TLC Screening for Antioxidant Activity of Extracts from Fifteen Bamboo Species and Identification of Antioxidant Flavone Glycosides from Leaves of Bambusa. textilis McClure
Abstract
:1. Introduction
2. Results and Discussion
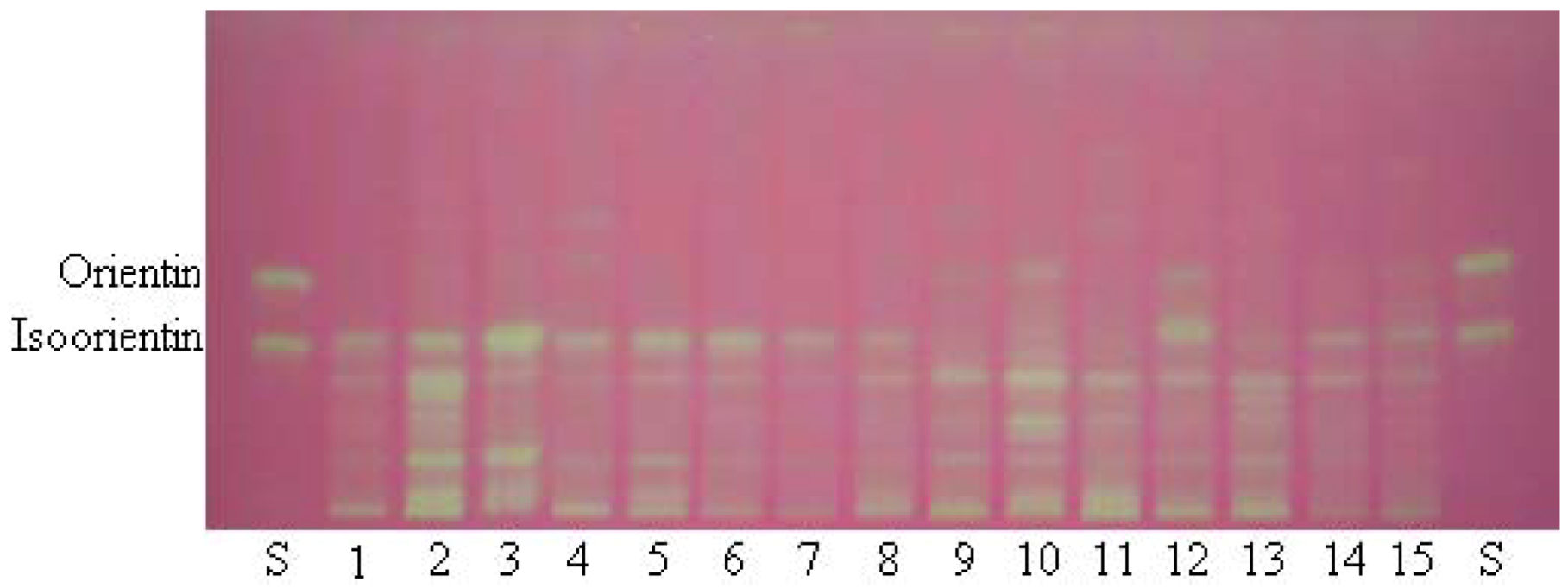
2.1. TLC Bioautography Assay
| No. | Family | Plant scientific name | Sampling Location | |
|---|---|---|---|---|
| 1 | Bambusa | B. multiplex cv. Alphonse-Karr | Nanjing, China | |
| 2 | Bambusa | B. textilis McClure | Nanchang, Jiangxi Province, China | |
| 3 | Bambusa | B. multiplex cv. Silverstripe | Nanchang, Jiangxi Province, China | |
| 4 | Bambusa | B. ventricosa McClure | Nanchang, Jiangxi Province, China | |
| 5 | Bambusa | B. multiplex var. multiplex | Nanjing, China | |
| 6 | Bambusa | B. multiplex cv. Fernleaf | Nanchang, Jiangxi Province, China | |
| 7 | Bambusa | B. multiplex var. riviereorum R.Maire | Nanchang, Jiangxi Province, China | |
| 8 | Bambusa | B. pervariabilis McClure | Nanning, Guangxi Province, China | |
| 9 | Bashania | B. fargesii (E.G. Camus) Keng f.et Yi | Nanjing, China | |
| 10 | Brachystachyurn | B. densiflorum (Rendle) Keng | Nanchang, Jiangxi Province, China | |
