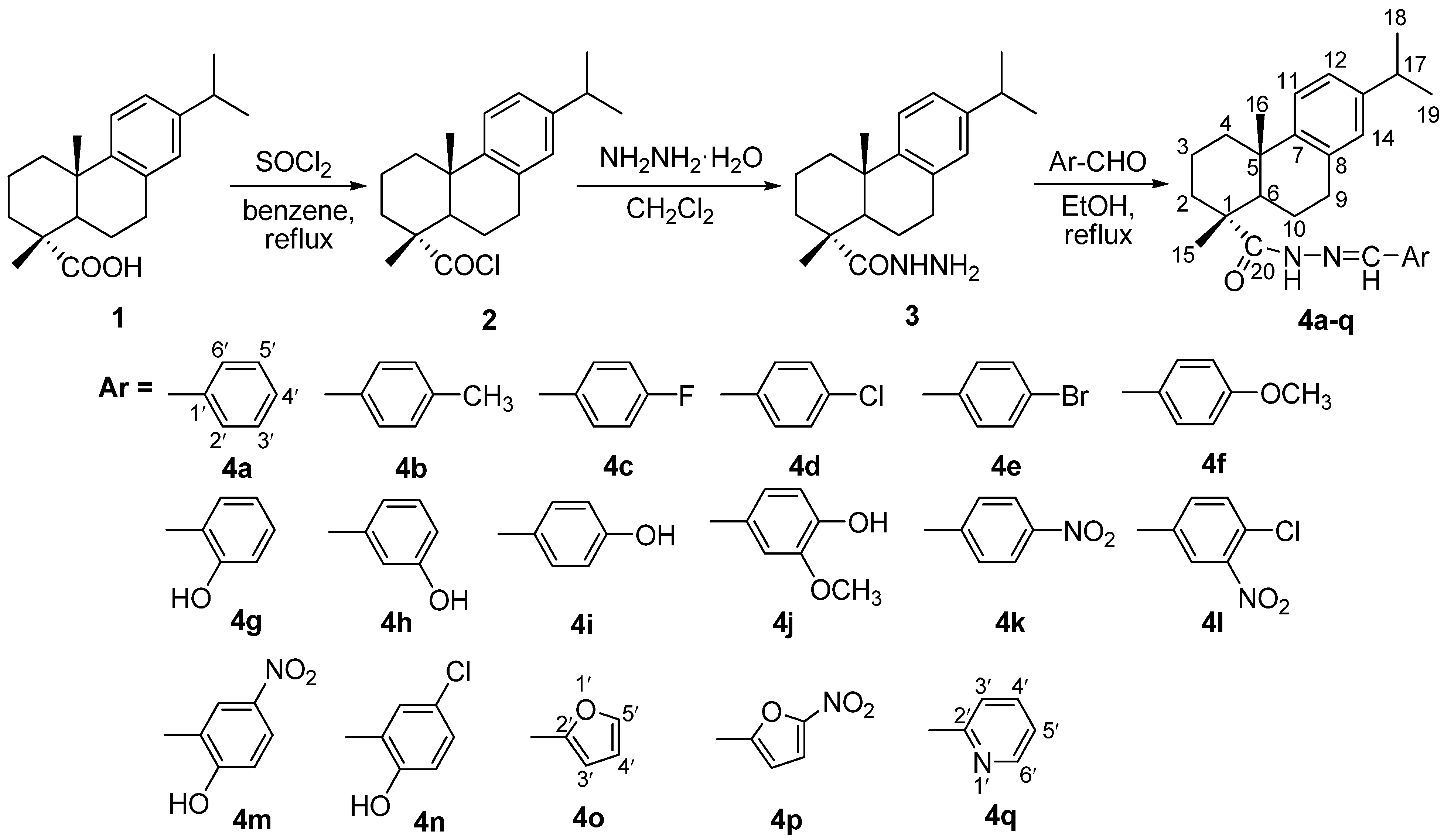
2.1. Synthesis
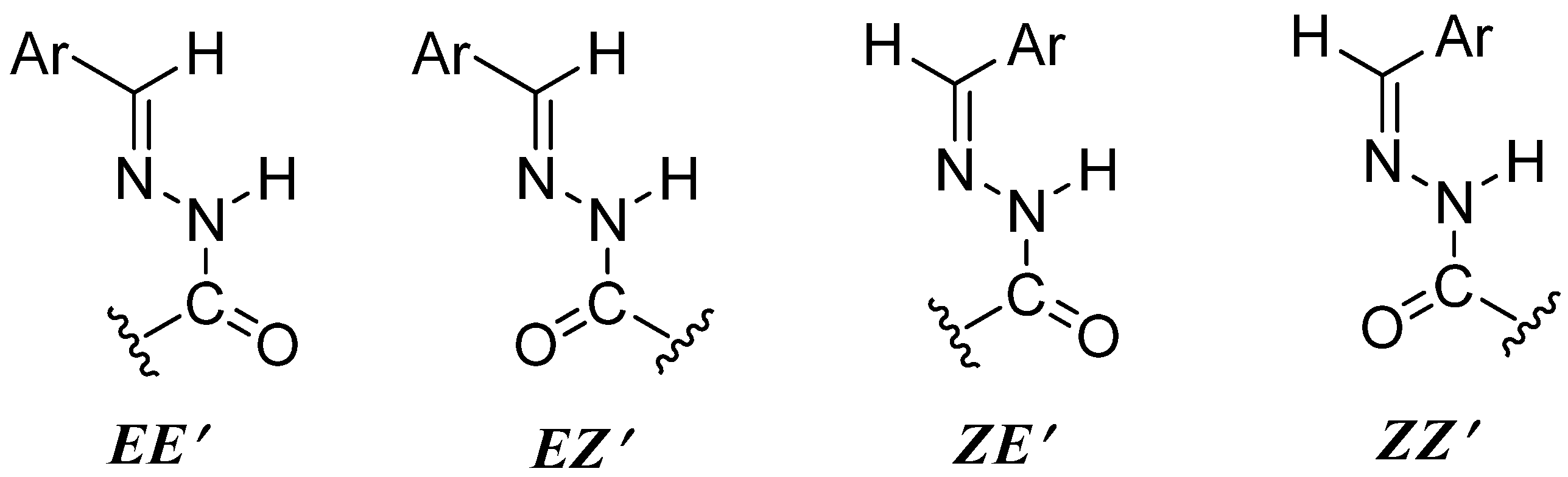
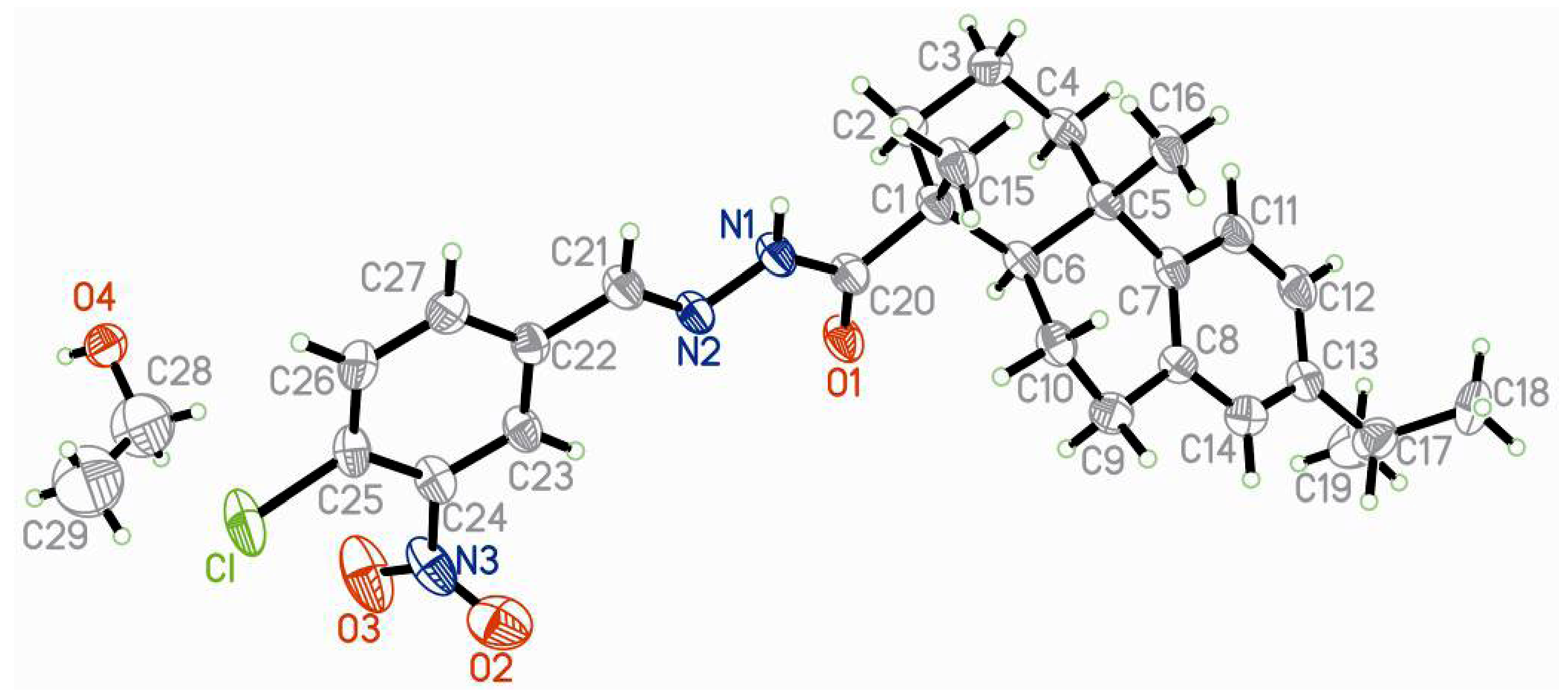
2.2. Structural Analysis
| Empirical formula | C29H38ClN3O4 | F(0 0 0) | 1128 |
| Formula weight | 528.07 | θ range for data collection (°) | 1.93–25.38 |
| Temperature (K) | 293(2) | Max. and min. transmission | 0.9674 and 0.9516 |
| Crystal size (mm3) | 0.30 × 0.20 × 0.20 | Index ranges | 0 ≤ h ≤ 15 |
| Crystal system | Orthorhombic | 0 ≤ k ≤ 15 | |
| Space group | P212121 | −21 ≤ l ≤ 21 | |
| a (Å) | 12.537(3) | Reflectons collected | 5870 |
| b (Å) | 13.097(3) | Independent reflections | 5374 |
| c (Å) | 17.855(4) | Rint | 0.0253 |
| α (°) | 90 | Data/restraints/parameters | 5374/3/328 |