| 11 | Chimonobambusa | Ch. quadrangularis (Fenzi) Makino | Nanchang, Jiangxi Province, China | |
| 12 | Chimonocalamus | Chi. delicatus Hsueh et Yi | Jinping, Yunnan Province, China | |
| 13 | Dendrocalamus | D. oldhamic McClure | Nanchang, Jiangxi Province, China | |
| 14 | Dendrocalamus | D. minor var. amoenus (Q.H.Dai et C.F.Huang) Hsueh et D.Z.Li | Yibin, Sichuan Province, China | |
| 15 | Fargesia | F. hsuehiana Yi | Jinping, Yunnan Province, China | |
2.2. Image Processing
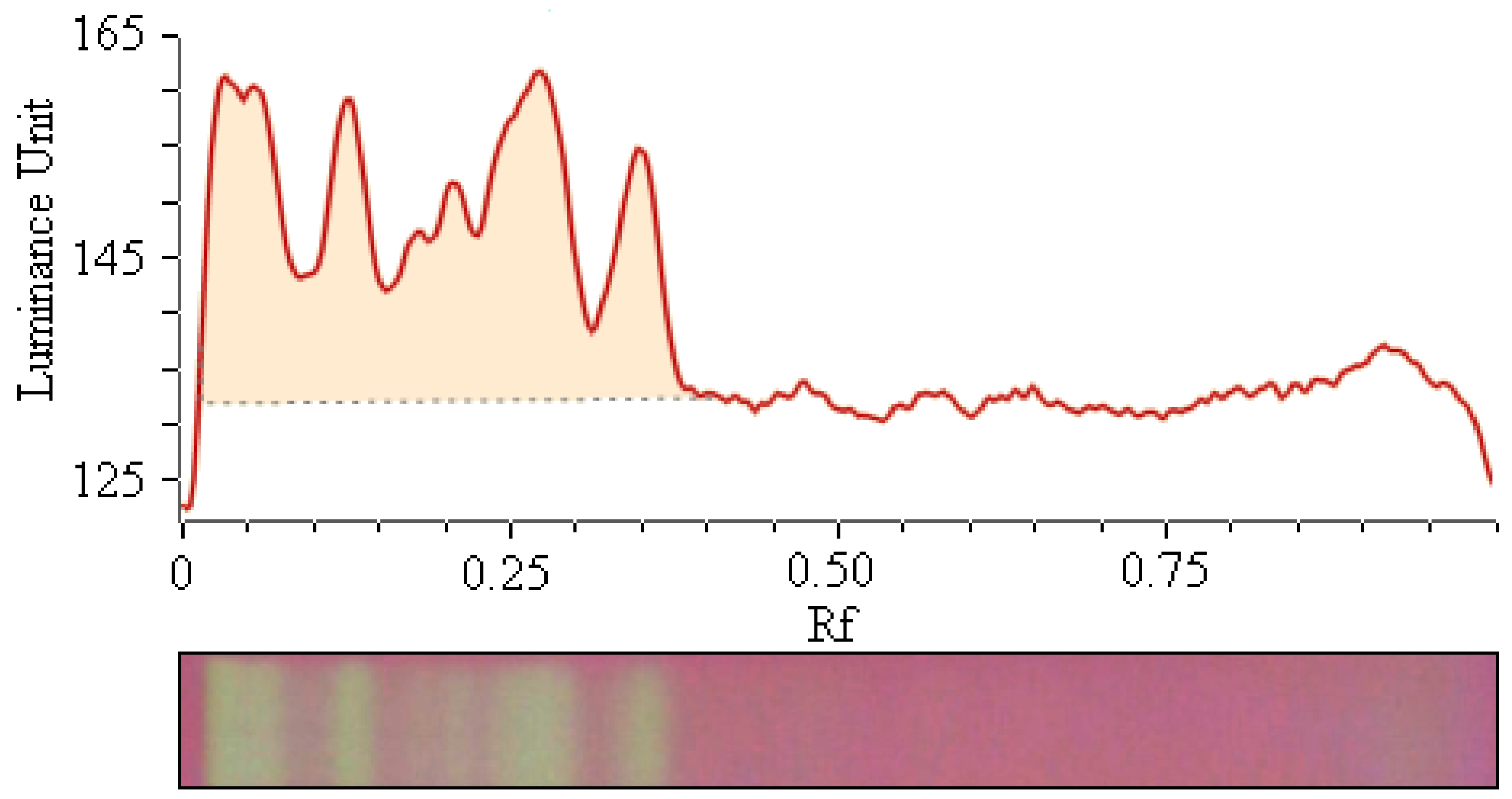
2.3. Comparison of Antioxidant Activity of Selected Bamboo Leaves

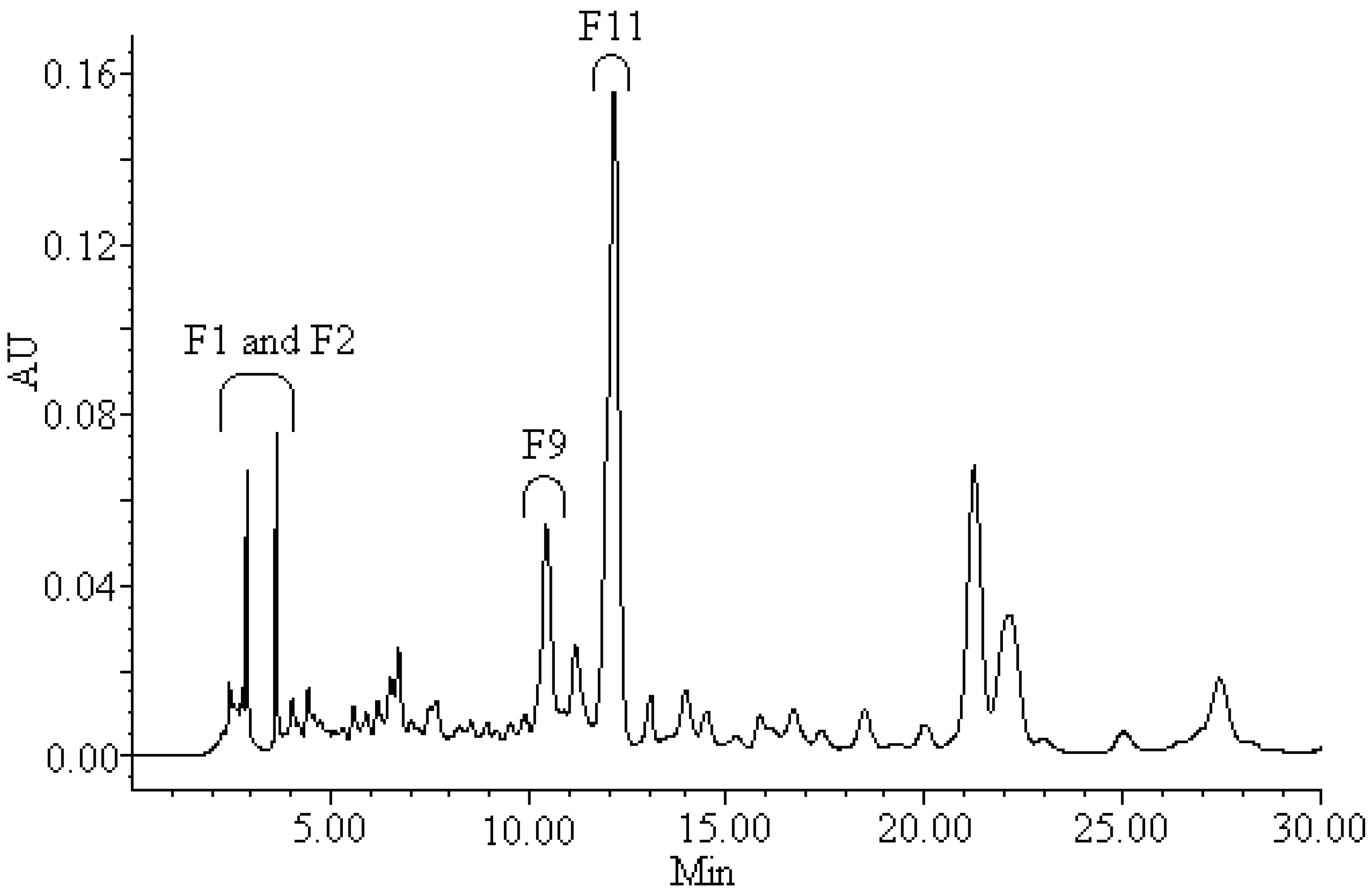
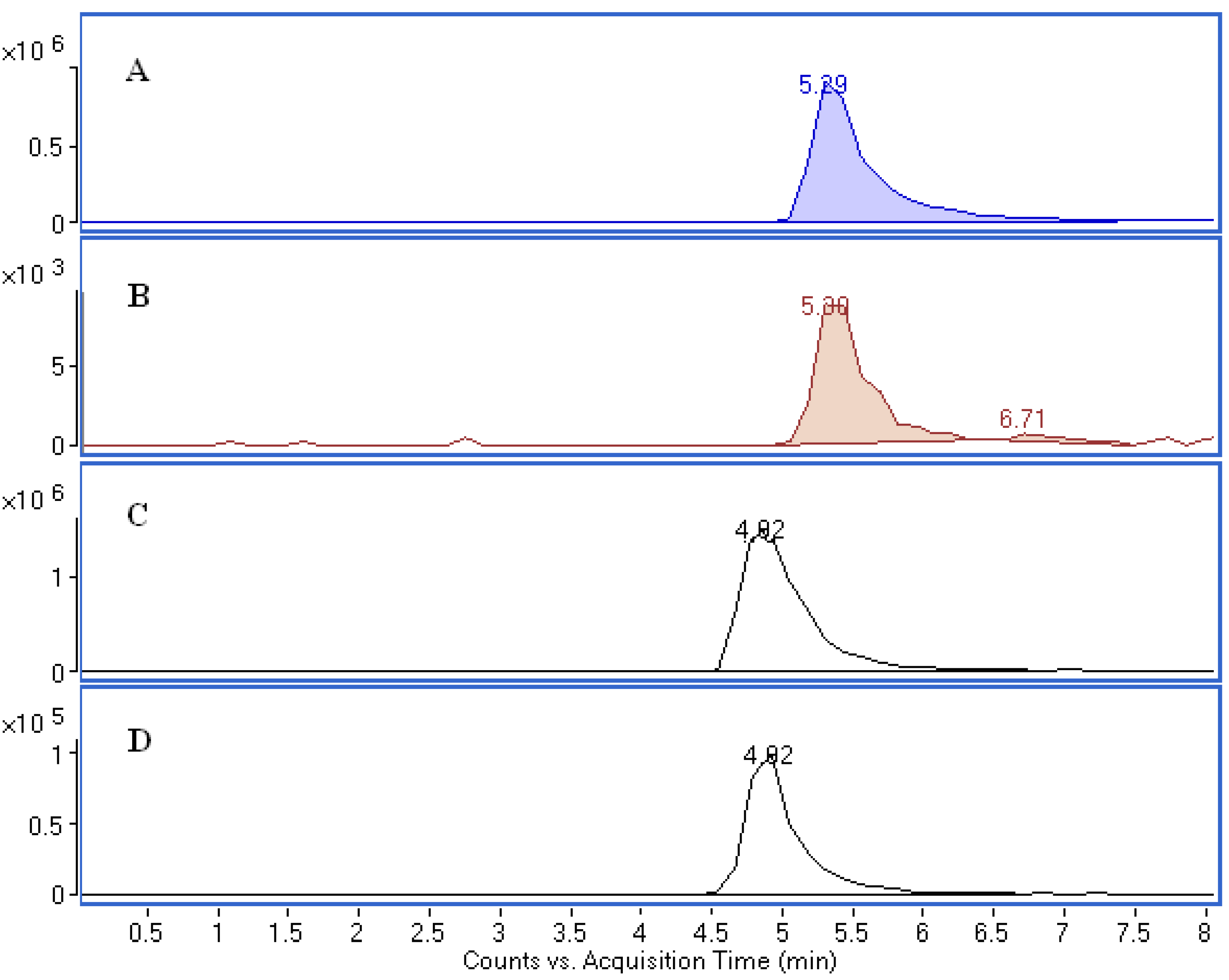
2.4. Screening and Isolation of Antioxidant Compounds

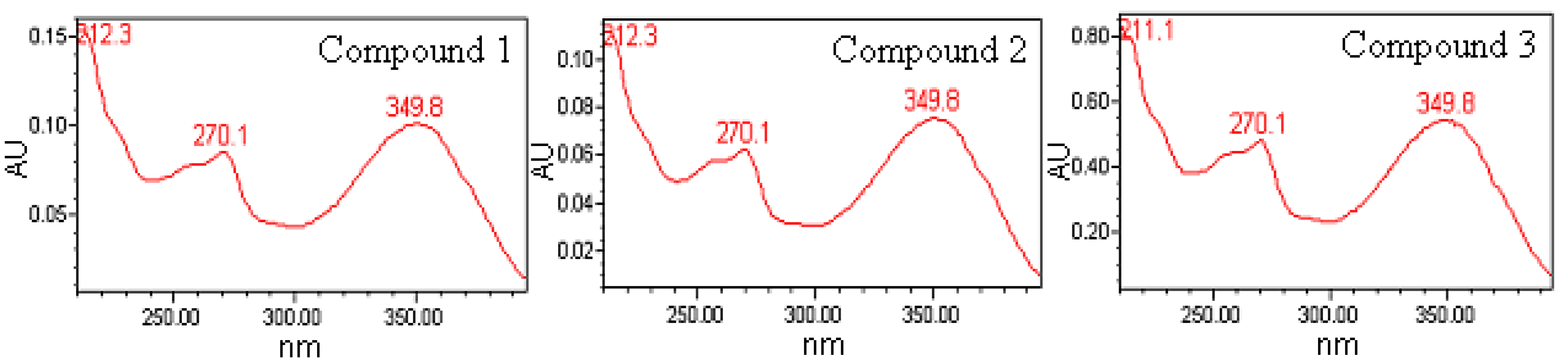
2.5. Structure Identification of the Three Antioxidant Compounds
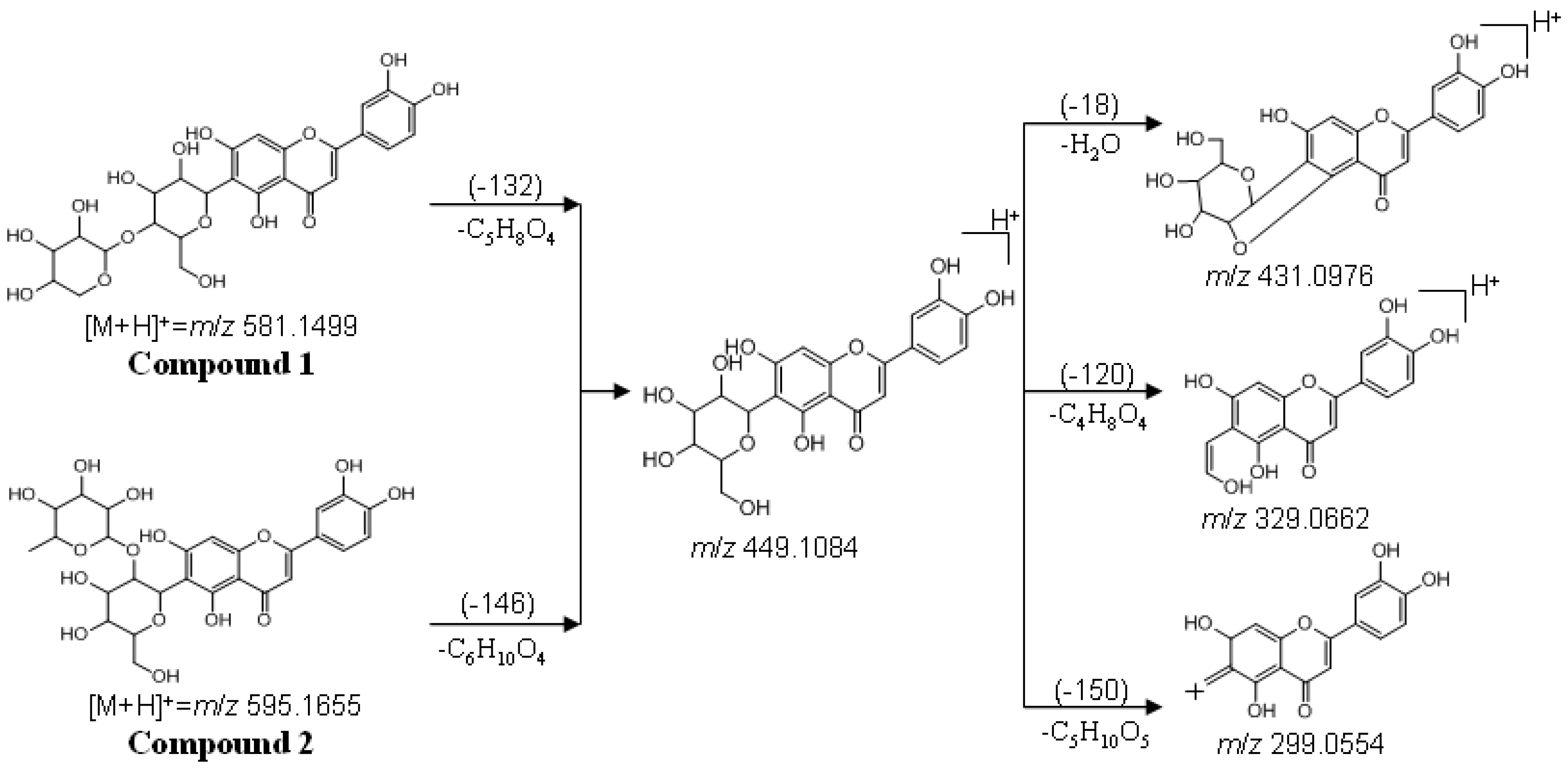
2.6. LC-Q-TOF-MS Analysis
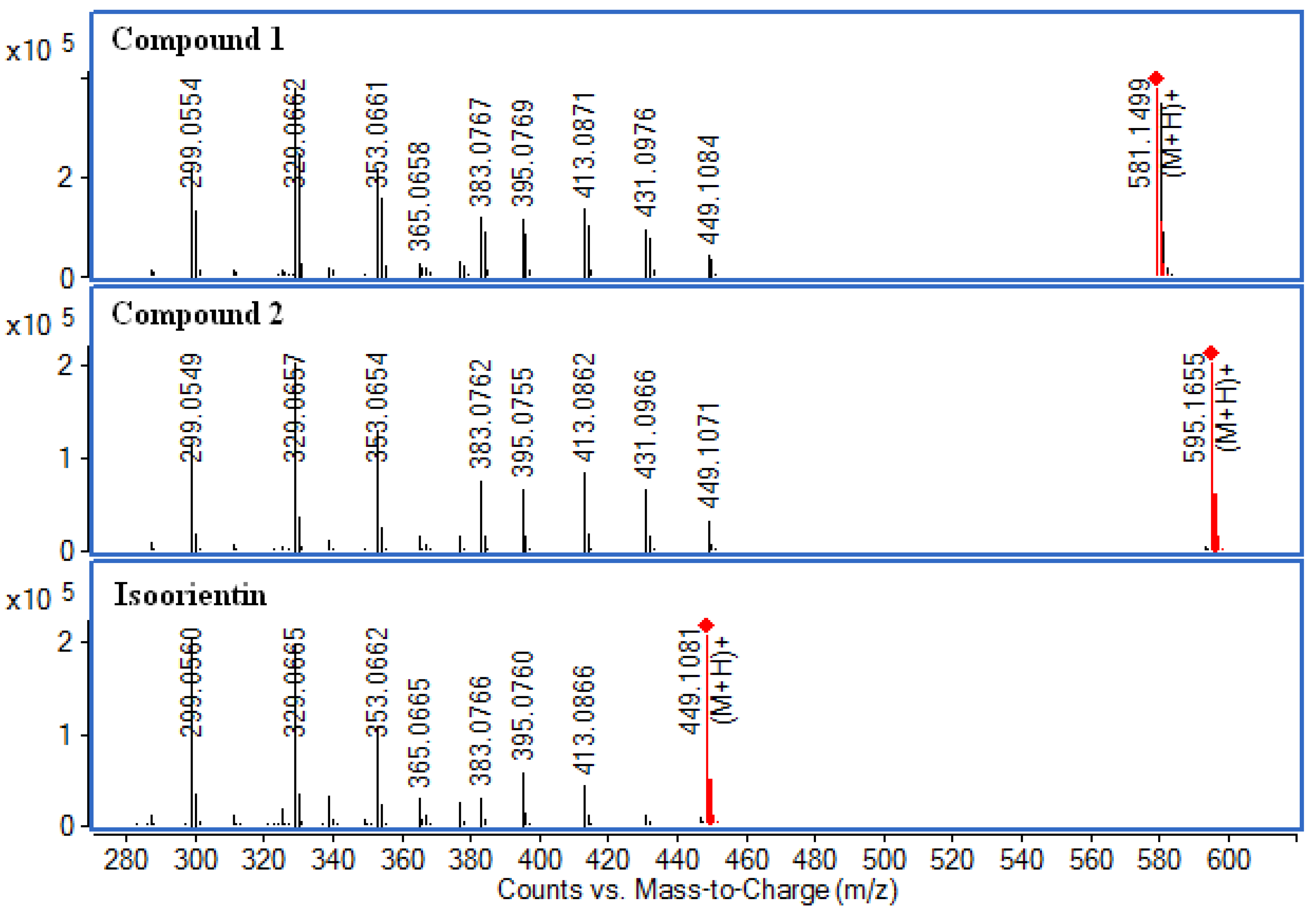
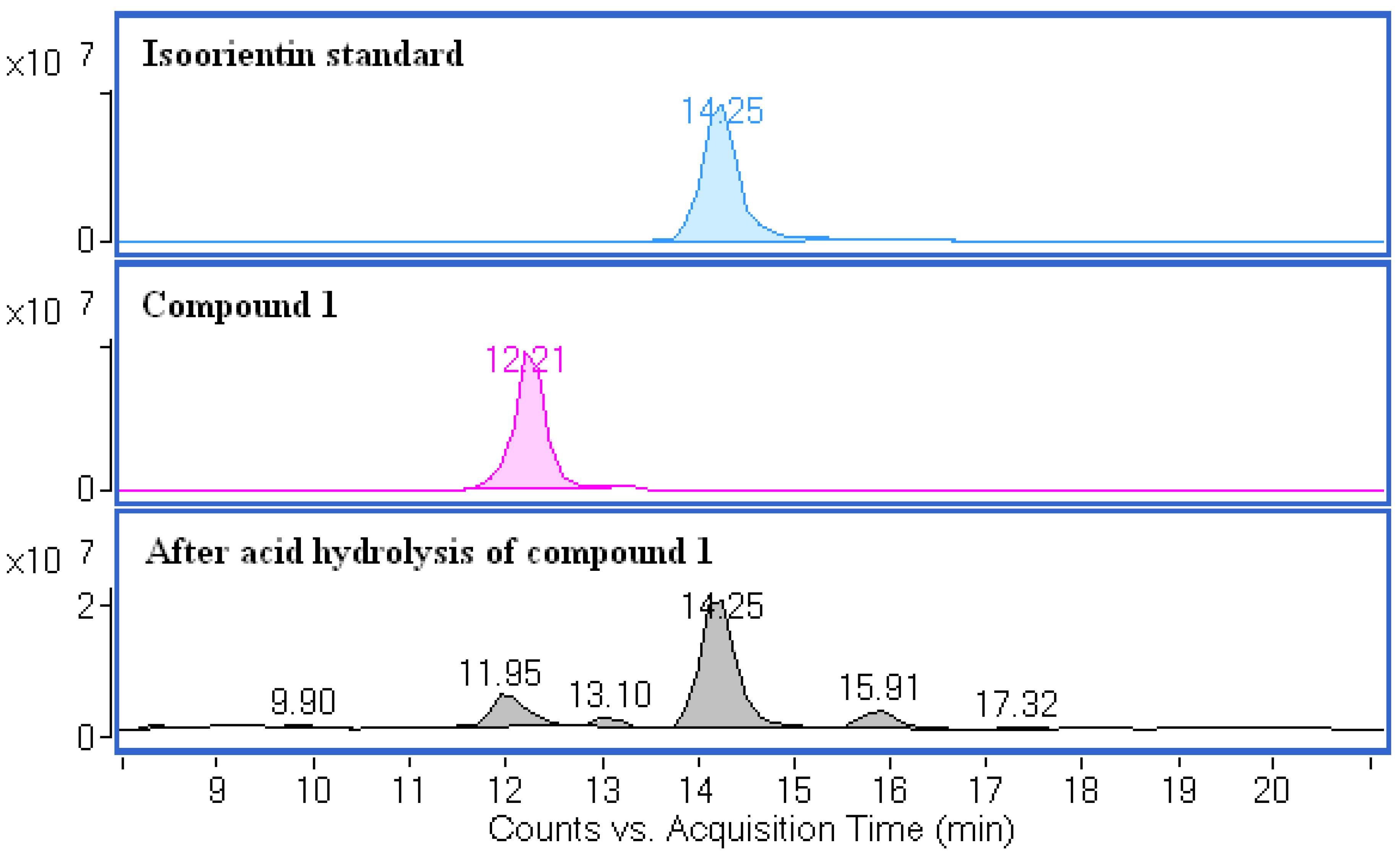
2.7. Acid Hydrolysis of Compound 1 and 2
3. Experimental
3.1. General
3.2. Materials
3.3. Chemicals
3.4. Standard and Sample Preparation
3.5. TLC Bioautography Assay