| β (°) | 90 | Goodness-of-fit on F2 | 1.001 |
| γ (°) | 90 | R1, wR2 [I > 2σ(I)] | 0.0592/0.1567 |
| V (Å3) | 2931.7(10) | R1, wR2 (all data) | 0.0912/0.1783 |
| Z | 4 | Flack parameter | −0.09(13) |
| Dx (g/cm−3) | 1.196 | (Δρ)max (eÅ−3) | 0.520 |
| μ (mm−1) | 0.167 | (Δρ)min (eÅ−3) | −0.373 |
2.3. Antibacterial Activity
| Test compounds | Minimum inhibitory concentration (μg/mL) | |||
|---|---|---|---|---|
| S. aureus | B. subtilis | E. coli | P. fluorescens | |
| 3 | 31.2 | 31.2 | 62.5 | 31.2 |
| 4a | 31.2 | 62.5 | >100 | 62.5 |
| 4b | >100 | 62.5 | >100 | >100 |
| 4c | 7.8 | 3.9 | 31.2 | >100 |
| 4d | 7.8 | 15.6 | 31.2 | 62.5 |
| 4e | 31.2 | 31.2 | >100 | >100 |
| 4f | >100 | 62.5 | >100 | >100 |
| 4g | 62.5 | 15.6 | 31.2 | 62.5 |
| 4h | 62.5 | 31.2 | >100 | >100 |
| 4i | >100 | >100 | >100 | >100 |
| 4j | >100 | 62.5 | >100 | >100 |
| 4k | 15.6 | 15.6 | 31.2 | 15.6 |
| 4l | 3.9 | 3.9 | 15.6 | 7.8 |
| 4m | 7.8 | 3.9 | 15.6 | 31.2 |
| 4n | 3.9 | 7.8 | 15.6 | 7.8 |
| 4o | >100 | 62.5 | >100 | >100 |
| 4p | 1.9 | 1.9 | 7.8 | 3.9 |
| 4q | 62.5 | >100 | >100 | >100 |
| Amikacin | 0.9 | 0.9 | 1.9 | 0.9 |
3. Experimental
3.1. General
3.2. Crystallographic Studies
3.3. Chemistry
3.3.1. Dehydroabietic Acid Hydrazide (3)
3.3.2. Gerneral Procedure for the Syntheses of N-acylhydrazone Derivatives 4a–q
3.4. Antibacterial Studies
4. Conclusions
Acknowledgements
- Sample Availability: Samples of the newly synthesized compounds (4a-q) are available from the authors.