3.6. TLC Images Processing and Evaluation of Antioxidant Activity
3.7. Preparation of Crude Extract of B. textilis McClure
3.8. HPLC Micro-fractionation
3.9. Isolation of Antioxidant Compounds from B. textilis McClure
3.10. Analytical Data
3.11. LC-Q-TOF-MS Analysis
3.12. Acid Hydrolysis of Compounds 1 and 2
4. Conclusions
Supplementary Materials
Acknowledgments
- Sample Availability: Samples of the compounds isoorientin 4"-O-β-D-xylopyranoside and isoorientin 2"-O-α-L-rhamnoside are available from the authors.
References
- Kweon, M.H.; Hwang, H.J.; Sung, H.C. Identification and antioxidant activity of novel chlorogenic acid derivatives from bamboo (Phyllostachys. edulis). J. Agric. Food Chem. 2001, 10, 4646–4655. [Google Scholar]
- Wang, J.; Yue, Y.D.; Jiang, H.; Tang, F. Rapid screening for flavone C-glycosides in the leaves of different species of bamboo and simultaneous quantitation of four marker compounds by HPLC-UV/DAD. Int. J. Anal. Chem. 2012, 2012, 205101:1–205101:8. [Google Scholar]
- Hoyweghen, L.V.; Karalic, I.; Calenbergh, S.V.; Deforce, D.; Heyerick, A. Antioxidant flavone glycosides from the leaves of Fargesia. robusta. J. Nat. Prod. 2010, 9, 1573–1577. [Google Scholar]
- Zhang, Y.; Bao, B.; Lu, B.; Ren, Y.; Tie, X.; Zhang, Y. Determination of flavone C-glucosides in antioxidant of bamboo leaves (AOB) fortified foods by reversed-phase high-performance liquid chromatography with ultraviolet diode array detection. J. Chromatogr. A 2005, 2, 177–185. [Google Scholar]
- Zhang, Y.; Jiao, J.; Liu, C.; Wu, X.; Zhang, Y. Isolation and purification of four flavone C-glycosides from antioxidant of bamboo leaves by macroporous resin column chromatography and preparative high-performance liquid chromatography. Food Chem. 2008, 3, 1326–1336. [Google Scholar]
- Park, H.S.; Lim, J.H.; Kim, H.J.; Choi, H.J.; Lee, I.S. Antioxidant flavone glycosides from the leaves of Sasa. borealis. Arch. Pharm. Res. 2007, 2, 161–166. [Google Scholar]
- Jasprica, I.; Bojic, M.; Mornar, A.; Besic, E.; Bucan, K.; Medic-Saric, M. Evaluation of antioxidative activity of croatian propolis samples using DPPH and ABTS + stable free radical assays. Molecules 2007, 5, 1006–1021. [Google Scholar]
- Zhao, J.; Zhang, J.S.; Yang, B.; Lv, G.P.; Li, S.P. Free radical scavenging activity and characterization of sesquiterpenoids in four species of curcuma using a TLC bioautography assay and GC-MS analysis. Molecules 2010, 11, 7547–7557. [Google Scholar]
- Kusznierewicz, B.; Piekarska, A.; Mrugalska, B.; Konieczka, P.; Namieśnik, J.; Bartoszek, A. Phenolic composition and antioxidant properties of polish blue-berried honeysuckle genotypes by HPLC-DAD-MS, HPLC postcolumn derivatization with ABTS or FC, and TLC with DPPH visualization. J. Agric. Food Chem. 2012, 7, 1755–1763. [Google Scholar]
- Olech, M.; Komsta, Ł.; Nowak, R.; Cieśla, Ł.; Waksmundzka-Hajnos, M. Investigation of antiradical activity of plant material by thin-layer chromatography with image processing. Food Chem. 2012, 1, 549–553. [Google Scholar]
- Cimpoiu, D.C. Analysis of some natural antioxidants by thin-layer chromatography and high performance thin-layer chromatography. J. Liq. Chromatogr. R. T. 2006, 7-8, 1125–1142. [Google Scholar]
- Badarinath, A.V.; Mallikarjuna, K.; Madhau Sudhana Chetty, C.; Ramkanth, S.; Rajan, T.V.S.; Guanaprahash, K. A review on in vitro antioxidant methods: comparisions, correlations and considerations. Int. J. Pharm. Tech. Res. 2010, 2, 1276–1285. [Google Scholar]