References and Notes
- Aksoy, D.Y.; Unal, S. New antimicrobial agents for the treatment of Gram-positive bacterial infections. Clin. Microbiol. Infect. 2008, 14, 411–420. [Google Scholar]
- Kumar, D.; Judge, V.; Narang, R.; Sangwan, S.; De Clercq, E.; Balzarini, J.; Narasimhan, B. Benzylidene/2-chlorobenzylidene hydrazides: Synthesis, antimicrobial activity, QSAR studies and antiviral evaluation. Eur. J. Med. Chem. 2010, 45, 2806–2816. [Google Scholar]
- Rollas, S.; Küçükgüzel, S.G. Biological activities of hydrazone derivatives. Molecules 2007, 12, 1910–1939. [Google Scholar]
- ink, R.; Kovač, A.; Tomašić, T.; Rupnik, V.; Boniface, A.; Bostock, J.; Chopra, I.; Blanot, D.; Mašič, L.P.; Gobec, S.; et al. Synthesis and biological evaluation of N-acylhydrazones as inhibitors of MurC and MurD ligases. ChemMedChem 2008, 3, 1362–1370. [Google Scholar] [CrossRef]
- Masunari, A.; Tavares, L.C. A new class of nifuroxazide analogues: Synthesis of 5-nitrothiophene derivatives with antimicrobial activity against multidrug-resistant Staphylococcus aureus. Bioorg. Med. Chem. 2007, 15, 4229–4236. [Google Scholar]
- Chornous, V.A.; Bratenko, M.K.; Vovk, M.V.; Sidorchuk, I.I. Synthesis and antimicrobial activity of pyrazole-4-carboxylic acid hydrazides and N-(4-Pyrazoyl)hydrazones of aromatic and heteroaromatic aldehydes. Pharm. Chem. J. 2001, 35, 203–205. [Google Scholar]
- Melnyk, P.; Leroux, V.; Sergheraert, C.; Grellier, P. Design, synthesis and in vitro antimalarial activity of an acylhydrazone library. Bioorg. Med. Chem. Lett. 2006, 16, 31–35. [Google Scholar]
- Tian, B.H.; He, M.Z.; Tan, Z.W.; Tang, S.X.; Hewlett, I.; Chen, S.G.; Jin, Y.X.; Yang, M. Synthesis and antiviral evaluation of new N-acylhydrazones containing glycine residue. Chem. Biol. Drug Des. 2011, 77, 189–198. [Google Scholar]
- Cui, Z.N.; Li, Y.; Ling, Y.; Huang, J.A.; Cui, J.R.; Wang, R.Q.; Yang, X.L. New class of potent antitumor acylhydrazone derivatives containing furan. Eur. J. Med. Chem. 2010, 45, 5576–5584. [Google Scholar]
- da Silva, Y.K.C.; Augusto, C.V.; Barbosa, M.L.D.; Melo, G.M.D.; de Queiroz, A.C.; Dias, T.D.M.F.; Bispo, W.; Barreiro, E.J.; Lima, L.M.; Alexandre-Moreira, M.S. Synthesis and pharmacological evaluation of pyrazine N-acylhydrazone derivatives designed as novel analgesic and anti-inflammatory drug candidates. Bioorg. Med. Chem. 2010, 18, 5007–5015. [Google Scholar]
- Dimmock, J.R.; Vashishtha, S.C.; Stables, J.P. Anticonvulsant properties of various acetylhydrazones, oxamoylhydrazones and semicarbazones derived from aromatic and unsaturated carbonyl compounds. Eur. J. Med. Chem. 2000, 35, 241–248. [Google Scholar]
- Ergenç, N.; Günay, N.S.; Demirdamar, R. Synthesis and antidepressant evaluation of new 3-phenyl-5-sulfonamidoindole derivatives. Eur. J. Med. Chem. 1998, 33, 143–148. [Google Scholar]
- Zapata-Sudo, G.; Pereira, S.L.; Beiral, H.J.V.; Kummerle, A.E.; Raimundo, J.M.; Antunes, F.; Sudo, R.; Barreiro, E.J.; Fraga, C.A.M. Pharmacological characterization of (3-thienylidene)-3,4-methylenedioxybenzoylhydrazide: A novel muscarinic agonist with antihypertensive profile. Am. J. Hypertens. 2010, 23, 135–141. [Google Scholar]
- Lee, W.S.; Kim, J.R.; Han, J.M.; Jang, K.C.; Sok, D.E.; Jeong, T.S. Antioxidant activities of abietane diterpenoids isolated from Torreya nucifera leaves. J. Agric. Food Chem. 2006, 54, 5369–5374. [Google Scholar]