- Wang, J.; Yue, Y.D.; Tang, F.; Sun, J. Screening and analysis of the potential bioactive components in rabbit plasma after oral administration of hot-water extracts from leaves of Bambusa. textilis McClure. Molecules 2012, 8, 8872–8885. [Google Scholar]
- Ruiz-Terán, F.; Medrano-Martínez, A.; Navarro-Ocaña, A. Antioxidant and free radical scavenging activities of plant extracts used in traditional medicine in Mexico. Afr. J. Biotechnol. 2010, 12, 1886–1893. [Google Scholar]
- Rumzhum, N.N.; Rahman, M.M.; Kazal, M.K. Antioxidant and cytotoxic potential of methanol extract of Tabernaemontana. divaricata leaves. Int. Curr. Pharm. J. 2012, 2, 27–31. [Google Scholar]
- Lapornik, B.; Wondra, A.G.; Prošek, M. Comparison of TLC and spectrophotometric methods for evaluation of the antioxidant activity of grape and berry anthocyanins. J. Planar Chromatogr. 2004, 3, 207–212. [Google Scholar]
- Ignatov, S.; Shishniashvili, D.; Ge, B.; Scheller, F.W.; Lisdat, F. Amperometric biosensor based on a functionalized gold electrode for the detection of antioxidants. Biosens. Bioelectron. 2002, 3, 191–199. [Google Scholar]
- Figueirinha, A.; Paranhos, A.; Pérez-Alonso, J.J.; Santos-Buelga, C.; Batista, M.T. Cymbopogon citratus leaves: characterization of flavonoids by HPLC-PDA-ESI/MS/MS and an approach to their potential as a source of bioactive polyphenols. Food Chem. 2008, 3, 718–728. [Google Scholar]
- Lv, Z.; Dong, J.; Zhang, B. Rapid identification and detection of flavonoids compounds from bamboo leaves by LC-(ESI)-IT-TOF/MS. BioResources 2011, 2, 1405–1418. [Google Scholar]
- Hasegawa, T.; Tanaka, A.; Hosoda, A.; Takano, F.; Ohta, T. Antioxidant C-glycosyl flavones from the leaves of Sasa. kurilensis var. gigantea. Phytochemistry 2008, 6, 1419–1424. [Google Scholar]
- Mu, J.; Uehara, T.; Li, J.; Furuno, T. Identification and evaluation of antioxidant activities of bamboo extracts. For. Stud. China 2004, 2, 1–5. [Google Scholar]
- Zeraik, M.L.; Yariwake, J.H. Quantification of isoorientin and total flavonoids in Passiflora. edulis fruit pulp by HPLC-UV/DAD. Microchem. J. 2010, 1, 86–91. [Google Scholar]
- Tian, R.T.; Xie, P.S.; Liu, H.P. Evaluation of traditional Chinese herbal medicine: Chaihu (Bupleuri radix) by both high-performance liquid chromatographic and high-performance thin-layer chromatographic fingerprint and chemometric analysis. J. Chromatogr. A 2009, 11, 2150–2155. [Google Scholar]
- Ragab, E.A.; Hosny, M.; Kadry, H.A.; Ammar, H.A. Flavanone glycosides from Gleditsia. caspia. J. Nat. Prod. 2010, 35–46. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, J.; Yue, Y.-D.; Tang, F.; Sun, J. TLC Screening for Antioxidant Activity of Extracts from Fifteen Bamboo Species and Identification of Antioxidant Flavone Glycosides from Leaves of Bambusa. textilis McClure. Molecules 2012, 17, 12297-12311. https://doi.org/10.3390/molecules171012297
Wang J, Yue Y-D, Tang F, Sun J. TLC Screening for Antioxidant Activity of Extracts from Fifteen Bamboo Species and Identification of Antioxidant Flavone Glycosides from Leaves of Bambusa. textilis McClure. Molecules. 2012; 17(10):12297-12311. https://doi.org/10.3390/molecules171012297
Chicago/Turabian StyleWang, Jin, Yong-De Yue, Feng Tang, and Jia Sun. 2012. "TLC Screening for Antioxidant Activity of Extracts from Fifteen Bamboo Species and Identification of Antioxidant Flavone Glycosides from Leaves of Bambusa. textilis McClure" Molecules 17, no. 10: 12297-12311. https://doi.org/10.3390/molecules171012297