- Chao, K.P.; Hua, K.F.; Hsu, H.Y.; Su, Y.C.; Chang, S.T. Anti-inflammatory activity of sugiol, a diterpene isolated from Calocedrus formosana bark. Planta Med. 2005, 71, 300–305. [Google Scholar]
- Savluchinske-Feio, S.; Curto, M.J.M.; Gigante, B.; Roseiro, J.C. Antimicrobial activity of resin acid derivatives. Appl. Microbiol. Biotechnol. 2006, 72, 430–436. [Google Scholar]
- Córdova, I.; León, L.G.; León, F.; San Andrés, L.; Luis, J.G.; Padrón, J.M. Synthesis and antiproliferative activity of novel sugiol β-amino alcohol analogs. Eur. J. Med. Chem. 2006, 41, 1327–1332. [Google Scholar]
- Gigante, B.; Santos, C.; Silva, A.M.; Curto, M.J.M.; Nascimento, M.S.J.; Pinto, E.; Pedro, M.; Cerqueira, F.; Pinto, M.M.; Duarte, M.P.; et al. Catechols from abietic acid: Synthesis and evaluation as bioactive compounds. Bioorg. Med. Chem. 2003, 11, 1631–1638. [Google Scholar]
- Fonseca, T.; Gigante, B.; Marques, M.M.; Gilchrist, T.L.; De Clercq, E. Synthesis and antiviral evaluation of benzimidazoles, quinoxalines and indoles from dehydroabietic acid. Bioorg. Med. Chem. 2004, 12, 103–112. [Google Scholar]
- Kang, M.S.; Hirai, S.; Goto, T.; Kuroyanagi, K.; Lee, J.Y.; Uemura, T.; Ezaki, Y.; Takahashi, N.; Kawada, T. Dehydroabietic acid, a phytochemical, acts as ligand for PPARs in macrophages and adipocytes to regulate inflammation. Biochem. Biophys. Res. Commun. 2008, 369, 333–338. [Google Scholar]
- Sepúlveda, B.; Astudillo, L.; Rodríguez, J.A.; Yáñez, T.; Theoduloz, C.; Schmeda-Hirschmann, G. Gastroprotective and cytotoxic effect of dehydroabietic acid derivatives. Pharmacol. Res. 2005, 52, 429–437. [Google Scholar]
- Gu, W.; Wang, S.F. Synthesis and antimicrobial activities of novel 1H-dibenzo[a,c]carbazoles from dehydroabietic acid. Eur. J. Med. Chem. 2010, 45, 4692–4696. [Google Scholar]
- Su, G.F.; Guan, Y.F.; Huang, W.Y.; Wang, H.S.; Pan, Y.M. Synthesis and crystal structure of 2-(4-decarboxydehydroabietyl)-5-p-tolyl-[1,3,4]-oxadiazole. Chinese J. Struct. Chem. 2009, 28, 881–885. [Google Scholar]
- Shingalapur, R.V.; Hosamani, K.M.; Keri, R.S.; Hugar, M.H. Derivatives of benzimidazole pharmacophore: Synthesis, anticonvulsant, antidiabetic and DNA cleavage studies. Eur. J. Med. Chem. 2010, 45, 1753–1759. [Google Scholar]
- Lee, S.; Jung, K.Y.; Park, J.; Cho, J.H.; Kim, Y.C.; Chang, S. Synthesis of potent chemical inhibitors of dynamin GTPase. Bioorg. Med. Chem. Lett. 2010, 20, 4858–4864. [Google Scholar]
- Han, C.R.; Song, Z.Q.; Shang, S.B.; Gao, H. Synthesis and characterization of novel dehydroabietyl-1,3,4-oxadiazoles (in Chinese). Xiandai Huagong 2007, 27, 42–44. [Google Scholar]
- Syakaev, V.V.; Podyachev, S.N.; Buzykin, B.I.; Latypov, S.K.; Habicher, W.D.; Konovalov, A.I. NMR study of conformation and isomerization of aryl- and heteroarylaldehyde 4-tert-butylphenoxyacetylhydrazones. J. Mol. Struct. 2006, 788, 55–62. [Google Scholar]
- Nakka, M.; Begum, M.S.; Varaprasad, B.F.M.; Reddy, L.V.; Bhattacharya, A.; Helliwell, M.; Mukherjee, A.K.; Beevi, S.S.; Mangamoori, L.N.; Mukkanti, K.; et al. Naproxen and ibuprofen based acyl hydrazone derivatives: Synthesis, structure analysis and cytotoxicity studies. J. Chem. Pharm. Res. 2010, 2, 393–409. [Google Scholar]
- Palla, G.; Predieri, G.; Domiano, P.; Vignali, C.; Turner, W. Conformational behaviour and E/Z isomerization of N-acyl and N-aroylhydrazones. Tetrahedron 1986, 42, 3649–3654. [Google Scholar]
- Unsal-Tan, O.; Ozden, K.; Rauk, A.; Balkan, A. Synthesis and cyclooxygenase inhibitory activities of some N-acylhydrazone derivatives of isoxazolo[4,5-d]pyridazin-4(5H)-ones. Eur. J. Med. Chem. 2010, 45, 2345–2352. [Google Scholar]
- Plech, T.; Wujec, M.; Siwek, A.; Kosikowska, U.; Malm, A. Synthesis and antimicrobial activity of thiosemicarbazides, s-triazoles and their Mannich bases bearing 3-chlorophenyl moiety. Eur. J. Med. Chem. 2011, 46, 241–248. [Google Scholar]
- Kossakowski, J.; Krawiecka, M.; Kuran, B.; Stefańska, J.; Wolska, I. Synthesis and preliminary evaluation of the antimicrobial activity of selected 3-benzofurancarboxylic acid derivatives. Molecules 2010, 15, 4737–4749. [Google Scholar]
- Upadhyay, A.; Srivastava, S.K.; Srivastava, S.D. Conventional and microwave assisted synthesis of Some new N-[(4-oxo-2-substituted aryl-1, 3-thiazolidine)-acetamidyl]-5- nitroindazoles and its antimicrobial activity. Eur. J. Med. Chem. 2010, 45, 3541–3548. [Google Scholar]
- Küçükgüzel, S.G.; Mazi, A.; Sahin, F.; Öztürk, S.; Stables, J. Synthesis and biological activities of diflunisal hydrazide-hydrazones. Eur. J. Med. Chem. 2003, 38, 1005–1013. [Google Scholar] [CrossRef]
- Viodé, C.; Bettache, N.; Cenas, N.; Krauth-Siegel, R.; Chauvière, G.; Bakalara, N.; Périé, J. Enzymatic reduction studies of nitroheterocycles. Biochem. Pharmacol. 1999, 57, 549–557. [Google Scholar]
- Quillardet, P.; Arrault, X.; Michel, V.; Touati, E. Organ-targeted mutagenicity of nitrofurantoin in Big Blue transgenic mice. Mutagenesis 2006, 21, 305–311. [Google Scholar]
- Bartel, L.C.; de Mecca, M.M.; Castro, J.A. Nitroreductive metabolic activation of some carcinogenic nitro heterocyclic food contaminants in rat mammary tissue cellular fractions. Food Chem. Toxicol. 2009, 47, 140–144. [Google Scholar]
- Chung, M.C.; Bosquesi, P.L.; dos Santos, J.L. A prodrug approach to improve the physico-chemical properties and decrease the genotoxicity of nitro compounds. Curr. Pharm. Design 2011, 17, 3515–3526. [Google Scholar]
- Halbrook, N.J.; Lawrence, R.V. The isolation of dehydroabietic acid from disproportionated rosin. J. Org. Chem. 1966, 31, 4246–4247. [Google Scholar]
- Sheldrick, G.M. SHELX-97; University of Gǒttingen: Gǒttingen, Germany, 1997. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gu, W.; Wu, R.; Qi, S.; Gu, C.; Si, F.; Chen, Z. Synthesis and Antibacterial Evaluation of New N-acylhydrazone Derivatives from Dehydroabietic Acid. Molecules 2012, 17, 4634-4650. https://doi.org/10.3390/molecules17044634
Gu W, Wu R, Qi S, Gu C, Si F, Chen Z. Synthesis and Antibacterial Evaluation of New N-acylhydrazone Derivatives from Dehydroabietic Acid. Molecules. 2012; 17(4):4634-4650. https://doi.org/10.3390/molecules17044634
Chicago/Turabian StyleGu, Wen, Rongrong Wu, Shilong Qi, Chenhai Gu, Fanjunnan Si, and Zhuhui Chen. 2012. "Synthesis and Antibacterial Evaluation of New N-acylhydrazone Derivatives from Dehydroabietic Acid" Molecules 17, no. 4: 4634-4650. https://doi.org/10.3390/molecules17044634




